Abstract
Minimum iliac vein caliber necessary to maintain normal peripheral venous pressure can be derived by the Poiseuille equation. Duplex was compared to intravascular ultrasound (IVUS) in the assessment of iliac vein stenosis in this single center retrospective study. Parallel IVUS and duplex caliber data for common iliac vein (CIV) and external iliac vein (EIV) in 382 limbs were separately compared. One or both segments were stenotic by IVUS criteria in 213 limbs. Neither segment was stenotic by IVUS in 22 limbs. Bland–Altman analyses and Passing–Bablok linear regressions were used. Duplex calibers were dimensionally smaller than corresponding IVUS images of CIV and EIV segments in Bland–Altman comparison by a mean of 54 mm2 and 34 mm2, respectively. Passing–Bablok regression suggested the difference was due to a systematic bias and not proportional. Duplex yields a smaller cross-sectional image of CIV and EIV compared to IVUS. Duplex is not a reliable diagnostic test for iliac vein stenosis.
Introduction
Iliac vein stenosis is being recognized more often and treated with stent placement in patients with advanced chronic venous disease (CVD). Intravascular ultrasound (IVUS) is increasingly used for diagnosis and procedural guidance during stent placement. IVUS appears to be superior to venography in this role. In the multicenter VIDIO trial, multiplanar venography was compared to IVUS in the detection of treatable iliac vein stenosis. 1 IVUS detected significant lesions not detected with three-view venography in 26% of 100 patients; investigators revised treatment plans after IVUS in 57% of cases because of the failure of venography to detect a significant lesion. In a recent blinded comparison of venography and IVUS from our institution, venography altogether failed to identify lesion existence in 19% of 155 limbs. 2 The maximal area stenosis was significantly higher with IVUS than with venography (69% vs 52%; p < 0.0001). Furthermore, venography missed the location of maximal stenosis in more than two-thirds of limbs. The iliac-caval confluence was located higher by as much as one vertebral body with IVUS than with venography in 74% of the patients. The distal landing zone defined with IVUS was lower than with venography in 64% of limbs. It appears that contrast-related obscuration of lesions and luminal landmarks is a major source of error with venography.
Diagnosis of stenosis in the arterial system is based on estimation of percentage stenosis relative to an adjacent ‘normal’ segment. This practice has followed in the venous system as well. A diffuse form of stenosis without focal cues (‘Rokitanski stenosis’) is uniquely present in iliac vein stenosis. The pathology arises from development of a restrictive fibrous envelope around the vein during thrombus evolution. This form of stenosis, which varies in severity, may be present in ≈25% of post-thrombotic iliac lesions and is easily missed on venography. 3 If the reference segment is diffusely stenotic, underestimation of the lesion will result (including in the arterial system from diffuse lesions of other pathology).
IVUS criteria to define and grade iliac vein stenosis are still being debated. Venous stenoses do not exhibit a ‘critical threshold’ (≈70–80%) often seen in arterial stenosis from compensatory vasodilation.4,5 Nevertheless, an 50% caliber stenosis relative to adjacent ‘normal’ segment is widely used. No basis for such a threshold has ever been published. The 50% threshold is also subject to the error from diffuse stenosis of the reference segment discussed above. Since the relationship between caliber and pressure is non-linear in veins, the 50% stenosis threshold is fundamentally erroneous.
Minimal caliber of the iliac vein segments to maintain normal venous pressure can be defined in absolute terms using the Poiseuille equation (F = ΔP*πr4/8lη). 6 Most terms in the equation specific for iliac vein are constants or known. Average flow is known as well. Because radius enters the equation in the fourth power, caliber variations to accommodate body mass-related flow variations are relatively minute. This allows a near universal definition of normal iliac vein caliber with a tight range. The minimum ‘normal’ diameter thresholds for common iliac vein (CIV) and external iliac vein (EIV) are 16 mm and 14 mm, respectively. A caliber less than stated is considered stenotic. The calculated normal sizes correspond to Young’s scaling law and IVUS measurements of non-stenotic iliac vein segments. These sizes also approximate visual observation of the iliac vein during open pelvic surgery.
These thresholds have not been validated by others but have been in use for the diagnosis and stent treatment of iliac vein stenosis in our clinic for the past 6 years with good clinical correlation.7,8 A caliber less than normal as defined is associated with peripheral venous hypertension in 76% of limbs. 9
The aim of this study is to compare the diagnostic accuracy of duplex with IVUS reference standard using the caliber method. This is a single center (three surgeons) retrospective analysis of prospectively collected data entered into an electronic medical record program.
Methods
Permissions
Informed consent from patients for the described procedures was obtained. Institutional Review Board permission was granted for analysis and publication of de-identified data.
Patients
A total of 3272 new patients were referred to our tertiary referral venous center over a 5-year period (2014–2018) for evaluation of symptoms suggestive of chronic venous disease (CVD). After clinical evaluation, patients with advanced CVD who had failed conservative therapy (typically after at least 6 months) underwent IVUS examination. All patients who had duplex assessment of iliac veins within 3 months before IVUS are included in the analysis.
Patient selection
Indications for IVUS/iliac vein stenting, preoperative work up, procedure details, stent surveillance, long-term clinical and quality of life Chronic Venous Insufficiency Quality of Life Questionnaire [CIVIQ-20] outcomes in various CVD subsets have been described in detail elsewhere.10–18 Briefly, clinical examination included CEAP (Clinical-Etiology-Anatomy-Pathophysiology) classification, venous clinical severity score (VCSS), visual analogue pain scores, physical grading of swelling (0 = none; 1 = pitting; 2 = ankle edema; 3 = gross), and quality of life measure (CIVIQ). Patients considered for IVUS underwent coagulation profile, duplex examination, ambulatory venous pressure measurement, and air-plethysmography. 19 Most IVUS candidates underwent either transfemoral venography, computed tomography (CT) venography or magnetic resonance (MR) venography, except when there was contrast allergy, renal impairment or when the patient was fragile. In latter cases, preoperative imaging was withheld in favor of a single stage IVUS followed by stenting when stenosis was found. Duplex was performed routinely in most patients but was not the determinant of proceeding with IVUS examination. The latter was based on clinical presentation fortified by venography or imaging (CT, MR) studies.
Intervention was limited to patients with CEAP ⩾ 3 with clinical correlation to a suspected obstructive lesion. Rarely, patients with ‘venous hypertension syndrome’ with no clinical signs but with underlying severe orthostatic venous hypertension underwent IVUS/stenting. 12 Asymptomatic or minimally symptomatic individuals were not investigated or stented for iliac vein obstruction. Incidentally found silent lesions, via duplex or other imaging technique, howsoever severe, were not candidates for interventions.
Inclusions
CIV and EIV segments were separately analyzed. The limb was included in the analysis when technically satisfactory IVUS and duplex data were available for at least one of the two segments.
Exclusions
Limbs with chronic total occlusions (CTO) were excluded.
Duplex-related exclusions
Out of 496 limbs, 59 (12%) were excluded because pre-IVUS duplex was older than 3 months before IVUS examination. CIV could not be adequately imaged in 101/496 (20%) limbs and EIV in 24/496 (5%) limbs due to bowel gas or obesity and were excluded.
Intravascular ultrasound (IVUS)-related exclusions
Out of 496 limbs, 137 (28%) were excluded from comparison analysis as the lumen caliber could not be completely imaged by IVUS due to a ‘missing border’ in one or more quadrants of the lesion image. 3 Of the 137 limbs with the missing border feature, 73 (53%) involved CIV, 33 (24%) involved EIV, and 31 (23%) involved both CIV and EIV. This is related to lack of a centering mechanism in the IVUS catheter yielding an incomplete image.
Data collection
Clinical parameters, duplex findings, and IVUS planimetry were contemporaneously entered into an electronic medical record program and analyzed retrospectively.
IVUS
An IVUS catheter (Visions PV .035; Philips Volcano Corporation, San Diego, CA, USA) was used. The caliber perimeter of the CIV and EIV were traced at their narrowest point with an electronic pen; the machine planimetry software provided the area.
Duplex
A color duplex instrument (Logiq 9; GE Medical Systems, Waukesha, WI, USA) with B-flow feature was used. Patients were examined during regular work hours without food or fluid restriction. They were asked to take one 125 mg tablet of simethicone 3 hours before the examination. Patients were examined in the recumbent position with a 5 HZ curvilinear probe (Model C1-6). B-mode settings: focus width 2; frame average 1; line density 1. The lumen caliber of CIV and EIV were initially defined by color duplex, rotating the probe till the image was clear of the overlapping artery and the vein caliber was maximized in width. The diameter of the lumen of each vessel at their narrowest point was then measured in B-mode or B-flow, whichever offered better image quality. The measurements are referred to as ‘B-mode measurements’ or generically as ‘duplex measurements’.
Technical
Inter-observer variation of duplex measurements among three technicians was checked in a blinded manner by different paired observations in 16 limbs. In addition, diameter measurement in longitudinal view was compared to that in transverse view in 20 limbs to determine variability with probe orientation.
Analytical methodology
The duplex measured caliber of CIV and EIV were compared to the IVUS reference standard for equivalence. The duplex caliber diameter was converted to area (πr2) for comparison with IVUS caliber area data. Reverse conversion of the IVUS planimetry area into diameter for comparison yielded a smaller area of the imputed circle than the pixel-based planimetry area and therefore was not used.
Statistics
Paired two-tailed t-tests were used to compare IVUS versus duplex caliber areas (mean ± SD). McNemar’s test for paired proportions was used to compare stenosis prevalence (%) between IVUS and duplex. The D’Agostino and Pearson test was used to test normality of caliber areas. Bland–Altman plots were used to compare duplex and IVUS caliber areas for agreement in predicting iliac vein stenosis. 20 CUSUM tests of linearity for CIV and EIV caliber areas were computed to confirm linearity. Respective caliber area data were analyzed using Passing–Bablok regression in order to determine proportional and systematic differences between IVUS and duplex methods. 21 Pearson correlation (r) was calculated for longitudinal versus transverse duplex diameter measurements. Intraclass correlation coefficients were calculated using a one-way random effects model (absolute agreement type) in order to determine inter-observer variability for CIV and EIV diameter measurements. Analyses were performed using commercial software (PRISM; La Jolla, CA, USA and MedCalc; Ostend, Belgium). Sample sizes varied due to missing data, which is reflected in the n-values provided with all tables and graphs. An alpha of p < 0.05 was used to indicate significance in all analyses.
Results
Dataset
A total of 265 CIV segments and 352 EIV segments in 382 limbs had matching duplex measurements and were available for analysis after exclusions. IVUS-defined stenotic thresholds were not met in 38 (14%) CIV segments, 71 EIV (20%) segments, and 22 limbs (6%) for both CIV and EIV segments. These limbs (negative IVUS subset) with ‘normal’ IVUS calibers are also included in the analysis. Both CIV and EIV segments were able to be included in 235 limbs.
Technical
The intraclass correlation coefficients (average measures) for diameter measurement of CIV and EIV in 16 paired blinded comparisons rotated among three technicians were r = 0.86 (CI: 0.60–0.95) and 0.92 (CI: 0.78–0.97), respectively. Measurement of duplex diameters in paired longitudinal and transverse views measured in 20 limbs were virtually identical (Pearson r = 0.96). All diameters reported in results below are from longitudinal views: caliber image was maximized by probe rotation. Localization of stenosis in transverse view was cumbersome because the probe had to be moved several times for panning the length of the two segments (≈15 cm).
Demographics
Demographic characteristics (Table 1) correspond to other large stent series. CEAP clinical class was 3–6 in 98% of limbs. Six (2%) patients (CEAP 0–2) were stented for severe orthostatic venous pain without clinical signs. 12
Demographics.
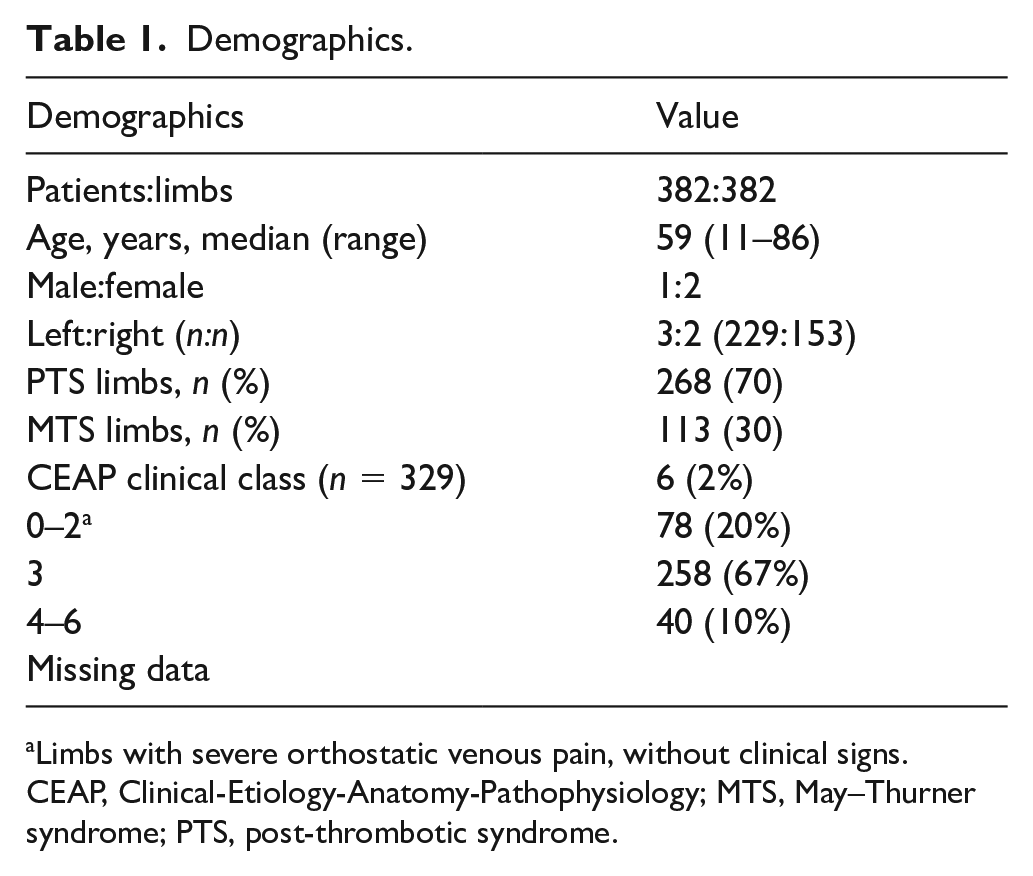
Limbs with severe orthostatic venous pain, without clinical signs.
CEAP, Clinical-Etiology-Anatomy-Pathophysiology; MTS, May–Thurner syndrome; PTS, post-thrombotic syndrome.
IVUS and duplex stenosis
The mean IVUS and duplex caliber values for CIV and EIV, as well as the prevalence distribution of caliber stenosis using the same IVUS threshold values for duplex (CIV: 200 mm2; EIV: 150 mm2), are shown in Table 2. Mean duplex lumen areas were significantly smaller and more prevalent than companion IVUS areas in both iliac vein segments. This results in a significantly higher stenosis prevalence percentage for duplex compared to IVUS when using identical stenosis thresholds. This favors high duplex false positives.
IVUS and duplex stenosis prevalence and mean caliber area (n = 235 a ).
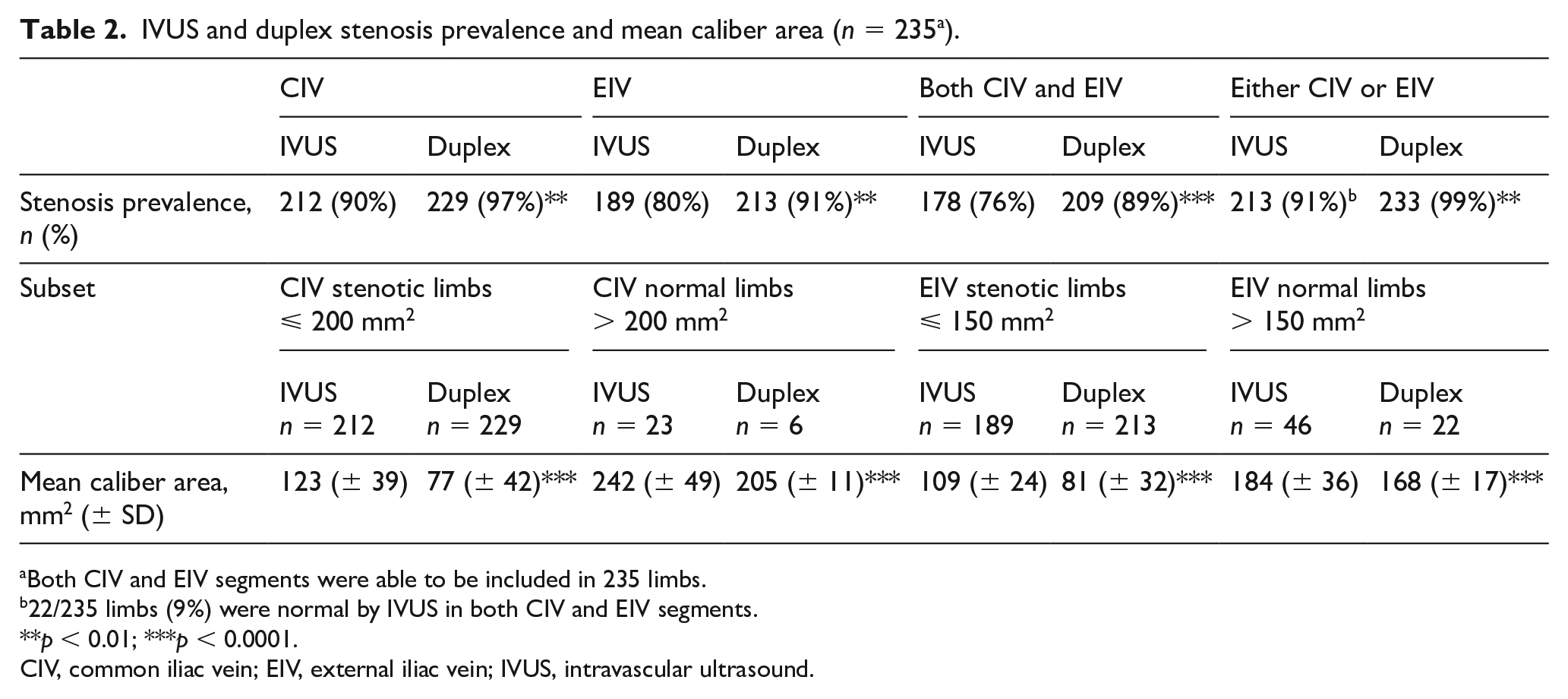
Both CIV and EIV segments were able to be included in 235 limbs.
22/235 limbs (9%) were normal by IVUS in both CIV and EIV segments.
p < 0.01; ***p < 0.0001.
CIV, common iliac vein; EIV, external iliac vein; IVUS, intravascular ultrasound.
Of the 235 limbs with paired IVUS and duplex measurements, prevalence of IVUS stenosis was the highest in CIV (90%) followed by EIV (80%); both CIV and EIV were stenotic together in 178 (76%) limbs. Both segments were normal on IVUS in 9% of the limbs. In 213 (91%) limbs, at least one of the two iliac vein segments were stenotic on IVUS. Duplex stenosis prevalence (using criteria identical to IVUS) was even worse than IVUS in every one of these categories. Duplex stenosis prevalence was 7% and 11% higher for CIV and EIV, respectively, and reached 99% prevalence when either CIV or EIV were considered in the diagnosis.
Comparison between IVUS and duplex
IVUS and duplex caliber areas used in the Bland–Altman plots passed the D’Agostino and Pearson normality test and were normally distributed for both CIV and EIV. 22 Bland–Altman plots of the mean difference of duplex lumen areas compared to IVUS calibers are shown in Figure 1A for CIV and Figure 1B for EIV, respectively. Duplex calibers have a negative bias (smaller) than corresponding IVUS calibers. On average, the duplex calibers are > 50 mm2 and > 30 mm2 smaller than IVUS measurements for CIV and EIV, respectively. Scatter plots of the duplex/IVUS relationship for the two segments (Figures 2A and 2B) show the datapoints crowded below the line of equality (i.e. IVUS measurements are higher than duplex).
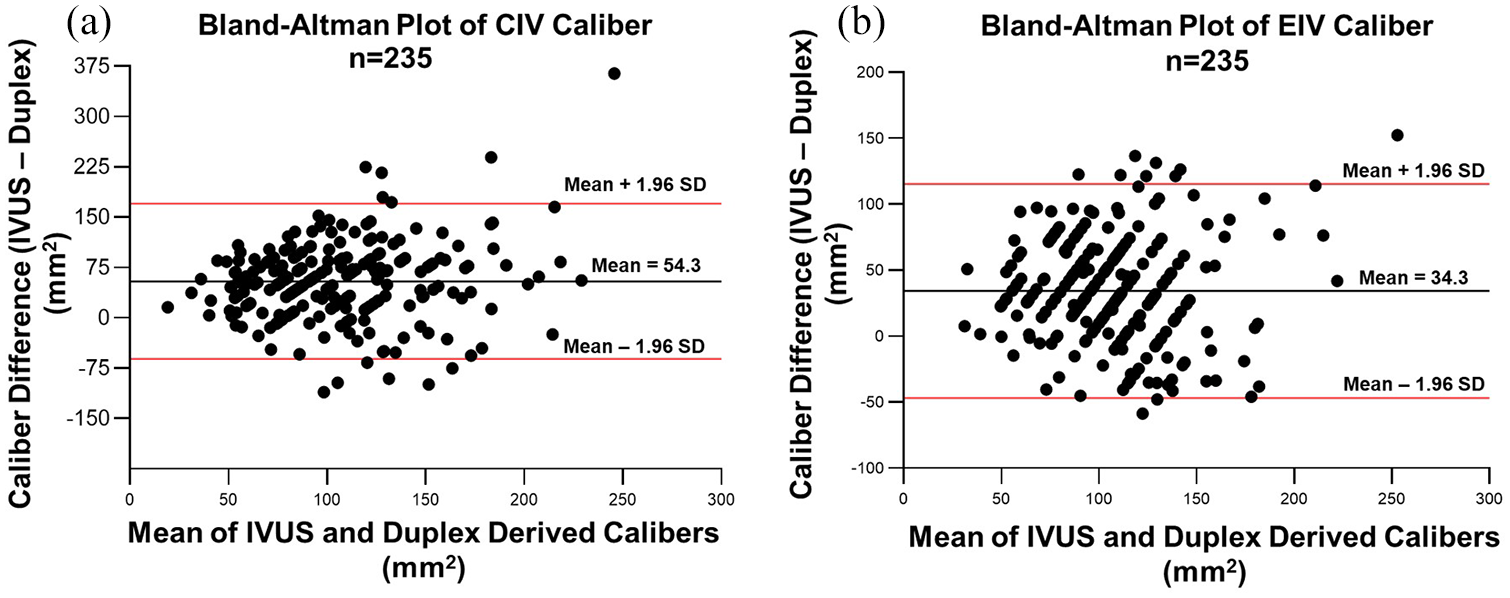
Bland–Altman plots of the difference in duplex-derived calibers compared to the IVUS reference standard for CIV (A) and EIV (B).
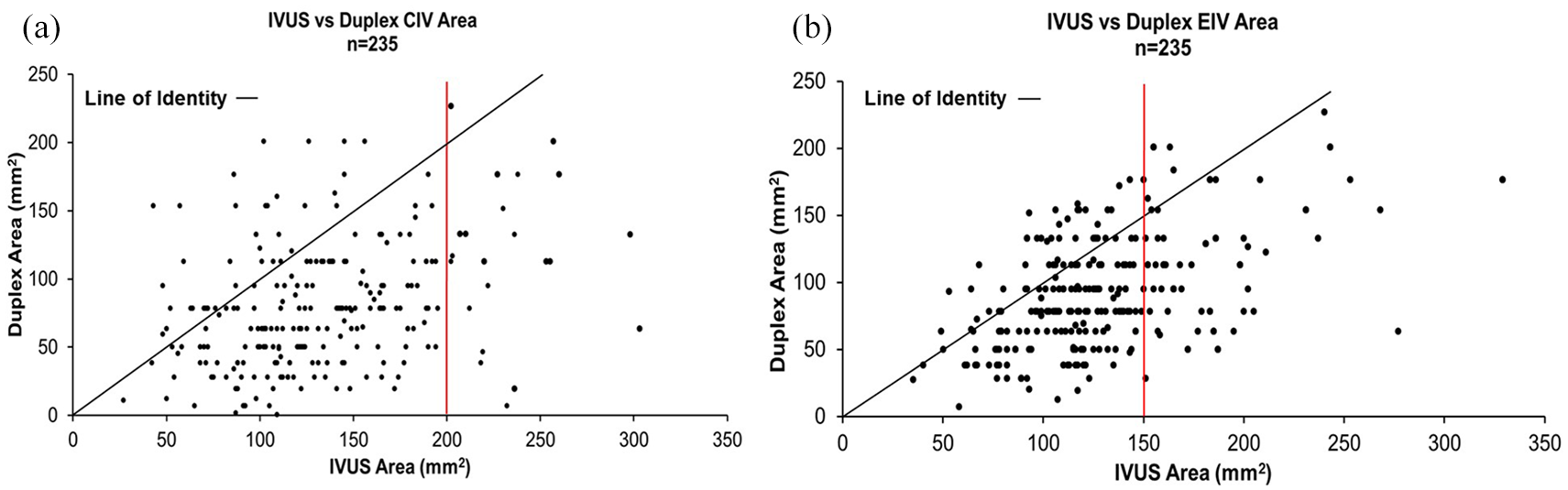
Scatter plots for IVUS versus duplex areas.
Both CIV (p = 0.22) and EIV (p = 0.49) caliber data passed the CUSUM test for linearity. Passing–Bablok regression analyses for CIV and EIV, respectively, are shown in Figures 3A and 3B. Since 0 is not in the CI of either segment, a systematic difference between IVUS and duplex metrics can be inferred. Since 1 is within the CI of both segments, a proportional difference is lacking (i.e. it cannot be mitigated by using a coefficient for correction). IVUS calibers of both CIV and EIV segments were ‘normal’ (non-stenotic) in 22/382 (6%) of limbs in this study: 14 had obesity with a mean BMI of 46.5 (± 17), three had elevated venous pressures due to cardiogenic causes, two had lymphedema, and three had post-thrombotic stenosis of common femoral and femoral veins without iliac involvement.
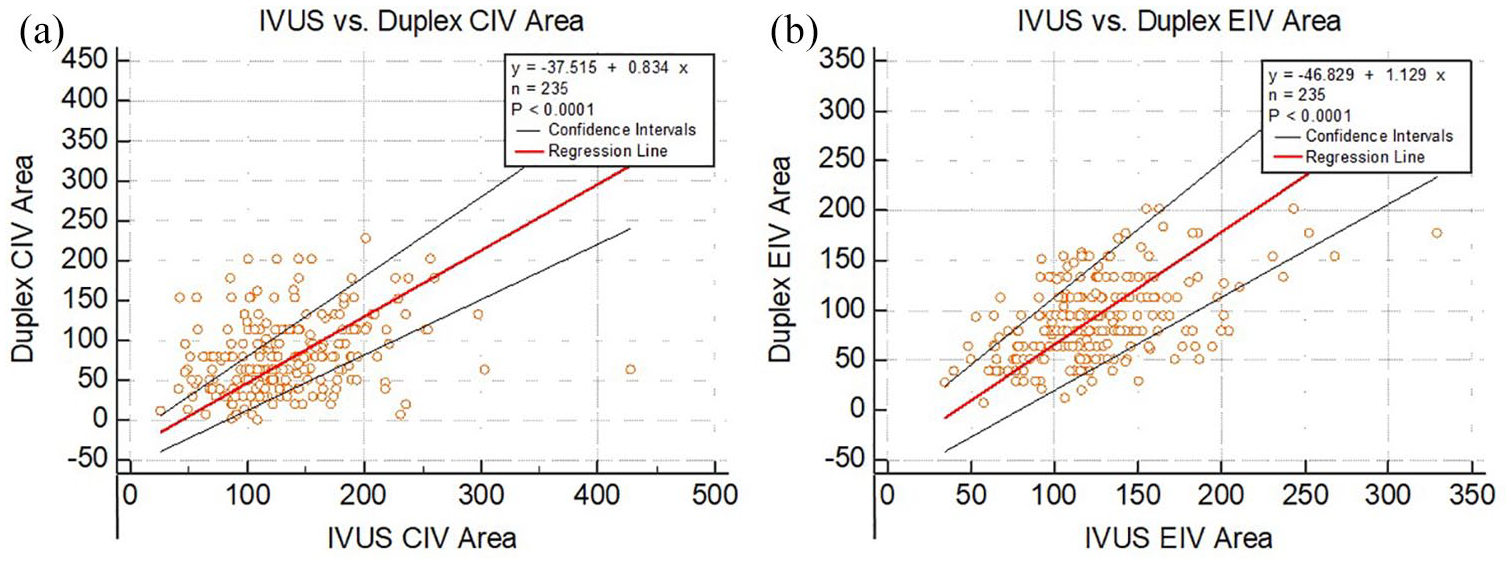
Passing–Bablok regression for comparison of IVUS and duplex-derived calibers for CIV and EIV, respectively.
Discussion
IVUS-defined stenosis thresholds and duplex
Stenotic thresholds per IVUS as described are attractive because they do not need a reference segment for comparison. They can apply to a wide range of patients with varying body mass or surface area. Cardiac output and regional flows such as the iliac have a narrow range (maximum ± 20%).23,24 This translates to a caliber variation of only ± 2.2% in the Poiseuille equation as radius is in the fourth power. This yields diameter ranges of 16–16.3 mm for CIV and 14–14.3 mm for EIV to accommodate body mass-related variations of flow in pressure neutral fashion; these thresholds equal caliber area ranges of 200–205 mm2 and 150–153 mm2, respectively.
Duplex was found to lack dimensional parity with these IVUS caliber metrics. This means that duplex application of this method is not a definitive test for clinical management of iliac vein stenosis. There are other described duplex methods for diagnosis of iliac vein stenosis that were not evaluated in the current study.25–27
IVUS is the most practical diagnostic arbiter, and the best procedural tool to guide stent placement at present.1,2,28 The diagnostic yield of IVUS is high in patients with advanced symptomatic manifestations of CVD.1,2,29,30 This is because iliac vein stenosis is a permissive lesion that remains silent in about 30% of the general population. Any additional insult such as general or surgical trauma to the limb, infection, thrombosis, lymphatic damage or onset of de novo reflux can precipitate symptoms. Much higher prevalence in the symptomatic subset compared to the silent cohort is a well-known characteristic of permissive lesions. For example, patent foramen ovale, a well-known permissive pathology, has nearly identical prevalence as iliac vein stenosis among silent and symptomatic populations. The high IVUS yield justifies its use for diagnosis and stent placement at the same sitting with appropriate consent. This is certainly the preferred approach in patients who have contrast allergies or renal impairment as the stenting procedure can be accomplished without use of any contrast. IVUS has its own deficiencies, such as its inability to provide a complete image of the lesion in a fraction of patients. In most of these cases, the presence of a high-grade lesion was apparent from the incomplete image even though its precise measurement for purposes of this study was not possible. In such cases, a balloon sizing maneuver is performed to assess lesion severity by noting the degree of waisting. 11 IVUS measurements will be normal in patients who have CVD or CVD-like manifestations due to pathologies other than iliac veins stenosis. The most common of these in our practice is morbid obesity with venous hypertension caused by increased intra-abdominal pressure.31,32 Post-thrombotic venous stenosis below the inguinal ligament without iliac venous involvement is a relatively rare cause. It is well known that a number of other systemic pathologies can mimic CVD-like manifestations; normal IVUS metrics of iliac veins should be expected in such cases. The association of CVD with obesity is complex. 13 Many limbs in obese patients also have organic post-thrombotic or non-thrombotic IVUS stenosis (in addition to compression), which are included in the dataset analyzed here.
CT venography has close metric parity with IVUS and has a low false-positive rate when both CIV and EIV calibers are separately assessed for stenosis (two segment method). 33 This is the preferred pre-IVUS test if one is required. It is our experience that most patients seem to desire a pretest. The majority of referrals from specialty physicians are accompanied by ancillary imaging studies, commonly duplex.
Caliber discrepancy between IVUS and duplex
An orthogonal orientation is necessary for accurate B-mode measurement. Doppler is more tolerant with angle of incidence for measurement but was not used in this study to avoid color overflow errors. The orthogonal probe orientation is impossible with a duplex surface probe on the abdominal wall due to the tortuous three-dimensional (3-D) anatomic course of the iliac vein within the pelvis. Even the endovenous IVUS catheter lacks perfect axial orientation as it has no centering mechanism. An oblong rather than a true perpendicular cross-section is obtained with either probe skewing the metric difference between the two probes for comparison. A smaller calculated area will result if the vein is inadvertently compressed during the examination or it collapses into an ellipse from low internal pressure. A number of other factors such as probe tilt to ‘clear’ the vein image of the overlapping artery, as shown in the Figure 4, may lead to mistaken measurement of a shorter secant or tangent than the true diameter. There are numerous other sources of dimensional errors inherent to the ultrasound technology itself and its application technique.34,35 The duplex technique used herein utilized a two-dimensional (2-D) probe for a 3-D image projected on a flat screen with the Z axis represented as layer thickness. Measurements in 2-D have been frequently observed to be smaller than 3-D measurements in a variety of other regions.36–38 Regardless of the precise reason, the metric disparity between B-mode and IVUS is an empiric fact that needs to be reckoned with in clinical decision making. Individual laboratories should cross-check the validity of their duplex iliac vein measurements against IVUS or CT venography-based measurements.
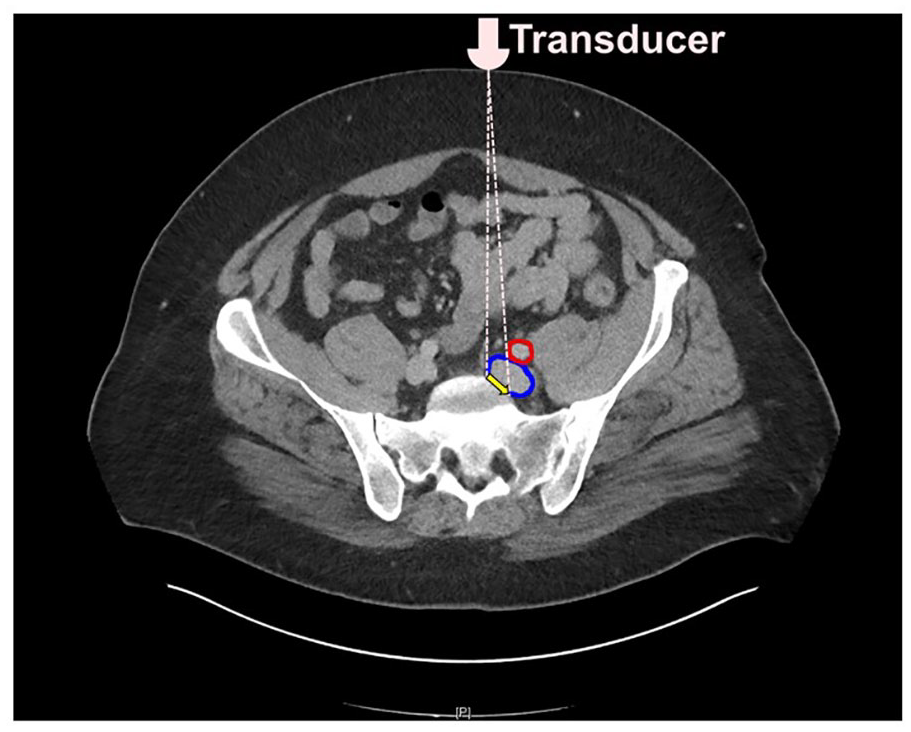
An orthogonal orientation is necessary for accurate B-mode measurement.
Study limitations
IVUS is used as the reference standard in this study. It is not free from its own limitations and errors. The quantitative impact of this error on current analyses is undetermined. The study has built-in selection bias as only symptomatic patients likely to have IVUS stenosis were selected. Randomized or case series selection based on duplex is not possible as equipoise between IVUS and duplex is absent. This analysis is retrospective from prospectively collected data.
Future directions
Chronic iliac vein stenosis is seldom an emergency threatening life or limb. Reconstructions in 3-D of the iliac vein confluence and the cava based on CT show promise, allowing accurate diagnosis and providing other useful information to pre-plan intervention. 39 The utility of 3-D ultrasound for iliac vein assessment remains to be examined.
Conclusion
Duplex was found to lack dimensional parity with IVUS in iliac vein caliber metrics in this study. Individual laboratories should validate their duplex iliac vein measurements with parallel IVUS or other morphometric standard before clinical application.
Footnotes
Acknowledgements
Critical assistance with statistical analysis was provided by Professor Jennifer Stafford, Mississippi College, Jackson, MS. Professor Fedor Lurie, University of Michigan, Ann Arbor, MI, gave valuable assistance regarding the ultrasound technology principles discussed.
Declaration of conflicting interests
Dr Raju receives royalty payments from Veniti, Inc. and holds US patents for venous stent design and IVUS (diagnostic). The remaining authors have nothing to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
