Abstract
There is a reported increased incidence of lower extremity amputations in individuals with diabetes who are treated with canagliflozin (an SGLT2 receptor inhibitor). It is unclear whether this is an unintended consequence of therapy, or whether canagliflozin can affect peripheral limb perfusion in the setting of underling arterial malperfusion. To evaluate this we explored the effect of canagliflozin on tissue recovery following unilateral hind-limb ischemia (HLI). Adult wildtype (+/+) and diabetic (db/db) mice were maintained on 8 weeks of a regular chow diet, or a chow diet containing canagliflozin (200 mg/kg). Following HLI, hind-limb appearance, function, and Doppler perfusion were serially evaluated. Gastrocnemius muscle fiber size and microvessel density were also evaluated 21 days following HLI. We observed that db/db that received a diet containing canagliflozin had significantly worse hind-limb function and appearance scores compared to both db/db mice that received a regular diet and +/+ mice that received a canagliflozin diet. At post-HLI day 21, db/db mice that received a canagliflozin diet also had decreased Doppler perfusion, gastrocnemius muscle fiber size, and microvessel density compared to +/+ mice that received a canagliflozin diet. These findings indicate that canagliflozin appears to impede ischemic peripheral tissue recovery and warrant further clinical investigation in individuals with diabetes and a history of peripheral artery disease.
Introduction
Sodium-dependent glucose co-transporter 2 (SGLT2) inhibitors are a class of pharmacological agents used for the treatment of type 2 diabetes.1–3 Their mechanism of action is to block renal glomerular reabsorption of glucose and promote glucosuria, hence improve serum euglycemia, decrease adipose mass, and reduce cardiovascular-associated mortality.1, 3–5 Canagliflozin (Invokana®, Janssen, Titusville, NJ) belongs to this class of medications and was US Food and Drug Administration (FDA)-approved in 2013 for the treatment of patients with type 2 diabetes and cardiovascular disease. 6 However, the CANagliflozin cardioVascular Assessment Study (CANVAS) program observed that even though canagliflozin improved cardiovascular and renal outcomes in patients with type 2 diabetes, it also led to an increased incidence of major and minor lower extremity amputations.7–9 Accordingly, the FDA issued a black box warning for this potential increased risk of amputations with the use of canagliflozin.8–10 The mechanism of action for this effect remains unclear; however, multiple studies have demonstrated that canagliflozin has pleotropic tissue effects that may affect ischemic tissue recovery.11–16
Notably, large-scale studies of other SGLT2 inhibitors, such as dapagliflozin and empagliflozin, have not observed a similar effect on subject amputation rates.3,17,18 This has led to wide speculation that specifically canagliflozin may have off-target effects on the peripheral vasculature.8,9 This notion was supported by recent studies demonstrating that unlike other SGLT2 inhibitors, canagliflozin can significantly inhibit endothelial cell (EC) tubule formation and proliferation, and promote cell cycle arrest. 11 However, these findings are in conflict with one report that suggests that a short acute oral course of canagliflozin can actually increase ischemic murine hind-limb perfusion. 19 Given these seemingly contradictory findings, we evaluated the impact of canagliflozin on hind-limb perfusion and angiogenesis in wildtype (+/+) and diabetic (db/db) mice, which are most representative of human metabolic syndrome and type 2 diabetes.20, 21 We hypothesized that canagliflozin impedes ischemic hind-limb recovery in the five +/+ and db/db mice.
Subjects and methods
Murine models and diets
We evaluated +/+ and db/db mice, on a C57BL6 background, from both sexes, that received either a regular chow diet (db/db, n = 7; +/+, n = 5) or a canagliflozin-containing diet (db/db, n = 8; +/+, n = 9). The canagliflozin-containing diet was prepared by mixing canagliflozin 200 mg/kg with the regular chow diet (Envigo, Madison, WI, USA). This provided an estimated canagliflozin dose of 0.8–1.6 ng per day. All mice were initiated on their respective diets at 6 weeks of age for a period of 8 weeks prior to a unilateral femoral artery ligation procedure (Figure 1). All animal care and handling procedures conformed to Institutional Animal Care and Use Committee (IACUC) guidelines.
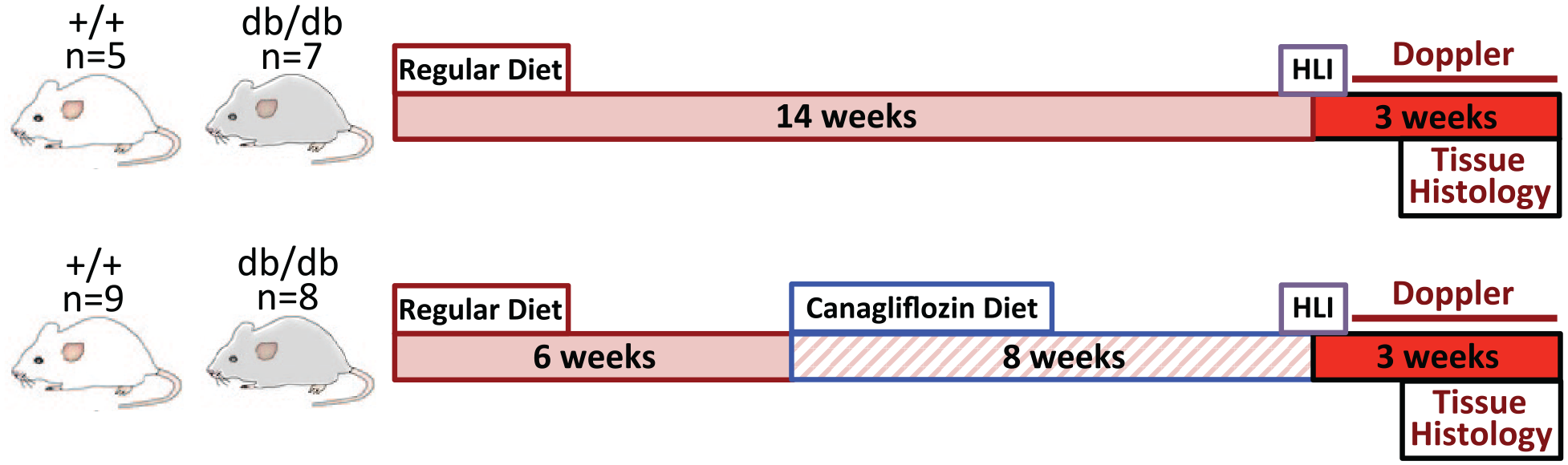
Study timeline. At 6 weeks from birth, both diabetic (db/db) and wildtype (+/+) mice were maintained on either a regular chow diet or a regular diet containing canagliflozin (200 mg/kg). A unilateral HLI procedure was performed in all mice at 14 weeks. Outcome measurements were collected on postoperative days 0, 7, 14, and 21. All mice were sacrificed on post-HLI day 21 (week 17) for muscle histology analysis.
Hind-limb ischemia (HLI) procedure
Unilateral femoral artery ligation was performed as previously described.22, 23 Briefly, following administration of inhaled anesthetic, a small oblique groin incision was made in the left adductor hind-limb region. The femoral artery was exposed proximal to the epigastric collaterals, ligated with 6-0 silk ties, and transected. The incision was then closed with interrupted non-absorbable sutures and the mice were closely monitored for 24 hours post-procedure.
Limb function and appearance scores
The db/db and +/+ mice were evaluated on postoperative days 3, 7, 14, and 21 for ischemic hind-limb function and appearance as previously described with some modifications. 23 Briefly, limb function was independently graded on a scale of 0 to 4 (4 = foot dragging, 3 = no foot dragging but no plantar flexion, 2 = abnormal plantar flexion, 1 = flexing toes with gentle traction of tail, and 0 = normal foot and leg function). Limb appearance was independently graded on a scale of 0 to 4 (4 = digit/foot auto-amputation, 3 = severe discoloration/gangrene, 2 = moderate discoloration, 1 = mild discoloration, 0 = normal appearance). A single experienced observer surveyed and collected serial limb function and appearance scores in a consistent fashion.
Doppler perfusion
Doppler perfusion assessments were performed as previously described. 23 Briefly, mice were subjected to an inhaled anesthetic and body temperature was maintained over a warming pad. A laser Doppler perfusion imager (model LD12-IR; Moor Instruments, Wilmington, DE, USA) modified for high resolution and depth of penetration (2 mm) with an 830-nm wavelength, infrared, 2.5-mW laser diode (100 μm beam diameter and 15 kHz bandwidth) was used to evaluate the bilateral hind-limbs (ischemic and non-ischemic). Perfusion was evaluated in the whole limb, gastrocnemius region, and hind-paw region on post-HLI days 0, 7, 14, and 21.
Gastrocnemius muscle fiber size
On post-HLI day 21, the mice were euthanized and perfused with intra-cardiac heparin solution. The gastrocnemius muscles were removed en bloc and embedded in optimal cutting temperature (OCT) compound. Muscle fiber size and morphology were evaluated using standard tissue sectioning and H&E staining methods.23, 24 Briefly, at least five 10 µm diameter sections obtained at interrupted intervals of 50 µm were obtained from the ischemic and non-ischemic hind-limb gastrocnemius muscle of +/+ and db/db mice. The relative muscle fiber area was evaluated by averaging three ×20 magnification images from three randomly selected regions (n = 3 per mouse group). The muscle fiber area was then measured using MatLab software (MathWorks, Natick, MA) and expressed in relative units between the mouse genotypes and diets.
Microvessel density staining
Gastrocnemius muscle sections were blocked with 5% goat serum (0.1% Triton) for 60 minutes. Muscle microvessel density was evaluated with Alexa Flor 594-conjugated Griffonia simplicifolia isolectin-1-B4 (1:100; Thermo Fisher Scientific, Waltham, MA, USA).23,24 Five ×20 images were selected from three randomly selected sections per group (n = 3 per mouse group). The microvessel capillary count was quantified using ImageJ software (National Institutes of Health, Bethesda, MD, USA), and expressed in relative units between the mouse genotypes and diets.
Statistical analysis
An unpaired two-tailed t-test with Welch’s correction was performed to evaluate differences in gastrocnemius muscle fiber size and microvessel density between +/+ and db/db mice, on either regular or canagliflozin diet. Non-parametric ordinary two-way analysis of variance (ANOVA) with Tukey’s multiple comparisons test was used to evaluate differences in hind-limb function and appearance scores, as well as relative changes in hind-limb Doppler perfusion over time between mouse and diet groups. All statistical analyses were performed using GraphPad Prism, Version 8.0 (GraphPad Software; San Diego, CA, USA). We considered p < 0.05 to be statistically significant, and also reported significance at p < 0.01 and p < 0.001. Error was presented as standard error of the mean. Both sexes were evaluated in murine studies but were underpowered to extrapolate statistical significance.
Results
Limb function scores
Among mice that received a regular diet, db/db mice had worse limb function scores compared to +/+ mice, particularly at post-HLI days 3 and 7 (p < 0.01, p < 0.001, respectively; Figure 2A). Similarly, db/db mice that received canagliflozin had worse limb function scores at all postoperative time points compared to +/+ mice that also received canagliflozin (Figure 2B). Interestingly, +/+ mice that received canagliflozin had worse limb function scores compared to +/+ mice that did not receive canagliflozin throughout the post-HLI recovery period (Figure 2C). A similar trend was observed in db/db mice that received canagliflozin, which demonstrated worse limb function scores compared to db/db mice that did not receive canagliflozin, particularly at post-HLI days 14 and 21 (Figure 2D).
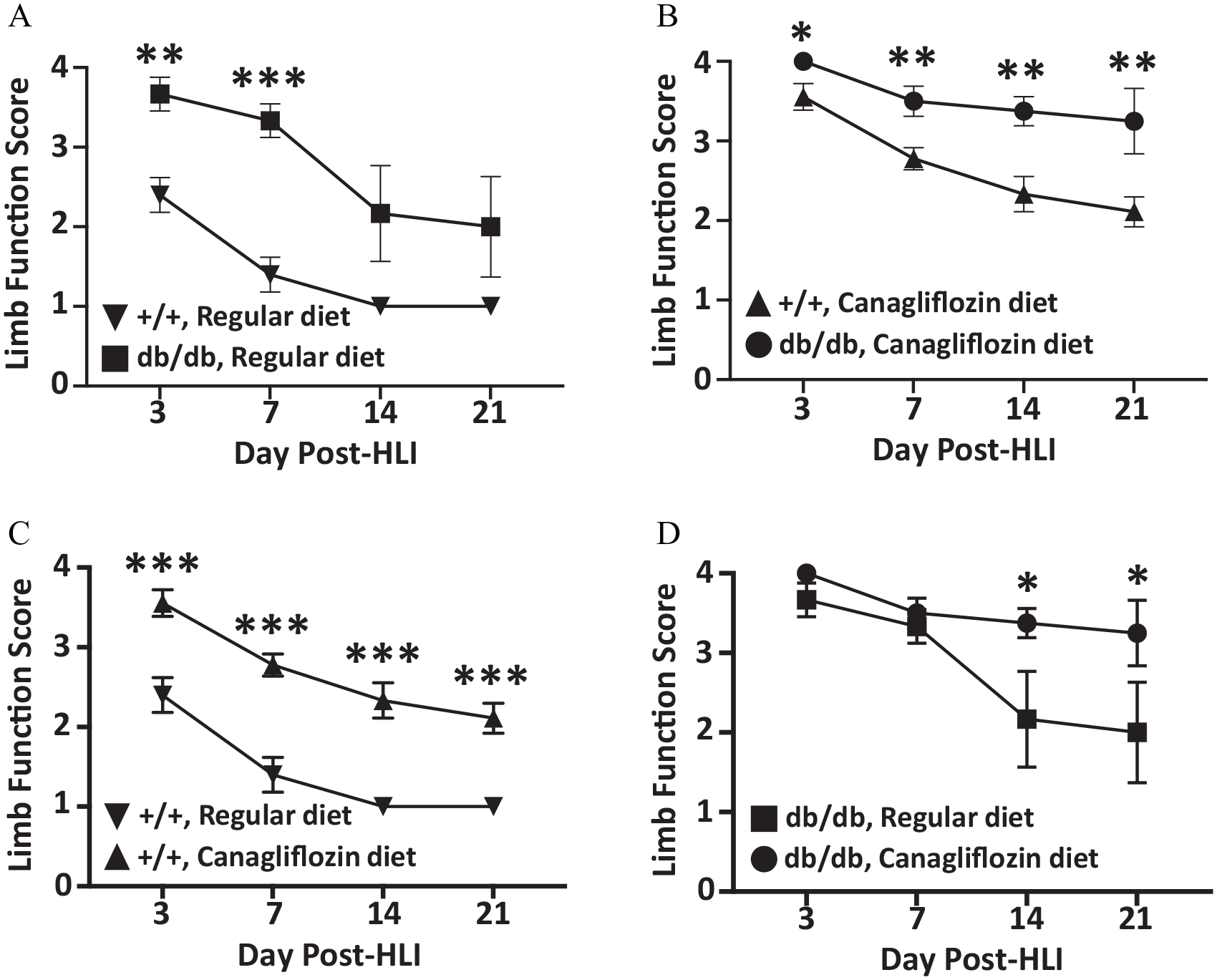
Ischemic hind-limb (HLI) function scores. (A) +/+ and db/db mice that received a regular diet. (B) +/+ and db/db mice that received a canagliflozin diet. (C) +/+ mice that received either a regular or canagliflozin diet. (D) db/db mice that received either a regular or canagliflozin diet. *p < 0.05, **p < 0.01, ***p < 0.001.
Limb appearance scores
Among mice that received a regular diet, db/db mice had worse limb appearance scores compared to the +/+ mice, particularly at post-HLI days 3 and 7 (p < 0.001, p < 0.01, respectively; Figure 3A). Similarly, db/db mice that received canagliflozin had worse limb appearance scores throughout the post-HLI recovery period when compared to +/+ mice that also received canagliflozin (Figure 3B). Initially, +/+ mice that received canagliflozin had worse limb appearance scores compared to +/+ mice that received a regular diet, but later time points demonstrated an improving limb appearance (Figure 3C). However, db/db mice that received canagliflozin demonstrated worse limb appearance (Figure 3D) and appearance scores compared to db/db mice that did not receive canagliflozin, particularly at post-HLI days 14 and 21 (p < 40.05; Figure 3E).
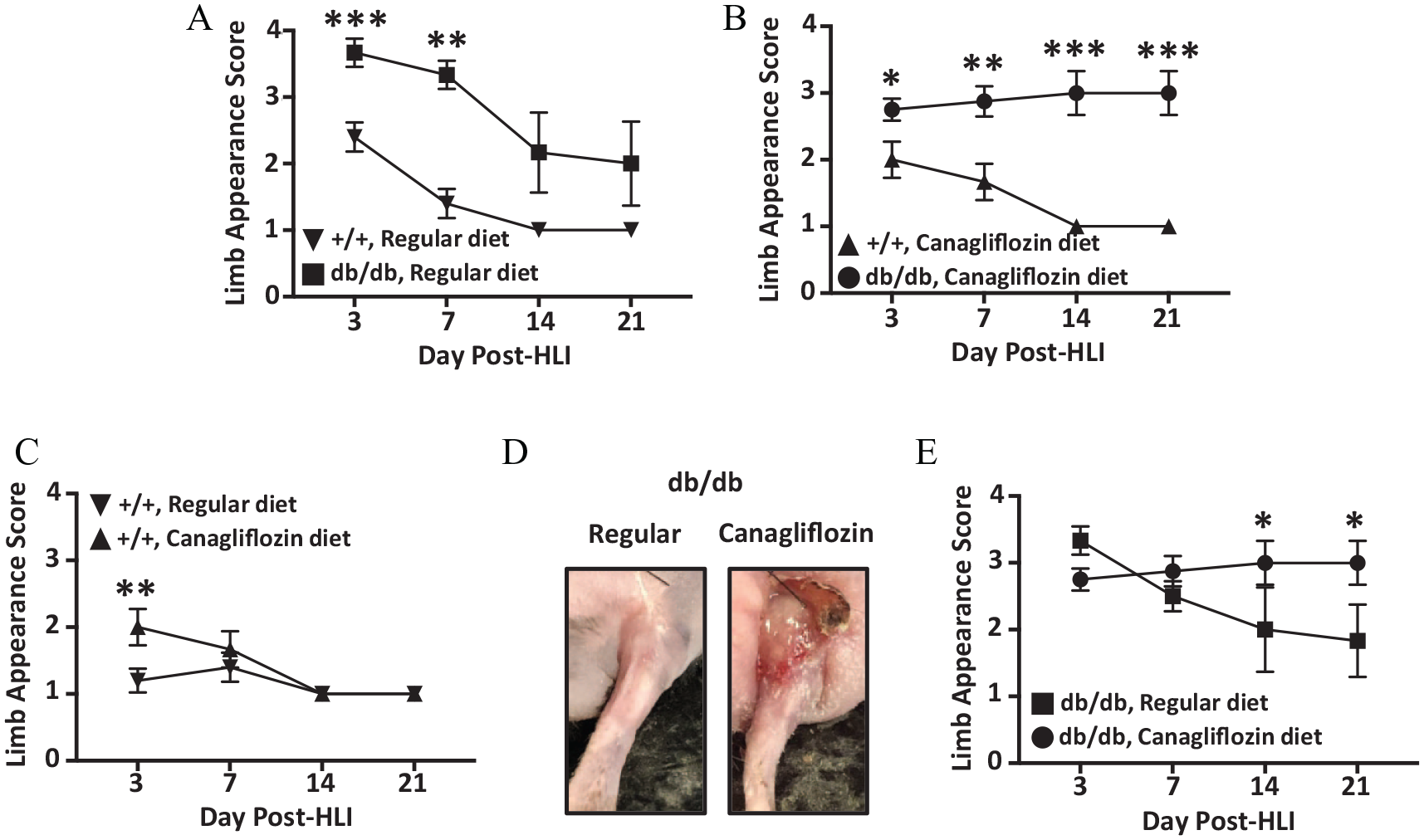
Ischemic hind-limb (HLI) appearance scores. (A) +/+ and db/db mice that received a regular diet. (B) +/+ and db/db mice that received a canagliflozin diet. (C) +/+ mice that received either a regular or canagliflozin diet. (D) Poorly healing ischemic hind-limb tissue in db/db mice that received a canagliflozin diet. (E) db/db mice that received either a regular or canagliflozin diet. *p < 0.05, **p < 0.01, ***p < 0.001.
Gastrocnemius muscle fiber size and microvessel density
Among +/+ mice, those that received canagliflozin had a smaller relative muscle fiber size compared to those maintained on a regular diet (Figure 4A), but the difference was not statistically significant (p = 0.09; Figure 4B). Among db/db mice, those that received canagliflozin had a significant decrease in ischemic hind-limb gastrocnemius muscle fiber sizes compared to those maintained on a regular diet (p < 0.05; Figure 4B).
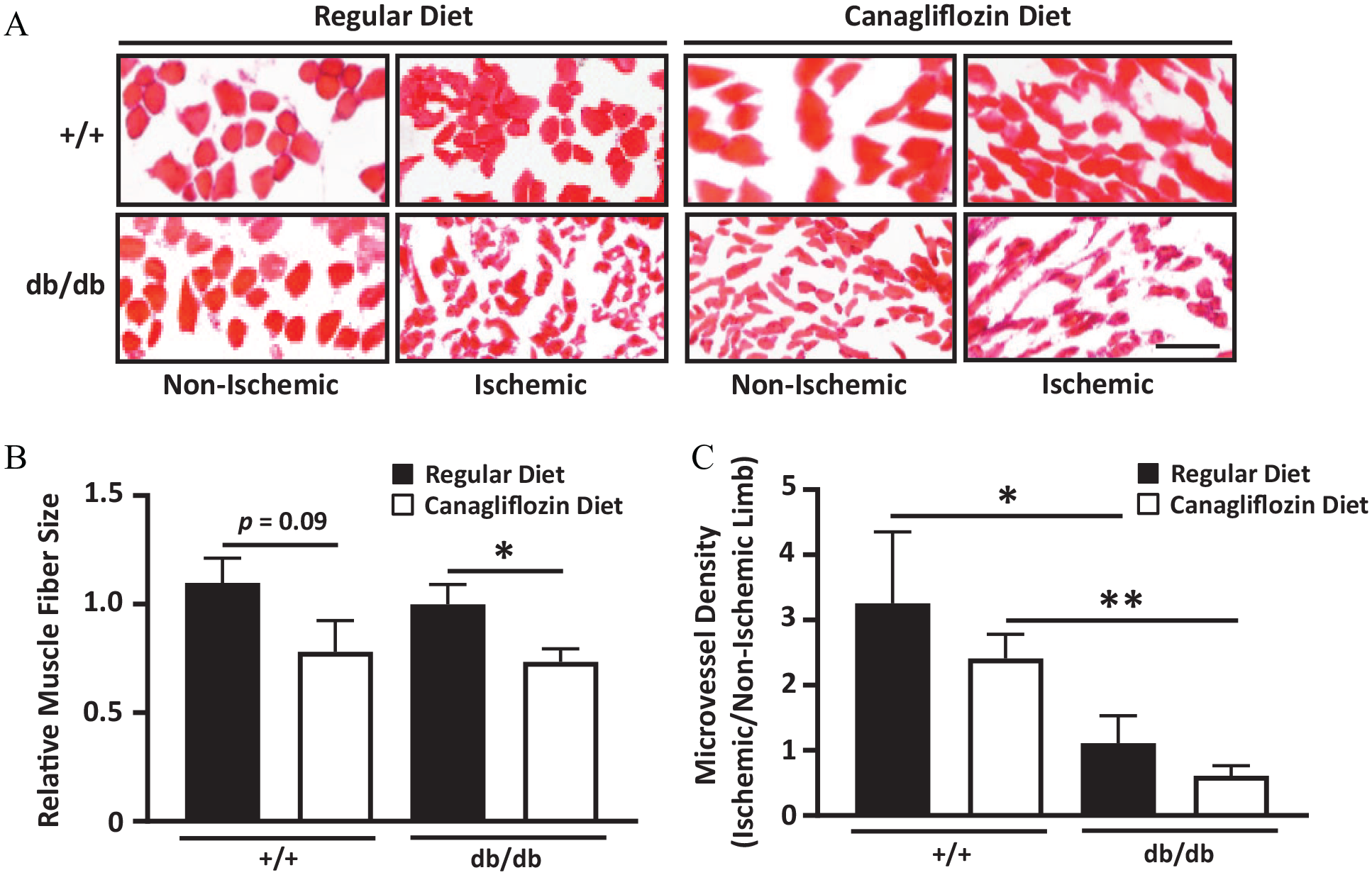
Ischemic hind-limb muscle fiber size and microvessel density. (A) Representative images of H&E-stained gastrocnemius muscle segments isolated from ischemic and non-ischemic hind-limbs of db/db and +/+ mice maintained on a regular diet or a canagliflozin-containing diet. (B) Relative muscle fiber size in +/+ and db/db mice that were maintained on either a regular diet or canagliflozin-containing diet. (C) Microvessel density ratio (ischemic/non-ischemic limb) in +/+ and db/db mice maintained on a regular or canagliflozin-containing diet. *p < 0.05, **p <0.01.
Among mice that received a regular diet, db/db mice had lower gastrocnemius muscle microvessel density compared to the +/+ mice (Figure 4C). Similarly, among mice that received canagliflozin, db/db mice had lower gastrocnemius muscle microvessel density compared to +/+ mice (p < 0.01; Figure 4C).
Doppler perfusion analysis
As expected, db/db mice demonstrated decreased hind-limb Doppler perfusion compared to +/+ mice maintained on a regular diet (Figure 5A; online supplemental Figure 1). Among both +/+ and db/db mice that received canagliflozin there was decreased perfusion in both ischemic and non-ischemic hind-limbs (Figure 5B). Malperfusion of the ischemic hind-limbs was greater among db/db mice (Figure 5B). Among +/+ mice that received canagliflozin, there was overall decreased Doppler perfusion in the whole hind-limb (Figure 5C), as well as in the gastrocnemius (Figure 5D), and distal hind-paw regions (Figure 5E). Similarly, among db/db mice that received canagliflozin, there was also overall decreased perfusion in the whole hind-limb (Figure 5F), gastrocnemius (Figure 5G), and distal hind-paw regions (Figure 5H). Both +/+ and db/db mice demonstrated significantly decreased perfusion in the gastrocnemius region on post-HLI day 21 (p < 0.05; Figures 5D and 5G), but only db/db mice demonstrated a significant difference in hind-paw perfusion (p < 0.05; Figure 5H).
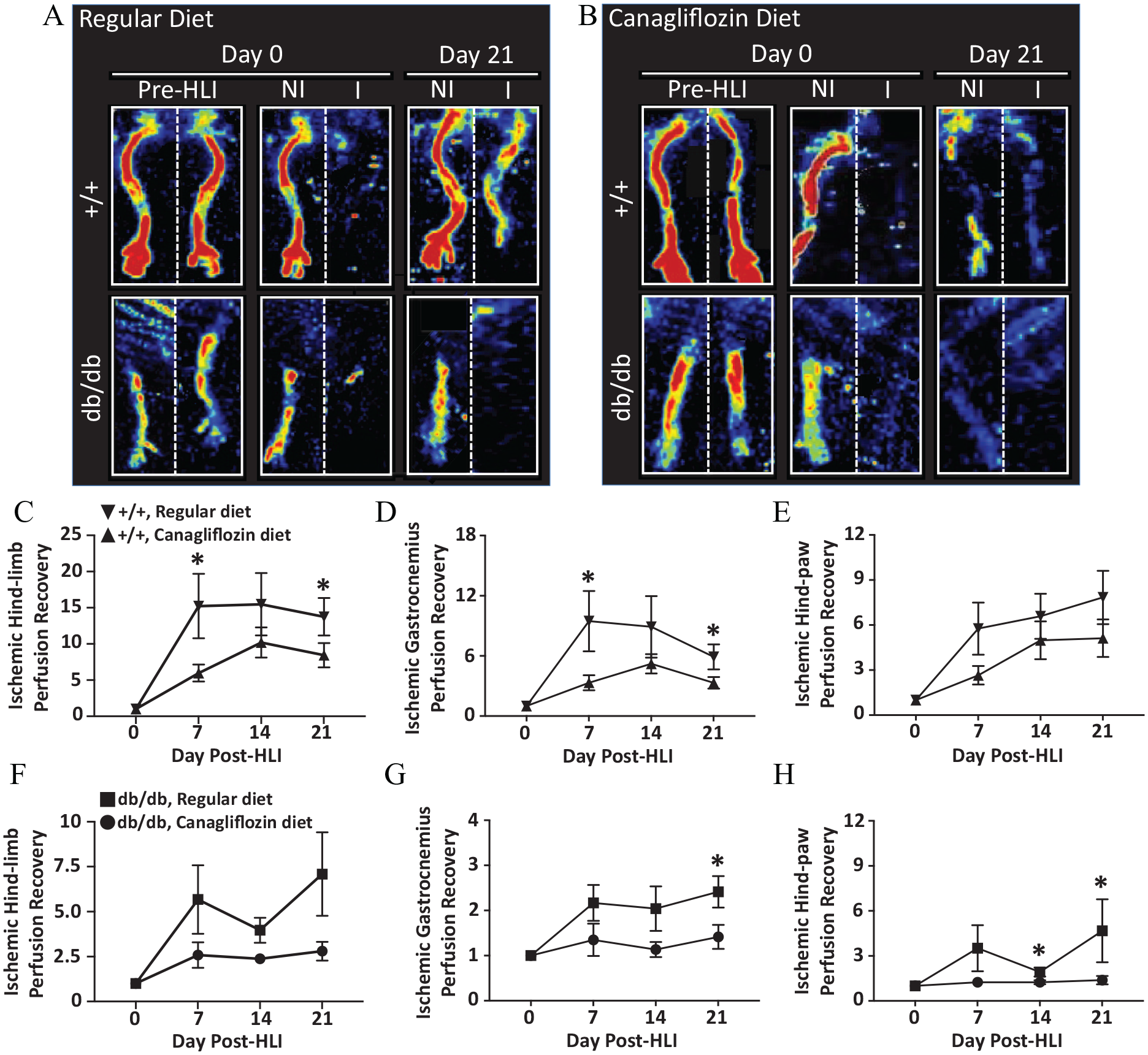
Hind-limb Doppler perfusion. (A and B) Representative Doppler perfusion images of ischemic and non-ischemic hind-limbs of +/+ and db/db mice maintained on either a regular diet or a canagliflozin-containing diet. The weekly relative change in perfusion of the whole hind-limb (C), hind-limb gastrocnemius region (D), and distal hind-paw region (E) in +/+ mice maintained on either a regular or canagliflozin-containing diet over a 21-day period. Similarly, the weekly relative change in perfusion of the whole hind-limb (F), gastrocnemius region (G), and distal hind-paw region (H) in db/db mice maintained on either a regular or canagliflozin-containing diet over a 21-day period. *p < 0.05.
Discussion
Our study is the first to demonstrate that canagliflozin appears to impede recovery of ischemic hind-limb tissue in diabetic mice. Following canagliflozin administration, we observed that both db/db and +/+ had worse limb function and appearance scores, smaller relative gastrocnemius muscle fiber sizes, decreased microvessel density, and decreased hind-limb Doppler perfusion. Most notably, these effects were particularly more pronounced in db/db mice that received canagliflozin.
Individuals with diabetes have an increased lifetime risk of lower extremity amputations.25, 26 In fact, it is estimated that somewhere in the world, an amputation is performed every 30 seconds in an individual with diabetes. 27 The risk of amputation is even more elevated in individuals with diabetes who have advanced peripheral artery disease (PAD).26,28 Individuals with diabetes who undergo amputation have higher disease-related morbidity and mortality, 25 and significantly reduced quality of life. 29 It is therefore critical to identify potential offending factors that may increase this in individuals with diabetes who are already at high risk of this outcome.
The CANVAS program integrated data from two trials involving > 10,000 participants with type 2 diabetes and high cardiovascular risk.4,7 Participants were randomized to either canagliflozin or placebo, and composite endpoints of death from cardiovascular causes, nonfatal myocardial infarction, and nonfatal stroke were evaluated over a mean follow-up of 188 weeks. 4 The study observed a 14.6% reduction (HR 0.86; 95% CI: 0.75–0.97) in the primary cardiovascular composite endpoint, and a 27% reduction in albuminuria (HR 0.73; 95% CI: 0.67–0.79) in individuals randomized to canagliflozin. However, there was an unexpected increase in the risk of lower extremity amputations (6.3 vs 3.4 participants per 1000 patient-years; HR 1.97; 95% CI: 1.41–2.75) with use of canagliflozin. The majority of amputations were observed to be at the level of the toe or metatarsal, but 29% of the amputations were classified as major amputations (above the ankle).4,9 The absolute risk of amputation was highest among patients who had a history of amputations (HR 20.9; 95% CI: 14.2–30.8) or a history of PAD (HR 3.1; 95% CI: 2.2–4.5). These findings strongly suggest that individuals with diabetes and underlying peripheral arterial malperfusion who received canagliflozin were at higher risk of lower extremity amputation.
The CREDENCE study is a subsequent trial that also randomized individuals with type 2 diabetes to canagliflozin to evaluate renal outcomes. 30 The trial was stopped early after a planned interim analysis observed a significant 34% decrease in renal composite endpoints (HR 0.66; 95% CI: 0.53–0.81) in individuals randomized to canagliflozin. 31 By this point the trial had enrolled 4401 participants with a median follow-up of 135.5 weeks. Interestingly, the trial observed no difference in amputation rates between individuals randomized to canagliflozin and placebo (5.4% vs 5.2%, respectively). 31 It is argued that the differences observed in amputation outcomes between CREDENCE and CANVAS may be due to differences in the study follow-up periods, size of the study population, and differences in enrolled patient demographics.8, 31 Additionally, the CREDENCE study did not screen or stratify participants by incidence or severity of PAD, and also had a protocol to stop the study drug in trial participants who developed conditions considered to be risk factors for potential amputation. 30
In our study we used db/db mice, which in adulthood develop the majority of the clinical features attributed to metabolic syndrome and type 2 diabetes.20,21 Like humans with type 2 diabetes, adult db/db mice develop obesity, elevated total plasma cholesterol, high serum low-density lipoprotein (LDL), and arterial atherosclerosis. 20 Previous reports demonstrate that db/db mice have delayed healing following HLI and reduced angiogenesis.32–34 Similarly, we observed that compared to +/+ mice, db/db mice demonstrated reduced limb function, worse appearance, and reduced perfusion – particularly in the most distal ischemic hind-paw tissue. Importantly, we also observed that oral administration of canagliflozin led to impaired ischemic hind-limb recovery in both +/+ and db/db mice. This effect was especially more pronounced in db/db mice, which demonstrated significantly reduced microvessel density and hind-limb perfusion. Naturally decreased angiogenesis and perfusion can explain why these mice also have worse appearance and function scores, as well as decreased gastrocnemius muscle fiber sizes.
A prior study also evaluated the effect of oral canagliflozin administration on hind-limb recovery following unilateral HLI. 19 The study evaluated the effect of a short 28-day course of canagliflozin upon induction of HLI in NOD/SCID (nonobese diabetic/severe combined immunodeficiency) mice. The study endpoints were hind-limb perfusion and microvessel density. This study observed that compared to vehicle control, canagliflozin actually increased hind-limb perfusion 28 days post-HLI, but no differences were observed on days 7, 14, and 21. Our study design and execution differs in several ways from this report, which explains the profound differences in results. First, we believe our db/db mouse model is more representative of human type 2 diabetes,20, 32 whereas NOD/SCID mice are more representative of autoimmune manifestations of type 1 diabetes. 35 NOD/SCID mice do not develop elevated total cholesterol, elevated serum triglycerides, and obesity, even when maintained on a high-fat chow diet. 36 Second, unlike db/db mice, NOD/SCID mice have a defective immune response, which blunts ischemia-induced inflammation and, as a result, angiogenesis.32, 33 Third, NOD/SCID mice have much shorter lifespans compared to db/db mice, which makes db/db mice more suitable for evaluation of chronic medication courses that are more representative for human application. Accordingly, our study pre-conditioned db/db mice with canagliflozin for 8 weeks prior to induction of HLI. We also included a vital +/+ control to help establish the effects of canagliflozin on the db/db pre-clinical diabetes model.
One of the hallmarks of diabetes is endothelial cell (EC) dysfunction, leading to macro- and microvascular complications such as retinopathy, coronary artery disease, and PAD.37–39 A recent study observed that unlike other SGLT2 inhibitors, canagliflozin can significantly decrease EC proliferation and tubule formation. 11 Canagliflozin appeared to halt DNA synthesis, causing cell cycle arrest, and a dramatic decrease in cyclin A expression in murine and human ECs. 11 These anti-proliferative effects suggest that canagliflozin has pleiotropic effects beyond its intended effects on SGLT2. Additionally, effects may be critical to the diminished response observed following HLI in both +/+ and db/db mice.
Study limitations
We acknowledge some limitations exist in our study. Like prior studies, we did not provide drug vehicle and sham surgical controls since the drug carrier and the femoral artery ligation surgical technique are traditionally thought to have minimal confounding effects.14,19 On the other hand, +/+ mice and mice that were maintained on a regular diet prior to HLI, provide essential controls in our study for contrasting outcomes to db/db mice and mice that were maintained on a canagliflozin-containing diet. Like other investigators, we also chose to administer canagliflozin via the chow diet to help ensure consumption of the drug. 14 We acknowledge that variations in eating habits may have affected precise drug dosing, but we expect that these variations are minor based on prior studies characterizing the eating habits of +/+ and db/db mice.40,41 We also observed little variance in study endpoints in both db/db and +/+ mice, and no difference in the weights of db/db mice maintained on either a regular or canagliflozin diet (online supplemental Figure 2). Lastly, we chose to administer to mice a daily canagliflozin dose that is several orders less than the daily clinical human dose.6, 9 Future studies may be needed to fully delineate the dose effects of canagliflozin and other SGLT2 inhibitors on hind-limb perfusion and tissue recovery.
Conclusion
We observed that canagliflozin significantly decreased perfusion, angiogenesis, and ischemic hind-limb recovery in db/db mice. Our study findings belong to a string of studies implicating canagliflozin to decreased EC function and peripheral vascular health, and to an increased risk of amputation. Additional studies are needed to evaluate potential off-target effects of canagliflozin in individuals with diabetes.
Supplemental Material
Supplemental_Figure_1 – Supplemental material for Canagliflozin impedes ischemic hind-limb recovery in the setting of diabetes
Supplemental material, Supplemental_Figure_1 for Canagliflozin impedes ischemic hind-limb recovery in the setting of diabetes by Margaret Nalugo, Nikolai Harroun, Chenglong Li, Larisa Belaygorod, Clay F Semenkovich and Mohamed A Zayed in Vascular Medicine
Supplemental Material
Supplemental_Figure_2 – Supplemental material for Canagliflozin impedes ischemic hind-limb recovery in the setting of diabetes
Supplemental material, Supplemental_Figure_2 for Canagliflozin impedes ischemic hind-limb recovery in the setting of diabetes by Margaret Nalugo, Nikolai Harroun, Chenglong Li, Larisa Belaygorod, Clay F Semenkovich and Mohamed A Zayed in Vascular Medicine
Footnotes
Acknowledgements
The authors thank Dr Xiaohua Jin for her critical suggestions and critiques.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by grants from the Vascular Cures Foundation Wylie Scholar Award (MAZ), American Surgical Association Research Fellowship Award (MAZ), Society for Vascular Surgery Foundation Research Investigator Award (MAZ), Washington University School of Medicine Diabetes Research Center: NIH/NIDDK P30 DK020589 (MAZ), NIH/NHLBI K08 HL132060 (MAZ), and NIH/NIDDK R01 DK101392 (CFS).
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
