Abstract
Introduction:
Vena cava filters have been used as a primary means to prevent symptomatic pulmonary embolism (PE) in trauma patients who cannot be anticoagulated after severe injury, but the economic implications for this practice remain unclear.
Methods:
Using a healthcare system perspective to analyze the a priori primary outcome of the da Vinci trial, we report the cost-effectiveness of using vena cava filters as a primary means to prevent PE in patients who have contraindications to prophylactic anticoagulation after major trauma.
Results:
Of the 240 patients enrolled, complete, prospectively collected, hospital cost data during the entire hospital stay − including costs for the filter, medical/nursing/allied health staff, medical supplies, pathology tests, and radiological imaging − were available in 223 patients (93%). Patients allocated to the filter group (n = 114) were associated with a reduced risk of PE (0.9%) compared to those in the control group (n = 109, 5.5%; p = 0.048); and the filter’s benefit was more pronounced among those who could not be anticoagulated within 7 days (filter: 0% vs control: 16%, Bonferroni-corrected p = 0.02). Overall, the cost needed to prevent one PE was high (AUD $379,760), but among those who could not be anticoagulated within 7 days, the costs to prevent one PE (AUD $36,156; ~ USD $26,032) and gain one quality-adjusted life-year (AUD $30,903; ~ USD $22,250) were substantially lower.
Conclusion:
The cost of using a vena cava filter to prevent PE for those who have contraindications to prophylactic anticoagulation within 3 days of injury is prohibitive, unless such contraindications remain for longer than 7 days. (
Introduction
The financial burden of venous thromboembolism − due to pulmonary embolism (PE) and deep vein thrombosis (DVT) − including loss in potential productivity in Australia was $1.72 billion (Australian dollars, AUD) per year according to the report by the Australian National Safety and Quality Health Service.1,2 The burden of venous thromboembolism in relation to healthcare resource utilization and return to work was similarly high in European Union countries. 3
Venous thromboembolism is common after major trauma; an older study reported that without prophylactic anticoagulation, up to 18% and 11% of major trauma patients would develop proximal DVT and PE, respectively. 4 The use of retrievable vena cava filters as a primary means to prevent symptomatic PE in patients who have contraindications to prophylactic anticoagulant is highly contentious. Although it may offer theoretical benefits in reducing PE, these filters are expensive and associated with serious complications including filter adherent to the vena cava wall and migration. 5 The da Vinci multicenter randomized controlled trial (RCT) was conducted to address this perplexing issue, by assessing if placement of a retrievable vena cava filter in trauma patients with a contraindication to standard prophylactic anticoagulant within 72 hours of admission would reduce the composite endpoint of mortality or symptomatic PE.6–8 Because vena cava filters are expensive, the cost-effectiveness of using a vena cava filter to prevent symptomatic PE − including the costs of the filter and the procedures to implant and retrieve the device, complications related to the filter, procedures needed to diagnose and treat DVT and PE, total length of intensive care unit (ICU) and hospital stay, procedures and drugs needed to treat bleeding complications − was also a primary endpoint of this trial. 6
We hypothesized that if vena cava filters are effective in reducing PE in trauma patients without known venous thromboembolism and the incidence of PE is sufficiently high, using a vena cava filter as a primary means to prevent symptomatic PE may reduce the net total healthcare cost, or at least be cost neutral. 9 This result is achievable, at least theoretically, when the incremental cost of the filter and the costs of the procedure to insert and retrieve the device are offset by the saving achieved by having fewer symptomatic PE and associated complications. A vena cava filter can be particularly cost-effective if it can also reduce fatal PE. In the current economic analysis, we report the cost-difference and cost-effectiveness analyses of using vena cava filters as a primary means to prevent symptomatic PE in patients who had contraindications to prophylactic anticoagulation within the first 72 hours of admission after major trauma.
Methods
This economic analysis was a co-primary endpoint of the da Vinci trial (Australian New Zealand Clinical Trials Registry no.: ACTRN12614000963628) and a prespecified substudy, which was approved by the relevant ethics committees of the participating centers. 6 Written informed consent was obtained before enrollment in the da Vinci trial from all patients who were assessed as being competent to provide consent. If patients were not competent to provide consent, their next of kin agreed to enrollment and signed an acknowledgment document; patients provided written informed consent after they regained competence. In brief, the da Vinci trial recruited and randomly allocated 240 severe trauma patients, who had contraindications to anticoagulant prophylaxis within 72 hours of admission to a trauma center, to receive either a vena cava filter or standard care alone. The primary composite-outcome of 90-day all-cause mortality or symptomatic PE was not significantly different between the two groups.7,8 The difference in symptomatic PE between the two treatment arms in patients who could not be anticoagulated within 7 days of injury – mainly due to severe brain contusions requiring ongoing surgical interventions − was prespecified as an a priori secondary outcome; in this subgroup, vena cava filters were effective in reducing symptomatic PE (n = 80; filter group: 0% vs control group: 14.7%, including one fatal PE; p = 0.01). Furthermore, the number of patients with thrombus entrapped in the vena cava filters in the filter group was identical to the number of patients with symptomatic PE in the control group, suggesting that the filters were achieving the desired effect in preventing embolization of lower limb thrombi to the pulmonary circulation. 7
In order to address the financial implications of using vena cava filters as a primary means to prevent symptomatic PE, we compared the total healthcare cost of using vena cava filters as a primary means to prevent PE with the total cost of not using the filters (i.e., cost-difference analysis), and also the net cost needed to achieve the net health benefits (i.e., cost-effective analysis or cost needed to prevent one symptomatic PE).
Cost-difference analysis
Comparing the total hospitalization costs per study patient between the two arms of the trial would indicate whether there was a net increase in cost, it was cost neutral, or there was a cost savings by using vena cava filters as a primary measure to prevent symptomatic PE in major trauma patients who had contraindications to prophylactic anticoagulation within the first 72 hours after trauma admission.
The current cost analysis was taken from the Australian health system perspective using the funding provided to each recruited patient according to the Australian Activity-Based-Funding (ABF) model. The Australian public healthcare system is a single-payer system; all cost items were actual costs incurred by each individual patient to the healthcare system and not charged to the patients or the insurance, although some of the costs could have been reimbursed subsequently by the insurance companies if patients were either privately insured or involved in a motor-vehicle accident. The total cost of using a vena cava filter for prophylaxis against symptomatic PE included the filter device itself, the consumables required for insertion and removal, the costs of personnel required for the procedure, and costs of complications. Because there was a potential in saving by reducing symptomatic PE, and its associated effect on length of ICU and hospital stay, or bleeding requiring blood transfusion, the cost components for both arms of the trial also captured costs beyond the cost of the filter alone. The total healthcare cost for each patient thus included the costs of the (a) length of ICU stay and hospitalization; (b) medical/nursing/allied health staff; (c) all pharmaceuticals; and (d) investigations and treatments for PE or DVT, such as X-rays, computed tomography (CT) pulmonary angiographies, ultrasonography scans, pathology tests, anticoagulants, and also blood products for bleeding (due to use of anticoagulants or other causes).
All cost data were drawn from the hospital finance department and all resources were standardized to 2020 Australian dollars, using financial resource data such as Australia’s Medicare Benefits Scheme (MBS) for medical procedures, the Australian Red Cross cost data for blood products, and the Pharmaceutical Benefits Scheme for pharmaceuticals. In brief, the cost of a retrievable vena cava filter was AUD $1800.00 (which included the full delivery system), and the cost of the filter removal kit was $1891.65 (which included the recovery cone at $1475.15, three dilators [5 Fr, 7 Fr, 9 Fr] at $10.5 each, a guidewire at $115.0, and a long 12 Fr sheath at $270). The MBS costs for chest X-ray, CT head (or chest or abdomen) scan with intravenous contrast, CT pulmonary angiography, bilateral limb Doppler ultrasound scan, insertion of a vena cava filter, and removal of the filter were $47.15, $567.75, $510.0, $169.5, $515.35, and $592.45, respectively. The costs of a unit of packed red blood cells, pooled platelets, (nonpooled) cryoprecipitate, and fresh frozen plasma were $377.36, $253.39, $167.33, and $154.54, respectively (https://www.blood.gov.au/national-product-list). In this study, one Australian dollar was converted to 0.72 US dollar (USD) when reporting the cost in US currency.
Cost-effectiveness analysis
The cost of using a vena cava filter to prevent one symptomatic PE was calculated using the mean cost-difference per study patient between the two arms of the trial multiplied by the number needed to treat (NNT) to prevent one PE by comparing the absolute risk difference in the incidence of symptomatic PE between the two arms of the trial. Cost per life-year gained (LYG) was estimated if there was an all-cause mortality benefit at day-90 after using the vena cava filters to prevent symptomatic PE. We also conducted a threshold analysis to assess when a vena cava filter could be considered cost-effective by assuming the usual limit of less than US$100,000 per LYG was acceptable. In this analysis, we assumed that those who survived the injury would survive until 4 years less than the average life expectancy of other Australians, which was 79.4 years for a male and 84.4 years for a female. 10 In estimating the cost-effectiveness of the filter in terms of quality-adjusted life-years (QALYs), we assumed the annual quality of life disutility after PE to be 0.03. 11
Statistical analysis
Categorical variables such as incidence of PE and DVT, and continuous variables including the total healthcare cost per patient were compared using chi-squared and Mann–Whitney tests, respectively. In reporting the cost-difference and cost-effectiveness analyses, the whole cohort as well as the a priori subgroup – those who had persistent contraindications to prophylactic anticoagulation for longer than 7 days − were analyzed and reported separately to assess whether the economic costs of using a vena cava filter as the primary means to prevent PE were different for patients who had different risks of developing symptomatic PE after major trauma.
We developed a logistic regression model to identify predictors for a delay (beyond 7 days) in receiving prophylactic anticoagulation, which constituted the cohort we used for the a priori secondary outcome. The Injury Severity Score (ISS), body mass index, use of an intracranial pressure monitor, and pattern of injury, including spinal cord injury with neurological deficits, complex pelvic fractures, lower limb fractures, major bleeding requiring a transfusion of more than six units of red blood cells within the first 24 hours, effacement of basal cistern, midline shift, or intracranial hematoma / cerebral contusions on the CT brain scan, were initially included in this logistic regression model. Predictors that were not associated with a p-value less than 0.10 were then removed to improve the precision of the final logistic regression model. In addition, we also wanted to assess whether the healthcare cost-difference between using and not using a filter to prevent symptomatic PE was related to the risk of developing PE, as measured by the Trauma Embolic Scoring System (TESS) score.12,13 Because healthcare costs were not normally distributed, we logarithmically transformed the total healthcare cost of each patient in this analysis.
In this study, all tests were two-tailed, based on intention-to-treat analysis and conducted using SPSS for Windows, version 22.0 (IBM Corp., Armonk, NY, USA; 2013) and MedCalc 19.7.4 (MedCalc Software Ltd, Ostend, Belgium; 2021). A p-value less than 0.05 was taken as significant, but to account for multiple statistical testing, we used Bonferroni-correction to adjust the p-value associated with the a priori secondary outcome.
Results
Study subjects and outcomes
Of the 240 patients included in the study, 223 (114 in the filter group and 109 in the control group) had complete financial data available for the current economic analysis. Seventeen patients (7.1%) were excluded because they were recruited into the study centers outside Western Australia or they had been repatriated back to their home states or overseas before 90 days after enrolment. Two patients in the analysis crossed over to the other arm of the trial but their cost and effectiveness outcomes were analyzed according to intention-to-treat in this economic analysis. The total healthcare cost for each patient increased with their risks of developing thromboembolism according to the TESS score (slope of overall regression line = 0.2, 95% CI: 0.13–0.20; p = 0.0001) (Figure 1).
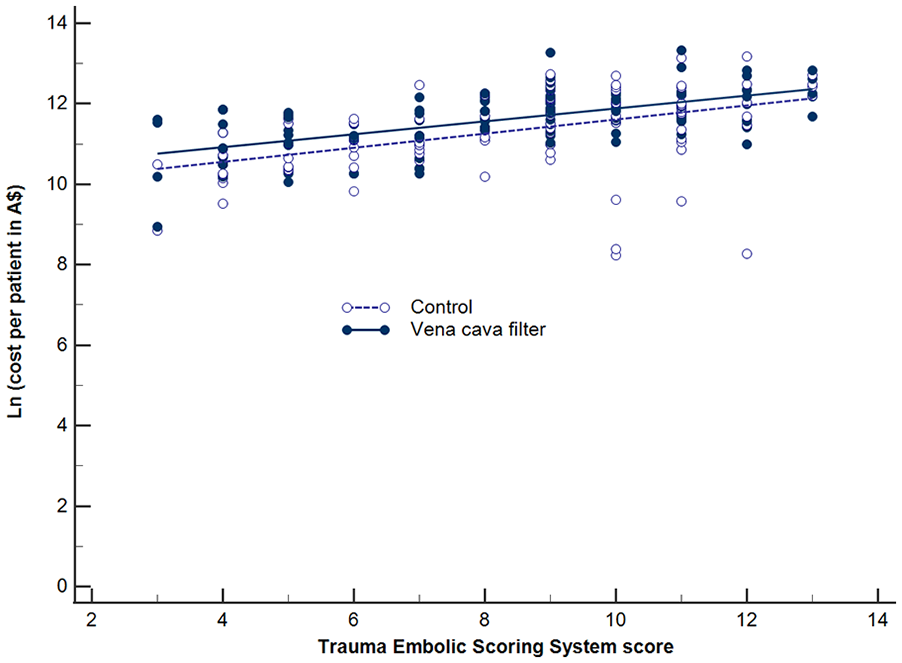
Difference in costs (in logarithmic scale) between those allocated to the vena cava filter and control groups in relation to increasing predicted risks of developing thromboembolism after trauma.
The baseline characteristics, including the severity of injury and risk of developing thromboembolism − estimated by the TESS scores − were comparable between patients allocated to either arm of the trial (Table 1). For the effectiveness endpoint, there was a significant reduction in the incidence of symptomatic PE in patients allocated to the filter group (0.9% vs 5.5%, p = 0.048) compared to the control group without increasing the incidence of lower limb DVT. The beneficial effect of the filter on risk of symptomatic PE was slightly different from the primary analysis due to a difference in the total number of patients analyzed (or the denominators used) in the current study. 8
Characteristics of the da Vinci trial patients included in the economic analysis (N = 223).
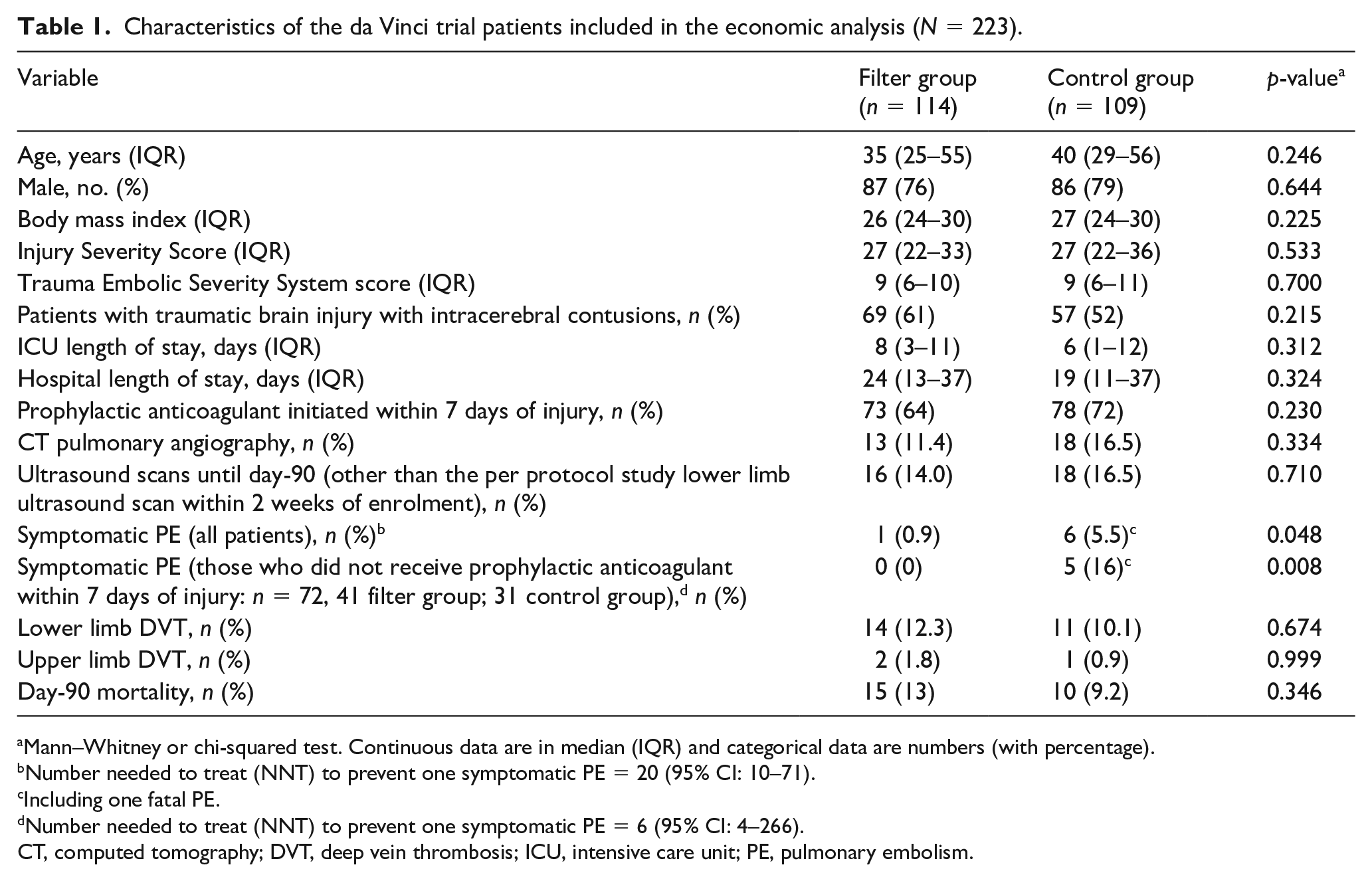
Mann–Whitney or chi-squared test. Continuous data are in median (IQR) and categorical data are numbers (with percentage).
Number needed to treat (NNT) to prevent one symptomatic PE = 20 (95% CI: 10–71).
Including one fatal PE.
Number needed to treat (NNT) to prevent one symptomatic PE = 6 (95% CI: 4–266).
CT, computed tomography; DVT, deep vein thrombosis; ICU, intensive care unit; PE, pulmonary embolism.
Logistic regression analysis showed that only four clinical factors were associated with an increased risk of having a delay (beyond 7 days after injury) in receiving prophylactic anticoagulation (Table 2). For this subgroup of patients, the benefit of the filter in reducing symptomatic PE was even greater (filter group: 0% vs control group: 16%, p = 0.008; Bonferroni-corrected p-value = 0.02).
Predictors for delaying initiation of prophylactic anticoagulation beyond 7 days after the injury (N = 240).
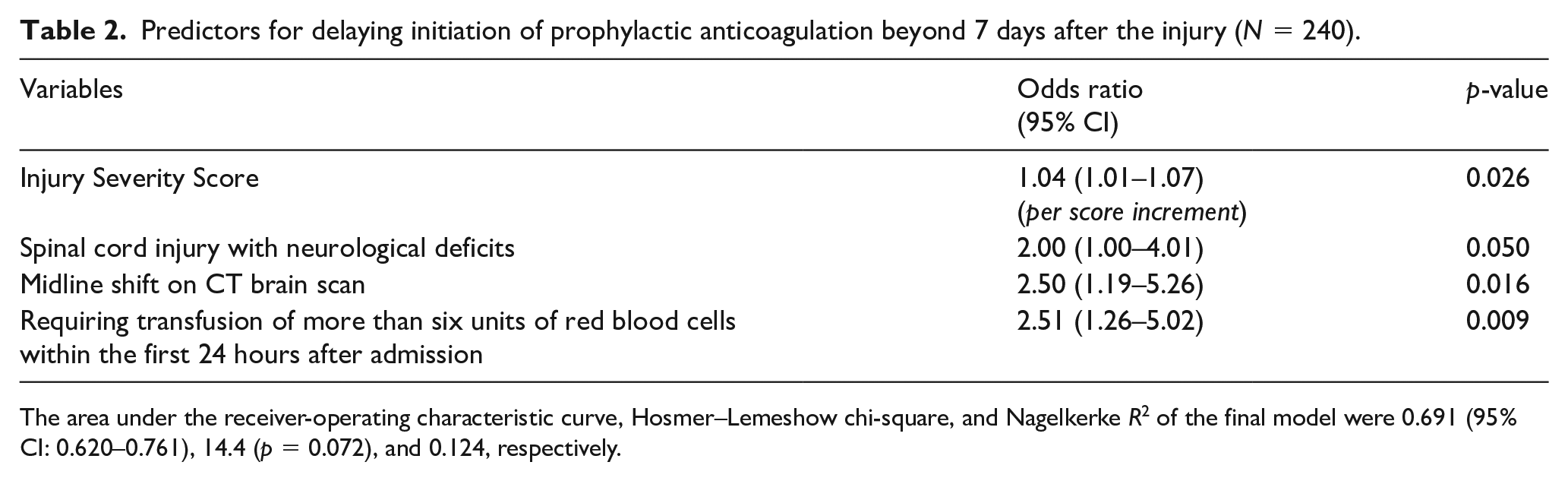
The area under the receiver-operating characteristic curve, Hosmer–Lemeshow chi-square, and Nagelkerke R2 of the final model were 0.691 (95% CI: 0.620–0.761), 14.4 (p = 0.072), and 0.124, respectively.
Cost-difference between with and without using vena cava filters
As expected, the vena cava filter and total radiology costs per patient were significantly higher among those allocated to the filter group (Table 3). The blood product and other hospital costs were, however, not significantly different between the two groups. The net total healthcare cost per patient was thus higher among those allocated to the filter group (AUD $140,112, IQR 74,890–186,189 or USD $100,881, IQR 53,921–134,056) than those in the control group (AUD $121,112, IQR 47,785–168,739 or USD $87,209, IQR 34,405–121,492). However, a relatively higher total healthcare cost per patient was not observed among those who remained untreated with prophylactic anticoagulation within 7 days of injury (filter: AUD $138,280 vs control: $132,254; p = 0.941) (Table 4), with the costs of using the filter offset by the savings from having a lower incidence of symptomatic PE and its complications. The cost-difference between using and not using a vena cava filter to prevent symptomatic PE was not significantly related to the patients’ risk of developing thromboembolism, as measured by the TESS score (difference in slopes = 0.01, standard error 0.04; p = 0.684) (Figure 1).
Major itemized cost of per study patient enrolled in the da Vinci trial (N = 223).
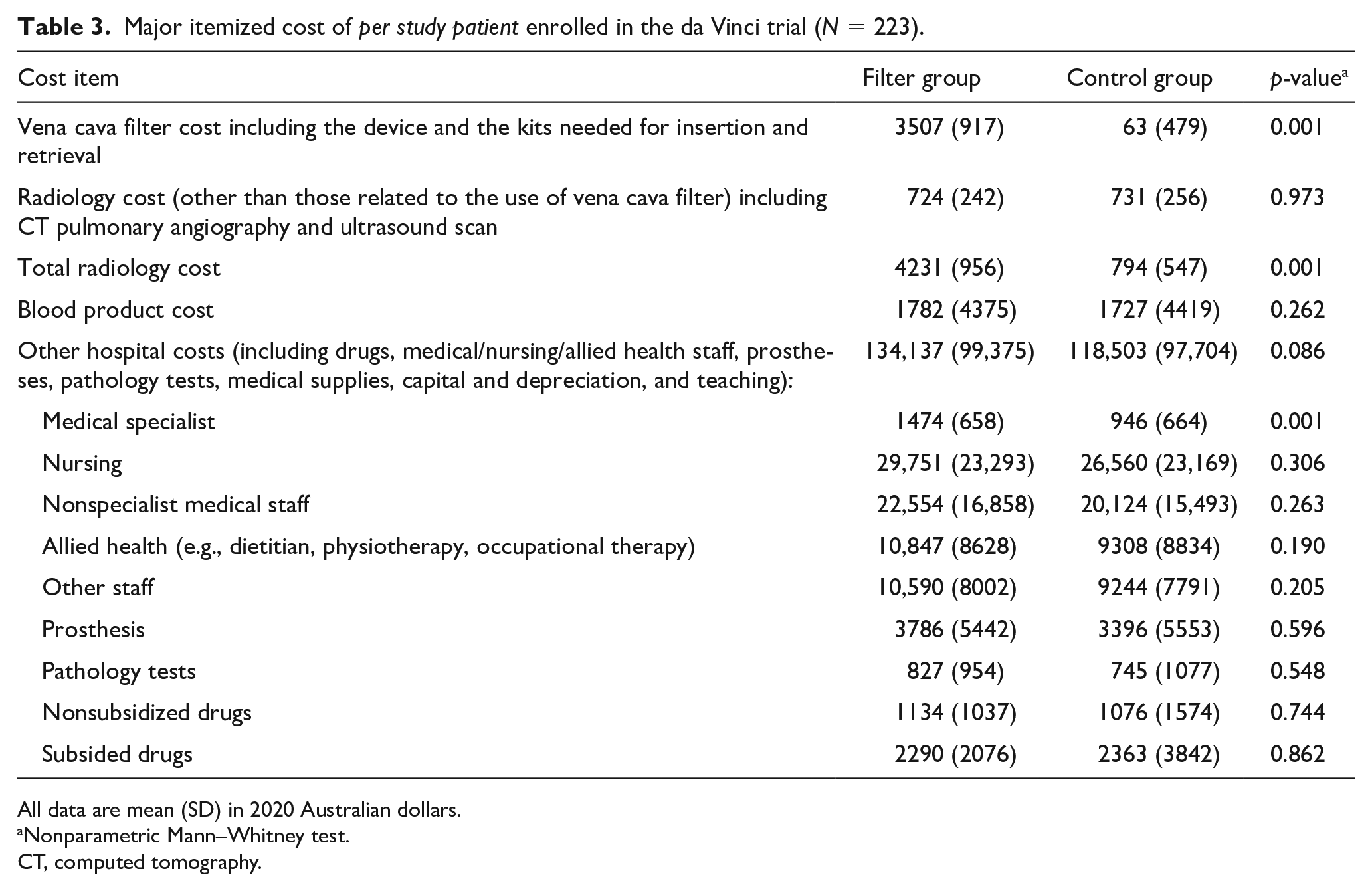
All data are mean (SD) in 2020 Australian dollars.
Nonparametric Mann–Whitney test.
CT, computed tomography.
Differences in total healthcare costs per patient between the vena cava filter and control groups, stratified by whether the patients were treated or untreated with prophylactic anticoagulant within 7 days of injury.
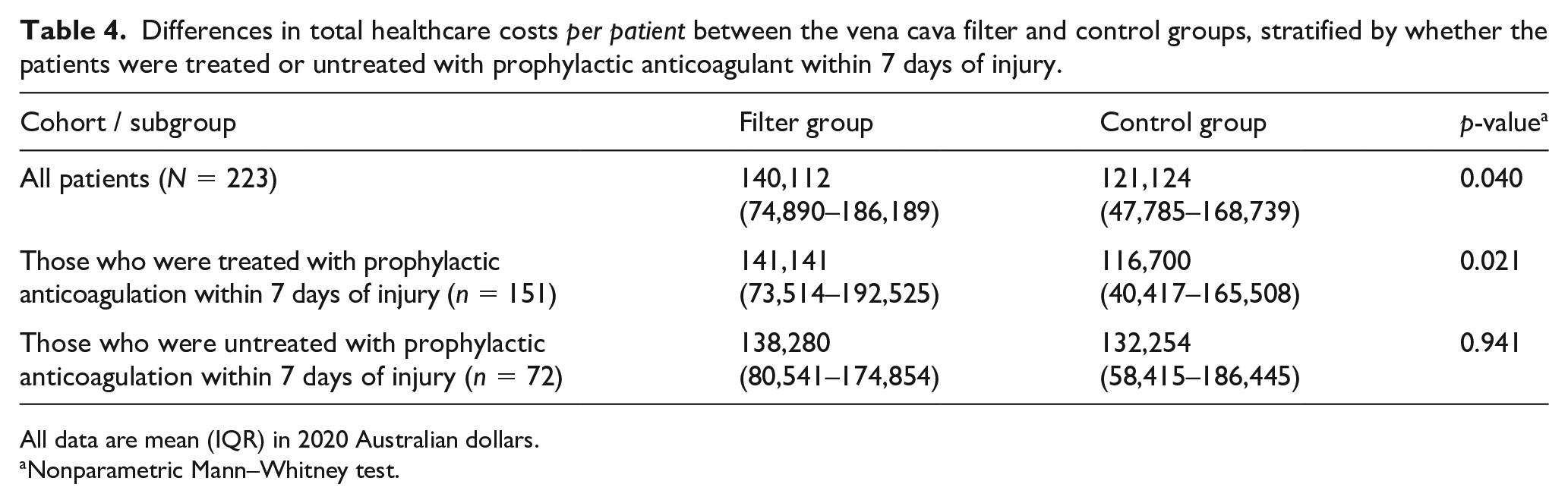
All data are mean (IQR) in 2020 Australian dollars.
Nonparametric Mann–Whitney test.
Cost-effectiveness of using vena cava filters to prevent symptomatic PE
When the whole cohort (N = 223) was considered, the cost of using a vena cava filter to prevent one symptomatic PE in major trauma patients with contraindications to prophylactic anticoagulation was estimated to be AUD $379,760 (or USD $201,427). The cost of using vena cava filters to prevent one symptomatic PE was substantially lower (AUD $36,156 or USD $26,032) when the analysis was restricted to those who had a prolonged period of contraindications to prophylactic anticoagulation (> 7 days) after the injury (n = 72).
The mortality difference at day-90 after enrolment between the arms was not observed (filter: 13% vs control: 9.2%, p = 0.346) and, hence, the actual cost per LYG was not estimated. Using the mean age of the male and female patients in our study for the threshold analysis, in order for the filter to be considered cost-effective (< USD $100,000 per LYG), it would require the filter to reduce the fatal PE rate by 0.42% compared to without using a filter. The cost per QALY gained for the whole cohort and the subgroup who could not be anticoagulated within 7 days were estimated to be AUD $324,581 (USD $233,698) and $30,903 ($22,250), respectively.
Discussion
This economic analysis showed that the healthcare system cost of using a retrievable vena cava filter to prevent symptomatic PE in patients who have contraindications to prophylactic anticoagulation is prohibitive, with the exception of those who continue to have such contraindications for a prolonged period of time (e.g., 7 days). These results have clinical implications and require further discussion.
First, apart from morbidity and mortality, venous thromboembolism also poses a substantial financial burden on the healthcare system.1–3 The treatment of an acute venous thromboembolism has been reported to be associated with an incremental direct medical cost of over USD $12,000 (2014 USD) within the first year. A substantial amount of the medical cost (USD $18,000–$23,000) is also needed to manage complications related to venous thromboembolism subsequent to the first year. 14 In two other studies, the mean total hospitalization costs for DVT and PE have been reported to be USD $9407 and between $8764 and $11,486, respectively.15,16 As such, the cost of using any strategies to prevent PE must be weighed against the total cost needed to manage PE and its associated complications. Preventing symptomatic PE in severe trauma patients who cannot be anticoagulated due to bleeding risk is particularly challenging because there are not many effective alternatives.11,17 Whether vena cava filters should be used in such situations remains unclear and highly contentious, resulting in wide practice variations in many trauma centers. 18 The da Vinci multicenter RCT provides the strongest evidence so far on the effectiveness of using vena cava filters as a primary means to prevent symptomatic PE in patients who have contraindications to prophylactic anticoagulation after major trauma. 8 Whether this strategy would significantly increase (or decrease) hospitalization cost, and whether it is cost-effective, remains uncertain. 19 The current economic analysis provides an important piece of information because the study design aimed to prospectively capture all components of healthcare costs of the study patients, including the costs of DVT, PE, transfusion for bleeding, anticoagulants, pathology tests, radiology imaging, personnel and procedures needed to insert and retrieve the filters, and managing the filter complications. Our results showed that using vena cava filters as a primary means to prevent symptomatic PE in major trauma patients who had contraindications to prophylactic anticoagulation after major trauma substantially increased the total hospitalization cost (USD $13,672 per patient). Furthermore, this strategy was also not cost-effective, with the cost needed to prevent one symptomatic PE being prohibitively high (USD $201,427) compared to the standard average cost needed to treat PE in hospitalized patients.9,15,16 Nonetheless, if we restrict the use of vena cava filters only to those who could not be anticoagulated for a prolonged period of time (e.g., 7 days) after major trauma (among whom the cumulative risk of symptomatic PE was high), the cost needed to prevent one symptomatic PE (USD $26,032) became substantially lower and the cost-effectiveness of using a vena cava prophylactically (USD $22,250 per QALY gained) would become more acceptable.9,10 The cost needed to prevent one PE by the filter for this highly selective subgroup of trauma patients will be even more acceptable if we also factor in the substantial direct and indirect costs of managing long-term complications of symptomatic PE (USD $18,000–$23,000).1,14
Second, there is an important caveat in interpreting the cost-effectiveness of using retrievable vena cava filters to prevent PE in major trauma patients. We noted that there was one patient allocated to the control arm who died from PE confirmed by the post-mortem examination (0.9% vs 0% in the filter group, 95% CI: –2.3% to +4.6%). 8 Comparing this result with our threshold analysis – which showed that a 0.42% reduction in fatal PE would make the filter cost less than US$100,000 per LYG – would suggest that if the reduction in fatal PE rate in our trial can be confirmed by a larger size RCT (with 867 subjects per group to achieve 80% power to detect ⩾ 0.9% difference in fatal PE rates between the two groups), 19 this would have a dramatic positive impact on the cost-effectiveness of using vena cava filters as a primary means to prevent PE after major trauma. This is because many trauma patients are young; any preventable deaths averted by a vena cava filter could result in a substantial gain in many productive life-years, 20 making use of the vena cava filter to prevent PE cost-effective from a societal perspective.1,2
Study limitations
Finally, we need to acknowledge the limitations of this study. The current economic analysis was only based on a healthcare system perspective up to day-90 after study enrolment without considering out-of-hospital healthcare costs, including medical investigation and drug costs that may be required after hospital discharge and 90 days after trauma. It is well-established that DVT and PE can have long-term consequences affecting patients’ quality of life, their ability and time taken to return to work, and hence also productivity for the society as a whole.1–3 Furthermore, although our a priori subgroup was prespecified in our trial design6,8 and we also adjusted our analysis of this subgroup by Bonferroni-correction, survivor bias may still exist, which may have inflated the potential benefit of the filter in preventing PE among those who survived longer than 7 days after study enrolment. 8
Conclusion
In summary, from a healthcare system perspective, the cost of using a retrievable vena cava filter (as a primary means) to prevent symptomatic PE in patients who have contraindications to prophylactic anticoagulation within the first 3 days after injury is prohibitive. For those who have a prolonged period (e.g., > 7 days) of contraindications to prophylactic anticoagulation, the high incidence of symptomatic PE (> 10–15%)8,9 may justify the costs of using a vena cava filter as a primary means to prevent PE, especially when the long-term medical and nonmedical costs of PE are also considered.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was jointly funded by the Trauma Research Program from the Royal Perth Hospital Medical Research Foundation, Translational Research Project from the Western Australian Department of Health, and Clinician Research Fellowship (round 3) from the Raine Medical Research Foundation. No financial or nonmonetary (in-kind) support was received from any companies that manufacture vena cava filters or from commercial entities. The funding agencies have no role in the design of the study, data analysis, interpretation of the results, and the decision to submit this manuscript for publication.
Data availability
Nonidentifiable study data will be available upon reasonable request by contacting the corresponding author after all the substudies of the da Vinci trial have been completed and published.
