Abstract
Chronic skeletal muscle ischemia protects the ischemic heart by preserving coronary flow and inducing arterioangiogenesis. We sought to determine the effect and the underlying molecular mechanisms of preconditioning (PreC) and postconditioning (PostC), applied in a model of chronic skeletal muscle ischemia. Male rabbits were divided into 3 series. In each series, the animals were subjected either to severe hind limb (HL) ischemia, by excision of the femoral artery, or to sham operation (SHO). After 4 weeks, all the animals underwent 30 minutes of regional heart ischemia and 3 hours reperfusion. The animals of the first series received no further intervention (HL and SHO groups), those of the second series underwent PreC (HL + PreC and SHO + PreC), and of the third series PostC (HL + PostC and SHO + PostC). Infarct size (I) and risk zones (R) were determined, and their ratio was calculated in percentage. Three additional series of experiments were performed with respective interventions up to the 10th minute of reperfusion, where sample tissue was obtained for assessment of protein kinase B (Akt), endothelial nitric oxide synthase (eNOS), glycogen synthase kinase 3β (GSK3β), p44/42, signal transducer and activator of transcription (STAT) 3, and STAT5. All groups demonstrated significantly smaller percentage of I/R compared with the SHO group (HL: 14.4% ± 3.7%, HL + PreC: 13.1% ± 1.0%, SHO + PreC: 21.3% ± 1.6%, HL + PostC: 18.0% ± 1.1%, and SHO + PostC: 24.3% ± 1.7%, P < .05 vs 35.7% ± 4.4% in SHO). The PreC and PostC did not further reduce the infarct size in HL groups. The Akt, eNOS, GSK3β, p44/42, and STAT3 were activated in all PreC or PostC groups regardless of the infarct size reduction. The STAT5 was activated only in the HL groups compared with the SHO groups. In conclusion, chronic skeletal muscle ischemia results in effective cardioprotection, which is not further enhanced with application of PreC or PostC. The Akt, eNOS, GSK3β, p44/42, and STAT3 may only be considered as indicators of the intracellular changes taking place during protection. Activation of STAT5 is possibly the end effector, which is responsible for infarct size reduction provided by chronic skeletal muscle ischemia.
Introduction
Ischemic conditioning is the protective mechanism, by which brief ischemia renders the heart resistant to potentially lethal ischemia and reperfusion. The 2 main mechanisms of conditioning are ischemic preconditioning (PreC) 1 and ischemic postconditioning (PostC), 2 both preventing the development of necrosis due to reperfusion injury. 3 Although effective, PreC and PostC necessitate invasive treatment applied directly to the myocardium rendering these interventions not practical or safe in everyday clinical practice. 4 An interesting concept to avoid these limitations is the so-called remote ischemic conditioning (RIC), that is, the application of ischemia in a remote vascular bed, conferring protection to the heart 5 without additional manipulation on the heart itself. This can be performed before, during, or after the index ischemia, characterized as remote ischemic PreC, Per-, or PostC, respectively. 4 Indeed, experimental studies have reported myocardial protection provided by remote ischemia of other organs or vascular territories 6 –12 resulting in a decrease in myocardial infarct size, normalization of biomarkers, improvement in endothelial dysfunction, and reduction in arrhythmic events. Among other interventions, acute limb ischemia represents a remote PreC stimulus, which confers protection to the heart by reducing the final infarct size. 8,13 –15 Therefore, any short remote ischemia has emerged as a novel and easily applicable therapeutic strategy for myocardial protection in patients undergoing surgical 16 –18 or percutaneous myocardial revascularization 19 as well as in patients with acute myocardial infarction (AMI). 20,21
Despite the great body of evidence regarding remote conditioning by short-lived periods of ischemia, there are no sufficient data regarding cardioprotection afforded by remote chronic ischemia. Chronic skeletal muscle ischemia represents a more exaggerated model of ischemia, which contributes to the reduction in reperfusion injury, to the development of collateral circulation by promoting angiogenesis/arteriogenesis, 22 and to the preservation of the coronary flow reserve in rats. 23 However, it is unknown whether this mode of application may act as a conditioning analog. The aim of the present study was to examine the effect of chronic hind limb ischemia on infarct size, to elucidate whether the application of PreC or PostC confers additional cardioprotection, and to investigate the intracellular myocardial signal transduction pathways that are involved.
Materials and Methods
Animals
The initial animal experiment cohort comprised 98 New Zealand White male rabbits weighing 2.6 to 3.4 kg each. Animals were housed in individual cages, in a climate-controlled environment with an artificial 12-hour light–dark cycle. All animals received proper human care in compliance with the Principles of Laboratory Animal Care formulated by the National Society for Medical Research and the Guide for the Care and Use of Laboratory Animals prepared by the National Academy of Sciences and published by the National Institute of Health.
Induction of Femoral Ischemia
The rabbits were randomly divided into 2 groups. In 36, severe permanent ischemia of the right hind limb (HL) was surgically induced as described previously, 24 while the other 32 were sham operated (SHO).
In brief, animals were anesthetized with a mixture of ketamine (50 mg/kg) and xylazine (2.5 mg/kg). A longitudinal incision was performed extending inferiorly from the inguinal ligament to a point just proximal to the patella. The femoral artery was dissected free along its entire length; all branches of the femoral artery (including the inferior epigastric, deep femoral, lateral circumflex, and superficial epigastric arteries) were also dissected free. The popliteal and saphenous arteries distally were further dissected. The arteries mentioned earlier as well as the external iliac artery were proximally ligated (4.0 silk; Ethicon, Sommerville, New Jersey). Finally, the femoral artery was completely excised from its proximal origin to its distal bifurcation (to the saphenous and popliteal arteries). All animals received close postoperative monitoring. Throughout the study duration, analgesia was administered subcutaneously as required by evidence of discomfort. Prophylactic antibiotics were administered subcutaneously for a total of 5 days postoperatively, and no HL necrosis was observed. The SHO animals received identical anesthesia and antibiotic regimen but without induction of limb ischemia. All animals were left to recover under close supervision and proper housing and care.
Induction of Myocardial Ischemia and Reperfusion
Four weeks after the first surgical procedure, all animals were subjected to 30 minutes regional myocardial ischemia/reperfusion. Sodium pentobarbitone at a dose of 30 mg/kg was injected into a peripheral ear vein for anesthesia. Then, the animals were intubated and connected to a positive pressure respirator for small animals (MD Industries, Mobile, Alabama) for mechanical ventilation at a rate adjusted to maintain normal blood gases. Two polyethylene catheters were inserted into the left jugular vein for fluids or additional infusion of anesthesia and into the carotid artery for continuous blood pressure monitoring via a transducer attached to a multichannel recorder (Model 6000; Nihon-Kohden, Japan). More information regarding the experimental conditions has been described in detail previously. 25 A bipolar chest lead was used for continuous electrocardiographic monitoring. The chest was opened with a left thoracotomy in the fourth intercostal space, and after pericardiotomy, the beating heart was exposed. A 3-0 silk suture was passed around a prominent coronary artery. Ischemia was induced by pulling the thread through a small piece of soft tubing, which was firmly positioned against the coronary arterial wall with the aid of a small clamp. At the end of the ischemic period, the snare was opened, the artery refilled, and the myocardium reperfused. 25 After the end of reperfusion, the animals were euthanized (by injecting an overdose of anesthetics), and the hearts were excised.
Experimental Protocol
In the first series of experiments, 68 rabbits were subjected to a 30-minute transient ischemia of the myocardium, followed by 3-hour reperfusion and were randomized into 6 groups as follows (experimental protocol presented in detail in Figure 1): Group SHO (n = 11): SHO animals with no additional intervention. Group HL (n = 14): animals with HL ischemia with no additional intervention. Group SHO + PreC (n = 10): SHO animals with 2 cycles of 5-minute ischemia separated by 10-minute reperfusion prior to the sustained myocardial ischemia. Group HL + PreC (n = 11): animals with HL ischemia with 2 cycles of 5-minute ischemia separated by 10-minute reperfusion prior to the sustained myocardial ischemia. Group SHO + PostC (n = 11): SHO animals with 6 cycles of 10-second ischemia separated by 10-second reperfusion immediately after the end of the sustained myocardial ischemia. Group HL + PostC (n = 11): animals with HL ischemia with 6 cycles of 10-second ischemia separated by 10-second reperfusion immediately after the end of the sustained myocardial ischemia.

Schematic presentation of the study protocol.
In a second series of experiments, 30 animals (5 per group) were subjected to the same interventions up to the 10th minute of reperfusion, where tissue samples from the ischemic area of myocardium were collected, emerged in liquid nitrogen, and stored at −80°C for Western blot analysis of protein kinase B (Akt), endothelial nitric oxide synthase (eNOS), glycogen synthase kinase 3β (GSK3β), p44/42, signal transducer and activator of transcription STAT3, and STAT5.
Measurement of Risk Zone and Infarct Zone
All harvested hearts were mounted on a reperfusion apparatus and perfused (50 mm Hg) retrogradely via the aorta with normal saline (10 mL/min) at room temperature for 2 minutes for blood removal. After all residual blood had been removed from the coronary arteries, the coronary ligature was retightened at the same site, and 5 mL of green fluorescent microspheres (2-9 μm diameter; Duke Scientific Corp, Palo Alto, California) suspended in saline were infused over 5 minutes for the separation of normally perfused area from the area at risk. Hearts were frozen for 24 hours in −20°C and cut into 3-mm thick sections from the apex to the base. The slices were stained with triphenyltetrazolium chloride (TTC) at 37°C and immersed in formaldehyde. The TTC reacts with dehydrogenase enzymes and nicotinamide adenine dinucleotide in viable tissue; the infarcted area was defined as the area which did not stain. The heart slices were immersed in 10% formaldehyde solution for 24 hours to delineate the infarcted areas more clearly. With a wavelength of 366 nm ultraviolet light, we separated the risk from the infarcted zone of the heart and traced all areas onto an acetate sheet in order to estimate the volumes of these areas in cubic centimeter. The tracings were subsequently scanned with the Abode Photoshop 6.0 and measured with the Scion Image program (Scion Image 4.0, Scion Corporation, Meyer Instruments, Inc., Houston). The areas of myocardial tissue at risk and the infarcted areas were automatically transformed into volumes. Infarct and risk area volumes were expressed in cubic centimeter, and the percentage of infarct to risk area ratio (I/R) was calculated. 25
Western Blot Analysis
Tissue from the ischemic area of myocardium was converted into powder with liquid nitrogen and dry ice. The sample powder was moisturized with lysis solution (1% Triton X100, 20 mmol/L Tris pH 7.4–7.6, 150 mmol/L NaCl, 50 mmol/L NaF, 1 mmol/L EDTA, 1 mmol/L ethylene glycol tetraacetic acid, 1 mmol/L glycerolphosphatase, 1% sodium dodecyl sulfate [SDS], 100 mmol/L phenylmethanesulfonyl fluoride, and 0.1% protease phosphatase inhibitor cocktail) and homogenized. After centrifugation at 11 000g for 15 minutes at 4°C, supernatants were collected. The supernatant was mixed with Dave buffer (4% SDS, 10% 2-mercaptoethanol, 20% glycerol, 0.004% bromophenyl blue, and 0.125 mol/L Tris/HCl). The samples were heated at 100°C for 10 minutes and stored at −80°C. The protein concentration was determined based on the Bradford dye-binding procedure. An equal amount of protein was loaded in each well and then separated by SDS-polyacrylamide gel electrophoresis 7.5% to 11% and transferred onto a polyvinylidene difluoride membrane. After blocking with 5% nonfat dry milk, membranes were incubated overnight at 4°C with the following primary antibodies: phospho-eNOS (Ser1177), eNOS, phospho-Akt (Ser473), Akt, phospho-STAT3 (Tyr 705), STAT3, phospho-GSK3β, GSK3β, phospho-p44/42 (Thr202/Tyr204), p44/42, phospho-STAT5 (Tyr694), and STAT5 (Cell Signaling Technology, Beverly, Massachusetts). Membranes were then incubated with secondary antibodies for 1 to 2 hours at room temperature (Biorad goat anti-mouse and goat anti-rabbit horseradish peroxidase) and developed using the GE Healthcare ECL Western Blotting Detection Reagents (Thermo Scientific Technologies, Thermo Fisher Scientific Inc.,Waltham). Relative densitometry was determined using a computerized software package (NIH Image, Image J 1.44P, National Institutes of Health), and the values for phosphorylated Akt, eNOS, GSK3β, p44/42, STAT3, and STAT5 were normalized to the values for total Akt, eNOS, GSK3β, p44/42, STAT3, and STAT5. 26
Statistical Analysis
All results are presented as mean ± standard error mean. Comparisons of numeric variables among the 6 groups were analyzed using 1-way analysis of variance model with Bonferroni correction and with Tukey post hoc analysis. A calculated P value of less than .05 was considered to be statistically significant.
Results
Hemodynamic Data
The hemodynamic variables (blood pressure) and heart rate at baseline, at the 20th minute of ischemia and at the 180th minute of reperfusion showed no differences between the groups (data not shown).
Infarct Size
All HL groups as well as the SHO + PreC and the SHO + PostC groups demonstrated significantly smaller percentage of I/R compared with the SHO group (14.4% ± 3.7% in HL, 13.1% ± 1.0% in HL + PreC, 21.3% ± 1.6% in SHO + PreC, 18.0% ± 1.1% in HL + PostC, and 24.3% ± 1.7% in SHO + PostC vs 35.7% ± 4.4% in SHO; P < .05 for all comparisons with the SHO group). Area at risk was not different among the groups (Table 1). The application of PreC and PostC resulted in significantly reduced infarct size in the SHO animals (SHO group vs SHO + PreC: P = .007; SHO group vs SHO + PostC: P = .05). On the contrary, in the animals that were subjected to HL ischemia, no differences were observed as a result of PreC or PostC (HL group vs HL + PreC group: P = .73, HL group vs HL + PostC group: P = .3, and HL + PreC group vs HL + PostC: P = .35; Figure 2).
Mean Value ± SEM in Cubic Centimeter of the Area at Risk, Infarct Size, and I/R in Percentage, in Various Groups.
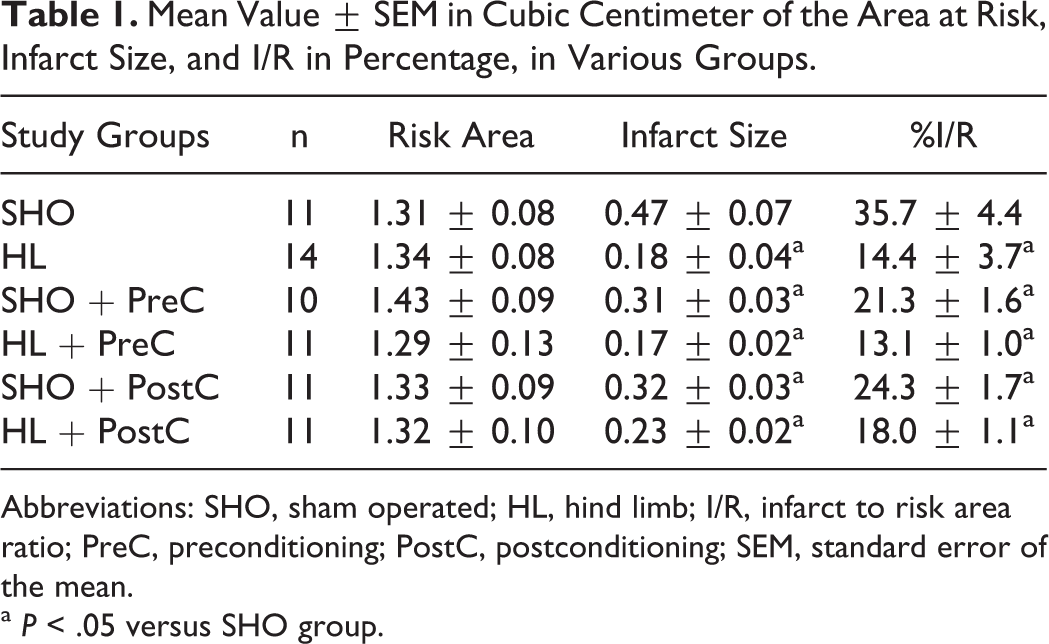
Abbreviations: SHO, sham operated; HL, hind limb; I/R, infarct to risk area ratio; PreC, preconditioning; PostC, postconditioning; SEM, standard error of the mean.
a P < .05 versus SHO group.

A, Percentage of infarct size to area at risk ratio (%I/R) and (B) percentage of area at risk to all myocardial area (%R/A) in different study groups (*P < .05 vs sham operated [SHO] group).
Western Blot Analysis
The Akt, eNOS, GSK3β, and p44/42 were phosphorylated in all groups, in which PreC or PostC was applied. A higher phosphorylation of Akt, eNOS, and p44/42 was observed in the HL groups when PreC and PostC were applied. The phospho-STAT3 levels did not differ significantly among groups. The STAT5 was activated in all HL groups compared with the SHO group. Total levels of proteins remain stable (Figure 3A–F, respectively).

Western blot analyses: (A) phospho-Akt and total Akt in different study groups (*P < .05 vs SHO and HL groups and *- P < .05 vs SHO + PreC and SHO + PostC); (B) phospho-eNOS and total eNOS in the study groups (*P < .05 vs SHO and HL groups and *- P < .05 vs SHO + PreC and SHO + PostC); (C) phospho-GSK3β and total GSK3β in different study groups (*P < .05 vs SHO and HL groups); (D) phospho-p44/42 and total p44/42 in different study groups (*P < .05 vs SHO and HL groups and *- P < .05 vs SHO + PreC and SHO + PostC); (E) phospho-STAT3 and total STAT3 in different study groups (*P < .05 vs SHO and HL groups); and (F) phospho-STAT5 and total STAT5 in different study groups (*P < .05 vs SHO, SHO + PreC, and SHO + PostC groups). eNOS, indicates endothelial nitric oxide synthase; GSK3β, glycogen synthase kinase 3β; HL, hind limb; PreC, preconditioning; PostC, postconditioning; SHO, sham operated; Akt, protein kinase B; STAT, signal transducer and activator of transcription.
Discussion
The findings of the present study indicate that (1) chronic HL ischemia decreases infarct size in an in vivo rabbit model after exposure to 30 minutes of myocardial ischemia and 3 hours of reperfusion demonstrating the cardioprotective effect of severe limb ischemia. (2) Simultaneous application of classic PreC and PostC does not enhance the cardioprotection afforded by skeletal muscle ischemia. (3) Phosphorylation of Akt, eNOS, GSK3β, p44/42, and STAT3 is present when classical conditioning is applied regardless of the infarct size limitation. (4) The STAT5 activation seems to be the main protein involved in cytoprotection derived from the application of remote conditioning by HL ischemia. (5) It seems that remote conditioning by severe HL ischemia does add to classic conditioning interfering with the intracellular mechanisms.
The HL ischemia is probably the most suitable form of remote conditioning due to the vulnerability of other remote candidate organs such as the kidneys or the intestine, which are susceptible to damage from even brief periods of ischemia. Furthermore, access to the blood supply of these organs poses significant difficulties. In contrast, the skeletal muscle is relatively resistant to damage from ischemia, is easily accessible, and a direct PreC effect as a result of skeletal muscle ischemia has been observed in animal models of skeletal muscle flaps. 27
These findings confirm our previous observations in rabbit 22 and rat 23 models of chronic HL ischemia. Although the cardioprotective effects of short-term skeletal muscle ischemia have been previously evaluated in experimental 8,12 –15 and clinical studies, 17,20 the present study is the first to examine the association of chronic severe skeletal muscle ischemia with the 2 well-established endogenous cardioprotective mechanisms of PreC and PostC. We have previously shown that protection from remote PostC was greater than that provided by classical PostC in anesthetized rabbits. 11 As a result, we had hypothesized that remote conditioning upregulates specific mediators or facilitates the intracellular pathways to a greater extent compared to that induced by PostC, and we had indicated the need for additional studies, which would identify the intracellular alterations occurring during those interventions. In the present study, we compare the intracellular signaling in the myocardium as a result of all the interventions in order to elucidate which mediators are common and which are correlated with infarct size limiting effect.
Although the mechanisms of typical PreC and PostC have been extensively studied, the protective mechanisms underlying remote conditioning are still poorly understood. 28 In particular and despite the progress in elucidating the triggers and cardiac signaling components contributing to the infarct-sparing effect of remote conditioning, the model of communication between the site of the protective stimulus and the target tissue remains unknown. Remote conditioning shares some similar intracellular signal pathways with PreC and PostC. The involved primary kinases include p38, p44/42 or JNK1/ 2, PI3K/Akt (RISK pathway), and PKC1. Although inhibition of mitochondrial permeability transition pore (mPTP) opening plays a central role in classical PreC and PostC, it is not understood whether mPTP inhibition contributes to RIC. 29 However, although most of the studies are in agreement for the role of RISK pathway in cardioprotection when remote ischemic PreC is applied, the importance of this pathway in the cardioprotection conferred by remote ischemic PostC has been questioned. 28,30,31
In the present study, we found that Akt, eNOS, GSK3β, and p44/42 were phosphorylated in groups that were pre- or postconditioned independent of the infarct size limitation. The HL ischemia in combination with PreC and PostC resulted in a greater activation of Akt, eNOS, and p44/42 compared with the SHO groups; however, in the HL group, there was no significant activation of Akt, eNOS, and p44/42 compared with the SHO group. These findings can be explained by the hypothesis that the combination of remote with local conditioning upregulates the above-mentioned mediators to a greater extent. However, the activations of Akt, eNOS, and p44/42 may be just stable and reliable markers of effective conditioning and not necessarily a critical component of cardioprotection itself, as previously showed. 32
The same observation was made regarding the activation of GSK3β and STAT3. Recent studies suggest an involvement of STATs in remote PreC by activation of STAT3. 33 In patients, the unique signaling signature of remote PreC seems to be the phosphorylation of STAT5 with no other secondary messenger or mediator playing a key role in cardioprotection induced by remote conditioning. 34 In agreement with these previous findings, we found that STAT5 is activated in HL groups only and in accordance with infarct size limitation indicating that the pathway activated in remote ischemic PreC induced by chronic skeletal muscle ischemia is possibly the STAT5 activation.
Interestingly, although the infarct size was not significantly different among the HL and the SHO groups with classical conditioning interventions, there was a trend for lower infarctions in the HL groups. This observation is in accord with the finding of higher STAT5 phosphorylation that was observed in all HL groups and forces us to suggest that remote conditioning interferes with the mechanism of classical conditioning.
In clinical practice, chronic HL ischemia cannot be applied to patients before AMI. However, there are many patients with coronary artery disease who have severe peripheral atherosclerosis leading to sequential subtotal lesions or even total occlusions of the iliac, femoral, tibial, or other arteries. Another group of patients are those who have been subjected to limb amputation for various reasons. According to our findings, such patients are already in a “conditioning state” and therefore more tolerant against myocardial ischemia/reperfusion regardless of the application of coronary conditioning. We may also think whether some experimental animal models are already protected at the time of applied interventions rendering the additional conditioning applications rather abundant. Of note, it has been recently shown that the commonly used platelet inhibitors act as conditioning mimetics. 35 Considering that virtually all patients with evolving ST-segment elevated myocardial infarction are treated with antiplatelet agents or receive a loading dose of antiplatelets before primary PCI, we may conclude that these patients are already “conditioned.” This may sufficiently explain the divergent results observed in some clinical studies.
There are many reports that question the equal potency of PreC and PostC, with the beneficial effect of the latter not having been validated in all laboratories or experimental models. 36 –39 In contrast, other reports consider PreC and PostC equally effective. 40 This variability should be taken into account. Some of these differences may be related to the above-mentioned “conditioning state” or more probably to the algorithms of PostC protocols, which may have yielded a different final infarct size. 41
In order to prove a clinical benefit of remote PreC with a mortality reduction in patients with threatened AMI, large, prospective, well-designed clinical studies are needed. Yet, in the meantime, the harmless procedure of pumping up and down a blood pressure cuff can and should be applied in the ambulance based on the knowledge that this intervention achieves a modest decrease in the ultimate infarct size without any serious adverse effects.
In conclusion, the present study shows that chronic HL ischemia reduces infarct size after myocardial ischemia and reperfusion. Additional application of classical conditioning does not enhance the infarct size limiting effects. The signaling pathway of remote cardioprotection is focused on the Janus kinase (JAK)/STAT pathway and specifically on STAT5. Future studies may extend our model of HL occlusion to further identify the role of JAK/STAT pathway.
Footnotes
Acknowledgment
We thank ELPEN Pharmaceutical Co Inc for the continuous support.
Authors’ Note
Sofia-Iris Bibli, Ioanna Andreadou, Efstathios K. Iliodromitis and Zenon S. Kyriakides contributed equally to this work. Efstathios K. Iliodromitis and Zenon S. Kyriakides are senior authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
