Abstract
Background:
Venoarterial extracorporeal membrane oxygenation support is a well-established tool in the care of severe refractory cardiac and respiratory failure. The application of this support may serve as a bridge to transplant, recovery or to implantation of a ventricular assist device. Venoarterial extracorporeal membrane oxygenation support can be administered through an open surgical access via the common femoral or axillary artery or a percutaneous approach using Seldinger technique. Both techniques may obstruct the blood flow to the lower limb and may cause a significant ischemia with possible limb loss. Malperfusion of the distal limb can be avoided using an ipsilateral distal limb perfusion, which may be established by adding a single-lumen catheter during venoarterial extracorporeal membrane oxygenation treatment to overcome the obstruction. The aim of this study is to distinguish the presence or absence of a distal limb perfusion regarding the incidence of distal limb ischemia. Furthermore, expected risk factors of open and percutaneous femoral venoarterial extracorporeal membrane oxygenation installation were evaluated for the development of distal limb ischemia.
Methods:
Between January 2012 and September 2015, 489 patients received venoarterial extracorporeal membrane oxygenation support at our institution. In total, 307 patients (204 male, 103 female) with femoral cannulation were included in the analysis. The cohort was distinguished by the presence (group A; n = 237) or absence (group B; n = 70) of a distal limb perfusion during peripheral venoarterial extracorporeal membrane oxygenation treatment. Furthermore, a risk factor analysis for the development of distal limb ischemia was performed.
Results:
The main indications for venoarterial extracorporeal membrane oxygenation therapy were a low cardiac output syndrome (LCOS) (53%) and failed weaning of extracorporeal circulation (23%). A total of 23 patients (7.49%) under venoarterial extracorporeal membrane oxygenation support developed severe distal limb malperfusion (3.38% in group A vs 21.42% in group B). Preemptive installation of distal limb perfusion extended the intervention-free intervals to 7.8 ± 19.3 days in group A and 6.3 ± 12.5 in group B. A missing distal limb perfusion (p = 0.001) was identified as a main risk factor for critical limb ischemia. Other comorbidities such as arterial occlusion disease (p = 0.738) were not statistically significantly associated. Surgical intervention due to vascular complications after extracorporeal membrane oxygenation explantation was needed in 14 cases (4.22% in group A and 5.71% in group B).
Conclusion:
We were able to identify the absence of distal limb perfusion as an independent risk factor for the development of critical distal limb ischemia during femoral venoarterial extracorporeal membrane oxygenation treatment. The application of a distal limb perfusion should be considered as a mandatory approach in the context of femoral venoarterial extracorporeal membrane oxygenation treatment regardless of the implantation technique.
Keywords
Introduction
Severe cardiac and pulmonary failure is associated with increased morbidity and mortality in critically ill patients. Venoarterial extracorporeal membrane oxygenation (VA-ECMO), via percutaneous cannulation or after open exposition of the femoral vessels, may be needed in patients with refractory cardiopulmonary failure. Nondurable mechanical circulatory support for the treatment of acute heart failure continues to rapidly evolve and has been ascribed an increasing role for acute circulatory support. 1 VA-ECMO circulation can be implemented as a bridge to recovery, a bridge to durable ventricular assist device or a bridge to transplant.2,3
VA-ECMO support can be installed in awake or intubated patients. 4 However, this procedure may lead to critical limb malperfusion caused by the occlusion of the femoral artery by the large-bore arterial cannula. If not treated instantly, the affected leg may have to be amputated.5,6
There are large discrepancies regarding the published incidence of ECMO-induced limb ischemia. The exact incidence is not routinely recorded in the Extracorporeal Life Support Organization (ELSO) Registry database. Small studies have reported an incidence of 30% to 50%,7–9 while one meta-analysis described critical limb ischemia as a result of VA-ECMO treatment in 10% of a total of 1,200 patients. 10 To prevent critical limb ischemia, an additional small-caliber cannula can be placed distal of the arterial VA-ECMO cannula to ensure distal limb perfusion (DLP). 11 Another interesting technique was published by Spurlock et al. 12 in 2012. They cannulated and perfused the ipsilateral posterior tibial artery to prevent limb ischemia during VA-ECMO treatment.
The aim of this study is to distinguish the presence or absence of a DLP regarding the incidence of distal limb ischemia. Furthermore, expected risk factors of open and percutaneous femoral VA-ECMO installation will be evaluated for the development of distal limb ischemia. Is there a preventive effect of the establishment of a DLP?
The benefits of a DLP to prevent ischemia caused by a VA-ECMO treatment have not been studied so far and that the evaluation of possible risk factors is still underreported.
Materials and methods
Patient cohort
Patients who received a VA-ECMO from January 2012 to September 2015 by the Department of Cardiothoracic, Transplantation and Vascular Surgery at Hannover Medical School (Germany) were included into the study. The institutional ethics committee waived the need for patient consent for this study. All data were retrieved by retrospective review of patient’s records. During the observation period, 489 patients received a VA-ECMO treatment in our institution. We included patients on VV-ECMO (veno-venous) treatment, who required switching to a V-V-A (veno-veno-arterial) strategy in the later clinical course.
Main including criteria were the implementation of an ECMO into the common femoral artery (CFA) and the common iliac vein. The prevalence of distal limb ischemia after open or percutaneous installation was recorded dependent on the existence of a DLP. The patients were followed until the hospital discharge or intra-hospital death. Patients who received further vascular surgery in our department due to vascular complications were considered till September 2016. Main exclusion criteria were as follows: isolated intraoperative VA-ECMO treatment, patient death during the first 12 hours of treatment, and duration of ECMO support for less than 12 hours as well as central or subclavian arterial cannulation. Infants with a cannulation of the carotid artery were neglected. In total, 307 patients fulfilled the including criteria.
The included patients on VA-ECMO support were divided into two groups. Patients with a DLP during VA-ECMO implantation formed group A. Patients under VA-ECMO treatment without a DLP formed group B.
The decision for ECMO implantation was made by consensus of cardiac surgeons, intensivists, and cardiologists. Our department is a tertiary referral center; therefore, 27 patients underwent VA-ECMO installation at external hospitals prior to transfer. All implantations were performed by surgeons of our department.
Medical records including radiologic imaging were retrospectively examined for the following variables: age, gender, body mass index (BMI), presence or absence of peripheral artery disease, open- versus percutaneous implantation, cannula size (arterial/venous), presence or absence of malperfusion or secondary intervention, time till intervention, duration of ECMO treatment, indication for ECMO support, and overall mortality. The patient cohort was further distinguished by presence of absence of general anesthesia.
In our retrospective single-center study, we evaluated all patients who received a VA-ECMO treatment from January 2012 to September 2015 by the Department of Cardiothoracic, Transplantation and Vascular Surgery at Hannover Medical School (Germany). Over these 45 months, 489 patients received a VA-ECMO mode treatment. During the study period, 307 patients underwent VA-ECMO treatment via CFA and fulfilled the including criteria.
Implantation techniques
Percutaneous VA-ECMO implantation using Seldinger’s approach
The following algorithm describes our local standard operating procedure. After a single bolus of 5,000 IU unfractionated heparin, two guidewires are inserted percutaneously into the CFA and the common femoral vein. In addition, one wire is placed in the CFA in distal direction to establish DLP. In complicated cases, ultrasound was used for placing the guidewires.
This procedure permits a prompt insertion of the arterial and venous cannulas in case of emergency and prevents an occlusion of the distal CFA following proximal cannulation (Figure 1).
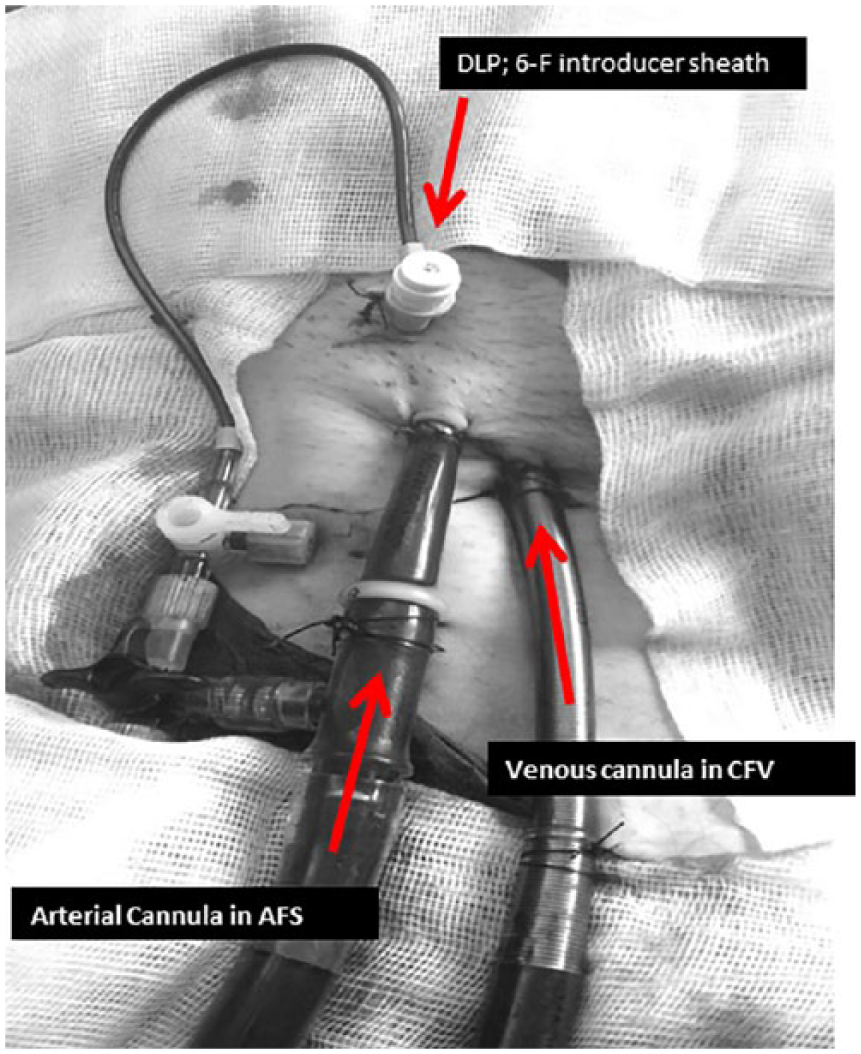
Percutaneously established arterial and venous cannulas in the common femoral artery and in the common femoral vein with additional 6-F introducer sheath placed in the CFA in distal direction.
The right positioning of the cannula was controlled by X-ray briefly after the VA-ECMO implantation. The targeted tip of the venous outflow cannula lay 1 cm above the diaphragm, in the right atrium.
Open surgical approach
The surgical access for arterial cannulation allows the complete visualization of the artery. Due to this reason, an adequate size of the arterial cannula can be selected. Arterial and venous cannulas were fixated using 5-0 Prolene purse strings.
DLP
For the DLP, a 6-F introducer sheath (Avanti®+; Cordis Corp., Miami, FL, USA) as a DLP catheter was inserted via Seldinger technique using the previously placed distal guidewire.
Explantation technique
The explantation of percutaneously inserted ECMO systems is generally performed in a standardized manner. The venous puncture is sealed with a 0.0 skin suture followed by a short period of manual compression. After the arterial cannula is being removed, compression is applied by a mechanical femoral compression system (FemoStop II Plus; Radi Medical Systems, Uppsala, Sweden) according to the manufacturer’s recommendations. Puncture position and ankle pulses should be examined every 60 minutes at least. As an exception, percutaneous inserted ECMO systems may be explanted via open access in the operation theater. In case of a vessel stenosis, atherosclerotic arterial wall conditions, or distal pulselessness, a reconstructive procedure was performed.
Materials
Arterial cannulation was performed using a Novaport® (Novalung GmbH, Heilbronn, Germany), a Bio-Medicus (Medtronic, Minneapolis, MN, USA) or a Fem-Flex II (Edwards Lifesciences, Irvine, CA, USA).
For the venous outflow, we used a FemTrak Cannula (Edwards Lifesciences).
The ECMO circuit consisted of Cardiohelp heart–lung support (HLS) system or a Maquet Rotaflow permanent life support (PLS) system with a centrifugal pump and the Quadrox membrane oxygenator (Maquet Cardiopulmonary AG, Hirrlingen, Germany).
A baseline-activated clotting time (ACT) was routinely measured prior the implantation procedure and for monitoring purposes. For further anticoagulation, continuous infusion of unfractionated heparin (15.000 IU/50 mL, according to patient weight) was started to reach a targeted ACT of 140-160 seconds during the first 24 hours after ECMO installation. After 24 hours, the targeted ACT lay between 160 and 180 seconds. Sufficient perfusion to the lower extremity was regularly controlled through Doppler sonography. Pulse of the tibial posterior artery and the dorsalis pedis artery was detected and marked using a skin pen. This procedure was performed every 2 hours. A continuous blood flow measurement of the DLP was not available.
Statistical analyses
Data were collected and analyzed using SPSS 20.0 (SPSS, Chicago, IL, USA). Categorical and continuous variables were summarized as percentages and means ± standard deviation and compared using Mann–Whitney U tests. Differences in categorical variables were assessed using X2 test (Fisher’s exact). statistical significance was evaluated with chi-square tests (Pearson chi-square, continuity correction, likelihood ratio, Fisher’s exact test, linear-by-linear association) and was assumed with a p of ⩽0.05.
Results
The mean age of all included patients was 49 ± 17 years in male and 54 ± 17 years in female patients. In total, 204 patients (66%) were male. The baseline demographics are shown in Table 1.
Patient demographics.
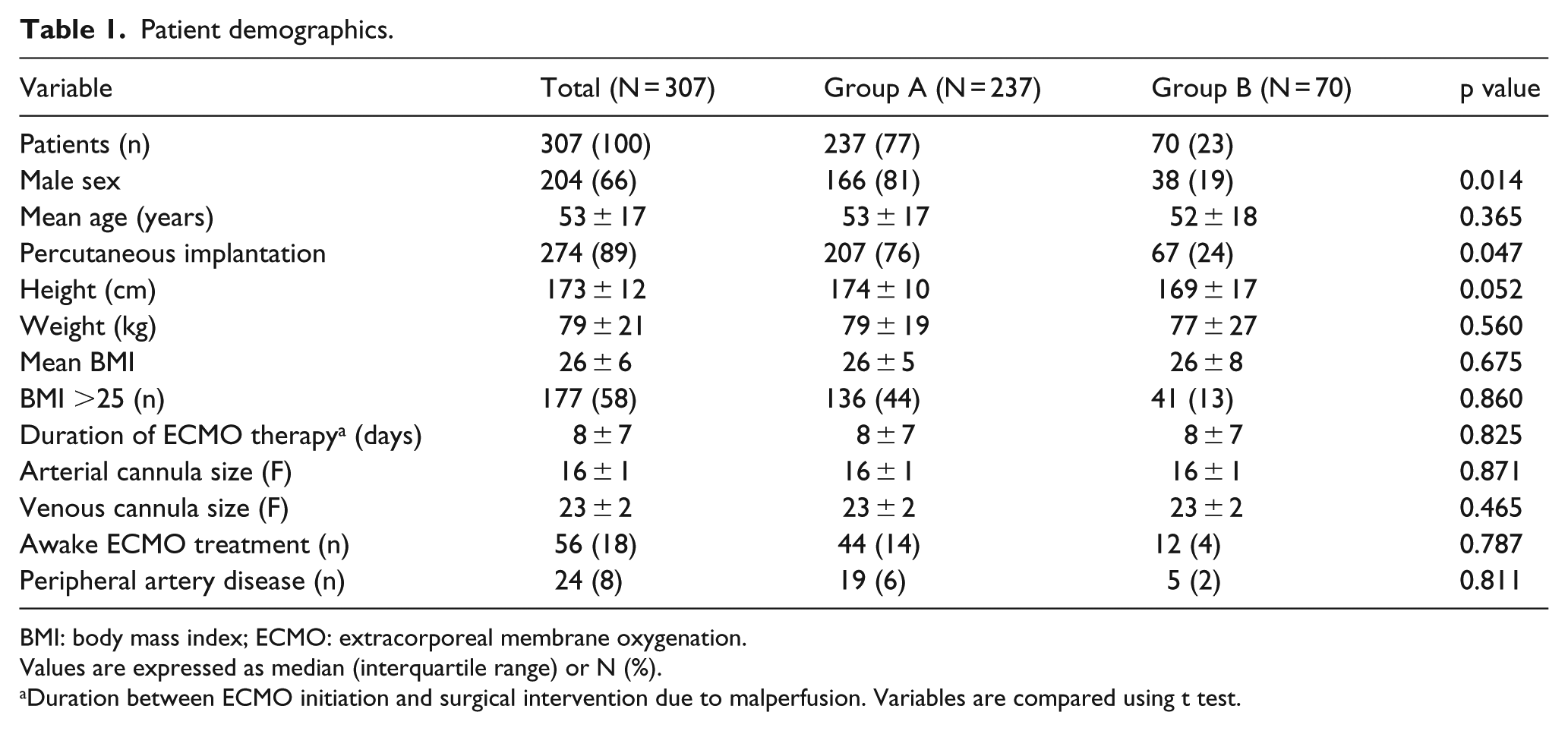
BMI: body mass index; ECMO: extracorporeal membrane oxygenation.
Values are expressed as median (interquartile range) or N (%).
Duration between ECMO initiation and surgical intervention due to malperfusion. Variables are compared using t test.
A total of 237 patients (77%) received a DLP during open or percutaneous VA-ECMO implantation and formed group A. At initiation of ECMO treatment, the insertion of the distal perfusion cannula was unfortunately not possible in 70 patients (23%) due to time constraints in case of emergency or due to anatomical reasons. These patients formed group B. Percutaneous cannulation was utilized in 207 patients (76%) of group A and 67 patients (24%) of group B (p = 0.047). Three patients (9%) had no DLP in case of open VA-ECMO implantation. The duration of VA-ECMO treatment was 8 ± 7 days for both groups.
The BMI of patients in group A or B was comparable (26 ± 5 group A; 26 ± 8 group B). Same arterial (16 ± 1 F) and venous (23 ± 2 F) cannula sizes were identified in both cohorts. Awake ECMO treatment was performed in 44 patients (19%) of group A and 12 patients (17%) of group B. Only marginal differences between group A and group B considering documented peripheral artery disease were seen (8% vs 7%, respectively; p = 0.811).
Indications for initial ECMO support included the following: failed weaning from extracorporeal circulation after open-heart surgery, ECMO support in association with a planned lung transplantation, LCOS, sepsis, severe pulmonary hypertension, pulmonary embolism, acute respiratory distress syndrome (ADRS), sepsis and intoxications.
The majority of (162 (53%)) patients required maintenance VA-ECMO therapy due to LCOS (group A: n = 129 (53%); group B: n = 33 (47%)). Pulmonary hypertension was the indication for ECMO implantation in 9 patients as well as massive pulmonary embolism in 10 patients. ECMO was applied for postcardiotomy shock in 71 patients (23%). Following lung transplantation, 39 patients needed ECMO support as well as 2 patients suffered from sepsis. ADRS was the indication in 11 patients and 3 patients suffered from severe intoxications prior to extracorporeal support. Indications for VA-ECMO treatment are shown in Table 2.
Indication for VA-ECMO treatment.
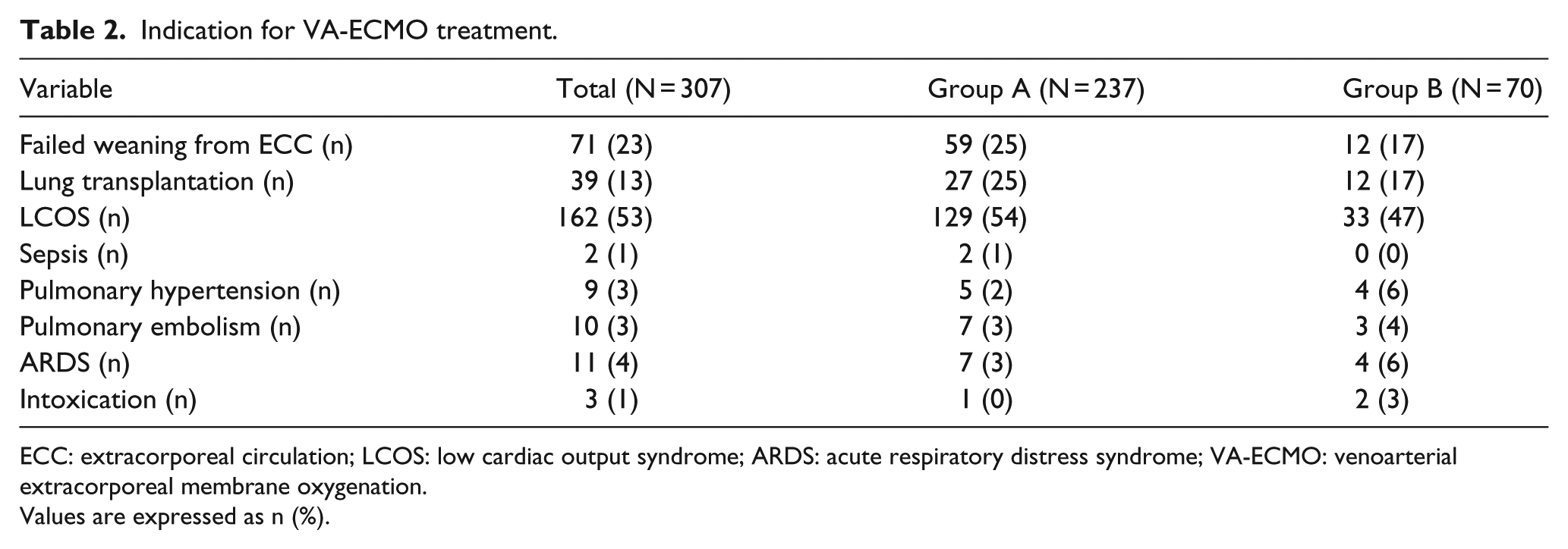
ECC: extracorporeal circulation; LCOS: low cardiac output syndrome; ARDS: acute respiratory distress syndrome; VA-ECMO: venoarterial extracorporeal membrane oxygenation.
Values are expressed as n (%).
In all, 15 patients received a DLP with a short delay and were included in group A. Indications for delayed establishments of a DLP have been mild clinical signs of ipsilateral malperfusion in 13 of 15 cases like a marbling and coldness of the distal limb. All patients received a DLP during the first hour after implantation.
Primary outcomes were defined as a VA-ECMO–induced malperfusion of the ipsilateral distal leg with the need for surgical treatment. Findings were distinguished according to the existence (group A) and the absence of a DLP (group B).
Clinical manifestations of malperfusion of the ipsilateral distal leg were observed in 23 of 307 (7.49%) of all patients (p < 0.001). Furthermore, a significant difference of a malperfusion rate was detected between both groups.
About 21.43% (15 of 70) of patients within group B versus 3.38% (8 of 237) in group A needed surgical intervention due to critical limb ischemia (p < 0.001) (Table 3). In all these cases, an ipsilateral decannulation and contralateral establishment of the VA-ECMO including a DLP were performed under direct visualization.
Selective cohort outcome.
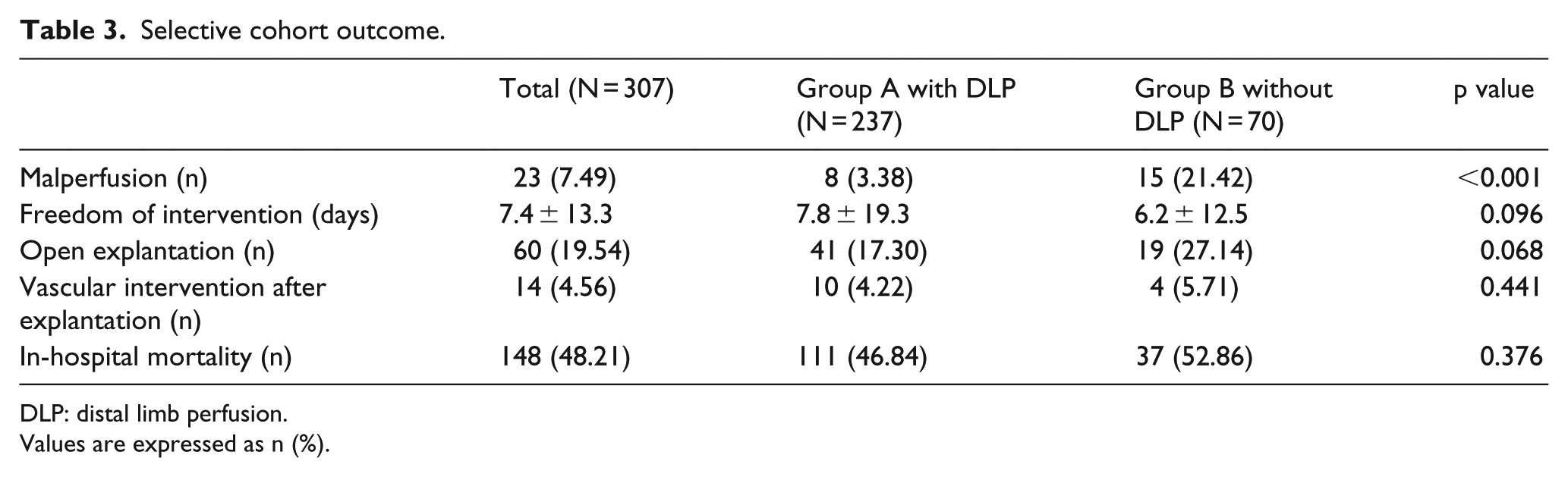
DLP: distal limb perfusion.
Values are expressed as n (%).
Overall intra-hospital mortality after initiation of VA-ECMO support was 48.2% (148 of 307). The mortality rate was 46.84% versus 52.86% for group A and group B, respectively (p = 0.376). One morbidly obese patient with a BMI over >35 and constrictive pericarditis on awake VA-ECMO treatment without DLP died after dislocation of the arterial cannula.
Freedom of intervention time is defined as the period between implantation and explantation or duration of ECMO support until surgical intervention. Freedom of intervention comparing both treatment groups is depicted in Figure 2.
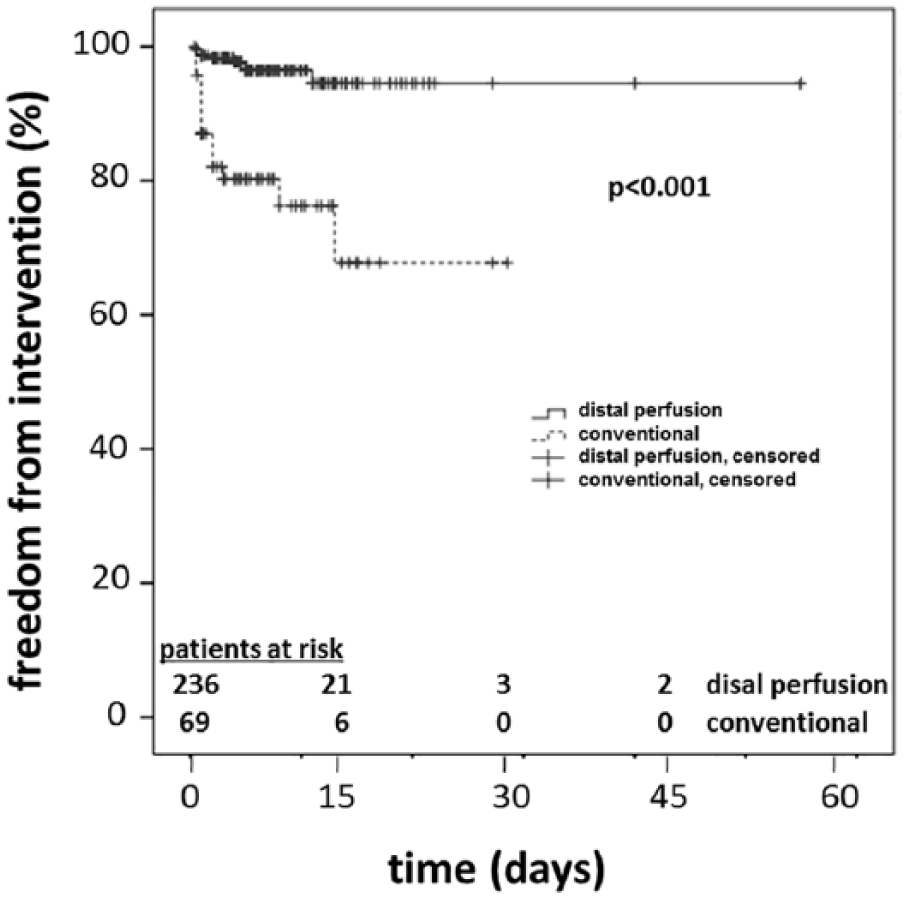
Freedom of intervention curve: patients with established distal limb perfusion and convention without perfusion.
Surgical intervention was needed after ended VA-ECMO treatment in a total of 14 patients (group A: 4.22%; group B: 5.71%). Within group A, surgery was indicated because of four cases with refractory bleeding, four cases with distal limb ischemia, and two cases with lymph fistulas. Within group B, surgery was indicated in three patients with clinical signs of critical limb ischemia and in one patient with a lymph fistula. Malperfusion was not observed in 33 patients, who received an ECMO implantation via open-access surgery.
Open-access explantation was performed in all cases following open-access implantation (Table 3). Open VA-ECMO explantation after ended treatment of percutaneously implanted cannulas was considered based upon the patients’ individual risk profile for the development of further critical limb ischemia. In this cohort, 41 patients (17.30%) on ECMO treatment with DLP had an elective open explantation without any signs of malperfusion. Of these 41 patients, 17 underwent a vascular reconstruction of an injured superficial femoral artery. In all cases, decannulation was performed under direct visualization after cutdown on the groin. Purse-string sutures were placed for hemostasis after extraction of the arterial and venous cannulas. Risk factor analysis of the complete cohort is presented in Table 4. Four patients under VV-ECMO treatment were converted to a VVA-ECMO regime. All patients initially received a DLP. There was no case of distal malperfusion documented in these patients.
Risk factor analysis—malperfusion: binary logistic regression analysis.
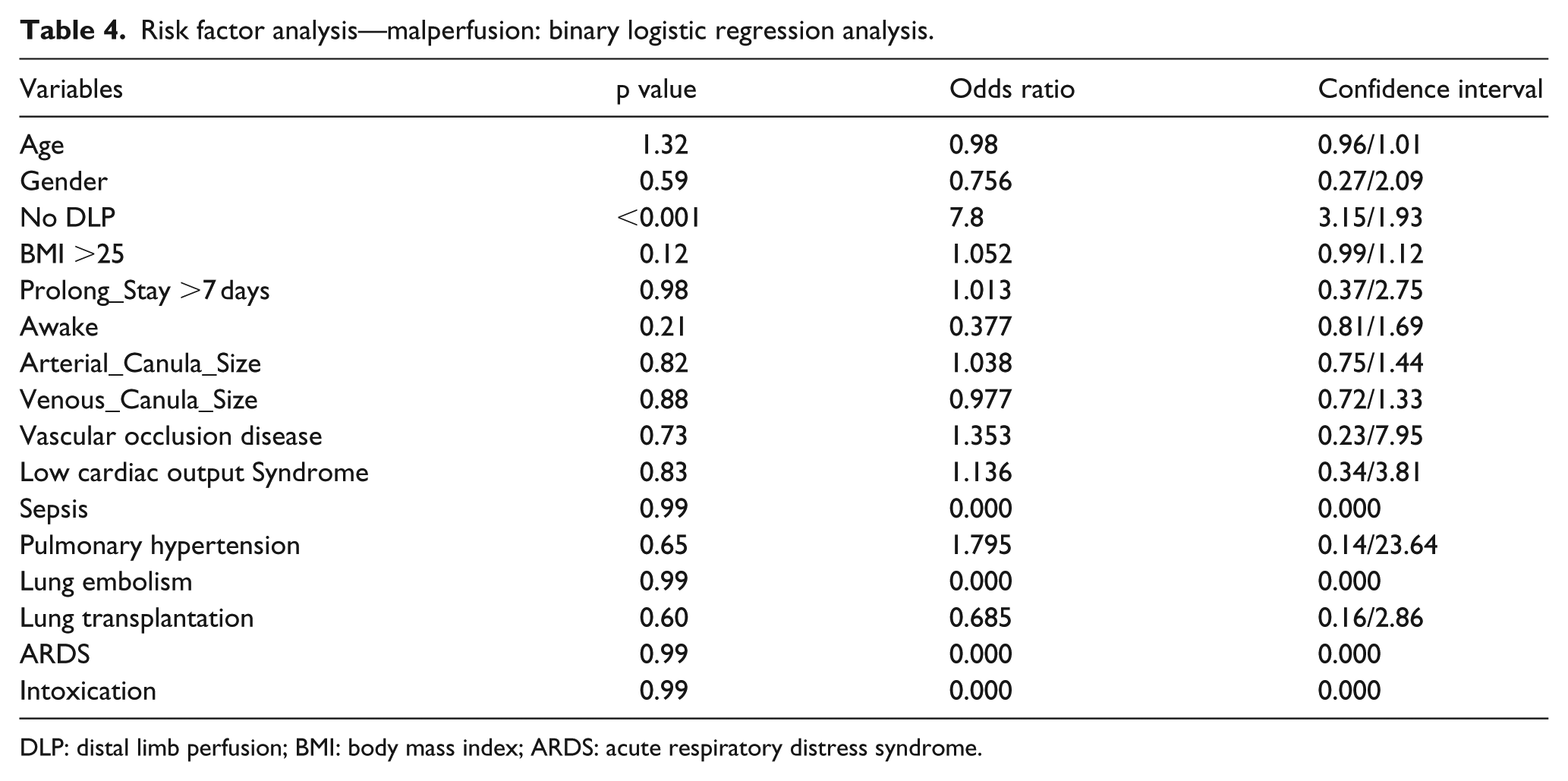
DLP: distal limb perfusion; BMI: body mass index; ARDS: acute respiratory distress syndrome.
Discussion
Distal malperfusion after VA-ECMO treatment is a feared side effect of this evolving technique. The incidence of distal malperfusion during femoral VA-ECMO treatment is still unknown. This retrospective study represents, to our knowledge, the largest single-center case series following up on the development of distal malperfusion in patients after peripheral VA-ECMO treatment. In clinical practice, a prompt decision for a VA-ECMO establishment has to be found in critically ill patients and the first priority is to treat the circulatory arrest. Due to this reason, there may not be time to perform a preceding duplex ultrasonography of the femoral arteries. At present, it is unclear in which patients the additional installation of a DLP should be considered and what the benefits are for the individual patient.
Existing meta-analysis of 12 studies including nearly 1,800 patients reports an incidence of limb ischemia after VA-ECMO treatment of 10%. 10 In our cohort, we found a comparatively lower ischemia rate of 7.48%. The variety of previously published rates of distal limb ischemia may be attributed to a significantly lower number of included patients and the isolated analysis of percutaneously implanted ECMO systems. 10
In contrast to earlier studies, we found a significant association between limb ischemia and the absence of a DLP in all ages. 13 We identified the absence of DLP as an independent risk factor for the development of distal limb ischemia (p < 0.001). Surprisingly, general cardiovascular risk factors like the presence of peripheral arterial occlusion disease, an enhanced BMI, or used cannula size could not be statistically linked to the development of distal malperfusion in our patients. This observation may be associated with a selection bias since obese patients and patients with an increased cardiovascular risk may be more likely to receive a preemptive DLP in daily routine.
We identified the benefit of a DLP with a prolonged freedom of intervention time (+1.5 days). However, the discrete deviation of the documented mortality rate (53% in patients without DLP vs 47% in patients with DLP) may not be attributed to ischemic complications rather than to the underlying disease.
In summary, DLP during peripheral VA-ECMO treatment should be considered as a mandatory approach to minimize the ischemia-induced complications and to avoid the additional morbidity in critically ill patients. Future studies should evaluate precise distal flow measurements and identify relevant diagnostic markers of critical limb ischemia after VA-ECMO installation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
