Abstract
Medial arterial calcification (MAC) is a known risk factor for cardiovascular morbidity. The association between vascular calcifications and poor outcome in several vascular districts suggest that infrapopliteal MAC could be a risk factor for lower-limb amputation (LLA). This study’s objective is to review the available literature focusing on the association between infrapopliteal MAC and LLA in high-risk patients. The PubMed and Embase databases were systematically searched. We selected original studies reporting the association between infrapopliteal MAC and LLAs in patients with diabetes and/or peripheral artery disease (PAD). Estimates were pooled using either a fixed-effects or a random-effects model meta-analysis. Heterogeneity was evaluated using the Q and I2 statistics. Publication bias was investigated with a funnel plot and Egger test. The trim-and-fill method was designed to estimate the possibly missing studies. Influence analysis was conducted to search studies influencing the final result. Test of moderators was used to compare estimates in good versus non-good-quality studies. Fifteen articles satisfied the selection criteria (n = 6489; median follow-up: 36 months). MAC was significantly associated with LLAs (pooled adjusted risk ratio (RR): 2.27; 95% CI: 1.89–2.74; I2 = 25.3%, Q-test: p = 0.17). This association was kept in the subgroup of patients with diabetes (RR: 2.37; 95% CI: 1.76–3.20) and patients with PAD (RR: 2.48; 95% CI: 1.72–3.58). The association was maintained if considering as outcome only major amputations (RR: 2.11; 95% CI: 1.46–3.06). Our results show that infrapopliteal MAC is associated with LLAs, thus suggesting MAC as a possible new marker of the at-risk limb.
Introduction
Medial arterial calcification (MAC), also known as Mönckeberg’s arteriosclerosis, is a non-atherosclerotic disease which affects the arterial tree diffusely, and is associated with aging, diabetes mellitus (DM), and chronic kidney disease (CKD).1–3 It results from an imbalance between pro-calcific and anti-calcific stimuli that induces differentiation of vascular smooth muscle cells and secretion of extracellular calcific micro-vesicles, thus creating calcification nests throughout the medial arterial layer. 4 Radiographically, MAC usually appears as linear, contiguous or rail-track patterns of arterial wall calcification. 5
While MAC has been historically thought of as a benign condition, studies on patients with DM or CKD have shown a strong association between MAC and cardiovascular events.6–8 The most common explanation of this association implies a reduction in arterial wall compliance, leading to high systolic and low diastolic blood pressure (BP), increased left ventricular strain and hypertrophy, and reduced perfusion of the coronary arteries during diastole. These conditions could explain the elevated risk of myocardial infarction, heart failure, and cardiovascular mortality seen in patients with MAC.3,5–8
MAC has also been shown to be associated with peripheral artery disease (PAD) and chronic limb-threatening ischemia (CLTI).9,10 Narula and colleagues found, in samples of patients submitted to lower-limb amputation (LLA) for various causes, a high prevalence of MAC in distal arteries. 11 In addition, distal MAC was more common in LLAs due to CLTI than in amputees for other reasons,11,12 suggesting that it might be a factor contributing to CLTI development. Histological studies have also revealed that most below-the-knee and below-the-ankle arteries, that could be collectively referred to as ‘infrapopliteal arteries’, tend to have a much higher burden of MAC than above-the-knee arteries.13,14
These data suggest that infrapopliteal MAC might be a potential risk factor for CLTI leading to amputation. This association might be of particular importance in patients with DM and PAD that present the highest risk of undergoing LLA. However, no single study is available providing enough evidence to support a definite role for MAC as a risk factor for LLA, neither in the general population nor in the subgroup of high-risk patients, and no systematic review focusing on this relationship has been conducted yet.
The aims of the present study were, therefore, to perform a systematic review and meta-analysis of the literature focusing on the association between the presence of infrapopliteal MAC and any kind of LLA in a population of patients with common risk factors for LLA (i.e. either DM or PAD), as well as in the subgroup of patients with DM and in the subgroup of patients with PAD. We also aimed to investigate the presence of an association between infrapopliteal MAC and the amputation level (i.e. above vs below the ankle).
Methods
Data source and search strategy
The authors declare that all supporting data are available within the article (and its online supplementary files).
We performed a systematic search of PubMed and Embase databases from inception of the database to the 31 January 2020. We searched for original studies on patients with DM and/or PAD, comparing patients with clinically evident infrapopliteal MAC versus patients with no clinically evident infrapopliteal MAC, in terms of LLAs. The exact search strategy is reported in online supplemental Table I. In addition, according to the Preferred Reporting Items for Systematic reviews and Meta-analyses (PRISMA) statement, 15 we screened the reference lists of the included articles for further eligible papers. Finally, we searched for possibly published editorials, letters, reviews or meta-analysis on this issue.
Study selection
The selection process is shown in Figure 1.
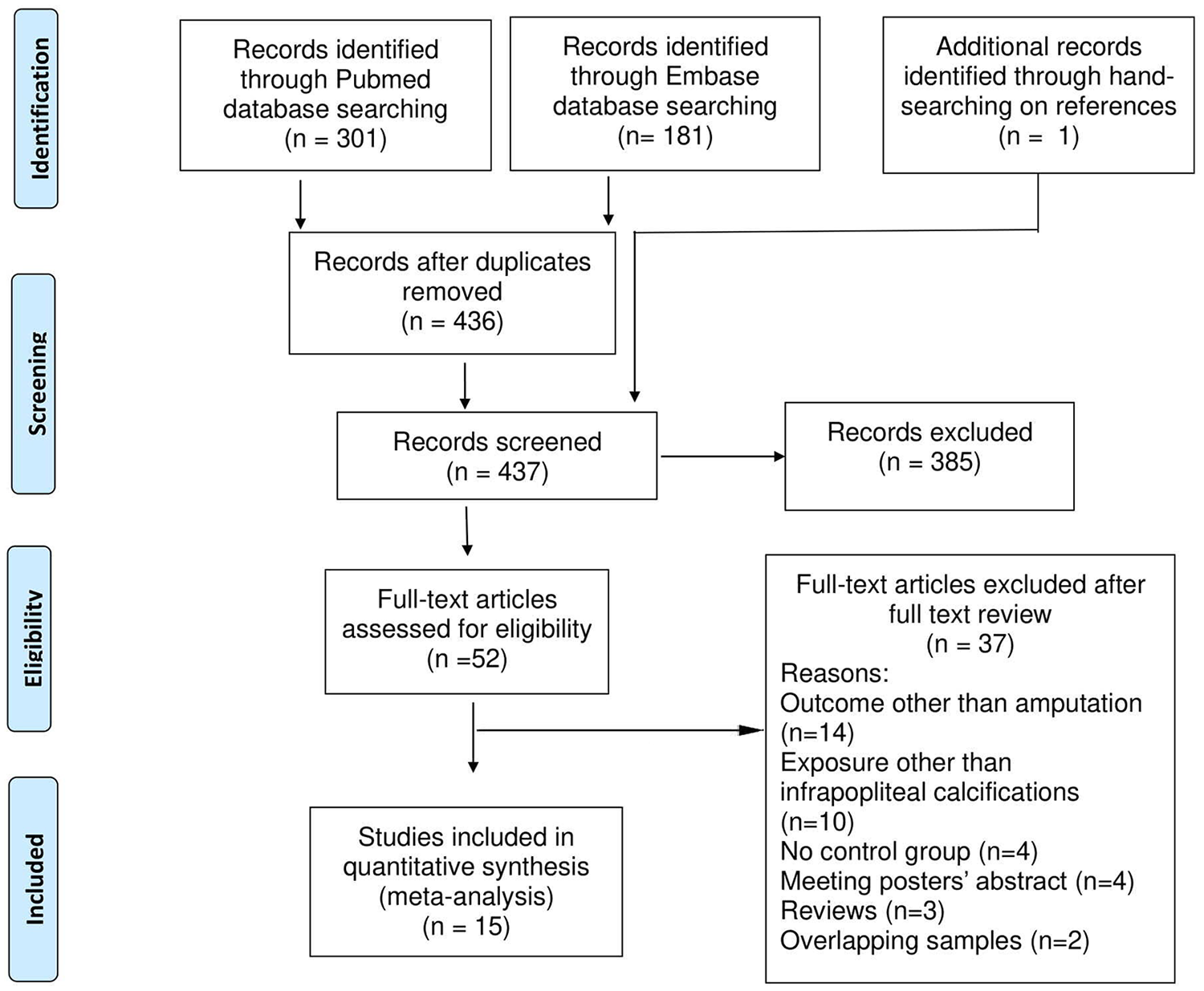
Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) diagram of study selection process.
Studies were reviewed for meeting the following inclusion criteria: (1) being a prospective or retrospective observational study (we excluded cross-sectional studies, case series, case reports, letters, and editorials); (2) recruiting patients with either PAD or DM, or both; (3) reporting data of at least one group of infrapopliteal MAC carriers, identified either by direct visualization of medial calcifications on imaging tests or indirectly by finding an ankle–brachial index (ABI) > 1.3 16 ; (4) reporting data of a control group with no infrapopliteal MAC; (5) considering as outcome any kind of LLA (i.e. either minor or major or both); (6) reporting point and interval estimates of the association between infrapopliteal MAC and any kind of LLA or at least providing in the full text enough data to calculate it.
Studies were at first evaluated by analyzing titles and abstracts. Those studies clearly not matching inclusion criteria were excluded. The remaining studies were accessed in full text for evaluation of inclusion criteria. If multiple papers referred to likely overlapping study groups (i.e. studies coming from the same research group with an overlapping recruitment period of patients with similar inclusion criteria), we selected the article with the most comprehensive data.
Data extraction and quality assessment
Two investigators (FL and AZ) independently reviewed all studies, matching selection criteria and extracted data using a data extraction form containing the following fields: (1) first author, year; (2) country and recruiting period; (3) study design; (4) study population; (5) definition and number of exposed/cases and controls; (6) events in exposed/cases and controls; (7) outcomes and relative measure of association between exposure and outcome (i.e. type of measure, value, 95% CI) – we considered as adequate, the relative risk (RR), the odds ratio (OR) or the hazard ratio (HR), and we considered OR and HR as an approximation of the RR, under the assumption of a rare outcome occurrence; (8) median follow-up; and (9) potential confounders adjusted for.
Where such information was neither reported nor calculable from the full text data, authors were contacted to request it.
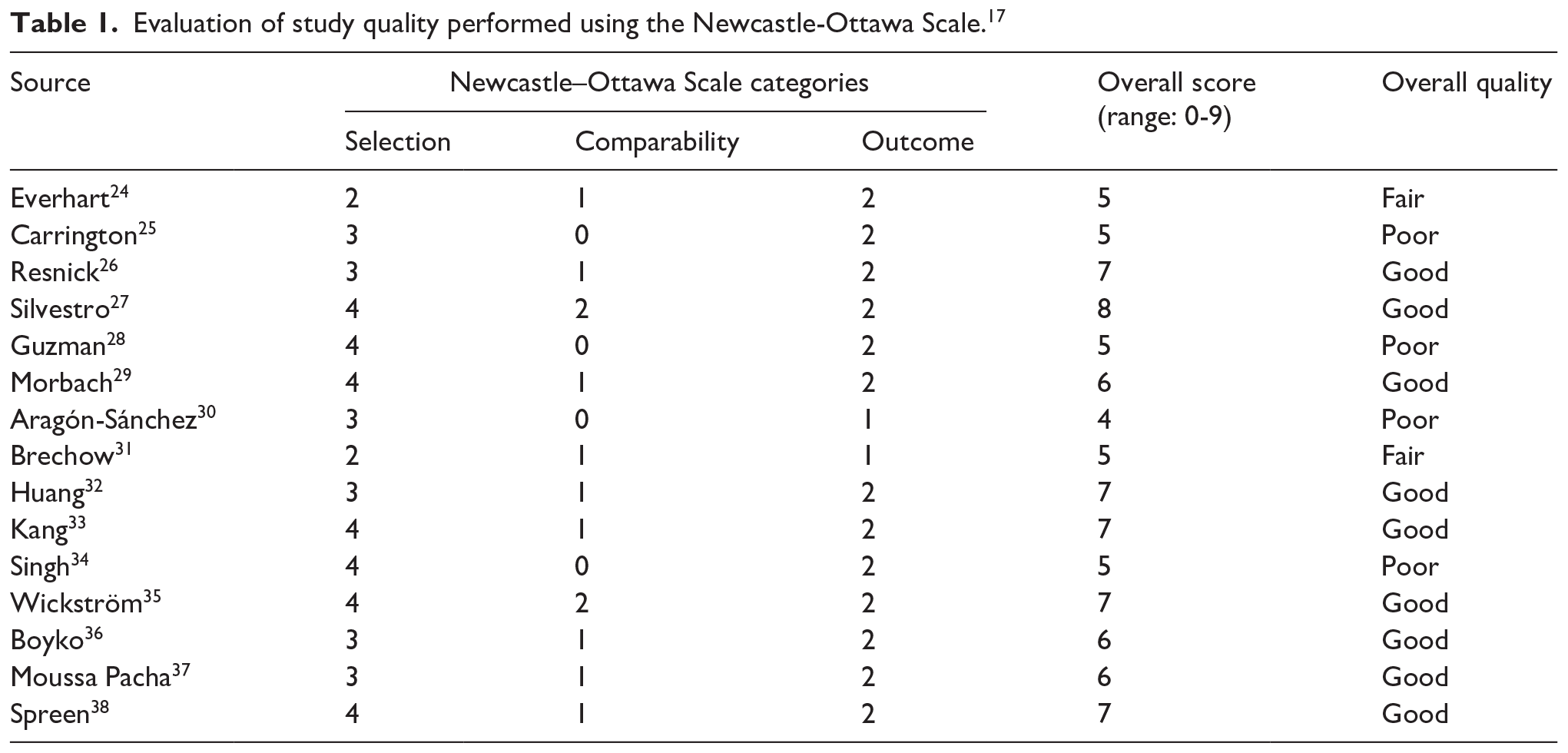
The risk of bias in the included studies was evaluated independently by two investigators (FL and AU) using the Newcastle–Ottawa Scale 17 (NOS), a tool that judges a study quality based on the evaluation of items regarding the selection of study groups, their comparability, and outcome assessment on a scale of 0–9. Disagreements were resolved by discussion between the two review authors; if no agreement could be reached, it was planned a third author (RF) would decide. Study quality was considered ‘good’ (i.e., reduced risk of bias) if a score of at least 3 in the selection domain and at least 1 in the comparability domain and at least 2 in the outcome domain of the NOS were reached, as previously described. 18
Statistical analysis
Original estimates were pooled using either a fixed- or a random-effects model. We tested the presence of between-studies heterogeneity using Q and I2 statistics. 19 We considered relevant statistical heterogeneity present when the I2 index was above 70%. In this case, results generated with a random-effects model were reported.
Forest plots were used to graphically show pooled estimates and relative 95% CIs. Publication bias was visually investigated with a funnel plot and formally tested with an Egger test. 20 The trim-and-fill method 21 was also used to estimate the number of possibly existing and missed studies as well as the adjusted pooled effect size, taking into account those missing studies.
We also performed an influence analysis to detect studies significantly conditioning the pooled estimate of the meta-analysis by calculating such estimate excluding one study at a time.
Moreover, we calculated a pooled estimate of effect size in high-quality studies versus fair-to-low quality studies, as well as in studies with MAC direct versus indirect diagnosis. Subsequently, we tested the absence of difference between these groups.
We also executed a separate analysis for the subgroup of patients with diabetes and for the subgroup of patients with PAD.
Considering that the overall LLA rate is the sum of major (i.e., above-the-ankle) and minor (i.e., below-the-ankle) amputation rate and that a higher minor amputation rate may not necessarily represent a negative outcome, as it may reflect earlier diagnosis and treatment of potentially limb threatening lesions, we performed a separate analysis including studies reporting specifically the major or the minor amputation rate. Data were analyzed using R, version 3.3 22 and the ‘metafor’ package. 23
Statistical significance was set at p < 0.05, and all tests were two-tailed.
Results
We identified 301 papers in PubMed and 181 in Embase. After deleting duplicates, we obtained 436 unique records that were reviewed by title and abstract (Figure 1). We excluded 385 articles that did not meet our inclusion criteria because they were not related to this study’s topic. One study was added after hand-searching references. This left 52 records for full text review. Among these, 37 articles were excluded (27% of fully reviewed papers were excluded because they did not study amputations as an outcome; Figure 1). Finally, 15 articles were selected for inclusion in the meta-analysis;24–38 their main characteristics are reported in online supplemental Table II.
Overall, 6489 participants (minimum 82, maximum 1830) were considered in the present systematic review with a median follow-up time of 36 months (minimum 0.8, maximum 264).
The majority of studies were from European (six studies)25,27,30,31,35,38 and North American (six studies)24,26,28,34,36,37 populations, followed by Asian population (two studies),32,33 while one study was multicentric (Germany, USA, and Tanzania). 29
Among included studies, three were conducted on asymptomatic outpatients with diabetes,24,26,36 three on inpatients admitted for diabetic foot-related causes,29–31 two studies included both symptomatic and asymptomatic patients with PAD,28,37 and six were conducted on symptomatic patients with PAD.27,32–35,38 One remaining study was a case–control study comparing lower-limb amputees and age-matched controls for the presence of incompressible arteries upon ABI testing. 25
The presence of infrapopliteal MAC was directly assessed by visualization of medial calcification on imaging tests in five studies,24,28,30,32,33 while it was indirectly deducted, by the presence of incompressible arteries upon ABI testing, in the remaining 10.25–27,29,31,34–38 All studies reported the occurrence rate of either major LLAs or any kind (i.e. major + minor) of LLAs. In 10 of the 15 included studies, the measure of association reported was the HR.24,28,29,32–38 There was only one study 27 reporting the RR, while in one further study 30 the RR and its corresponding CI were calculated from data reported in the full text. Furthermore, in three studies,25,26,31 the OR was used as a measure of association.
In two studies,32,35 it was not possible to retrieve crude numbers of events in MAC carriers and controls; however, a measure of association between exposure and outcome was available together with a CI and a list of confounders adjusted for in the statistical model used.
The risk of bias in each study, evaluated with the NOS is reported in Table 1. Of the included studies, 60% were of good quality, while 13% were of fair quality, and 27% were poor; thus showing suboptimal quality for 40% of included studies.
Evaluation of study quality performed using the Newcastle-Ottawa Scale. 17
Overall risk of lower-limb amputation (LLA)
The pooled adjusted RR of LLA in infrapopliteal MAC carriers versus non-carriers, obtained from a fixed-effects (FE) model, was 2.27 (95% CI: 1.89–2.74; p < 0.0001; I2 = 25.3%, Q-test p = 0.17). Results are graphically shown in a forest plot in Figure 2. The overall RR estimate was not significantly affected by the quality of included studies (pooled estimated RR in good-quality studies: 2.15; 95% CI: 1.72–2.69; pooled estimated RR in fair-to-low quality studies: 2.56; 95% CI: 1.83–3.59; test of moderators: p = 0.48) (online supplemental Figure I). We performed additional analysis to evaluate the potential modificatory effects on the overall RR estimate exerted by the MAC diagnostic method used or by the presence of an adjustment for confounding factors like blood pressure (BP) or CKD in the estimation model used by any single study.
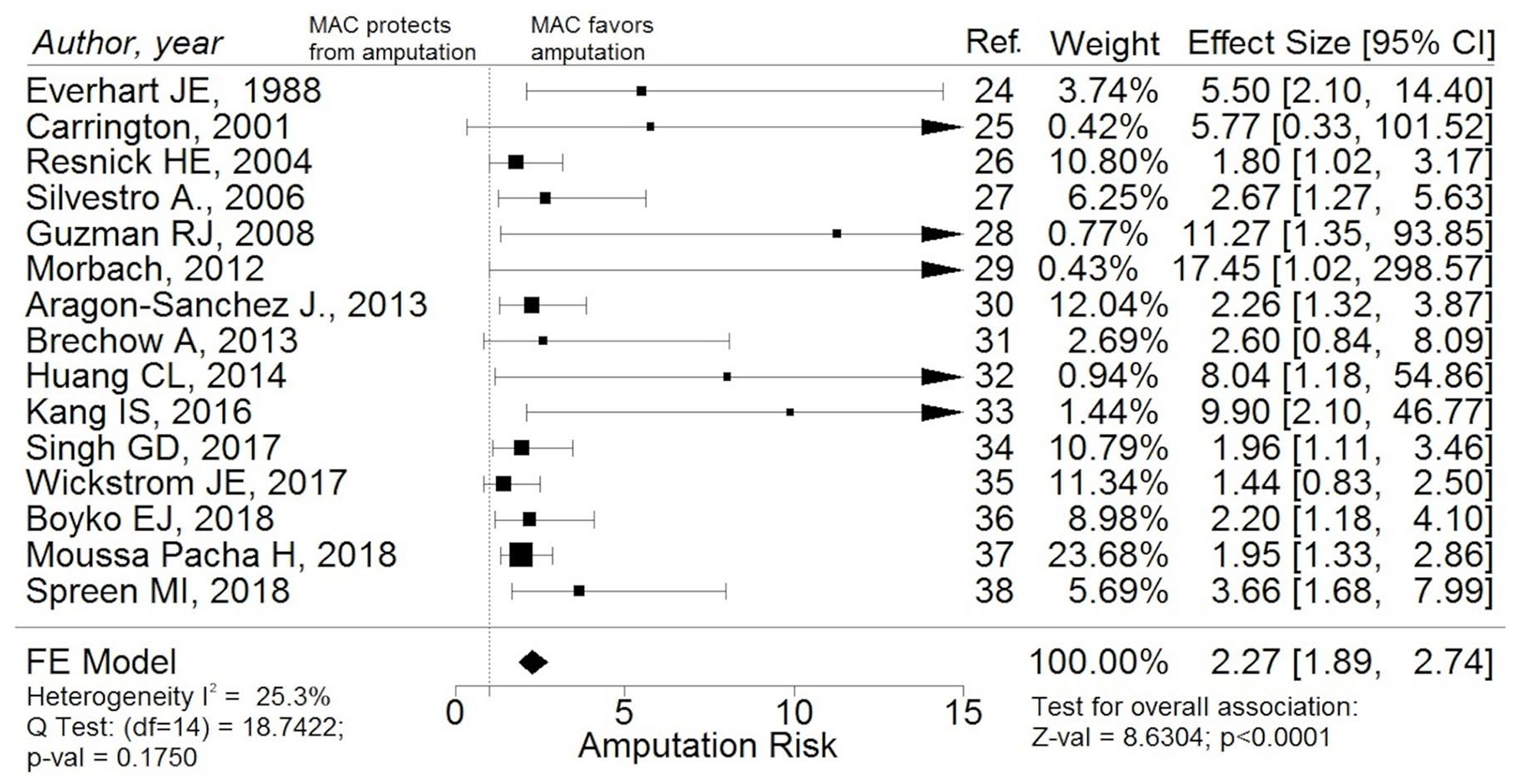
Forest plot with studies investigating the association between infrapopliteal MAC and overall LLA risk in patients with either diabetes or PAD or both: the presence of infrapopliteal MAC is significantly associated with increased risk of LLAs of any kind.
The pooled amputation RR estimate in the subgroup of studies in which MAC diagnosis was made through direct visualization of calcifications was higher than in the subgroup in which MAC diagnosis was made indirectly through finding elevated ABI and measured, respectively, 3.43 (95% CI: 2.23–5.26) and 2.06 (95% CI: 1.68–2.54) (test of moderators: p < 0.05) (online supplemental Figure II). There was no evidence of effect modification when comparing pooled estimates generated in BP-controlled versus non-BP-controlled studies, as well as in CKD-controlled versus non-CKD-controlled studies (online supplemental Figures III and IV).
The risk of publication bias has been evaluated graphically with a funnel plot (Figure 3A) that shows a clear asymmetry towards studies reporting MAC as a significant risk factor of LLA. The regression test for funnel plot asymmetry was highly significant (p = 0.0004); however, the pooled estimate was still statistically significant after applying the trim-and-fill method to account for publication bias (2.06; 95% CI: 1.72–2.46) (Figure 3B).
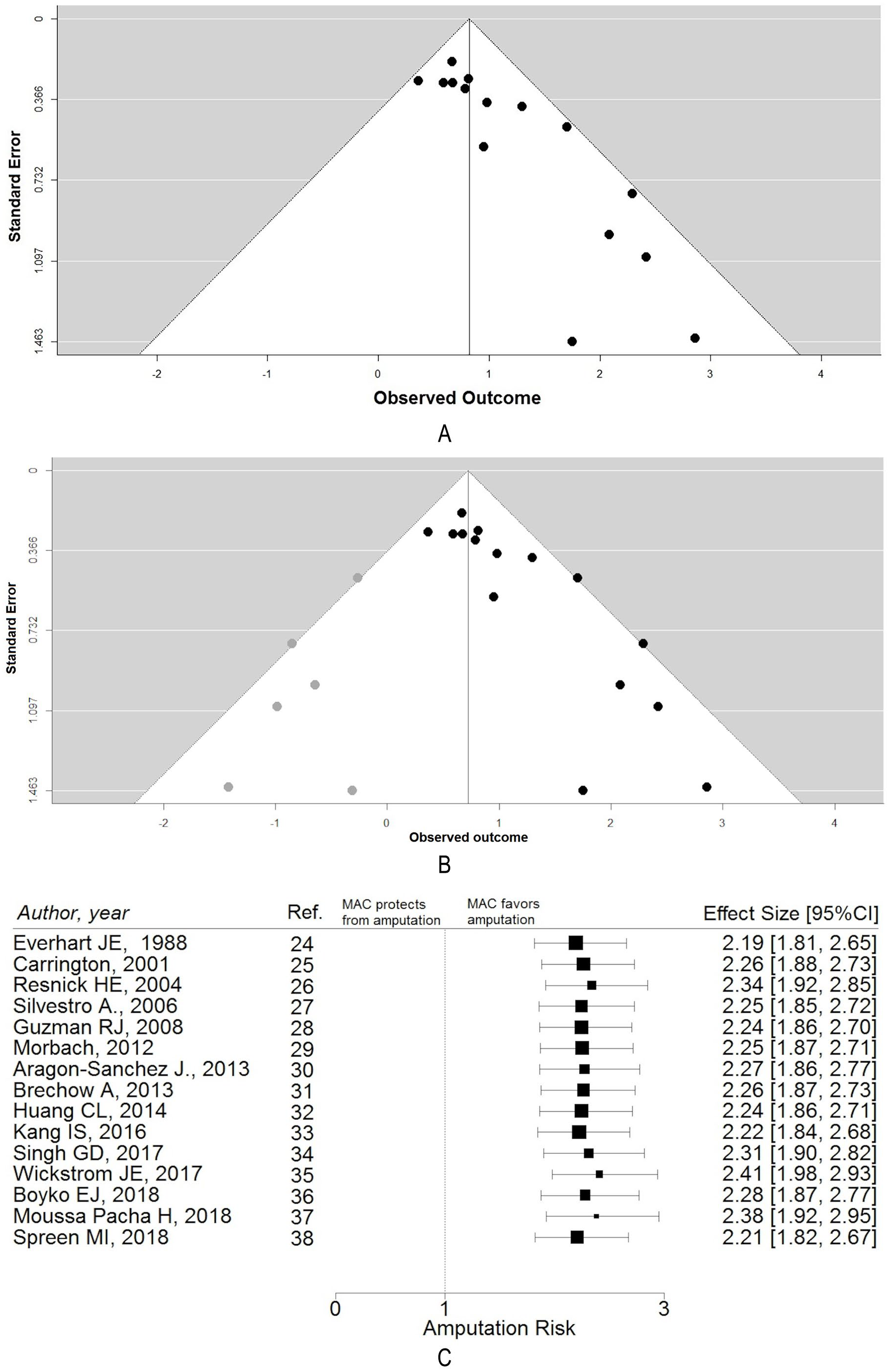
(A) Funnel plot of included studies showing significant publication bias; (B) trim-filled funnel plot; (C) influence analysis: overall amputation RR estimate excluding each study at a time by the final computation.
The influence analysis showed that the final estimate was not significantly different after excluding each of the studies by the pooled estimate calculations (Figure 3C).
Risk of LLA in patients with diabetes
Restricting the analysis to studies performed on a population of patients with diabetes, we found a statistically significant association between infrapopliteal MAC and occurrence of LLA (pooled estimated RR: 2.37; 95% CI: 1.76–3.20; p < 0.0001) with non-significant statistical heterogeneity (I2 = 17%; Q-test = 5.85) (Figure 4). The result was not influenced by the method used for MAC diagnosis (pooled estimated RR in studies with MAC direct visualization 2.79; 95% CI: 1.75–4.46; pooled estimated RR in studies with indirect MAC diagnosis 2.45; 95% CI: 1.09–5.49; test of moderators: p = 0.785). Moreover, the RR estimate was overlapping when considering only studies on symptomatic patients (2.46; 95% CI: 1.52–3.96).
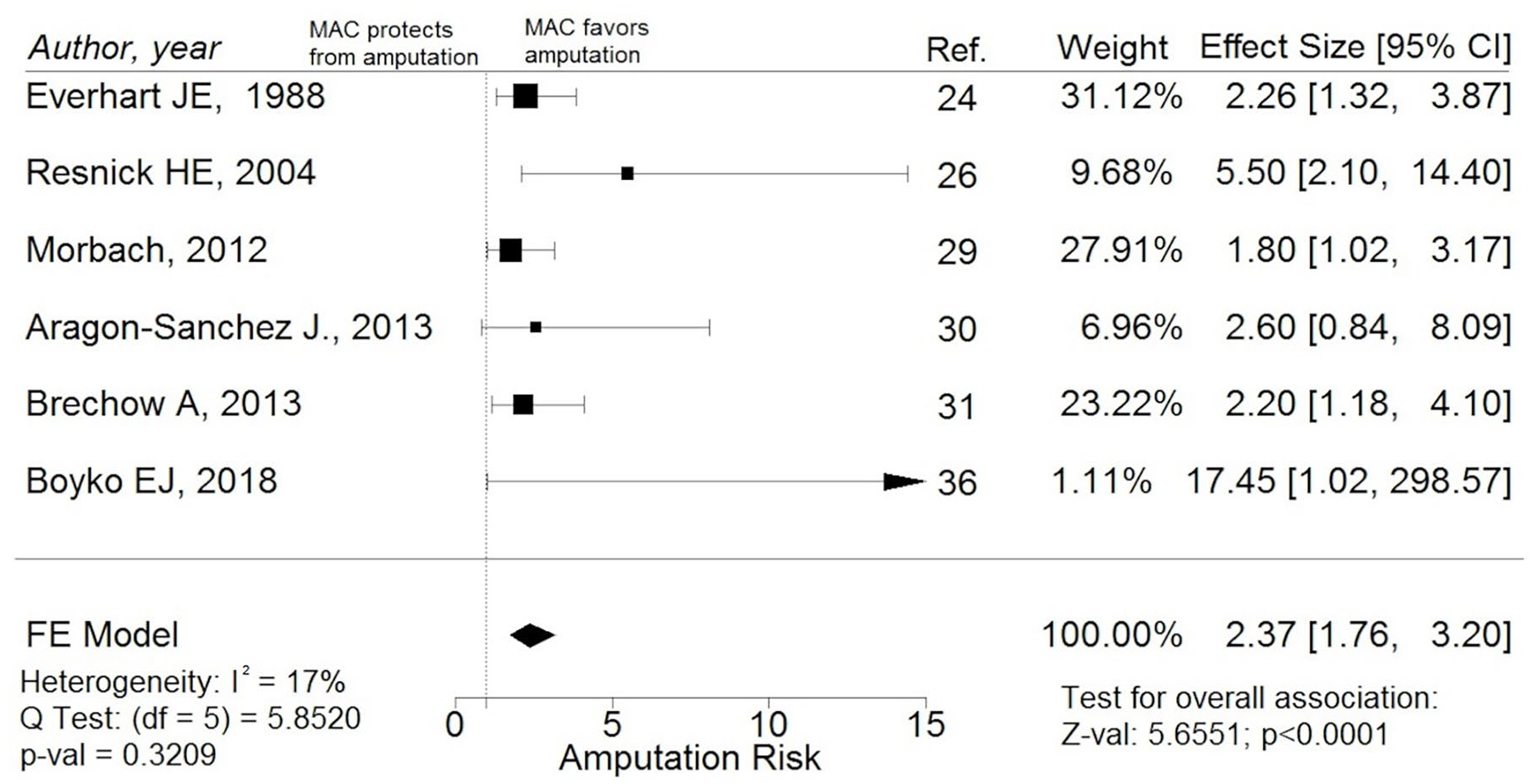
Forest plot reporting studies investigating the association between infrapopliteal MAC and LLA risk in a subgroup of studies on patients with diabetes mellitus: the presence of infrapopliteal MAC is significantly associated with increased risk of any kind of LLA in this subgroup.
Risk of LLA in patients with PAD
Performing the analysis only on studies recruiting patients with PAD, we found a statistically significant higher risk of any kind of LLA in infrapopliteal MAC carriers versus non-MAC carriers. The pooled estimated RR was 2.48 (95% CI: 1.72–3.58, p < 0.001) with only moderate statistical heterogeneity (I2 = 43.2%, Q = 12.3) (Figure 5). This result might have been influenced by the method used for MAC diagnosis (pooled estimated RR in studies with MAC direct visualization 9.60; 95% CI: 3.36–27.42; pooled estimated RR in studies with indirect MAC diagnosis 2.02; 95% CI: 1.58–2.59; test of moderators: p < 0.01). Moreover, the estimate was not different when considering only studies on symptomatic patients (2.29; 95% CI: 1.68–3.12).
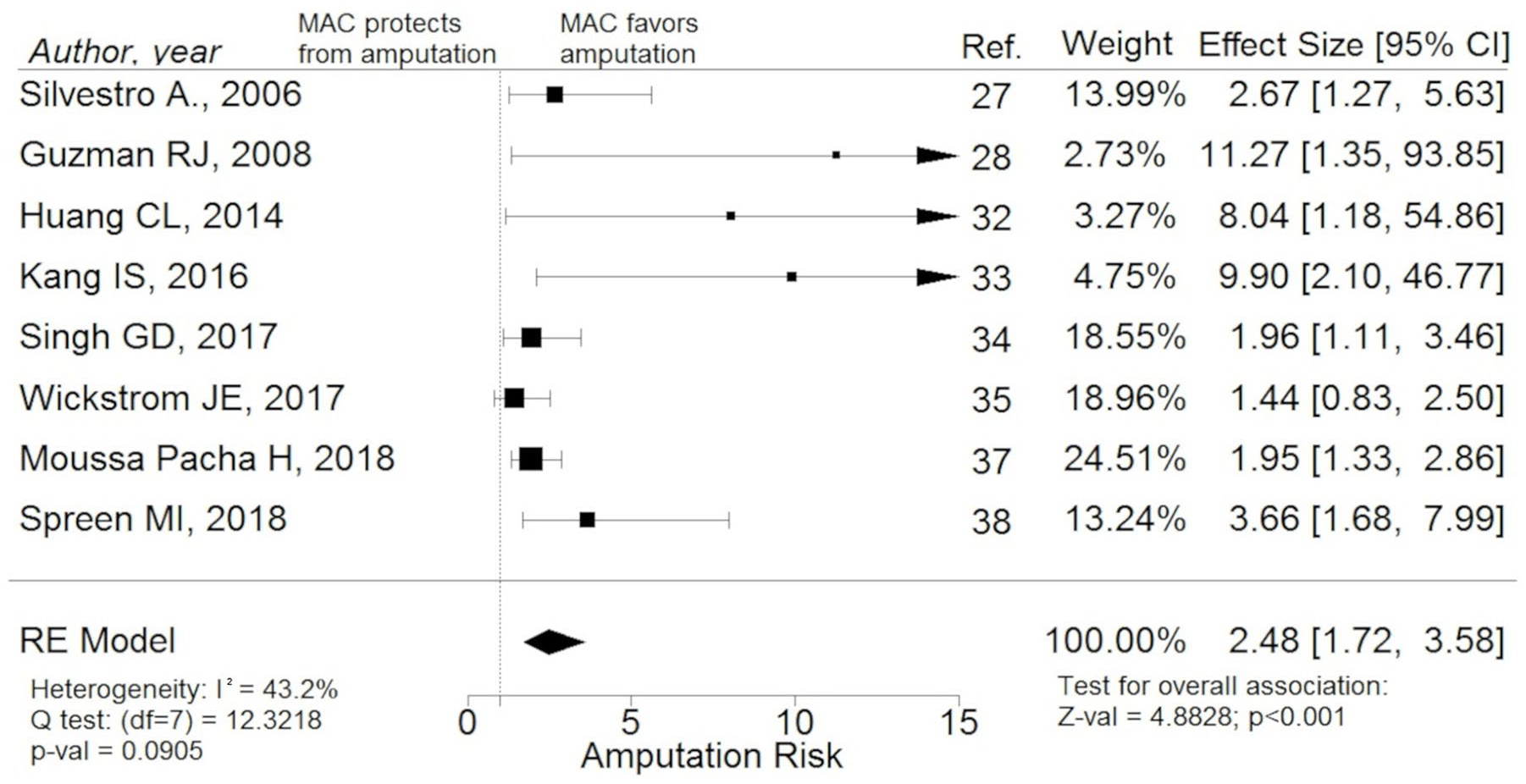
Forest plot reporting studies investigating the association between infrapopliteal MAC and LLA risk in a subgroup of studies on patients with PAD: the presence of infrapopliteal MAC is significantly associated with increased risk of any kind of LLA in this subgroup.
Risk of major LLA in study population
As the overall LLA rate is determined by both minor and major amputations, we decided to focus on those studies reporting major LLA as a separate outcome. We found that our pooled estimate of the major LLA risk ratio between infrapopliteal MAC carriers and non-carriers was still significantly higher in the infrapopliteal MAC carrier group (RR 2.21; 95% CI: 1.65–2.96, p < 0.001; I2 = 17%, Q = 9.65) (Figure 6). The estimate was not influenced by the method used for MAC diagnosis (pooled estimated RR in studies with MAC direct diagnosis 2.49; 95% CI: 1.48–4.19; pooled estimated RR in studies with MAC indirect diagnosis 2.19; 95% CI: 1.62–2.97; test of moderators: p = 0.675).
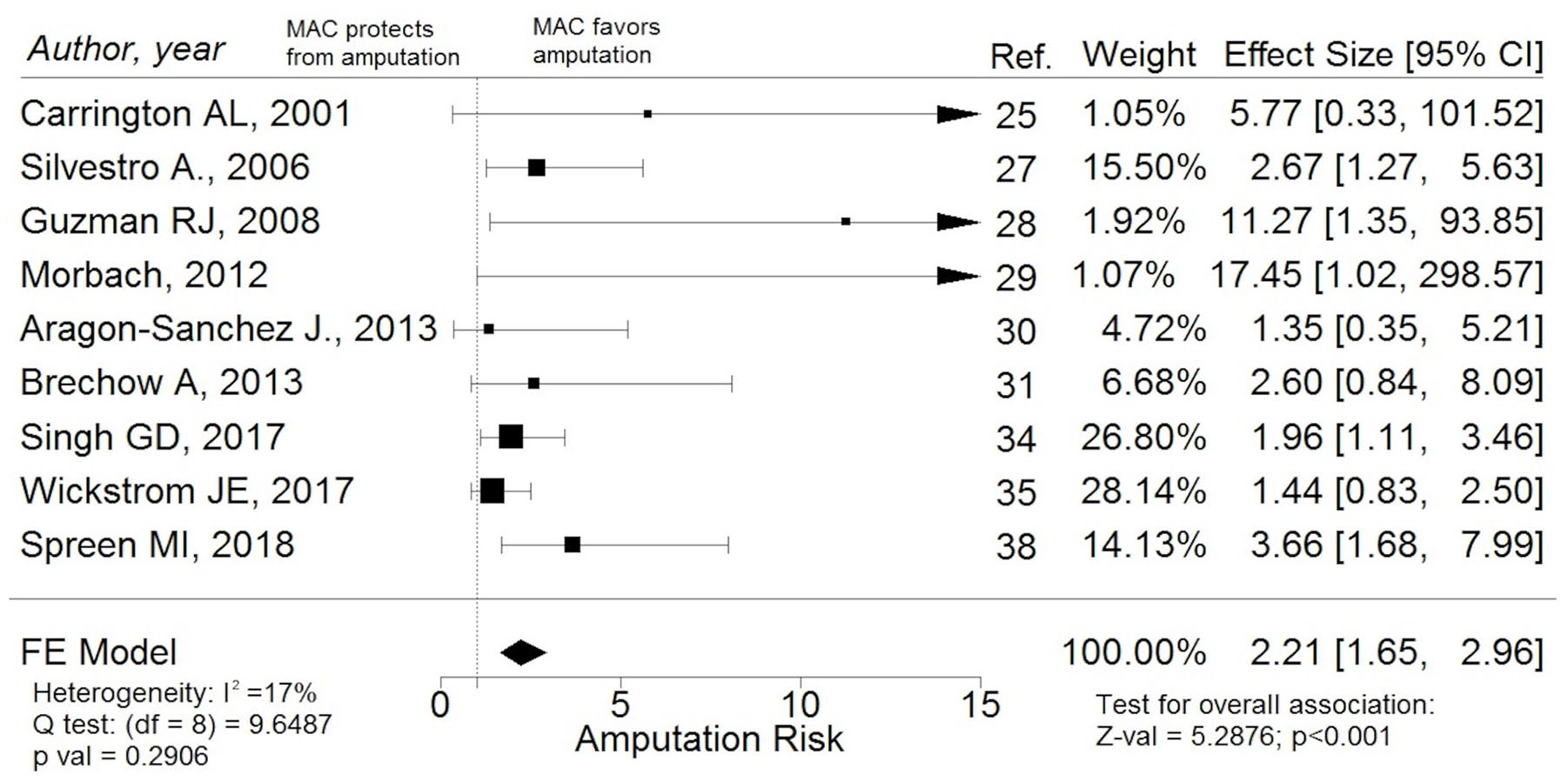
Forest plot reporting studies of association between infrapopliteal MAC and major amputation risk: the presence of infrapopliteal MAC is significantly associated with increased risk of lower-limb major amputation.
Risk of minor LLA in study population
None of the included studies reported a measure of the association between infrapopliteal MAC and minor LLAs. However, one study 30 reported enough data to allow calculation of the RR (2.53; 95% CI: 1.36–4.74).
Discussion
The aim of this systematic review and meta-analysis was to synthesize the available literature on the risk of LLA in infrapopliteal MAC carriers. Overall, studies reporting the measure of association between MAC and amputation risk were few and their quality, evaluated with the NOS, has been estimated as ‘good’ in only 9 out of 15. Despite this, the estimate of the association was quite consistent among them, thus allowing for low-to-moderate statistical heterogeneity.
The influence analysis showed that only one study had a significantly heavier weight than others; 37 however, the overall association between MAC and risk of LLA did not significantly change when removing it by the final computation, as well as removing each of the other studies, thus suggesting a good consistency for our final results. Moreover, our sensitivity analysis showed that the pooled estimate was not significantly affected by lower quality studies.
The final result of our study demonstrates a strongly significant association between infrapopliteal MAC and amputation risk in patients with baseline risk factors for LLA that include a background of DM and/or PAD. However, the method used for MAC diagnosis did influence the final overall estimate as well as the pooled estimate in the subgroup of patients with PAD (test of moderators: p < 0.05), with studies in which MAC was diagnosed through direct visualization pointing towards a stronger effect of MAC in predicting LLAs. This finding indicates that our main results are to be interpreted as a conservative estimate of the true effect of MAC as a predictor of LLA, which could potentially be higher when MAC diagnosis is made by directly visualizing calcifications on imaging tests.
The association between MAC and LLAs is kept in the subgroup of studies reporting as outcome the major amputation rate, thus indicating that the clinical finding of infrapopliteal MAC could be associated with a worsened final outcome. This is particularly important as MAC may be present independently from the findings commonly used to diagnose CLTI. The global guideline for the management of CLTI 39 defines the ‘threatened limb’ by the presence of either one among ABI < 0.4; ankle systolic pressure < 50 mmHg; toe systolic pressure < 30 mmHg; and transcutaneous oxygen partial pressure (TcPO2) < 30 mmHg. Our study shows that, in addition to these well-established criteria, the presence of infrapopliteal MAC, either directly visualized on imaging tests or indirectly deducted by finding a high ABI, qualifies as a further marker of the at-risk limb. Patients with infrapopliteal MAC may therefore likely benefit from a tight follow-up (i.e. more frequent clinical foot evaluations) aiming to prompt treatment of ulcer-predisposing conditions (like hyperkeratosis or nail disorders) and very early recognition of foot ulcers or infections, as well as to ensure adequate preventive footwear is used. Moreover, identifying patients at higher risk of failure could allow for better targeting of resources that might be destined primarily to such patients. Nevertheless, nothing can be inferred from our meta-analysis about the predictive role of infrapopliteal MAC in the general population, as the included studies were enrolling only patients with known baseline risk factors for LLA. Moreover, given the paucity of reported data on minor amputations, nothing could be inferred on their association with MAC.
The mechanism by which MAC is linked to limb failure is still unknown. MAC has been proposed to yield to a reduction of arterial elasticity and thus to a decreased ability to smooth down the intermittent and pulsatile arterial blood flow. 40 However, it cannot be excluded that medial calcific deposits might favor an obstructive effect, particularly in small calliper distal foot vessels, impairing blood distribution to distal foot areas (like toes, the forefoot or the heel) that might, in turn, result in being more susceptible to ischemic damages. It might be speculated that the association demonstrated in this meta-analysis may not reflect a true pathogenetic link between medial calcifications and limb failure but rather a statistical association between a marker of poor general health (i.e., MAC) and a specific clinical outcome (i.e., amputations). However, Chantelau 10 and Ferrier, 41 in small studies, showed that, in patients with diabetes, MAC-positive feet were associated with a small arteries disease, affecting particularly the metatarsal arteries, and potentially reducing forefoot tissue perfusion, thus suggesting a pathogenetic role for MAC in favoring small arteries occlusion and setting the basis for further studies aiming to answer this question.
Limitations
We acknowledge that this study has many limitations. Firstly, while the I2 statistic showed low statistical heterogeneity, this estimate has to be interpreted cautiously given the small number of included studies. 42 Furthermore, the population sampled in the included studies were clinically heterogenous, ranging from totally asymptomatic patients to severely ischemic ones. Therefore, we could assume that the true effect size varies from study to study and that the effect sizes included in our meta-analysis are just a random sample of the infinite number that could have been observed. We addressed this heterogeneity by comparing the pooled estimate generated with a fixed-effect model, legitimated by a low degree of statistical heterogeneity, with the estimate generated with two kinds of random-effects model (i.e., DerSimonian–Laird and Sidik–Jonkman). The estimates generated with random-effects models were clearly overlapping each other and to the estimate generated using a fixed-effect model thus suggesting the true effect size is likely to be close to our final estimates.
Secondly, data were affected by publication bias, as shown by the funnel plot and the formal Egger test. We addressed this limit by using the trim-and-fill method that yielded to an effect size that kept its significance and did not show any major changes in its magnitude after such correction. This gives confidence regarding the true existence of an association between infrapopliteal MAC and amputation risk.
Lastly, the quality of the included studies was not good in six out of 15. However, we addressed this by comparing the pooled estimates generated in the subgroups of good versus non-good-quality studies with a test of moderators that showed no significant differences to be present. This finding suggests that the final overall estimate was not significantly influenced by non-good-quality studies.
Future research directions
Future research will be needed to clarify the mechanism underlying MAC association with poorer limb prognosis. Specifically, it would be informative to compare the angiographic perfusion of heavily calcified distal foot arteries with non-calcified arteries of patients with comparable baseline conditions. If a direct pathogenetic connection between MAC and poor distal perfusion could be demonstrated, then MAC could become a possible new target for pharmacologic therapy aiming at reducing amputation risk.
Drugs to reduce MAC load are already in the early stages of the pharmacological pipeline. 43 The Yamani-15/5 solution has been shown, after selective instillation, to be effective in restoring patency to calcified peripheral arteries of amputated limbs. 44 This drug is now being investigated in a phase 1 clinical trial in patients deemed to require leg amputation (NCT02539303). 45 Moreover, a recent preclinical study showed that the tissue-nonspecific-alkaline-phosphatase inhibitor SBI-425 was able to nearly halt MAC formation and increase survival in the CKD-Mineral-and-Bone-Disorder mouse model. 46 Future research will be necessary to show whether, in humans, drugs acting on the balance between pro-calcific and anti-calcific factors may actually affect the building up of medial calcifications and, in turn, have any effect on clinically relevant outcomes, like amputations.
Furthermore, future research should investigate if the association we demonstrated for the lower limb is true also in other vascular beds, as this effect may likely not be lower-limb-specific. While cardiac calcifications are well known to worsen the general cardiovascular risk, little is known about other territories such as the upper limb or visceral districts.
Conclusions
This study shows that, in patients with either DM or PAD or both, having infrapopliteal MAC associates with a higher risk of undergoing a LLA of any kind. Further analysis showed that this association is kept in the subgroup of patients with DM as well as in the subgroup of patients with PAD. Moreover, the association is also significant when considering only major amputations as outcome.
Our data show an association between infrapopliteal MAC and LLAs, thus suggesting MAC as a possible new clinical marker of the at-risk limb that could allow better stratification of high-risk patients.
This result is strengthened by a low statistical between-studies heterogeneity and by the absence of high influence studies. However, the robustness of the overall effect is weakened by the presence of clinical heterogeneity between studies’ populations, by a relevant publication bias, and by a study quality that resulted in being ‘good’ in nine out of 15 included studies.
Future research will be needed to investigate the association between MAC and poor outcomes in vascular beds other than the lower limb, as well as to establish the pathophysiological pathways underlying the observed association that could potentially become, in the future, a new possible pharmacological target aiming at limb salvage.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X20979738 – Supplemental material for Association of infrapopliteal medial arterial calcification with lower-limb amputations in high-risk patients: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X20979738 for Association of infrapopliteal medial arterial calcification with lower-limb amputations in high-risk patients: A systematic review and meta-analysis by Fabrizio Losurdo, Roberto Ferraresi, Alessandro Ucci, Anna Zanetti, Giacomo Clerici and Antonella Zambon in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the ‘Master in Epidemiologia e Biostatistica per la Ricerca Clinica’ program held at the University of Milan-Bicocca, Milan, Italy.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
