Abstract
Critical limb ischemia (CLI) constitutes the most advanced form of peripheral artery disease (PAD) and is characterized by ischemic rest pain, tissue loss and/or gangrene. Optimized medical care and risk factor modification in addition to revascularization could reduce the incidence of cardiovascular events and major adverse limb events, improving patients’ quality of life and promising higher survival rates. Adequate adherence to cardioprotective medications, including antithrombotic therapy (e.g., antiplatelets, anticoagulants), cholesterol-lowering agents (e.g., statins, proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors), angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs), and smoking cessation should be strongly encouraged for patients with CLI. This review examines these guideline-recommended therapies in terms of cardiovascular and limb-related risk reduction in patients with CLI.
Keywords
Introduction
Critical limb ischemia (CLI) constitutes the most advanced form of peripheral artery disease (PAD) and is characterized by ischemic rest pain, tissue loss, and/or gangrene. 1 CLI is associated with high mortality and amputation rates within the first year of diagnosis. 2 The goals of CLI care include urgent revascularization to prevent disabling amputation or to limit the level of amputation, 3 with endovascular procedures increasingly utilized. 4 However, CLI is frequently accompanied by several other comorbidities, including coronary artery disease (CAD), heart failure, advanced renal disease, and diabetes mellitus. The presence of each of these comorbidities is associated with significantly greater risk for mortality and adverse limb events. 5 Thus, although revascularization constitutes the initial approach for the treatment of CLI, medical therapy and risk factor modification are crucial for disease management and prevention of major adverse cardiovascular events.6,7
Inadequate risk factor identification and modification, as well as suboptimal therapy for comorbid conditions among patients with CLI, have been associated with up to 26% all-cause mortality rates within the first year of diagnosis. 4 A study of 142 patients with CLI (169 limbs), who were unsuitable candidates for any type of revascularization, demonstrated that during a 1-year follow-up 23% of the limbs required major amputation. 8 Optimal medical care and risk factor modification in addition to revascularization could reduce the incidence of cardiovascular events and major adverse limb events (MALE), improving patients’ quality of life and promising higher survival rates.6,7 Therefore, adequate adherence to cardioprotective medication, 9 antithrombotic therapy (e.g. antiplatelets, anticoagulants), 10 cholesterol-lowering agents (e.g. statins, proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors), 11 angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs) 12 and smoking cessation has been strongly encouraged for patients with CLI. 4 This review will examine these guideline-recommended therapies in terms of cardiovascular and limb-related risk reduction in patients with CLI.
Systematic review
The PubMed/Medline, Scopus, and Cochrane Central databases were searched independently by two investigators in blinded fashion for English-language randomized controlled trials (RCTs) or observational studies reporting on guideline-recommended therapies for patients with PAD. The search was conducted up to August 2020. Two reviewers independently extracted the relevant data from the eligible studies. Data were extracted for the following baseline variables: therapeutic agent, follow-up, main results, and conclusion from each individual study. Owing to between-study differences in design, comparisons made, and reporting methods, the findings were summarized from each study, but the results were not pooled. Additionally, when CLI-specific data were limited, overall PAD data or experience from the coronary arteries were summarized.
Lipid-lowering therapy
Lipid-lowering therapy reduces the incidence of adverse cardiovascular events in patients with stable CAD and/or other atherosclerotic risk factors. Thus, as PAD has been associated with higher cardiovascular mortality risk than CAD, 13 American College of Cardiology and American Heart Association (ACC/AHA) guidelines have adopted clear recommendations for statin therapy in such patients. 14 The current role of statins and of newer antilipidemic therapies (i.e. PCSK9 inhibitors) for the treatment of CLI is discussed.
Statin therapy
Statins are 3-hydroxy-3 methyl-glutaryl-coenzyme A (HMG-CoA) reductase inhibitors that block cholesterol production. 15 Statins also exert a beneficial pleiotropic effect on function of the endothelial cells, the microvasculature and the atherosclerotic plaque, reducing vascular inflammation and the risk for thrombus formation. 16 Such effects may contribute to reduction of not only periprocedural but also late-term adverse vascular events. 17 High-intensity statins (i.e. atorvastatin 40–80 mg and rosuvastatin 20–40 mg) are recommended for patients with PAD up to 75 years of age, while older patients should be treated with at least moderate-intensity statin therapy (e.g. atorvastatin 10–20 mg, simvastatin 20–40 mg, pravastatin 40–80 mg). 18 If low-density lipoprotein (LDL) cholesterol blood levels remain ⩾ 70 mg/dL, adding ezetimibe is reasonable. 19 In cases of intolerance to statin therapy, a nonstatin cholesterol-lowering agent should be offered. 18
A recent meta-analysis of 19 studies investigated cardiovascular and limb-related risk for adverse events among patients with CLI treated with statins (n = 12,292) versus no statins (n = 12,513). 11 The study demonstrated that patients with CLI treated with statins were 25% (hazard ratio (HR): 0.75; 95% CI: 0.59–0.95; I2 = 79.0%) and 38% (HR: 0.62; 95% CI: 0.52–0.75; I2 = 53.2%) less likely to undergo major amputation or to die, respectively. 11 Additionally, statin use was associated with improved primary or primary-assisted patency during the follow-up periods of the individual studies. 11 Thus, in accordance with previous reports, the authors suggested statin therapy for patients with CLI in order to decrease major adverse cardiovascular events and prevent/delay disabling amputations. 20 However, the authors underlined the need for further studies that would provide data about specific statin subtypes and optimal dosage for certain high-risk populations (e.g. elderly), in an effort to optimize medical therapy for patients with CLI. 11 Contemporary studies investigating the outcomes of statin therapy in patients with CLI are presented in Table 1.21–39
Studies investigating the outcomes of statin therapy in patients with CLI.
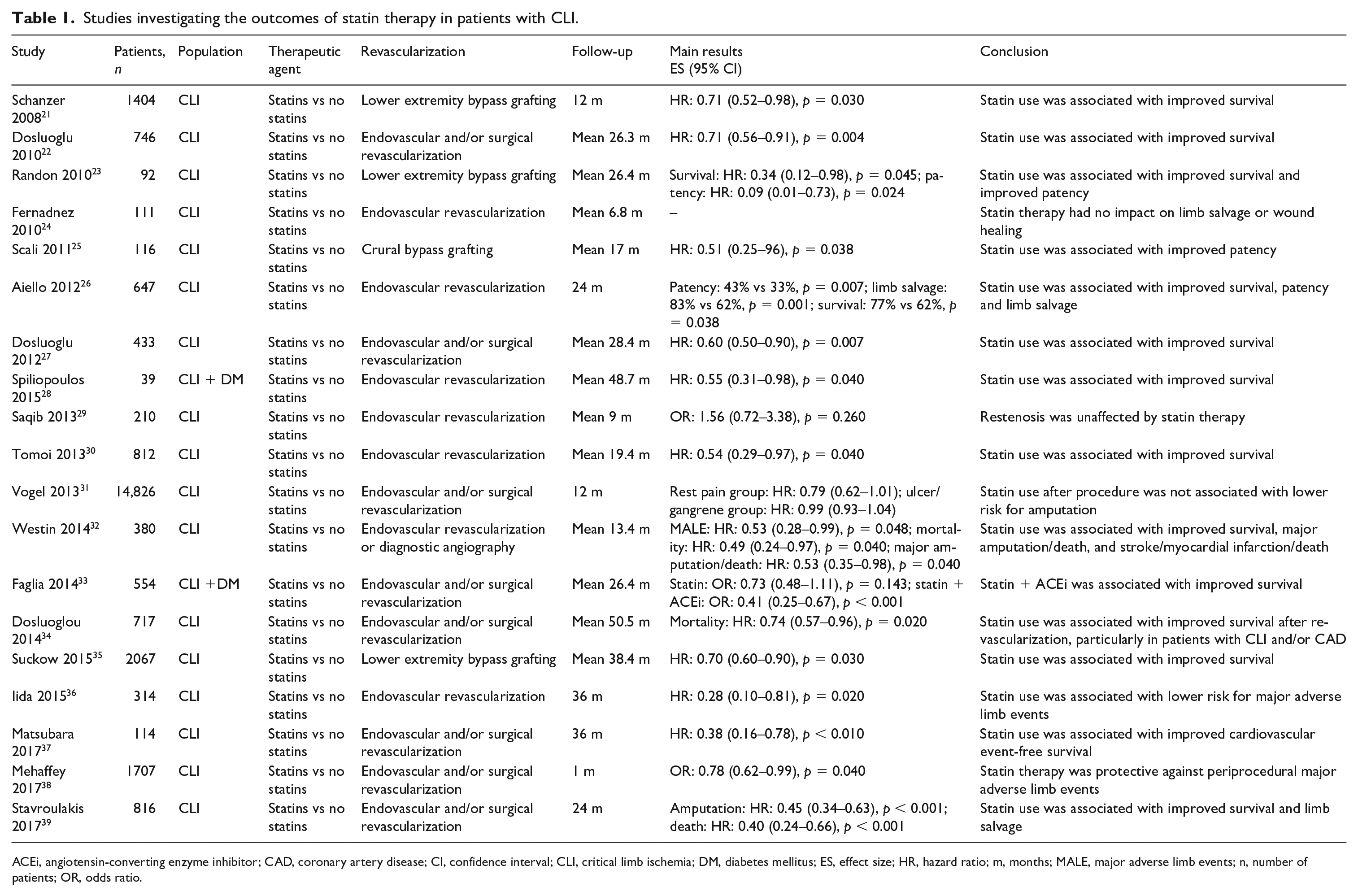
ACEi, angiotensin-converting enzyme inhibitor; CAD, coronary artery disease; CI, confidence interval; CLI, critical limb ischemia; DM, diabetes mellitus; ES, effect size; HR, hazard ratio; m, months; MALE, major adverse limb events; n, number of patients; OR, odds ratio.
Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibition
The primary mechanism of regulation of serum LDL cholesterol levels involves the number of LDL receptors on the outer cell membrane of hepatocytes. 40 Thus, having fewer LDL cholesterol receptors leads to higher levels of circulating LDL cholesterol. 40 PCSK9 is a serine protease that is involved in destruction of LDL cholesterol receptors on hepatocytes and as such it is indirectly involved in the regulation of serum LDL levels. 40 Thus, high PCSK9 activity is associated with higher serum LDL cholesterol levels. 40 Currently, two PCSK9 inhibitors, which are both human monoclonal antibodies targeting the PCSK9 enzyme, are available (i.e. alirocumab, evolocumab), 41 offering outstanding serum LDL cholesterol reduction (by approximately 60%). 42 Although several randomized trials demonstrated that LDL cholesterol-lowering therapy in patients with PAD is associated with reduced major adverse cardiovascular events, 43 only a few investigated whether these therapies affect the incidence of MALE over time. 20
The ‘Further Cardiovascular Outcomes Research With PCSK9 Inhibition in Subjects With Elevated Risk’ (FOURIER trial) randomized trial investigated the safety and efficacy of evolocumab in patients with PAD (n = 3642), including its effect on MALE during a median follow-up of 2.2 years. 44 The study demonstrated a 21% lower risk for the composite of cardiovascular death, myocardial infarction (MI), hospital admission for unstable angina, coronary revascularization, and stroke among evolocumab users versus placebo (HR: 0.79; 95% CI: 0.66–0.94; p < 0.001). 44 In addition, during follow-up, patients taking evolocumab were at 17% lower risk for cardiovascular death, MI, and stroke (HR: 0.73; 95% CI: 0.59–0.91, p = 0.004). 44 Moreover, evolocumab versus placebo was associated with a strong trend for lower risk of MALE among patients with PAD (HR: 0.63; 95% CI: 0.39–1.03; p = 0.063), with lower achieved LDL cholesterol levels being associated with lower limb-related risk, even at very low LDL cholesterol levels. 44
Thus, the authors suggested that LDL cholesterol reduction to very low levels, potentially with the use of PCSK9 inhibitors, should be considered for all patients with PAD. 44 The European Society of Cardiology/European Atherosclerosis Society Task Force consensus recommended the use of PCSK9 inhibitors in patients with PAD receiving maximum statin/ezetimibe dose, whose circulating LDL cholesterol levels remain above 139 mg/dL (3.6 mmol/L) or above 100 mg/dL (2.6 mmol/L) for certain higher-risk populations (e.g., patients with CLI), in order to attain a goal LDL below 70 mg/dL. 45 However, additional studies should investigate to what extent disease severity and CLI may affect this benefit of PCSK9 inhibitors, and whether PCSK9 inhibition is a cost-effective treatment modality for the management of symptomatic PAD. 42
Antihypertensive therapy with ACE inhibitors or ARBs
Hypertension has been associated with adverse cardiovascular events and progression of atherosclerotic disease. 46 Thus, antihypertensive therapy has been recommended as secondary prevention strategy for patients with PAD. 47 Previously published data from human and animal studies have provided evidence that ACE inhibitors may have a protective role in atherosclerosis by improving endothelial function.48,49 A post hoc analysis of the ‘Heart Outcomes Prevention Evaluation’ (HOPE) study demonstrated that patients with both clinical or subclinical PAD, based on ankle–branchial index (ABI) and claudication symptoms, benefited from ramipril, in terms of all-cause mortality (p < 0.001) and major adverse cardiovascular events (p < 0.001). 50 Contemporary studies reporting on the outcomes of ACE inhibitors or ARBs among patients with CLI are presented in Table 2.33,50–56
Studies investigating the outcomes of ACEi/ARBs therapy in patients with CLI.
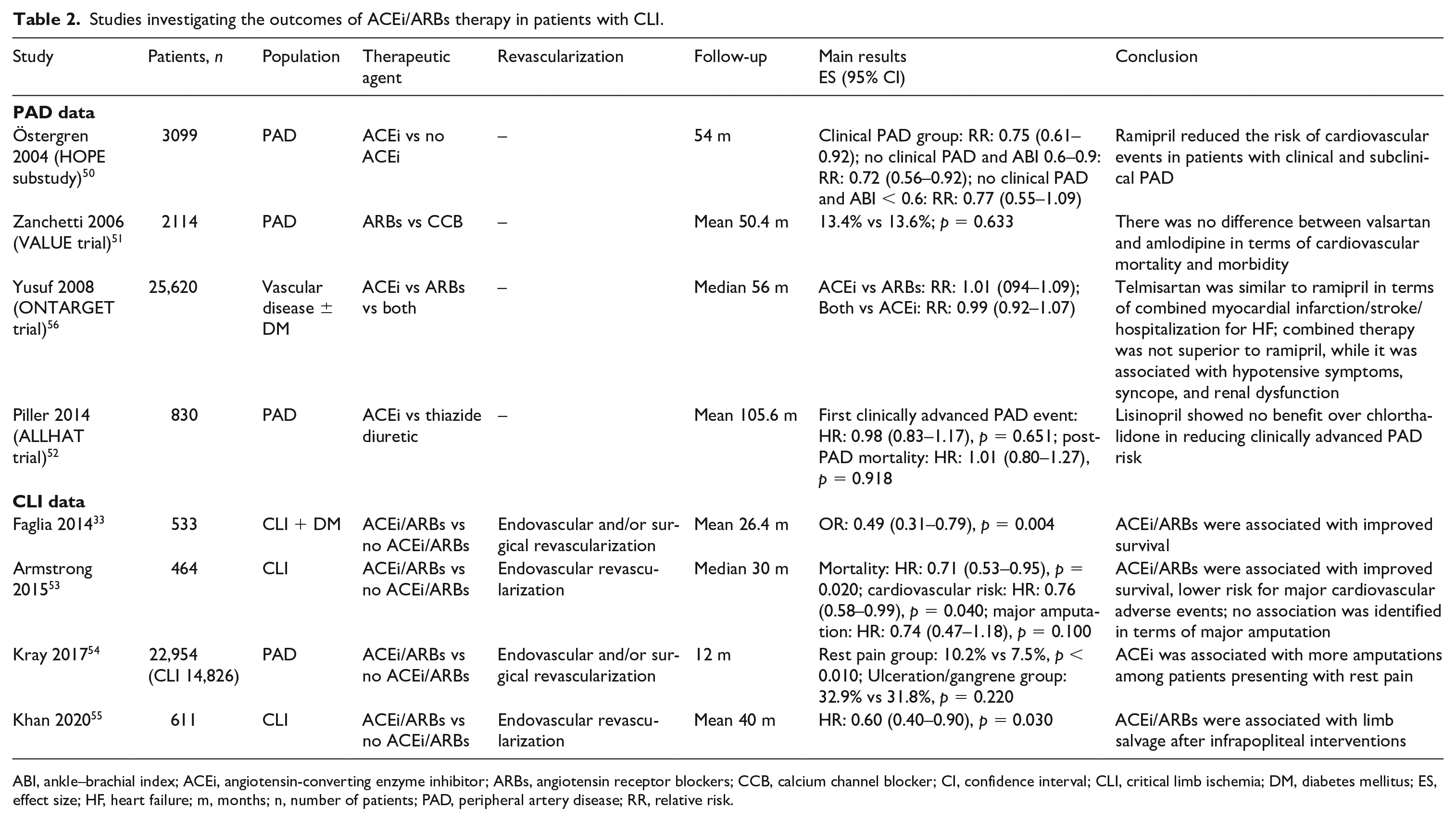
ABI, ankle–brachial index; ACEi, angiotensin-converting enzyme inhibitor; ARBs, angiotensin receptor blockers; CCB, calcium channel blocker; CI, confidence interval; CLI, critical limb ischemia; DM, diabetes mellitus; ES, effect size; HF, heart failure; m, months; n, number of patients; PAD, peripheral artery disease; RR, relative risk.
The protective effect of renin-angiotensin system inhibitors has been well-established for patients with advanced atherosclerotic disease. However, the safety and efficacy of these antihypertensive medications for secondary prevention among patients with CLI is still unknown. Bodewes et al. studied 1161 patients with CLI undergoing first-time surgical or endovascular revascularization, investigating whether ACE inhibitors or ARBs use was associated with long-term mortality benefit. 57 During a median follow-up of 1.5 years (IQR: 0.4–3.8), patients on renin-angiotensin system inhibitors were at 22% lower risk for late-term all-cause mortality (HR: 0.78; 95% CI: 0.65–0.94), with higher baseline dosages associated with lower mortality risk (HR: 0.70; 95% CI: 0.57–0.86). 57 However, the study failed to show any statistically significant reduction in risk for MALE, including major amputation, and reintervention among patients on renin-angiotensin system inhibitors versus patients on no renin-angiotensin system inhibitors. 57 Therefore, the authors suggested that although ACE inhibitors/ARBs were associated with a cardioprotective effect (especially at high doses), no significant benefit was observed in terms of limb-related risk among these high-risk patients. 57
A multicenter retrospective analysis evaluated the effect of ACE inhibitors or ARBs in 464 patients with CLI, undergoing either diagnostic angiography or endovascular intervention. 53 After propensity score weighting for baseline characteristics, patients taking ACE inhibitors or ARBs were at 24% lower risk for major adverse cardiovascular events (HR: 0.76; 95% CI: 0.58–0.99; p = 0.04) and 29% lower risk for all-cause mortality during 3-year follow-up (HR: 0.71; 95% CI: 0.53–0.95; p = 0.02). 53 However, the study did not find any statistically significant association between ACE inhibitors or ARBs and the risk for limb-related outcomes. 53 Therefore, the authors suggested that the primary benefit of renin-angiotensin system inhibitors may stem from physiologic mechanisms promoting perfusion in vascular beds other than lower limb arteries. 53 Angiogenesis, 58 improved endothelial function/vasodilation, 59 and stabilization of the atherosclerotic plaque 60 have been hypothesized to be mechanisms responsible for the beneficial effect of ACE inhibitors or ARBs among patients with PAD. 58 However, it is possible that these mechanisms cannot significantly alter the limb-related outcomes of advanced stage disease, including patients with CLI.
A recently published retrospective study of 611 patients (311 on ACE inhibitors/ARBs vs 300 no ACE inhibitors/ARBs) evaluated the safety and efficacy of ACE inhibitors or ARBs in terms of limb-related outcomes. 55 In accordance with previous reports, the study failed to show any statistically significant benefit of ACE inhibitor or ARB use in terms of limb salvage over 5-year follow-up. 55 However, a stratified analysis based on lesion location (i.e. femoropopliteal and infrapopliteal disease) showed that patients with below the knee lesions exhibited better limb salvage rates when prescribed ACE inhibitors/ARBs (75% vs 61%; p = 0.005). 55 The authors suggested that calcified infrapopliteal lesions might benefit more from ACE inhibitors/ARBs due to improvements in arterial stiffness. 55 Thus, although there is a clear cardioprotective effect of renin-angiotensin system inhibitors among patients with CLI, more data are warranted regarding the effect of ACE inhibitors/ARBs in the peripheral circulation.
Antithrombotic therapy
Antiplatelet agents have been shown to reduce major adverse cardiovascular events in patients with PAD, 61 with TASC guidelines recommending single antiplatelet therapy. 62 However, there have been concerns that the absolute adverse event rate reduction might be limited with antiplatelet monotherapy. 61 The ACC/AHA guidelines suggest at least 1 month of combined therapy with aspirin and P2Y12 inhibitors (e.g. clopidogrel) after endovascular procedures for lower limb revascularization. 63 Data regarding the additive benefit of dual-antiplatelet therapy (DAPT) and the optimal duration of DAPT are sparse, 61 with the level of evidence for this recommendation being low. 63 Based on expert consensus, DAPT for 1–6 months seems to be reasonable for secondary prevention among patients with symptomatic disease undergoing revascularization procedures. 64 Furthermore, recent data have suggested that low-dose rivaroxaban on a background of aspirin may reduce cardiovascular and limb-related risk in patients with PAD and CLI.
Antiplatelet monotherapy
The ‘Critical Limb Ischemia Prevention Study’ (CLIPS) was a multicenter RCT trial that investigated the risk of combined MI, stroke, vascular death, and CLI among patients with PAD taking 100 mg/daily aspirin versus placebo. 65 The study showed 64% versus 24% risk reduction for adverse vascular events or progression to CLI among patients on aspirin versus placebo, respectively (p = 0.022). 65 Thus, the authors suggested that aspirin should be routinely considered for patients with PAD. 65 A meta-analysis of six primary and 16 secondary prevention trials studied the incidence of adverse vascular events with long-term aspirin versus placebo. 66 The subanalysis of the primary prevention studies showed 12% reduction in adverse vascular events in favor of aspirin versus placebo (0.51% vs 0.57% per year; p < 0.001), mainly attributed to lower rates of MI. 66 Among the secondary prevention studies, aspirin versus placebo was associated with a 6.7% versus 8.2% per year rate of serious adverse vascular events (p < 0.001) 66 . However, the authors underlined that aspirin therapy should be always weighed against the potential risk for major bleeding. 66
The ‘Clopidogrel versus Aspirin in Patients at Risk for Ischemic Events’ (CAPRIE) study evaluated the relative efficacy of 75 mg/day clopidogrel versus 325 mg/day aspirin, in terms of ischemic stroke, MI, and vascular death among patients with atherosclerotic disease, including patients with PAD. 67 The study demonstrated 8.7% reduction in major adverse cardiovascular events with clopidogrel versus aspirin (p = 0.043), although the bleeding risk remained statistically similar between the two groups over a mean follow-up of 1.9 years. 67
Ticagrelor constitutes an alternative thienopyridine derivative to clopidogrel that has been shown to reduce major adverse cardiovascular events among patients with PAD. 68 The ‘Platelet Inhibition and Patient Outcomes’ (PLATO) trial was the first study to examine the safety and efficacy of ticagrelor in patients with acute coronary syndrome, demonstrating superiority of ticagrelor over clopidogrel in terms of adverse cardiovascular events prevention. 69 A post hoc analysis of the PLATO trial demonstrated a strong trend towards reduction of cardiovascular death, MI or stroke with ticagrelor versus clopidogrel, although no statistical significance was reached. 70
The ‘Examining Use of Ticagrelor in Peripheral Artery Disease’ (EUCLID) trial compared ticagrelor monotherapy with clopidogrel monotherapy in 13,885 patients with symptomatic PAD and an ABI ⩽ 0.8. 68 Both drugs exhibited similar efficacy profiles with no statistically significant difference between ticagrelor and clopidogrel in terms of combined cardiovascular death, MI, or ischemic stroke (ticagrelor: 10.6% vs clopidogrel: 10.8%; HR: 1.02; 95% CI: 0.92–1.13; p = 0.650). 68 Approximately 5% of the subjects enrolled in the EUCLID trial presented at baseline with CLI. 68 A EUCLID subanalysis showed that patients with CLI had significantly higher rates of cardiovascular death and morbidity compared to patients with milder PAD disease, underlining the need for further research efforts to reduce the risk in patients with CLI. 71
Dual-antiplatelet therapy
Although antiplatelet therapy has been shown to reduce cardiovascular and limb-related risk, 61 this effect may be limited with monotherapy. 61 It has been estimated that in up to 30% of the patients treated with clopidogrel, the antiplatelet effect is limited due to higher metabolism, attributed to either drug–drug interactions or genetic polymorphisms 72 and as such DAPT has become the standard of care in the treatment of patients with CAD undergoing revascularization procedures. 73 Thus, as patients with PAD may exhibit higher clopidogrel resistance rates than patients with CAD, 74 DAPT has gained a crucial role in secondary prevention after endovascular therapy for PAD.
A propensity scores matched analysis of 629 patients with symptomatic PAD compared DAPT with aspirin and clopidogrel to aspirin monotherapy over a follow-up period up to 3 years. 61 The study showed an association between DAPT and lower risk for major adverse cardiovascular events (adjusted HR: 0.65; 95% CI: 0.44–0.96; p = 0.030) and all-cause mortality (adjusted HR: 0.55; 95% CI: 0.35–0.89; p = 0.020). 61 Similarly, a post hoc analysis of 3096 patients with PAD from the ‘Clopidogrel for High Atherothrombotic Risk and Ischemic Stabilization, Management and Avoidance’ (CHARISMA) trial showed that DAPT with aspirin and clopidogrel versus aspirin reduced the hospitalization rates for ischemic events (HR: 0.81; 95% CI: 0.68–0.95; p = 0.011) and the incidence of MI (HR: 0.63; 95% CI: 0.42–0.96; p = 0.029). 75 The rates of fatal, severe or moderate bleeding were statistically similar between the two groups, although minor bleeding events were more common among the combined therapy group (aspirin and clopidogrel: 34.4% vs aspirin and placebo: 20.8%; OR: 1.99; 95% CI: 1.69–2.34; p < 0.001). 75
A secondary analysis of the ‘Prevention of Cardiovascular Events in Patients With Prior Heart Attack Using Ticagrelor Compared to Placebo on a Background of Aspirin–Thrombolysis In Myocardial Infarction 54’ (PEGASUS-TIMI 54) trial investigating 1143 patients with known PAD and prior MI, showed that ticagrelor significantly reduced the risk for MALE (HR: 0.65; 95% CI: 0.44–0.95; p = 0.026) and adverse cardiovascular events (HR: 1.60; 95% CI: 1.20–2.13; p = 0.001) compared to placebo, on a background of aspirin monotherapy. 76 Although no CLI-specific data are available, a superior benefit of ticagrelor to clopidogrel has not been demonstrated for patients with symptomatic PAD, with major bleeding rates being similar between the two groups. 68
Although the Global Vascular Guidelines recommend treatment of all patients with CLI with an antiplatelet agent, long-term use of DAPT is not currently indicated (grade 2; C level of evidence). 4 The efficacy of DAPT depends on several factors, including baseline patient, lesion, and procedural characteristics. 4 Thus, based on expert consensus, specific populations including patients with complex disease (e.g. chronic total occlusions, restenosis, long lesions, failed previous interventions, etc.) and patients at low risk for major adverse bleeding events (e.g. younger age, normal renal function, etc.) may benefit more from DAPT, frequently employed for 1–6 months after peripheral intervention. 77 Contemporary recommendations for antiplatelet therapy in patients with PAD are presented in Table 3.78–81
Contemporary recommendations for antiplatelet therapy in patients with PAD.
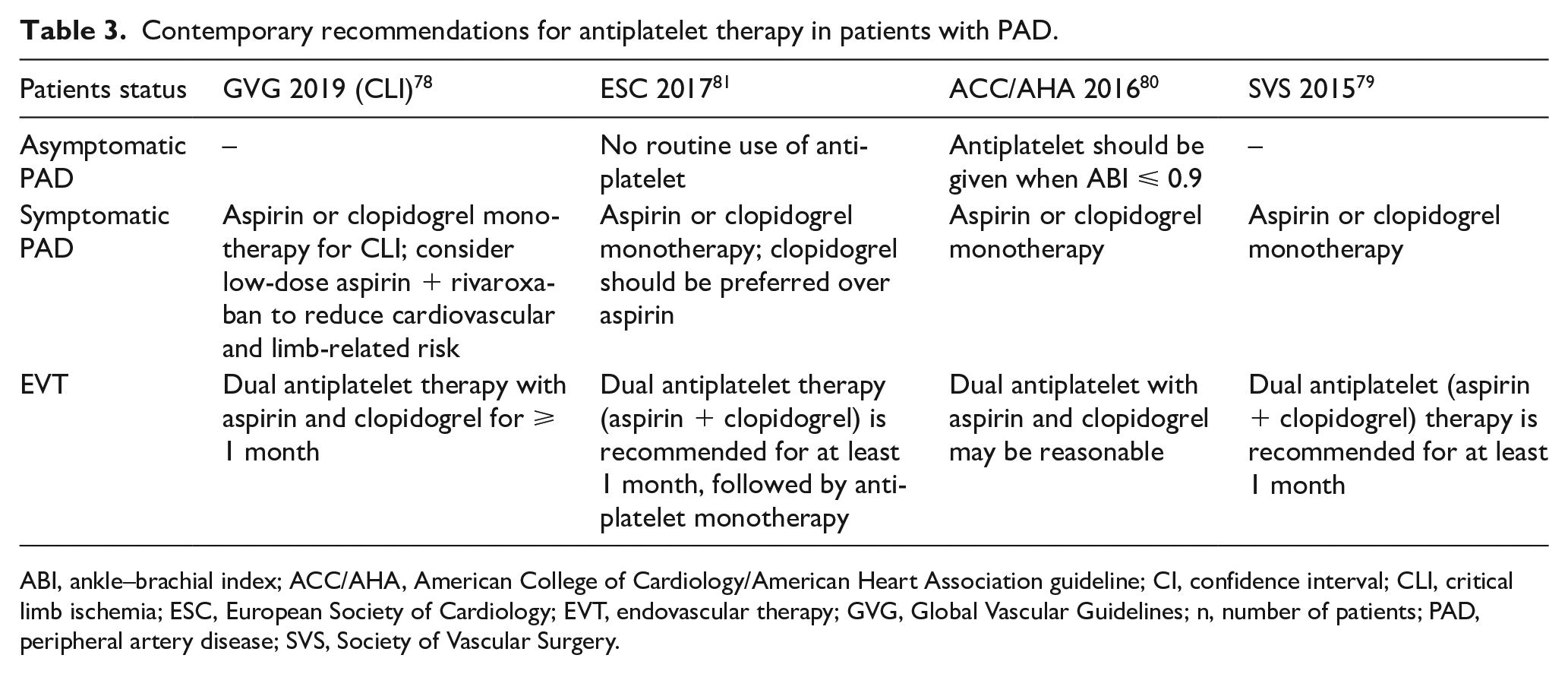
ABI, ankle–brachial index; ACC/AHA, American College of Cardiology/American Heart Association guideline; CI, confidence interval; CLI, critical limb ischemia; ESC, European Society of Cardiology; EVT, endovascular therapy; GVG, Global Vascular Guidelines; n, number of patients; PAD, peripheral artery disease; SVS, Society of Vascular Surgery.
Direct oral anticoagulants
Anticoagulants prevent thrombus formation by affecting the coagulation cascade. 82 Overall, systemic anticoagulation with the conventional vitamin K antagonists (i.e. warfarin) has not shown superiority to antiplatelet agents in terms of cardiovascular event reduction, while warfarin has been associated with life-threatening complications. 83 Direct oral anticoagulant agents (DOAC) have been developed, such as low-dose rivaroxaban (2.5 mg twice daily), that selectively inhibit coagulation factor Xa, reducing thrombin formation. The recent ‘Cardiovascular Outcomes for People Using Anticoagulation Strategies’ (COMPASS) trial was a randomized double-blind controlled trial that compared combined therapy with low-dose rivaroxaban and aspirin to monotherapy with aspirin in terms of MALE and all-cause mortality in patients with stable CAD and/or PAD.84,85 Overall, the study demonstrated a statistically significant adverse cardiovascular event and mortality benefit for the combined group (HR: 0.74; 95% CI: 0.65–0.86; p < 0.001).84,85 Among the 6391 patients with lower extremity PAD, 128 patients experienced MALE over a 21-month median follow-up, with statistically significant fewer events in the rivaroxaban + aspirin group versus aspirin only group (HR: 0.57; 95% CI: 0.37–0.88; p = 0.010). 86
The ‘Vascular Outcomes Study of Aspirin Along with Rivaroxaban in Endovascular or Surgical Limb Revascu-larization for Peripheral Artery Disease’ (VOYAGER PAD) trial specifically evaluated the outcomes of low-dose rivaroxaban added to aspirin in patients undergoing surgical or endovascular revascularization for PAD. 87 In the VOYAGER PAD trial, 6564 patients (23% had CLI) underwent randomization, with 3286 and 3278 assigned to the rivaroxaban and placebo groups, respectively. 88 The combined therapy group was at 15% lower risk for the combined outcome of acute limb ischemia, major amputation, MI, ischemic stroke, and cardiovascular death, over a 3-year follow-up (HR: 0.85; 95% CI: 0.76–0.96, p = 0.009). 88 Major bleeding according to ‘Thrombolysis in Myocardial Infarction’ classification was not statistically different between the two groups at 3-year follow-up. 88 However, when major bleeding was defined by the International Society on Thrombosis and Hemostasis, the risk for major bleeding complications was significantly higher among the rivaroxaban + aspirin versus aspirin alone group. 88
The study results have been promising regarding the protective effect of combined low-dose rivaroxaban and aspirin. However, this benefit has not yet been specifically established for patients with CLI. Understanding the role of lesion characteristics, type of revascularization technique, and degree of PAD severity on clinical outcomes is needed in order to define the benefit of DOAC agents for patients with CLI. Thus, further prospective studies and subanalyses of the VOYAGER PAD trial are necessary to determine the safety and efficacy of DOACs combined with aspirin after revascularization in this high-risk population. Studies reporting on the outcomes of DOACs for patients with PAD are presented in Table 4.86,88–94
Studies reporting on the outcomes of DOACs for patients with PAD.
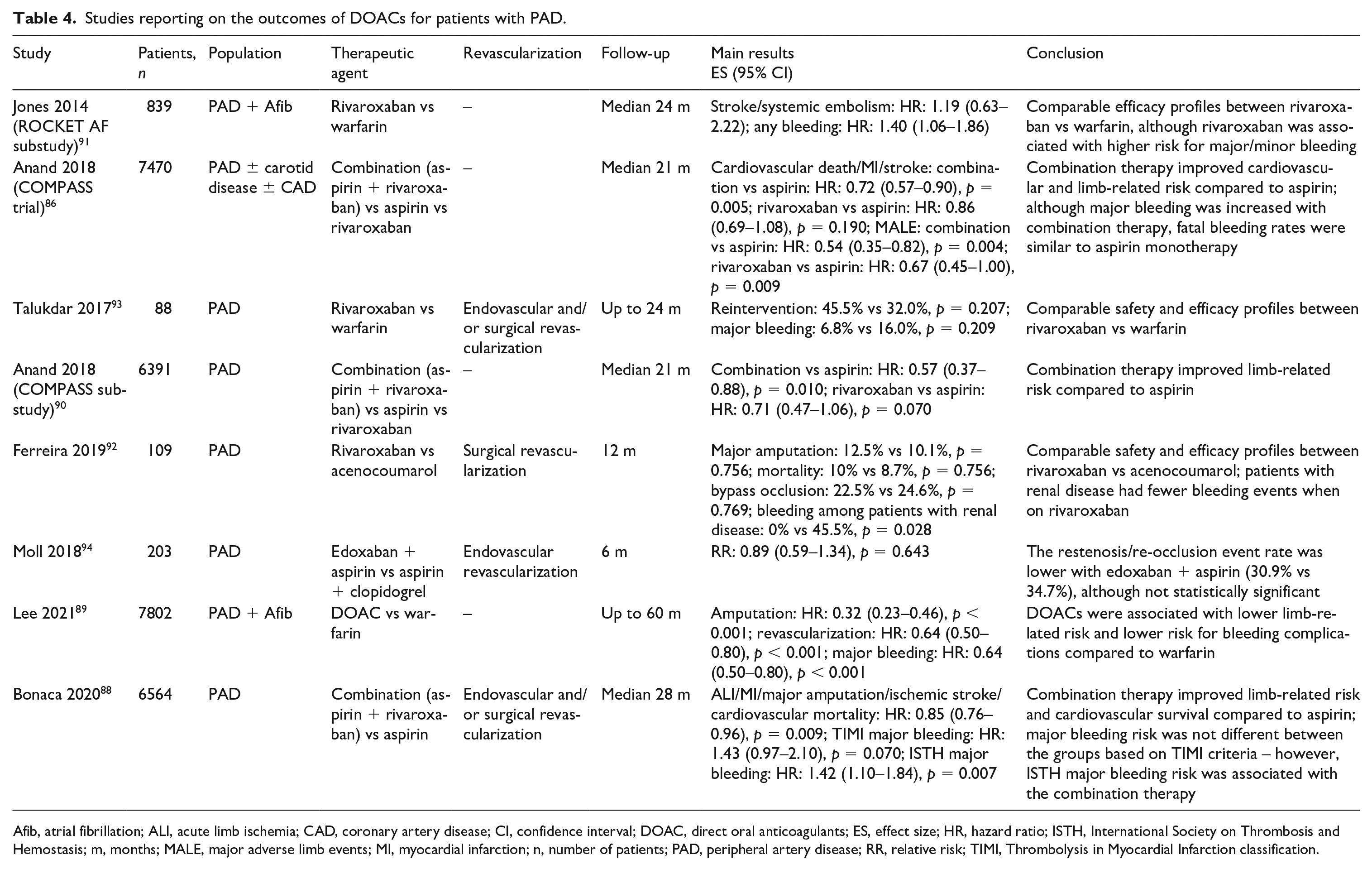
Afib, atrial fibrillation; ALI, acute limb ischemia; CAD, coronary artery disease; CI, confidence interval; DOAC, direct oral anticoagulants; ES, effect size; HR, hazard ratio; ISTH, International Society on Thrombosis and Hemostasis; m, months; MALE, major adverse limb events; MI, myocardial infarction; n, number of patients; PAD, peripheral artery disease; RR, relative risk; TIMI, Thrombolysis in Myocardial Infarction classification.
Cilostazol
Cilostazol is an antiplatelet agent with unique properties that include: (i) reversible inhibition of platelet aggregation and prevention of thrombus formation; (ii) vasodilatory effect; and (iii) antiproliferative properties. 95 Its mechanism of action has been attributed to targeted phosphodiesterase III inhibition. 96 Cilostazol has been studied in several patient populations with atherosclerosis, with promising favorable safety and efficacy. 95 The bleeding profile of cilostazol has been comparable to placebo, even when combined with additional antithrombotic agents. 95
Cilostazol has been studied in patients with claudication, with modest improvements in maximum and pain-free walking distance. 97 A small study of 14 patients (20 limbs) with PAD of Rutherford classification 3 or 4 demonstrated that cilostazol increased skin perfusion pressure by improving the microvascular circulation. 98 Additionally, RCTs have provided evidence that cilostazol significantly reduces re-occlusion rates after endovascular therapy for femoropopliteal lesions. 99 Similarly, a meta-analysis of four studies, with each study including mostly patients with claudication (approximately 75%), showed that cilostazol was associated with reduced in-stent restenosis rates after femoropopliteal stenting. 100 Therefore, cilostazol has been recommended as guideline-directed therapy for PAD. 101
However, owing to the absence of RCTs including exclusively patients with CLI, it is still unclear whether cilostazol could be associated with improved clinical outcomes. 102 Patients with CLI benefit from life-long antiplatelet therapy, but only a few prospective studies included patients with CLI treated with cilostazol. 103 A study by Soga et al. investigated the effect of cilostazol on the outcomes of endovascular therapy for infrainguinal lesions among patients with CLI. 102 The study found that the amputation-free survival and limb salvage rates were superior among the cilostazol-treated group during a follow-up of 5 years. 102 However, the overall rates of survival or freedom from further revascularization were similar between the groups. 102 Additionally, a retrospective analysis of 63 patients with CLI (68 limbs) undergoing infrapopliteal endovascular revascularization found that cilostazol was independently associated with lower restenosis/re-occlusion and target lesion revascularization (TLR) rates over a 3-month follow-up. 104 Nonetheless, at present no clear recommendation can be made regarding the benefit of cilostazol in patients with CLI and, as such, larger prospective studies are needed to evaluate the effect of this antiplatelet agent in this high-risk subgroup.
Smoking cessation
The Global Vascular Guidelines strongly recommended that smoking cessation interventions should be offered to all patients with CLI who either smoke or use alternative tobacco products. 4 Additionally, physicians are strongly recommended to perform screening for smoking or tobacco use at every visit of all patients with CLI. 4 A Swedish study of 343 patients with intermittent claudication investigated the effects of smoking in terms of adverse cardiovascular events. 105 The 10-year survival was 11% and 53% among smokers and nonsmokers, respectively. 105 The study also demonstrated a significant association between smoking and disease progression and, as such, higher risk for MI (p < 0.050) and cardiac death (p < 0.050). 105 An observational analysis cohort of 739 patients with symptomatic PAD, including patients with CLI, showed that patients who quit smoking were at statistically significant lower risk for all-cause mortality and lower limb amputation compared to active smokers during a follow-up period of up to 5 years. 7 Thus, this study provided significant evidence that smoking cessation not only decreases cardiovascular events, but also improves amputation-free survival compared to continuing smoking. 7
Continued smoking is also likely associated with delayed wound healing in patients with CLI. In a study including 264 patients with CLI, active smoking was associated with higher risk for incomplete wound healing during 6-month (HR: 4.54; 95% CI: 1.41–14.28; p = 0.012) and 9-month (HR: 2.32; 95% CI: 1.11–4.76; p = 0.026) follow-up. 106 Although the exact cause for this association is not fully understood, several mechanisms have been proposed to explain this observation, involving increased oxidative stress and endothelial dysfunction. 107 Nicotine acts as a direct vasoconstrictor, having an additive effect to decreased perfusion of the lower extremities and, as such, further impairing the process of wound healing. 107 Moreover, it has been shown that smokers have a higher level of circulating carboxyhemoglobin, which reduces oxygen transportation to ischemic ulcerated tissues. 108
Additional medical considerations
Pain management
Inadequate pain management could have a major impact on patients’ quality of life, significantly impairing daily function. The clinical decision making for appropriate pain control is complicated due to the lack of RCTs conducted for patients with CLI and the fact that neuropathic pain often coexists with ischemic rest pain, especially in diabetic patients. 4 Thus, in the absence of specific evidence, the Global Vascular Guidelines recommended an individualized approach to pain management, weighing the benefits (e.g. improvement of functional capacity) and the harms (e.g. constipation, dizziness, drowsiness) of such medical therapy. 4 The agents prescribed and the dose should be proportional to the patient’s discomfort. 8 When the pain is not adequately addressed, the patient should be referred to specialized clinics with pain management services and alternative treatment options. 109
Nutrition
Smoking cessation, stopping recreational drugs, and maintaining a normal weight with a healthy diet and/or exercise are important lifestyle modifications for all patients. 110 Controlling such risk factors is thus crucial in the management of CLI to improve cardiovascular health and/or limb-related outcomes. 111 A diet rich in carbohydrates and saturated fatty acids has been associated with increased risk for major adverse cardiovascular events and all-cause mortality, likely attributed to accelerated atherosclerosis. 112 Although there are no specific data for patients with CLI, it is generally accepted that reduction of the intake of saturated fatty acids and increased consumption of monounsaturated fats, ω-3 fatty acids, antioxidants, and other plant oils could limit atherosclerosis progression and thus cardiovascular events. 113 Weight loss interventions when necessary and optimized dietary plans, providing adequate nutritional support, should be strongly recommended in patients with CLI.
Exercise
Previous epidemiologic studies have shown that sedentary lifestyle is associated with worsening of claudication in patients with PAD and a decline in functional capacity. 114 Specifically, the daily number of hours spent sitting and outdoor walking speeds have been associated with faster decline of functional status and greater reduction in calf muscle density. 114 A systematic review by Lane et al. including 30 studies demonstrated that exercise programs for patients with PAD, who are considered to be fit for such interventions, could add significant benefit to usual care and could significantly improve the quality of life of these patients. 115 Although no CLI-specific studies exist, it is generally accepted that exercise-based rehabilitation could reduce cardiovascular morbidity and mortality, 116 while also delaying the disease progression and/or improving walking time and distance. 115 Therefore, it would be a reasonable approach if walking-based exercise programs were offered to patients with CLI if cleared for weight-bearing. 8
Future developments
Patients with CLI may require multiple surgical or endovascular interventions to maintain limb perfusion, while after a certain point amputation seems inevitable. Thus, alternative gene and cellular therapies promoting angiogenesis have been investigated for patients with no further options for revascularization, offering the potential to facilitate wound healing and delay/prevent amputation. The concept behind these investigational approaches is to induce/facilitate growth of new blood vessels from already existing blood vessels either by delivering several growth factors (e.g. vascular endothelial growth factor, fibroblast growth factor) or by injecting undifferentiated or partially differentiated cells that will eventually differentiate into vascular cells. 117
Autologous stem cell therapy for patients with advanced PAD has been gaining increased interest, with a previously published study showing that bone marrow mononuclear cell transplantation could be considered safe and facilitate angiogenesis in CLI cases. 118 A recent meta-analysis of 23 RCTs, comprising 962 patients, studied the outcomes of autologous stem cell therapy for the treatment of patients with CLI during an average follow-up period of up to 12 months among the individual studies. 119 The meta-analysis showed that cell therapy was associated with improved wound healing, increased angiogenesis of the target area, and fewer amputations. 119 Interestingly, the study also demonstrated better ABI values and improved pain-free walking distance for the cell therapy group. 119
The ‘ACP-01 in Patients With Critical Limb Ischemia’ study is a phase 2 study, which is designed to investigate the outcomes of intramuscular injection of ACP-01 (blood-derived autologous stem cells) versus placebo as last resort, in patients with CLI that are not candidates for any other revascularization intervention. 120 A total of 95 patients will be randomized to treatment with ACP-01 or placebo at a 2:1 ratio and will be followed for adverse events for at least 26 weeks. 120 The results of this RCT would provide further data about whether cellular therapy with blood-derived autologous stem cells has a role in the treatment of CLI.
Cellular and/or gene therapy seem to have promising results, but these therapeutic approaches are not harmless (e.g. risk for ‘off-target’ angiogenesis, tumorigenesis, proliferative retinopathy). Furthermore, additional large-scale prospective studies are warranted in order to determine the best cell source (e.g. blood-derived, placenta-derived, etc.), the appropriate dosage, and the optimal route of administration, as all these variables might affect the outcomes.119,121
Key points to remember
Optimized medical care and risk factor modification in addition to revascularization could reduce the incidence of cardiovascular events and MALEs, improving patients’ quality of life.
A recently published meta-analysis demonstrated that patients with CLI treated with statins were 25% and 38% less likely to undergo major target limb amputation or to have a fatal adverse event, respectively.
Statin therapy for patients with CLI could decrease cardiovascular events and prevent/delay disabling amputations.
Renin-angiotensin system inhibitors exert a cardioprotective effect among patients with CLI, but more data are needed to determine the safety and efficacy of ACE inhibitors/ARBs in terms of limb-related risk.
Antiplatelet agents have been shown to reduce cardiovascular events in patients with PAD, but data regarding the additive benefit of DAPT are sparse.
Recent data have provided evidence that low-dose rivaroxaban on a background of aspirin may reduce cardiovascular and limb-related risk in patients with PAD and CLI.
Smoking cessation, stopping recreational drugs, and maintaining a normal weight with a healthy diet and/or exercise are important lifestyle modifications for all patients with atherosclerotic disease, including patients with CLI.
General considerations for the treatment approach of patients with CLI are presented in Figure 1.

A proposed treatment approach for patients with critical limb ischemia (CLI).
Conclusions
All patients with CLI should receive medical preventive measures, including lifestyle modification and optimized medical management of atherosclerosis progression. Statins have been shown to reduce the risk for major cardiovascular and limb-related adverse events and as such are strongly recommended by current guidelines for patients with CLI. Future studies are needed to clarify the role of newer nonstatin agents, including PCSK9 inhibitors, in the management of advanced PAD. Hypertension management with ACE inhibitors or ARBs play an important role in the management of CLI, as it has been associated with improved survival.
Additionally, wide application of DAPT or antithrombotic therapy with low-dose rivaroxaban might improve the efficacy and long-term patency of endovascular interventions, while also minimizing all-cause mortality. However, additional studies are warranted to determine the optimal therapeutic antithrombotic regimens for patients with CLI undergoing revascularization. Finally, successful smoking cessation has been associated with lower mortality rates, better wound healing, and improved amputation-free survival. Thus, smoking cessation should be strongly encouraged in all patients with PAD, regardless of the degree of disease severity. Patients with CLI are at extremely high risk for cardiovascular events and death. Thus, they should receive aggressive guideline-directed medical therapy with a focus on cardiovascular and limb-related risk reduction and improving quality of life. A multidisciplinary team approach for those patients could improve limb salvage rates, facilitating clinical decision making regarding best intervention. Additionally, multidisciplinary teams could help to better address cardiovascular risk modification and wound management, promising increased amputation-free survival.
Footnotes
Declaration of conflicting interests
Ehrin Armstrong serves as a consultant to Abbott Vascular, Boston Scientific, Cardiovascular Systems Incorporated (CSI), Gore, Medtronic, Philips, PQ Bypass, and Shockwave. Stefanos Giannopoulos has no relationships to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
