Abstract

The majority of patients with iliofemoral deep vein thrombosis (DVT) have an underlying iliac vein compression. 1 Approximately 50% develop the post-thrombotic syndrome (PTS) if treated conservatively. 2 As in May–Thurner syndrome, common iliac vein lesions are typically located close to the iliocaval confluence and are caused by iliac vein compression from overriding iliac arteries against the spine. Conventional non-oblique stents had been used for many years to treat iliac vein compression syndromes by extending the stent across the iliocaval confluence into the inferior vena cava, thereby jailing the contralateral outflow. The risk of contralateral DVT with a conventional stent was 10% after 5 years. 3 A self-expandable nitinol stent with a 35° oblique-shaped proximal end (sinus-Obliquus; Optimed, Ettlingen, Germany) was designed for the treatment of common iliac vein compression to minimize contralateral outflow obstruction (figure in the online supplemental material).
In the prospective, international, monitored, single-arm TOPOS (Treatment Of Post-thrombotic syndrome with the Oblique Stent) study, we included 60 consecutive PTS patients who required endovascular therapy and who were treated with the self-expandable oblique stent for unilateral common iliac vein obstruction. TOPOS recruited from experienced centers that performed at least 30 deep venous interventions per year. We have described the study specifics and methodology previously. 4 All participants provided written informed consent and the study was approved by local research ethics committees. In brief, stent extension of the oblique stent in or below the external iliac vein was permitted using the sinus-XL Flex or sinus-Venous stents (both from Optimed) in the case of insufficient stent inflow. TOPOS did not mandate the use of intravascular ultrasound (IVUS). Follow-up visits were scheduled for 3, 6, 12, and 24 months following the procedure, each of which included a duplex ultrasound exam for stent patency evaluation. The course of clinical signs and symptoms was estimated with the Villalta score and revised venous clinical severity score (rVCSS), as well as the patient based visual analog scale for pain (VAS) and the chronic lower limb venous insufficiency questionnaire (CIVIQ-20).5–7 Recently, all remaining study participants had completed the 12-month follow-up visit.
The primary outcome was the primary patency rate at 12 months. The secondary outcomes were primary assisted patency, secondary patency, and changes in signs and symptoms at 12 months relative to baseline using the aforementioned clinical scores. Stent patency rates have been outlined in accordance with the reporting standards of the Society for Vascular Surgery. 8 In case of an inconclusive ultrasound exam, it was adjudicated for the worst-case scenario (non-patency). We calculated 12-month incidences for the primary and secondary outcomes accompanied by the corresponding 95% CI. The mean values of continuous dependent variables, namely the clinical scores for PTS, were compared at baseline and follow-up by analysis of covariance (ANCOVA). SAS software, Version 9.4 (SAS Institute Inc., Cary, NC, USA) served for statistical analysis.
Two patients withdrew informed consent, and five patients were lost to follow-up. At baseline, 43% of patients had a mild (Villalta score: 5–9 points), 30% had a moderate (10–14 points), and 27% had a severe PTS (> 14 points) according to the Villalta classification. At baseline, 92% of patients reported difficulties while exercising, 87% had pain in their ankles or thighs, and 13% had active venous ulcers. Seventy percent of patients had extensive post-thrombotic disease requiring stent extension below the inguinal ligament. Patient characteristics and procedural details are displayed in Table 1. Type and duration of antithrombotic therapy was left to the treating physician. At 6 months, 56 patients had ongoing oral anticoagulation therapy (OAC): 47 had direct oral anticoagulants (DOACS), nine vitamin-K antagonists (VKA), and 12 additional platelet inhibitors. At 12 months, 45 patients had ongoing OAC: 37 had DOACs, eight VKA, and six additional platelet inhibitors.
Study data
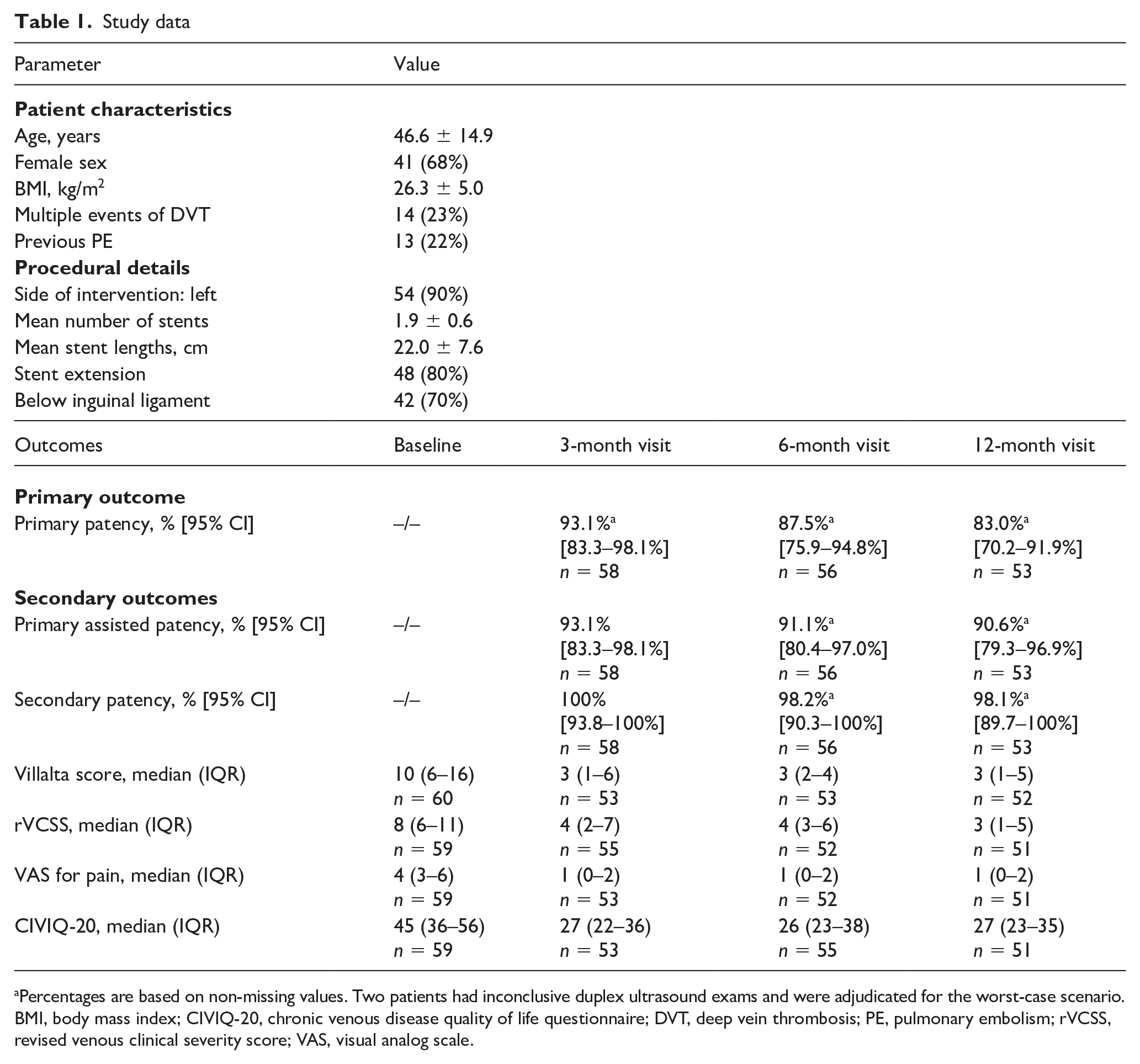
Percentages are based on non-missing values. Two patients had inconclusive duplex ultrasound exams and were adjudicated for the worst-case scenario.
BMI, body mass index; CIVIQ-20, chronic venous disease quality of life questionnaire; DVT, deep vein thrombosis; PE, pulmonary embolism; rVCSS, revised venous clinical severity score; VAS, visual analog scale.
At 12 months, primary, assisted primary, and secondary patency rates were 90% (95% CI 55.5–99.7%), 100% (95% CI 69.2–100%), and 100% (95% CI 69.2–100%) in patients with the self-expandable oblique stent only, and 81.4% (95% CI 66.6–91.6%), 88.4% (95% CI 74.9–96.1%), and 97.6% (95% CI 87.4–99.9%) in patients with stent extension below the inguinal ligament, respectively. Overall, eight (13%) patients (seven with stent extension) underwent re-intervention to maintain treatment success (three were treated for stent stenosis, five for stent occlusion); six had therapeutic-dose anticoagulation therapy at time of diagnosis. The primary and secondary outcomes are displayed in Table 1. At 12 months, 60% of patients were free from PTS; 18% had mild, 7% moderate, and 2% severe PTS. Mean improvement in Villalta, rVCSS, CIVIQ-20, and VAS from baseline to follow-up was 8.4 ± 6.9 (95% CI 6.5–10.3; p < 0.0001; n = 52) points, 5.1 ± 3.5 (95% CI 4.1–6.1; p < 0.0001; n = 51) points, 15.6 ± 12.4 (95% CI 12.1–19.1; p < 0.0001; n = 50) points, and 2.9 ± 2.2 (95% CI 2.3–3.5; p < 0.0001; n = 50) points, respectively. At 12 months, 37% of patients reported difficulties while exercising, 38% pain in their ankles or thighs, and none had active venous ulcers. No patient developed contralateral DVT, pulmonary embolism, or major bleeding during 12 months of follow-up. Routine assessment of stent fracture was not ascertained.
In conclusion, endovascular therapy of PTS with the self-expandable oblique venous stent and provisional extension resulted in a high primary patency rate and improvement of PTS and quality of life through 1 year. Lower patency rates were observed in patients with stent extension, which is most likely related to post-thrombotic femoral vein inflow. 9 The 2-year follow-up of TOPOS will permit an indirect comparison of patency rates with historical data of conventional non-oblique stents.
Supplemental Material
sj-png-1-vmj-10.1177_1358863X211017658 – Supplemental material for Twelve-month clinical outcomes of a hybrid oblique self-expanding nitinol stent used for the treatment of post-thrombotic syndrome with common iliac vein compression: The TOPOS study
Supplemental material, sj-png-1-vmj-10.1177_1358863X211017658 for Twelve-month clinical outcomes of a hybrid oblique self-expanding nitinol stent used for the treatment of post-thrombotic syndrome with common iliac vein compression: The TOPOS study by Tim Sebastian, Stefano Barco, Michael Lichtenberg, Stefan Stahlhoff, Oliver Schlager, Houman Jalaie, Rick de Graaf, Christian Erbel, Alexander Massmann, Marc Schindewolf and Nils Kucher in Vascular Medicine
Footnotes
Acknowledgements
The TOPOS study was conducted at eight European centers: Clinic of Angiology of the University Hospital Zurich (Switzerland); Department of Angiology, Arnsberg Medical Center, Arnsberg (Germany); Division of Angiology, Depart-ment of Medicine II, Medical University of Vienna (Austria); European Vascular Center Aachen-Maastricht, University Hospital RWTH Aachen (Germany); Department of Radiology, Interventional Radiology and Nuclear Medicine, Clinic of Friedrichshafen (Germany); Department of Cardiology, Angiology, and Pneumology, Heidelberg University Hospital, Heidelberg (Germany); Clinic for Diagnostic and Interventional Radiology, Saarland University Medical Center, Homburg (Germany); and Division of Angiology, Swiss Cardiovascular Center, Inselspital, Bern University Hospital, Bern, Switzerland.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Stefano Barco reports institutional grants from Bayer HealthCare and Sanofi; personal fees from Boston Scientific, LeoPharma, and Bayer HealthCare; and travel/congress costs from Daiichi Sankyo, outside the submitted work. Houman Jalaie reports personal fees from BD Bard, Boston Scientific, Medtronic, Optimed, Walk Vascular, and Bentley. Nils Kucher reports personal fees from Sanofi, Boston Scientific, BARD, and Optimed; and institutional grants from the Swiss Science Foundation, Bayer HealthCare, Sanofi, Pfizer, and Concept Medical. The other authors report no conflict of interest on the submitted work.
Funding
This investigator-initiated study was funded by Optimed Medizinische Instrumente GmbH. A clinical research organization (Pharmalog, Munich, Germany) performed statistical analysis based on the statistical analysis plan prepared by the investigators. Optimed had no involvement in the collection, analysis or interpretation of data.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
