Abstract
Peripheral artery disease (PAD) affects a growing number of women, with a prevalence equal to that of men. Still, little is known about sex-specific differences in disease manifestations of PAD and how these differences in presentation impact outcomes of surgical and endovascular intervention. Women have also been consistently underrepresented in clinical trials evaluating treatment options for vascular disease. Thus, many of the practice guidelines in place today were generated from clinical trial data that either included a small number of women or excluded them entirely, and, consequently, whose findings may not be applicable to female patients. This review discusses key differences in outcomes based on sex for intervention of lower extremity, carotid, and abdominal aortic aneurysmal (AAA) disease. Elucidating sex-specific differences in disease manifestation and outcomes will provide insight into ways to optimize patient care, particularly for women afflicted with vascular disease.
Keywords
The University of Virginia School of Medicine designates this journal-based CME activity for a maximum of 1 AMA PRA Category 1 Credit™ per article. Participants should claim only the credit commensurate with the extent of their participation in the activity. Learners are expected to read this article, along with any references and supporting material as appropriate, and complete the online post-test questions with an 80% pass rate to receive credit. CME credit may be claimed immediately after completing the quiz. To register and begin earning free CME credit, visit the Society for Vascular Medicine’s website at vascularmed.org/VMJ-CME. This activity expires 2 years after the publication date, on
The faculty, staff and planning committee of the University of Virginia Office of Continuing Medical Education have no financial affiliations to disclose. The CME planning committee disclosed the following: Heather Gornik has disclosed research support from AstraZeneca and CVR Global, intellectual property rights from Summit Doppler Systems, Inc., and intellectual property rights and stock/ownership from FlexLife Health. Aditya Sharma has disclosed research support from National Institute of Health Sciences, AstraZeneca, Biomet Biologics, and Portola Pharmaceuticals. Valerie Clark has no financial affiliations to disclose.
Introduction
Cardiovascular disease is a significant global health concern and is associated with an increased risk of myocardial infarction, stroke, and overall mortality. Identifying the predictors of adverse outcomes is crucial to reduce overall cardiovascular disease burden. Women with coronary artery disease are known to have worse overall outcomes than men.1–4 Whether this relationship applies to other vascular diseases is largely understudied. Historically, women have been consistently underrepresented in clinical trials evaluating treatment options for vascular disease. Thus, many of the practice guidelines in place today were generated from clinical trial data that either included a small number of women or excluded them entirely, and, consequently, whose findings may not be applicable to female patients.
In recent years, endovascular intervention has emerged as an alternative therapeutic option to open surgery for treatment of vascular disease. Endovascular therapy is appealing due to its less invasive nature and cost-effectiveness.5–8 As utilization of endovascular therapy increases, it is important to consider the role of sex in outcomes and how these outcomes compare to traditional therapies. This review will discuss key differences in outcomes based on sex for intervention of lower extremity, carotid, and abdominal aortic aneurysmal (AAA) disease. Elucidating sex-specific differences in disease manifestation and outcomes will provide insight into ways to optimize patient care, particularly for women afflicted with vascular disease.
Lower extremity intervention
A previous misconception about peripheral artery disease (PAD) was that it affected predominantly men, though it is now appreciated that there is roughly an equal sex-related distribution.9,10 Increased recognition of PAD in the female population has allowed for great strides to be made in improving patient care. In the past, women were less likely to undergo intervention for lower extremity PAD. 11 However, recent reports indicate equal rates of endovascular management between men and women, 12 and that treatment type is associated with disease severity as opposed to patient sex. 13 Still, little is known about sex-specific differences in disease manifestations of PAD and how these differences in presentation impact outcomes of lower extremity intervention. Consequently, the American Heart Association (AHA) recently issued a ‘Call to Action’ to propagate research initiatives focusing on sex-specific differences in disease characteristics. 9
Women undergoing lower extremity revascularizations typically present at an older age and with more complex disease compared to their male counterparts.9,13 The sex-specific differences in disease severity and age at presentation may be due to the fact that more women with PAD are asymptomatic until later in the disease process, which results in a delay to diagnosis,14–17 or differences may be related to socioeconomic factors that prevent access to quality care. Multiple studies have demonstrated that patients with PAD are generally under-treated with guideline-recommended therapies, including antiplatelet agents and statins; this under-treatment is roughly similar between men and women with PAD. In response to the AHA ‘Call to Action’ statement, Dreyer and colleagues conducted a prospective study evaluating the relationship between sex and cardiovascular outcomes and self-reported health status following a diagnosis of PAD. 18 They found that women with PAD were of a poorer socioeconomic status compared to men, a finding that has been illustrated in other studies.19–23 Additionally, female sex was an independent predictor of worse physical and mental health status. This may have important implications for outcomes, given that depression has been associated with worse limb outcomes and increased risk for cardiovascular events in patients undergoing lower extremity revascularization.24,25
The impact of sex on outcomes of lower extremity revascularization for treatment of PAD is poorly understood (Table 1). Most of the literature evaluating sex-related disparities focuses on outcomes of surgical intervention. Despite a vast number of publications, there is a lack of consensus on the impact of sex on postoperative mortality, with several studies reporting no significant difference between sexes following surgical bypass11,26–30 and others suggesting worse outcomes for women. 12 In regards to surgical bypass graft patency, women are frequently noted to have inferior patency compared to their male counterparts. In a recent study by Duffy and colleagues, women with critical limb ischemia (CLI) had inferior patency rates following lower extremity bypass. 31 Although limb salvage and mortality rates were similar, women also had worse functional outcomes, as was seen in other studies.26,27 Similarly, in a study by Nguyen et al., 32 black race and female sex were associated with negative outcomes, including reduced patency. Inferior lower extremity bypass outcomes in women may in part result from more tapering of the great saphenous vein (GSV), which is most commonly used for bypass grafts. 33
Key lower extremity revascularization studies.
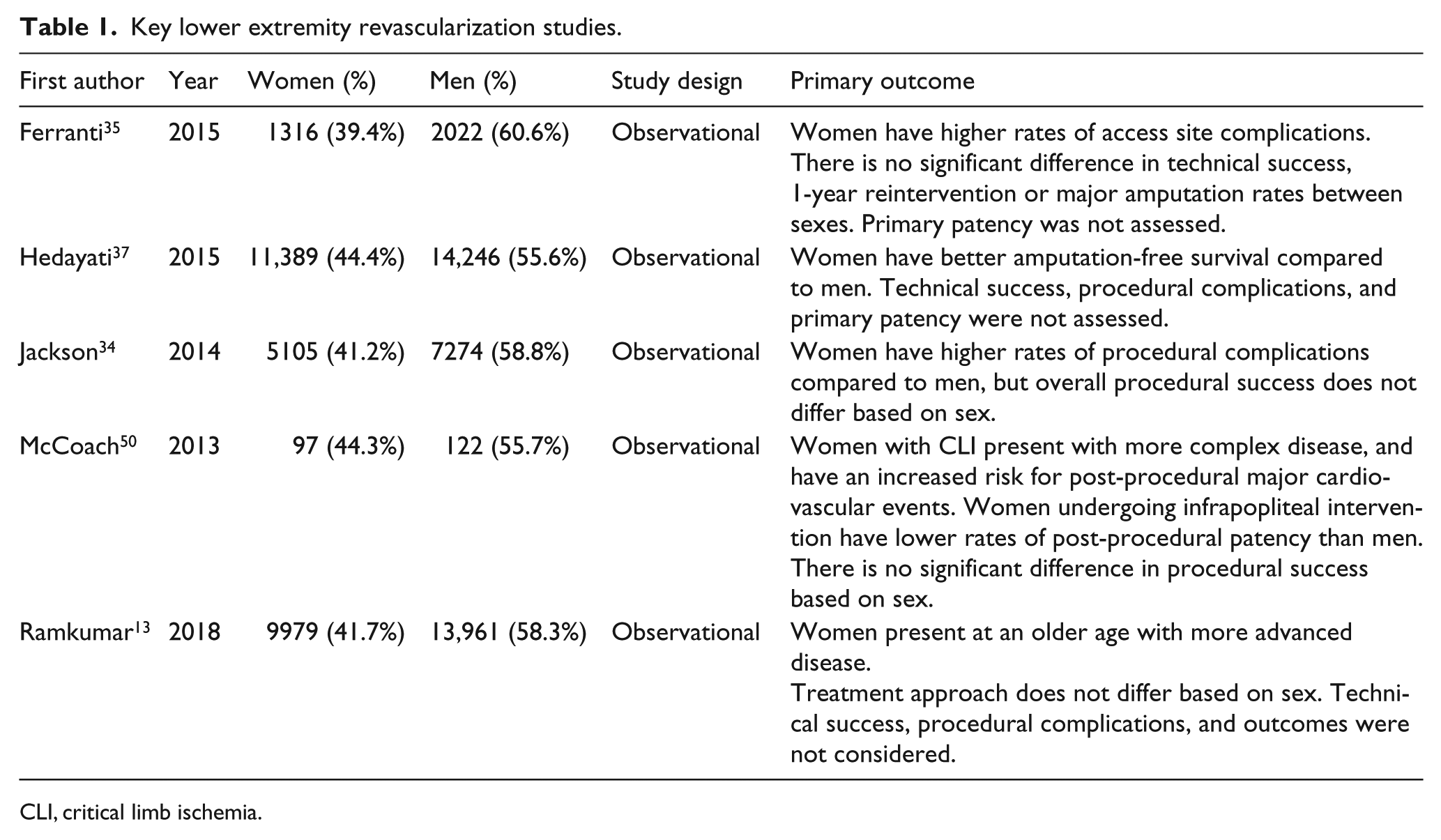
CLI, critical limb ischemia.
Although there is still an ongoing debate about the optimal treatment modality for lower extremity PAD, endovascular therapy has been largely adopted as the ‘first-line therapy’ for many patients with lower extremity disease. Published data evaluating sex-specific differences in outcomes for endovascular treatment are limited, though some studies report worse outcomes for women. In particular, women have been found to have higher rates of procedural complications than men. A recent study by Jackson and colleagues analyzed data from the Blue Cross and Blue Shield of Michigan Cardiovascular Consortium PVI Registry to compare the effects of sex on outcomes of lower extremity peripheral vascular intervention (PVI). 34 Women had higher rates of procedural complications, including vascular complications, transfusions, and embolism. Although technical success was achieved more frequently in females, the higher complication rate neutralized overall procedural success between sexes. Aside from procedural complications, no significant differences were observed in relation to in-hospital events, including mortality, myocardial infarction, stroke/transient ischemic attack (TIA), or amputation rates. Findings of increased procedural complications in women were corroborated by a large-scale cohort study by Ferranti et al., 35 which noted higher rates of access site complications in women undergoing lower extremity intervention. Similarly, Vouyouka and colleagues noted increased rates of bleeding complications in women. 36
While women are reported to have increased procedural complications, data suggest similar rates of major adverse limb events (MALE) and reintervention between sexes. Ferranti and colleagues reported outcomes out to 1 year post-procedure. After controlling for procedural indication (including claudication or CLI), there were no significant sex-related differences in reintervention or major amputation rates. 35 In a state-wide database study evaluating the effect of sex on PAD patients treated with endovascular intervention between 2005 and 2009, there was no significant difference in 12-month reintervention rates based on sex. 37 Interestingly, although more women presented with CLI, amputation-free survival was better among women compared to men (hazard ratio (HR) 0.84, 95% confidence interval (CI) 0.76–0.93).
A consensus on the impact of sex on patency after endovascular reintervention has yet to be attained. Several studies reported similar post-procedural patency between males and females undergoing lower extremity intervention.38–44 Pulli et al. 45 evaluated the outcomes of endovascular treatment of infrainguinal arterial obstructive disease and found no significant sex-specific differences in either primary or secondary patency. However, there was a trend towards poorer results for women who did not reach statistical significance. Conversely, a single-center study of patients who underwent infrageniculate percutaneous revascularization found that male sex was an independent predictor of decreased patency. 46
Studies evaluating the relationship between sex and long-term survival following intervention for lower extremity intervention have also yielded conflicting reports. Ferranti et al. 35 reported similar rates of mortality between males and females with claudication or CLI. This finding is supported by a study by DeRubertis and colleagues, in which there was no significant difference in 30-day mortality between males and females treated with endovascular intervention for infrageniculate disease. 39 While these studies did not find significant differences in mortality, a study reporting on data from three state-wide databases found significantly higher rates of procedural mortality in women. 12 Hedayati and colleagues also noted lower 12-month survival rates for women. 37 These findings were corroborated by a sub-analysis of a study by Vouyouka and colleagues focusing exclusively on endovascular treatment for lower extremity PAD. 36 Conversely, Hultgren and colleagues reported better long-term survival in females compared to males treated with surgical or endovascular procedures for lower limb ischemia. 30 Further research is needed to delineate the relationship between sex and long-term survival post-intervention.
Disease severity remains a frequent confounding factor influencing treatment outcomes based on sex. Women typically present with more advanced disease and are more likely to have critical limb ischemia (CLI) compared to men.31,34,37 CLI is the most advanced stage of PAD and is associated with increased risk for an array of adverse events including limb loss, cardiovascular events, myocardial infarction, stroke, and death.47–49 A recent single-center study by McCoach and colleagues reported sex-specific differences in long-term outcomes for lower extremity intervention for CLI patients. 50 The study reported higher rates of femoropopliteal or multilevel infrainguinal disease in women, consistent with prior reports of more complex disease presentation in women. In terms of outcomes, the overall cohort had equal rates of patency, though analysis of those treated for infrapopliteal lesions revealed decreased primary patency in women. There was no difference in amputation rates during follow-up (HR 0.94 for women, 95% CI 0.45–1.94, p=0.9). 50 However, there was an increased rate of subsequent major adverse cardiovascular events in women (HR 1.81, 95% CI 1.08–3.05, p=0.02). 50
The sex-related differences observed after revascularization of lower extremity PAD may be due to a multitude of other factors. Women have distinct anatomical differences compared to men. In general, women have smaller vessels, which increases procedural difficulty and impacts post-procedural surgical graft patency. In large trials of antiplatelet and antithrombotic agents, there have been no definite differences in sex-related outcomes, suggesting that differential outcomes after revascularization are not related to differences in drug efficacy between sexes. However, the microvasculature is impacted by sex, such that women with PAD have greater impairment of microvascular function. 51 This may contribute to greater functional impairment in females and negatively impact the utility of exercise therapy for the treatment of intermittent claudication, 52 resulting in a greater need for revascularization. Another potential explanation for worse outcomes in women following lower extremity intervention may be differences in underlying inflammatory processes. Women are thought to have a distinct inflammatory phenotype, such that they have higher C-reactive protein (CRP) levels compared to men.53–57 In a study by Hiramoto and colleagues, women with higher CRP and lower fibrinogen had significantly worse graft patency after undergoing lower extremity bypass for PAD. 58 However, this trend was not seen in males. Interestingly, despite the fact that women are of poorer socioeconomic status and overall physical and mental health, they have similar long-term outcomes as their male counterparts, as defined by objective measures such as mortality and amputation rates. Notably, functional status and quality of life (QOL) measures have not been taken into consideration in the reporting of outcomes post-revascularization. Given the fact that women with PAD present with worse QOL scores and poorer health status,59,60 and benefit less from supervised exercise therapies, 52 assessing change in QOL and functional status post-intervention may provide insight into the true benefit of intervention. Thus, additional exploration is needed into the sex-related disparities that exist and the implications of outcomes for intervention of lower extremity disease.
Carotid intervention
Carotid revascularization for atherosclerotic disease reduces the risk of recurrent stroke and may reduce the risk of stroke among selected asymptomatic patients. 61 Revascularization options for carotid disease include carotid endarterectomy (CEA) and carotid artery stenting (CAS). In recent years, research has focused on evaluating the optimal treatment modality for carotid disease and identifying predictors of outcomes.
Treatment selection for carotid disease depends on age, past medical history, degree of stenosis, and symptom severity. 62 The role of sex in the clinical decision-making process is unclear. While the most recent US AHA/ASA guidelines for the prevention of stroke in cases of atherosclerotic carotid disease do not explicitly state recommendations based on patient sex, they list female sex as a potential predictor of negative outcomes of CEA. 62 Similarly, the 2017 European guidelines comment on different outcomes for women, such as restenosis following intervention and a disproportionate susceptibility to stroke following CEA that appears to decrease over time. 63 These practice guidelines are based largely on data from several multicenter randomized control trials comparing the efficacy of CEA to medical therapy for treatment of carotid disease. However, women were consistently underrepresented in these studies.
The Asymptomatic Carotid Atherosclerosis Study (ACAS) was one such study to compare outcomes of CEA to medical therapy for patients with asymptomatic carotid artery disease (Table 2). The ACAS study reported a trend towards a decreased benefit of CEA for the reduction of perioperative stroke or death in women who did not reach statistical significance. 64 However, there were relatively few perioperative events (n=10/281 women, n=9/544 men treated with CEA), limiting the power of the analysis. In the case of symptomatic patients, the MRC European Carotid Surgery Trial (ECST) found surgery to be the optimal treatment option for patients with symptomatic stenosis of greater than 80%. 65 However, women were at a greater risk for 30-day stroke and mortality. Thus, the researchers concluded that sex should be an important consideration during treatment selection and that CEA may be better reserved for women with a greater degree of stenosis. Conversely, other trials including NASCET + ACE 66 and both initial and 10-year follow-up outcomes from ACST 67 reported no significant differences in outcomes based on sex. Of note, these studies contained low numbers of female patients. Equal sex distribution of rates of postoperative stroke and mortality after CEA have also been reported in population-based, 68 large cohort, 69 and single-center studies. 70 This lack of conclusive evidence has limited the ability to make specific recommendations based on sex, though data suggest women are less frequently referred for CEA.71,72
Key carotid revascularization studies.
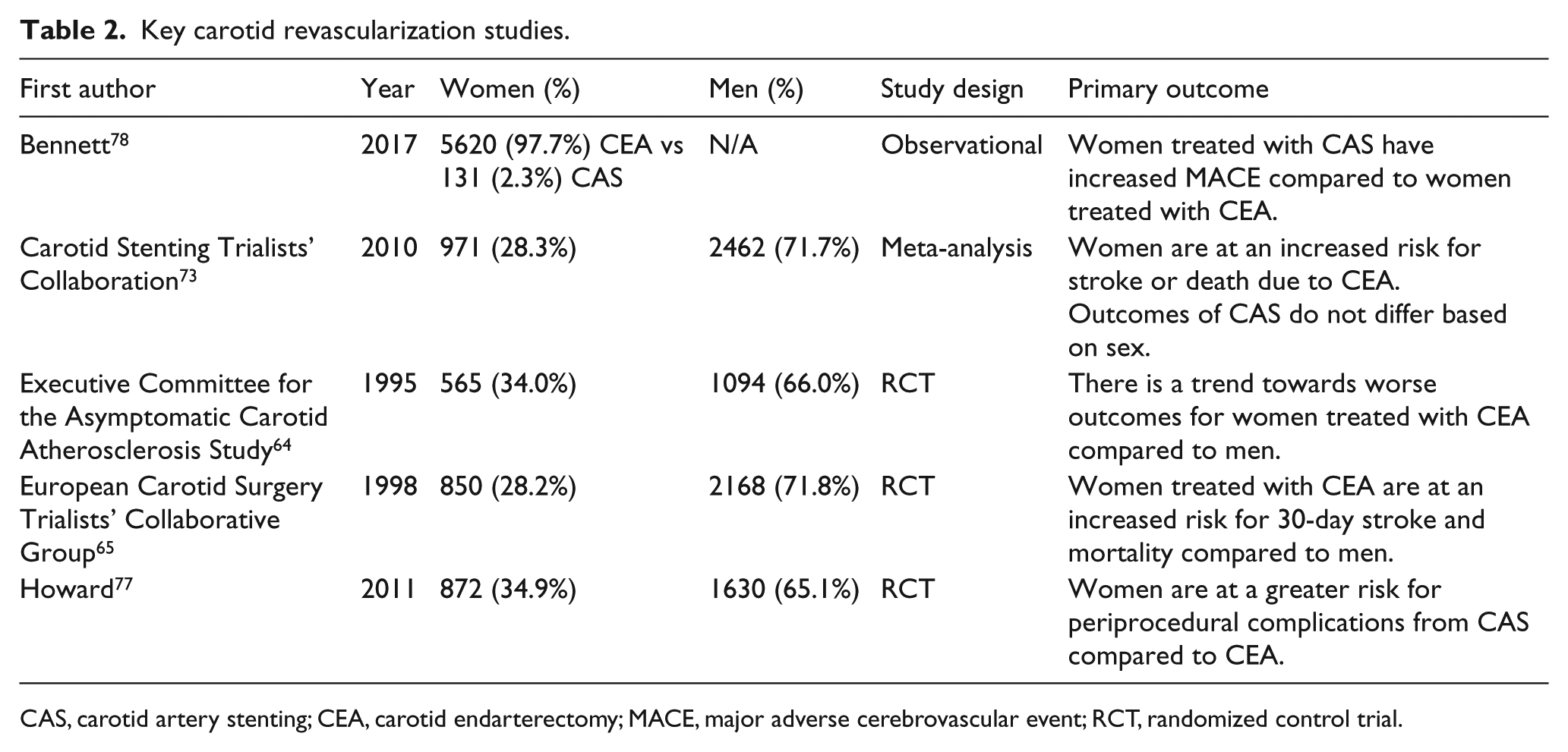
CAS, carotid artery stenting; CEA, carotid endarterectomy; MACE, major adverse cerebrovascular event; N/A: not applicable; RCT, randomized control trial.
Carotid angioplasty and stenting (CAS) has recently evolved as an alternative therapy for patients with carotid disease. Several multicenter randomized control trials were conducted to compare the efficacy of CAS to CEA. The majority of these trials did not comment on the impact of sex on patient outcomes. However, a meta-analysis 73 of individualized patient data from the Endarterectomy versus Angioplasty in Patients with Symptomatic Severe Carotid Stenosis (EVA-3S) trial, 74 the Stent-Protected Angioplasty versus Carotid Endarterectomy (SPACE) trial, 75 and the International Carotid Stenting Study (ICSS) 76 was conducted in 2010. A total of 2462 men (1230 treated with CAS; 1232 CEA) and 971 women (495 CAS; 476 CEA) were included in the analysis. The researchers concluded that women were at a greater risk for stroke or death due to CEA compared to men, but outcomes of CAS were equal between sexes. 73
The randomized Carotid Revascularization Endarterectomy Trial (CREST) 77 was designed to evaluate predictors of outcomes for CAS compared to CEA. CREST’s study design also incorporated a subgroup analysis to compare sex-specific differences in outcomes between CEA and CAS. Results from the subgroup analysis found that women assigned to the CAS treatment arm were more likely to experience periprocedural complications compared to those who underwent CEA (HR 2.63, 95% CI 1.23–5.65, p=0.013). 77 There was no significant difference in outcomes between males undergoing CAS or CEA (HR 1.39, CI 0.78–2.48, p=0.26). In a study by Bennett and Scarborough using data from the American College of Surgeons National Surgical Quality Improvement Program (ACS NSQIP), CAS was associated with a higher incidence of major adverse cerebral events (MACE) compared to CEA in females (290/5620, 5.2% CEA; 16/131, 12.2% CAS; p < 0.001), which remained significant even after propensity-score matching. 78 The small cohort size in the propensity-matched analysis limited the ability to assess components of MACE, but there was a trend towards higher rates of myocardial infarction (MI)/arrhythmia, stroke, and death. Similar findings were noted in a systematic review comparing sex-specific outcomes of CEA to CAS by Hedayati and colleagues, in which the researchers concluded that CAS is associated with worse outcomes for females. 79
The mechanisms contributing to differential outcomes based on sex are poorly understood. Some proposed explanations include differences in vascular anatomy, disease progression, and treatment approach. The narrower diameter of carotid vessels in women may increase the technical difficulty of vascular intervention and risk of procedural complications. Additionally, a single-center study by Spyris et al. 80 found that women with preoperative neurologic symptoms had a greater mean particle size of embolic debris compared to asymptomatic women undergoing CAS. This may suggest that symptomatic women are at a greater risk for perioperative complications of CAS. Several studies have also described differences in carotid plaque morphology between male and females, such that women have more stable plaques than men.81–83 These data suggest that men may benefit more from vascular intervention compared to their female counterparts. In the past, studies reported that women with carotid disease were more likely to be managed medically compared to men, with vascular intervention reserved for women with more advanced disease. Whether disparate intervention rates based on sex are due to differences in disease incidence 84 or in treatment approach remains unclear.
Abdominal aortic aneurysm intervention
Abdominal aortic aneurysms (AAA) have an unequal sex distribution, affecting males to females in roughly a 5:1 ratio. 85 Women with AAA are typically older86,87 and are thought to have worse outcomes than men. Owing to a relatively low prevalence in women, the United States Preventative Services Task Force does not recommend that women undergo screening for AAA. 88 The Society for Vascular Surgery only recommends that women over age 65 undergo screening if they have a family history of AAA or history of smoking. 89 Collectively, this leads to an underrepresentation of women in studies evaluating treatment options for AAA and a paucity of data on optimal management strategies (Table 3).
Key AAA studies.
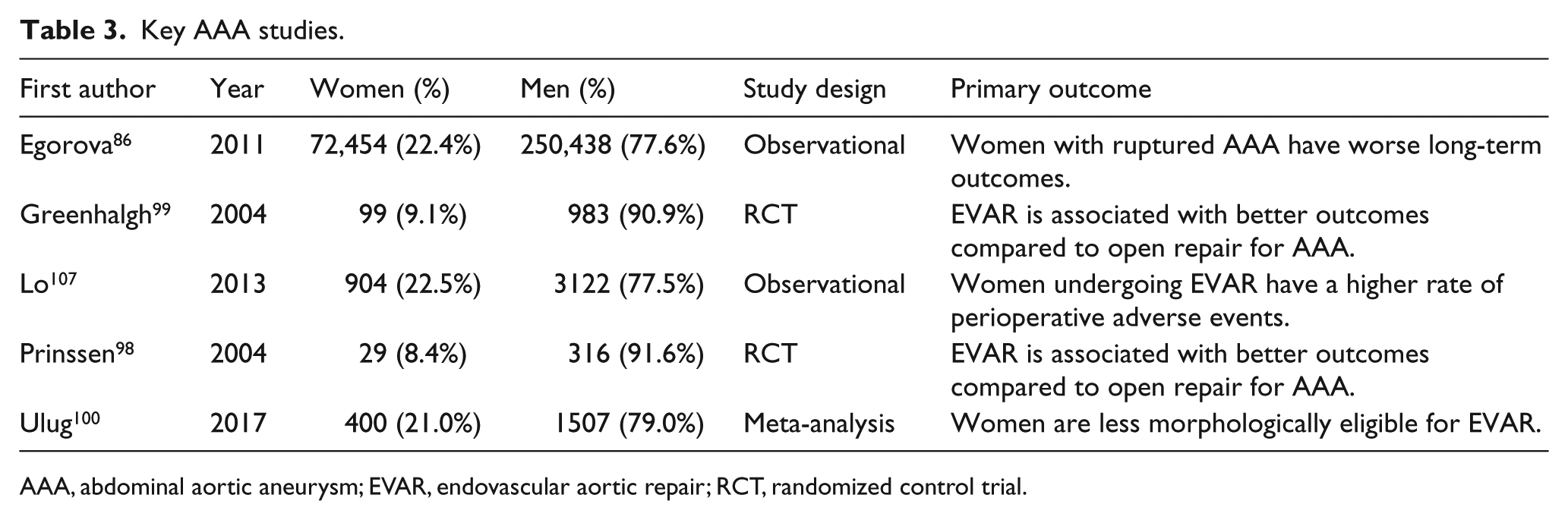
AAA, abdominal aortic aneurysm; EVAR, endovascular aortic repair; RCT, randomized control trial.
Although fewer women are affected, data suggest the prognosis for women with AAA is worse than that of men.90,91 Notably, women have a higher risk of rupture for a given aneurysm size. 87 Women with ruptured AAA are less likely to be repaired operatively90,92–95 and those who do undergo revascularization have inferior outcomes compared to men. Both perioperative and 30-day mortality rates are higher in women when treated with either open surgical or endovascular aortic repair (EVAR) for ruptured AAA.96,97
Data support the use of EVAR over open surgical repair for the treatment of AAA. Two large randomized trials98,99 comparing EVAR to open repair reported significantly lower rates of periprocedural and 30-day mortality in patients treated with EVAR. Consistent with clinical trials assessing vascular disease in other territories, these studies contained a disproportionate number of males compared to females: the Dutch Randomized Endovascular Aneurysm Management (DREAM) trial 98 included only 29 (8.4%) women and the Comparison of endovascular aneurysm repair with open repair in patients with abdominal aortic aneurysm (EVAR-1) trial 99 reported on outcomes for 99 (9.1%) women.
The enhanced perioperative and short-term outcomes associated with EVAR creates a conundrum for treating women with AAA: although women have a higher risk of rupture of untreated AAA and the outcomes for EVAR are better than open repair, they are often ineligible for EVAR due to differences in vascular morphology. A recent systematic review and meta-analysis performed by Ulug and colleagues evaluated the effects of sex on eligibility for EVAR or open repair for AAA and 30-day mortality rates. 100 The analysis consisted of five key studies101–105 reporting on sex disparities in the suitability for EVAR. Results from the meta-analysis found that the rate of morphological eligibility for EVAR was significantly lower in women compared to men (34% women, 54% men; OR 0.44, 95% CI 0.32–0.62), 100 a statistic that has not drastically changed since the early 2000s. 106 Women typically have shorter and more angulated necks and smaller iliac access sites, making them ineligible for device-specific instructions for use (IFU) criterion for EVAR. 103
Women who do undergo EVAR are reported to have worse perioperative outcomes compared to men. Analysis of the impact of sex on outcomes from treatment of AAA in patients within the Vascular Study Group of New England (VSGNE) database concluded that women had a higher rate of perioperative adverse events. 107 These findings were reproduced by a single-center study by Gloviczki et al. 108 Of note, both studies concluded that clinical presentation, concomitant health conditions, and age were better predictors of outcomes than sex. Other single-center studies109,110 have found a similar increase in rates of perioperative complications in women compared to men. While the majority of the literature is in agreement that perioperative complications from EVAR are higher in women, there is contradictory data on perioperative morality. The VSGNE and single-center studies found higher perioperative complication rates, but mortality rates were similar between sexes.107–110 Conversely, several large-scale studies have reported higher rates of perioperative mortality in women following EVAR.86,95,111 In their meta-analysis, Ulug and colleagues also reported that 30-day mortality rates for both EVAR and open repair were roughly twofold higher in women. 100 As proposed in a review by Lo and Schermerhorn, 91 these differences may be attributed to distinctions in study populations, making it difficult to discern the relationship between mortality and sex.
Sex-related disparities in outcomes for intervention of AAA may be attributed to several factors. The screening recommendations for women are limited; it is plausible that this leads to a delay to diagnosis in women and causes them to present with more advanced disease. Additionally, cardiovascular risk factors are not managed as efficiently in women,112,113 which may lead to increased risk of rupture or worse revascularization outcomes. Sex-based anatomical structure variations may also account for differences in AAA outcomes. The challenging vasculature in women with AAA not only makes them more frequently unsuitable candidates for EVAR, but also makes them more prone to procedural complications. Additionally, data suggest that measurement of aneurysm size should take into account body surface area; at the time of intervention, women often have larger aneurysms relative to body surface area, which may put them at a greater risk of rupture.114,115 Other studies suggest differences in biochemical properties between men and women may account for disparate outcomes. 116
New lower profile devices may help extend access of care to women who were previously ineligible for EVAR. The prospective, multicenter Evaluation of FemaLes who are Underrepresented Candidates for Abdominal Aortic AneurYsm Repair (LUCY) study aims to evaluate the impact of female sex on outcomes of EVAR using the TriVascular Ovation system, a low profile device. 117 Preliminary results show comparable results between men and women, with relatively low 30-day major adverse events and endoleak rates. Results from this and other studies evaluating lower profile devices may identify novel ways to reduce sex disparities for treatment of AAA.
Conclusion
This review highlights the importance of significant sex-related differences in outcomes following vascular intervention. Women undergoing intervention for lower extremity, carotid, and AAA have higher rates of procedural complications. In cases of lower extremity PAD, women present with more advanced disease, including higher rates of CLI. Women have lower rates of graft patency following surgical bypass, whereas it is unclear how endovascular intervention impacts differences in patency between sexes. Women with carotid disease may be at a greater risk for stroke when treated with CAS. The risk of rupture is higher in women with AAA, and they are less suitable candidates for EVAR. Differences in presentation, including the tendency to present at an older age and with more advanced disease, and differences in vascular morphology may account for inferior outcomes for women with vascular disease.
These findings have several implications for the clinical care of women with vascular disease. First, increased awareness of sex-related differences in disease manifestation and outcomes is needed. Sex should be an important consideration when evaluating treatment options for patients with vascular disease. Sex-specific intervention programs, including supervised exercise therapy, may be beneficial in minimizing disparities. This study also illustrates the need for advances in the development and research of procedural devices, such as low profile devices for EVAR, in order to allow for the inclusion of more women. Alarmingly, significant knowledge gaps still persist, reiterating the importance of enhancing research initiatives to investigate sex differences. Further research including large-scale clinical trials and cohort studies will be imperative to establish conclusive data and guide the clinical decision-making process for women with vascular disease.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Ehrin Armstrong serves as a consultant/advisory board member for Abbott Vascular, Boston Scientific, Cardiovascular Systems, Janssen, Medtronic, and Philips; Ellen Brinza has no disclosures.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
