Abstract
Peripheral artery disease (PAD) is estimated to affect approximately 8.5 million individuals in the US above the age of 40, and is associated with significant morbidity, mortality, and impairment. Despite the significant adverse limb and cardiovascular (CV) outcomes seen in patients with PAD, there is typically less attention paid to risk factor modification relative to other atherosclerotic diseases such as coronary artery disease (CAD) or stroke. In the current literature, statins have been shown to reduce mortality, major adverse CV events, major adverse limb events, and improve symptomatic outcomes in patients with PAD. In addition, proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors are emerging as an additional lipid-lowering therapy for patients with PAD. However, despite current guideline recommendations based on growing evidence, patients with PAD are consistently undertreated with lipid-lowering therapies. We provide an extensive literature review and evidence-based recommendations for the use of statins and PCSK9 inhibitors in patients with PAD.
Introduction
Peripheral artery disease (PAD) is a highly prevalent atherosclerotic disorder that is associated with significant long-term disability, morbidity, and mortality.1,2 The prevalence of PAD ranges from 15% to 20% in individuals over 65 years of age. 3 In addition, there are 148,000 major amputations performed annually in the US due to PAD. 4 Annual mortality is higher among patients with PAD (8.2%) than after a myocardial infarction (MI) (6.3%). 5 Despite these significant limb and cardiovascular (CV) outcomes, lipid-lowering medications are underutilized in patients with PAD compared to patients with other atherosclerotic diseases, including coronary artery disease (CAD) and stroke.6,7 Current guidelines recommend statin therapy for all individuals with PAD.8,9 Unfortunately, much of the data regarding lipid-lowering therapy for patients with PAD have been extrapolated from studies that mainly included patients with CAD. Furthermore, fewer studies have specifically examined limb outcomes.
Since US Food and Drug Administration (FDA) approval of lovastatin in 1987, statins have been used for the prevention of plaque accumulation and reduction of CV mortality.10,11 Many clinical trials have shown the benefit of statins in reducing low-density lipoprotein cholesterol (LDL-C) levels as well as CV morbidity and mortality. 12 Furthermore, additional trials have demonstrated that high-intensity statin therapy confers an additional reduction in the CV risk. 13
Many years after the discovery of statins, proprotein convertase subtilisin/kenxin type 9 (PCSK9) inhibitors have emerged as another group of medications that help reduce LDL-C. 14 Large-scale trials found that evolocumab and alirocumab, two different PCSK9 inhibitors, not only reduced the LDL-C level significantly, but also reduced MACE in patients with CV disease.15,16
In addition to statins and PCSK9 inhibitors, ethyl esters are also emerging as a potential lipid-lowering therapy for patients with CAD as well as PAD. Furthermore, ezetimibe has been shown to improve clinical outcomes by reducing MI and stroke when used in combination with a statin. 17 While other lipid-lowering therapies have been utilized, including resins, fibrates, selective cholesterol absorption inhibitors, and marine-derived omega-3 polyunsaturated fatty acids, there has been no convincing evidence that these therapies significantly impact CV outcome. Consequently, these lipid-lowering medications will not be covered in this review.
While the importance of cholesterol management using statins and PCSK9 inhibitors has been extensively described in patients with CAD, there is a lack of literature regarding cholesterol management in patients with PAD. This paper reviews current literature on the impact of statins and PCSK9 inhibitors on PAD outcomes. This information is intended to increase awareness and use of such therapies, as well as decrease adverse CV outcomes, in patients with PAD.
Methods
A narrative review of all existing evidence on the effectiveness of lipid-lowering medications, specifically statins and PCSK9 inhibitors, in patients with PAD was conducted. Public PubMed databases were searched using the terms (‘PAD’) AND (‘statins’, ‘PCSK9’, OR ‘lipid-lowering medication’) for all full-text articles written in English between January 1, 2003 and December 31, 2019. Search results yielded 237 articles published during this time interval. Articles with no distinct PAD group evaluation or no direct treatment with lipid-lowering medications were excluded. Endpoints of interest were all-cause and CV mortality, major adverse cardiovascular events (MACE), PAD-related symptoms, major adverse limb events (MALE), and exercise capacity.
Statins
Most studies evaluating mortality, MACE, MALE, and symptomatic endpoints have shown the benefit of statin therapy in patients with PAD (Table 1). However, most of these data have been abstracted from studies that mainly enrolled patients with CAD. In addition to randomized controlled trials, many observational and retrospective reviews have provided insight into the benefit of statin therapy in patients with PAD.
Statin therapy on MACE, MALE, and symptomatic outcomes in patients with PAD.
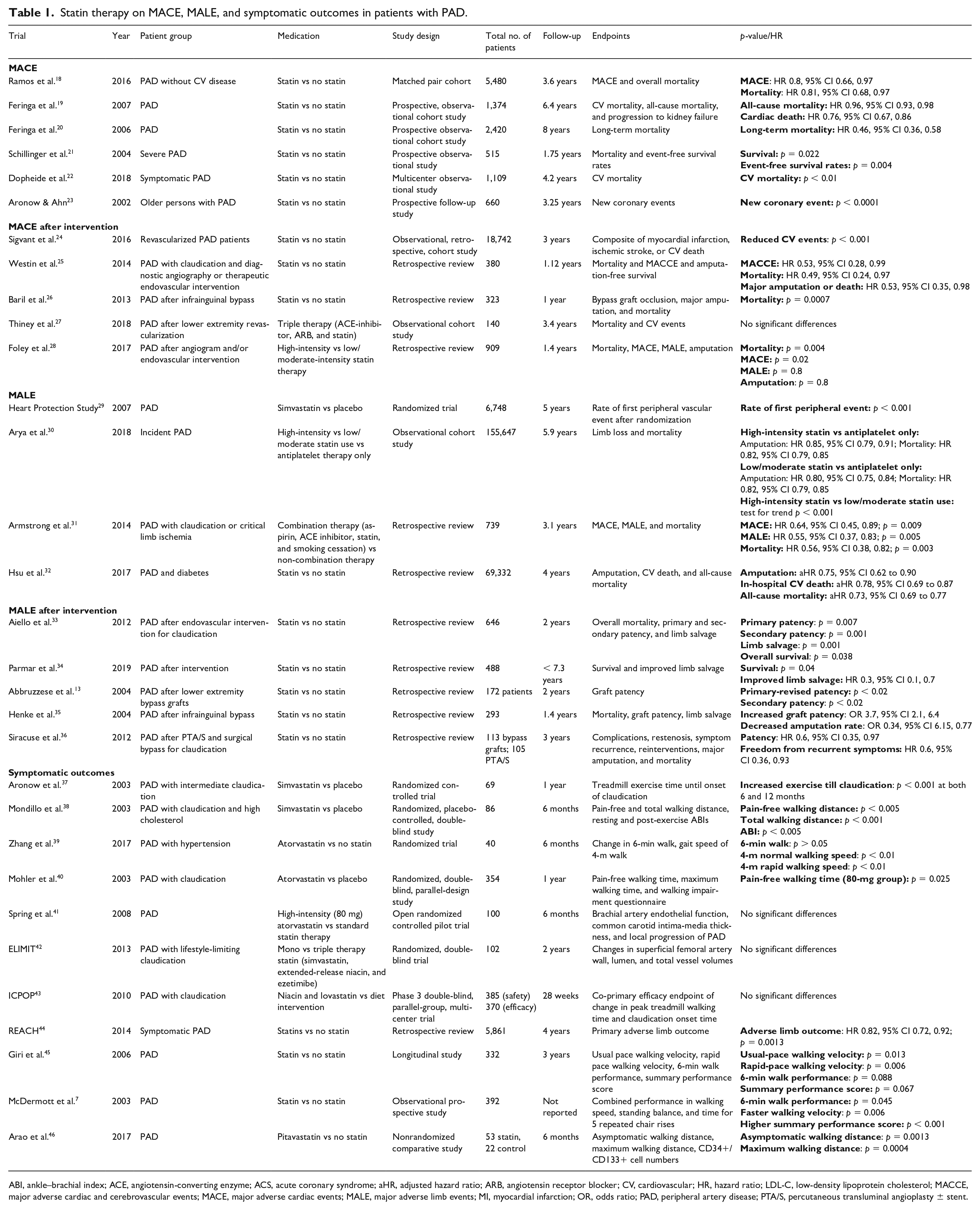
ABI, ankle–brachial index; ACE, angiotensin-converting enzyme; ACS, acute coronary syndrome; aHR, adjusted hazard ratio; ARB, angiotensin receptor blocker; CV, cardiovascular; HR, hazard ratio; LDL-C, low-density lipoprotein cholesterol; MACCE, major adverse cardiac and cerebrovascular events; MACE, major adverse cardiac events; MALE, major adverse limb events; MI, myocardial infarction; OR, odds ratio; PAD, peripheral artery disease; PTA/S, percutaneous transluminal angioplasty ± stent.
Major adverse cardiovascular events (MACE)
Statin use has been associated with decreased MACE and mortality in patients with PAD based on observational analyses. A matched pair cohort study which included patients with PAD without CAD (n = 5480) investigated the use of statins on MACE and overall mortality. 18 At a mean follow-up of 3.6 years, statin therapy was associated with lower MACE and overall mortality compared to no statin. In addition, a prospective observational cohort study investigating statin use in patients with PAD (n = 1374) by Feringa et al. showed that higher dose statin treatment and lower LDL-C levels were independently associated with lower all-cause and cardiac mortality after 6.4 years of follow-up. 19 Another prospective observational cohort study of patients with PAD (n = 2420) investigated the use of statins, angiotensin-converting enzyme (ACE) inhibitors, beta blockers, and aspirin on long-term mortality. 20 After a follow-up period of 8 years, all medications were associated with decreased risk of long-term mortality, with statins having the most benefit. Within a prospective study published by Schillinger et al., 515 patients with severe PAD on statin therapy were found to have improved intermediate-term survival when compared with those not taking statins at 1.75 years of follow-up. 21 Dopheide et al. demonstrated that while patients with PAD were remarkably undertreated with statins, symptomatic patients with PAD treated with statins had lower CV mortality at a median follow-up of 4.2 years. 22
A study by Aronow and Ahn that focused on statin use in patients with PAD over the age of 80 also reported better outcomes with statin therapy. 23 Of the 660 patients with PAD with a mean age of 80, statin therapy was associated with a lower incidence of new coronary events after 3.25 years of follow-up.
MACE after intervention
Statin therapy has also been associated with decreased adverse CV outcomes and death, specifically in patients with PAD who underwent an endovascular intervention or bypass. A retrospective observational cohort study utilizing the Swedish National Registry for Vascular Surgery Database identified 18,742 patients with PAD with a prior revascularization to investigate the outcome of initiating statin therapy after revascularization. 24 Statin therapy was found to be associated with lower CV events, including MI, ischemic stroke, and CV death at 3 years of follow-up.
Another retrospective review of 380 patients with PAD investigated the impact of statin prescription on major adverse CV and cerebrovascular events (MACCE) after an endovascular intervention. 25 At a mean follow-up of 1.1 years, statin prescription (65%) was associated with lower rates of mortality, MACCE, and greater amputation-free survival. Baril et al. also reported a lower 1-year mortality associated with statin use at time of infra-inguinal bypass surgery in 323 patients with PAD. 26
In addition to statin use alone, a study by Thiney et al. investigated the effect of triple therapy, consisting of ACE inhibitors, angiotensin receptor blockers, and statins, from time of discharge to end of follow-up, on mortality and CV events after lower extremity revascularization. 27 While this observational cohort study of 140 patients with PAD demonstrated a lower additional vascular surgery rate in patients on triple therapy, no significant difference in mortality or CV events was found.
A study by Foley et al. investigated the use of high-intensity statin within 2 months prior to intervention and throughout follow-up in patients with PAD who underwent angiography and/or endovascular intervention compared with standard statin therapy. 28 Of the 909 study group with PAD, high-intensity statin therapy was used in 124 patients, and was associated with improved survival and lower MACE compared with low or moderate intensity statin therapy at a mean follow-up of 1.4 years.
Major adverse limb events (MALE)
In addition to MACE and mortality, limb survival is an important outcome in patients with PAD. The Heart Protection Study randomized 20,536 subjects, 6748 of whom carried a diagnosis of PAD, to either simvastatin 40 mg daily or placebo with a mean follow-up of 5 years. 29 In the overall study population, simvastatin was associated with a 16% relative risk reduction in peripheral vascular events, which was driven primarily by a 20% reduction in noncoronary revascularization (lower extremity arterial procedures and carotid interventions). However, within the subset of patients with PAD, there were no differences in the rates of amputation between the two groups.
Arya et al. investigated statin use on limb loss and mortality in a large observation cohort study of patients with PAD (n = 155,647), which utilized national US Veterans Affairs data. 30 At a mean follow-up of 5.9 years, any statin use was associated with a reduction in limb loss and mortality compared to antiplatelet therapy alone, with high-intensity statins showing the greatest reduction in adverse outcomes.
In addition to statin use alone, Armstrong et al. investigated the benefit of placing patients with claudication or critical limb ischemia (n = 739) on a combination therapy of all four guideline-recommended therapies of aspirin, ACE inhibitor, statin, and smoking cessation. 31 Compared to patients on less than all four therapies, this retrospective study demonstrated that using all four therapies was associated with decreased rates of mortality, MACE, and MALE after a mean follow-up of 3.1 years.
Specifically in patients with PAD and diabetes, a study by Hsu et al. also investigated statin therapy on vascular outcome. 32 In this large retrospective analysis, which utilized the nationwide diabetes mellitus database in Taiwan, patients with PAD and diabetes (n = 69,332) were evaluated for statin use. Statin therapy was associated with decreased risk for amputation, in-hospital CV death, and all-cause mortality.
MALE after intervention
A retrospective analysis by Aiello et al. assessed overall mortality, primary and secondary patency, and limb salvage rates in patients with PAD who underwent endovascular intervention for critical limb ischemia. 33 Of the 646 patients, 319 were on a statin treatment at time of intervention and were found to have better survival, patency, and limb salvage rates at 2 years of follow-up. Parmar et al. also demonstrated that statin use throughout follow-up was associated with improved survival and limb salvage in 488 patients with PAD after a surgical or endovascular intervention. 34
Improved graft patency after lower extremity bypass surgery in patients with PAD has also been reported with statin therapy. A retrospective analysis that specifically investigated the effect of statins on graft patency after infra-inguinal bypass (n = 172 patients with 189 grafts) showed better graft patency in patients receiving statin therapy after 2 years of follow-up. 13 A similar study by Henke et al. investigated the use of statins in patients with PAD (n = 193) after infra-inguinal bypass. 35 After 1.4 years of follow-up, patients receiving statins at any point was associated with improved graft patency and limb salvage, but not survival.
Statin therapy was also associated with better outcomes after percutaneous transluminal angioplasty ± stent and bypass for critical limb ischemia. 36 After 3 years of follow-up, 113 patients underwent bypass grafting and 105 underwent percutaneous transluminal angioplasty ± stent. Those who received statin therapy at time of intervention had lower rates of restenosis and symptom recurrence. However, statins were not found to affect re-intervention rates.
Symptomatic outcomes
In addition to MACE and MALE, the effect of statin therapy on PAD symptoms has also been evaluated. A randomized trial involving patients with PAD and claudication (n = 69) by Aronow et al. investigated the effect of simvastatin on treadmill exercise time until the onset of claudication. 37 Patients who received 40 mg of simvastatin daily versus placebo had a significant increase in treadmill exercise time until onset of claudication (24% at 6 months and 42% at 12 months). A similar randomized trial by Mondillo et al. that included patients with PAD with claudication and high cholesterol also investigated the effect of simvastatin on walking outcomes. 38 Eighty-six patients were randomized to receive either 40 mg simvastatin daily or placebo. After 6 months, patients taking simvastatin showed a significant increase in pain-free and total walking distance, as well as resting and post-exercise ankle–brachial indexes (ABIs). Another randomized trial by Zhang et al. investigated the effect of atorvastatin on change in walking speed for a 6-minute walk and gait speed of 4-meter walk in patients with PAD (n = 40) and hypertension. 39 While there was no difference in walking speed for the 6-minute walk, there was a significant increase in gait speed of the 4-meter walk after 6 months.
Randomized trials have also investigated the role of statins in improving PAD symptoms. A large randomized trial by Mohler et al. investigated the effect of high and standard dose atorvastatin on walking outcomes in patients with PAD-related claudication. 40 Patients were randomized (n = 354) to either 10 or 80 mg atorvastatin daily, or placebo. After 1 year of follow-up, the high but not the low-dose atorvastatin group demonstrated significant improvement in pain-free walking distance compared with placebo. In a similar trial by Spring et al., patients with PAD were randomized to high-intensity statin (80 mg atorvastatin) versus conventional medical treatment. 41 There was no significant difference in brachial artery endothelial function, common carotid intima–media thickness, and local progression of PAD between the two groups.
A randomized trial by Brunner et al. investigated the effect of mono (simvastatin 40 mg daily) versus triple therapy (simvastatin 40 mg daily, extended-release niacin 1500 mg daily, and ezetimibe 10 mg daily) in patients with PAD-related limiting claudication. 42 There was no significant difference in superficial femoral artery wall, lumen, and total vascular volume between the two groups. In addition, a randomized trial by Hiatt et al. found no significant difference in claudication between patients taking niacin plus lovastatin compared with those who were on dietary intervention only. 43
Within the REACH Registry (Reduction of Atherothrombosis for Continued Health), statin therapy was evaluated in 5681 patients with symptomatic PAD. 44 Statin therapy was associated with a 14% relative risk reduction in the composite endpoint of worsening claudication, critical limb ischemia, peripheral revascularization, and amputation at 4 years of follow-up. A longitudinal observational study by Giri et al. investigated the use of statins in patients with and without PAD on walking variables including walking pace, walking velocity, and 6-minute walk performance. 45 Of the total 544 patients, 332 were diagnosed with PAD. After a follow-up of 3 years, statin therapy was associated with less annual decline in lower extremity performance including walking distance and velocity.
A prospective observational study by McDermott et al. investigating the effect of statin use on lower-limb functioning in patients with PAD (n = 392) also found statin use to be associated with improved walking performance, consisting of walking distance and velocity, independent of cholesterol levels. 7 Arao et al. evaluated the effect of short-term pitavastatin on asymptomatic walking distance and maximum walking distance in patients with PAD. 46 Of the 75 patients, use of pitavastatin in 53 patients was associated with improved asymptomatic walking distance and maximum walking distance.
PCSK9 inhibitors
PCSK9 inhibitors have been shown to be effective lipid-lowering agents in patients with CAD. However, they have been less extensively studied than statin therapy in PAD. Although there have been no randomized controlled trials that investigated PCSK9 inhibitors specifically in patients with PAD, there have been recent promising data abstracted from studies on patients with CAD showing CV benefits in the subset of patients with PAD (Table 2).
PCSK9 inhibitors on MACE, MALE, and LDL-C reduction in patients with PAD.

ACS, acute coronary syndrome; CV, cardiovascular; HR, hazard ratio; LDL-C, low-density lipoprotein cholesterol; MACE, major adverse cardiac events; MALE, major adverse limb events; MI, myocardial infarction; PAD, peripheral artery disease; VTE, venous thromboembolism.
FOURIER is a randomized, double-blind, placebo-controlled trial that enrolled 27,564 patients with atherosclerotic CV disease and LDL-C levels ⩾ 70 mg /dL who were receiving statin therapy. 15 Patients were assigned to receive subcutaneous evolocumab (either 140 mg every 2 weeks or 420 mg monthly) or matching placebo. Relative to placebo, evolocumab significantly reduced the risk of composite of CV death, MI, stroke, hospitalization for unstable angina, or coronary revascularization, as well as the composite of CV death, MI, or stroke. In a subanalysis of 3642 patients with confirmed PAD who were also on optimized statin therapy, 70% of whom where on high-intensity statin therapy, evolocumab treatment reduced the relative risk of MALE by 42% and MACE by 21%. 47 Moreover, the absolute reduction in risk for CV death, MI, stroke, hospitalization for unstable angina, and revascularization was greater in the PAD subset than in the overall FOURIER cohort (3.5% vs 1.6%).
ODYSSEY OUTCOMES is a multicenter, randomized, double-blind, placebo-controlled trial that investigated alirocumab, a different PCSK9 inhibitor, in 18,924 patients who had an acute coronary syndrome (ACS) between 1 and 12 months prior. 16 In addition, these patients had a LDL-C level of at least 70 mg/dL and were receiving statin therapy at either high-intensity or maximum tolerated dose. A composite of death from coronary heart disease, nonfatal MI, fatal or nonfatal ischemic stroke, or unstable angina requiring hospitalization occurred in 903 patients (9.5%) in the alirocumab group and in 1052 patients (11.1%) in the placebo group (HR 0.85, 95% CI 0.78 to 0.93; p < 0.001). A total of 334 patients (3.5%) in the alirocumab group and 392 patients (4.1%) in the placebo group died (HR 0.85, 95% CI 0.73 to 0.98). A sub analysis of patients with a history of PAD or VTE within the ODYSSEY OUTCOMES trial investigated the effect of alirocumab on PAD and VTE outcomes. 48 Compared to placebo, alirocumab demonstrated lower PAD-related events in 246 patients with PAD (HR 0.69, 95% CI 0.54 to 0.89; p = 0.004).
Inclisiran is another lipid-lowering therapy that targets the PCSK9 protein. The ORION trial was a phase two, multicenter, randomized trial that evaluated the effectiveness of inclisiran, a chemically synthesized small interfering RNA designed to target PCSK9 messenger RNA, on LDL-C levels. 49 Patients (n = 501) were assigned to receive either a single dose of placebo or 200, 300, or 500 mg of inclisiran on day 1, or two doses of placebo or 100, 200, or 300 mg of inclisiran on day 1 and day 90. After 180 days, all groups that received inclisiran had significantly lower LDL-C levels than placebo.
Ezetimibe
Ezetimibe is a nonstatin cholesterol-lowering medication that has been shown to be an effective supplementary therapy for patients with PAD when initiated along with statin therapy. The IMPROVE-IT trial evaluated patients (n = 18,144) over the age of 50 who had been hospitalized for an ACS event. 17 Patients were randomized to either ezetimibe (10 mg per day) ± simvastatin (40 mg per day) or placebo ± simvastatin (40 mg per day), with primary endpoints being a composite of CV death, MI, stroke, unstable angina leading to hospitalization, and coronary revascularization ⩾ 30 days post-randomization. After a median follow-up of 6 years, patients taking ezetimibe in addition to statin therapy demonstrated a significant reduction of the primary end point events by 7% compared to placebo (incidence-rate ratio (RR) 0.91, 95% CI 0.85 to 0.97; p = 0.007). Approximately 8% of patients in each treatment group had a history of PAD.
Furthermore, a subanalysis of the IMPROVE-IT trial evaluated the effectiveness of ezetimibe in patients with polyvascular disease including PAD (n = 1005), or previous stroke or transient ischemic attack (n = 1071). 50 Patients with polyvascular disease demonstrated higher rates of CV events (39.8%) than those without polyvascular involvement (29.6%). The benefit of ezetimibe was consistent in patients with and without polyvascular disease and type 2 diabetes.
Ethyl esters
Ethyl esters are also emerging as a therapy that can decrease cardiovascular morbidity and mortality by lowering both LDL-C and triglyceride levels. Icosapent ethyl has been studied in patients with CV disease and has demonstrated additional risk reduction in CV morbidity and mortality in addition to statin therapy. However, randomized trials on icosapent ethyl have only investigated patients with CAD.
The Reduction of Cardiovascular Events with Icosapent Ethyl-Intervention Trial (REDUCE-IT) was a phase 3 randomized controlled trial in which 8179 patients with CAD were assigned to receive either icosapent ethyl 4 g/day (2 g twice daily with food) or placebo. 51 At a mean follow-up of 4.9 years, patients taking icosapent ethyl were significantly less likely to experience a composite endpoint consisting of CV death, nonfatal MI, nonfatal stroke, coronary revascularization, or unstable angina (HR 0.75, 95% CI 0.68 to 0.83; p < 0.001). These promising results for icosapent ethyl in CAD patients indicate its potential as a lipid-lowering strategy for patients with PAD.
Clinical applications and guidelines
Guidelines
The 2018 AHA/ACC Guideline on the Management of Blood Cholesterol grouped PAD under major clinical atherosclerotic CV diseases (ASCVD) along with history of MI, stable or unstable angina, or coronary other arterial revascularization, stroke, transient ischemic attack. Furthermore, symptomatic PAD (history of claudication with ABI < 0.85, or previous revascularization or amputation) in addition to another ASCVD, or in addition to other high-risk conditions (age ⩾ 65, familial heterozygous hyperlipidemia, history of coronary revascularization, diabetes mellitus, hypertension, chronic kidney disease with an estimated glomerular filtration rate (eGFR) of 15–50 mL/min/1.73 m2, current smoking, persistent elevated LDL-C ⩾ 100 mg/dL or history of congestive heart failure), was considered very high risk for future ASCVD events. The ACC/AHA guideline indicated that in patients who are 75 years of age or younger with clinical ASCVD, including PAD, high-intensity statin therapy should be initiated or continued with the aim of achieving a 50% or greater reduction in LDL-C levels. 8 In patients with clinical ASCVD like PAD, in whom high-intensity statin therapy is contraindicated or who experience statin-associated side effects, moderate intensity statin therapy should be initiated or continued with the aim of achieving a 30–49% reduction in LDL-C levels. In patients with clinical ASCVD like PAD, who are judged to be very high risk for future ASCVD and who are on maximally tolerated LDL-C-lowering therapy with an LDL-C level of 70 mg/dL or higher or a non-high-density lipoprotein cholesterol level of 100 mg/dL or higher, it is reasonable to add a PCSK9 inhibitor following a clinician–patient discussion on the net benefit, safety, and cost. In patients with clinical ASCVD including PAD, who are on maximally tolerated statin therapy, are judged to be at very high risk for future ASCVD events, and have an LDL-C level of 70 mg/dL or higher, it is reasonable to add ezetimibe therapy. In patients older than 75 years of age with clinical ASCVD including PAD, it is reasonable to initiate moderate or high-intensity statin therapy after evaluation of the potential for ASCVD risk reduction, adverse effects, and drug–drug interactions, as well as patient frailty and preferences (Figure 1).
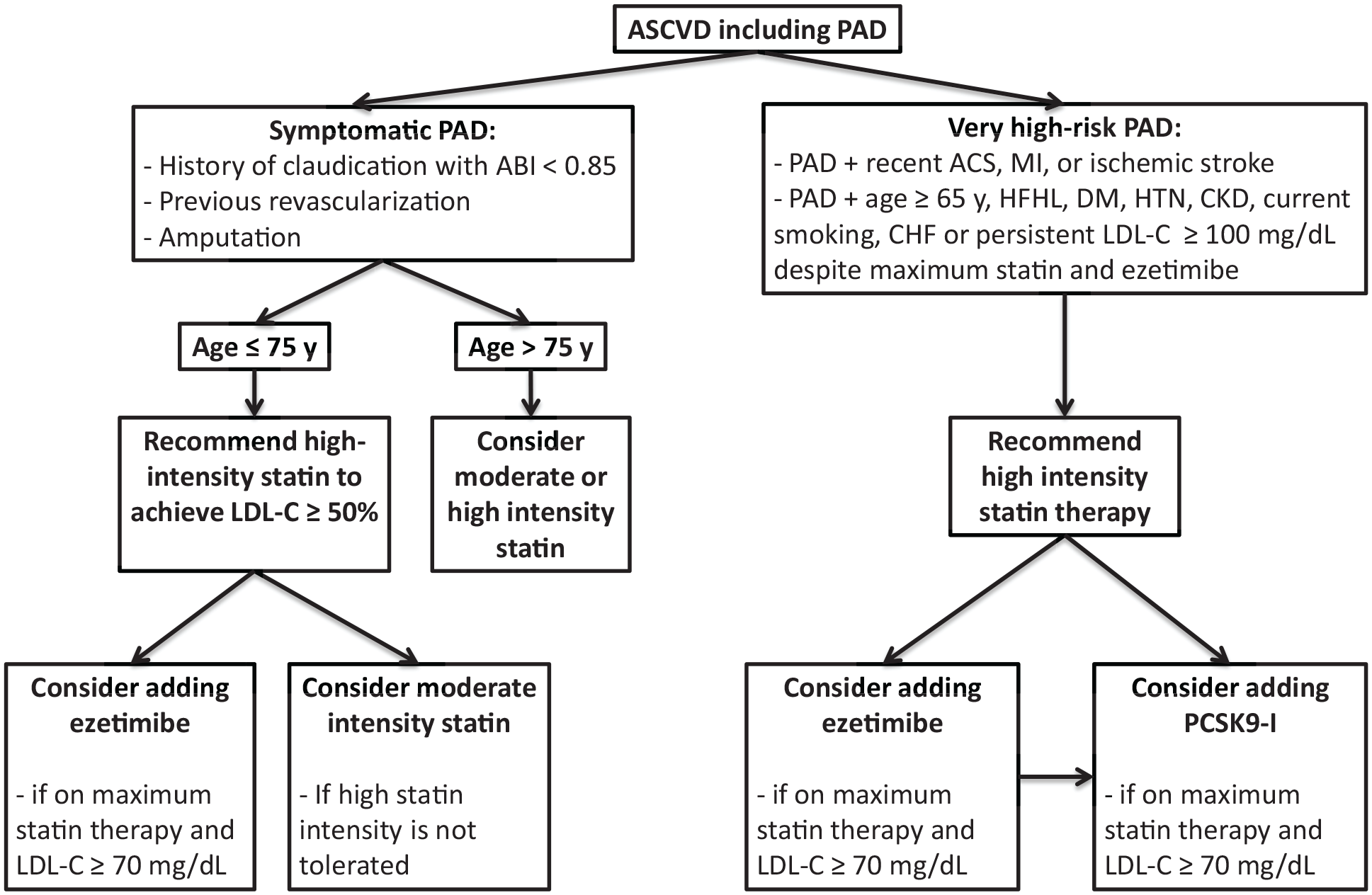
Summary of ACC/AHA 2018 lipid-lowering guideline recommendations for patients with ASCVD including PAD. 8
The 2019 ESC/EAS Guidelines for the Management of Dyslipidemias indicate that patients with PAD are at very high risk for ASCVD events, and should be managed with a therapeutic regimen to achieve ⩾ 50% LDL-C reduction and an LDL-C goal of < 1.4 mmol/L (< 55 mg/dL). Patients with no current statin use will likely require high-intensity LDL-lowering therapy. Patients with current LDL-lowering treatment will require an increased treatment intensity. 52 In patients with PAD, lipid-lowering therapy, including a maximum tolerated dose of statin, plus ezetimibe or a combination with a PCSK9 inhibitor if needed, is recommended to reduce the risk of ASCVD events.
Barriers
Despite current guidelines and evidence of the benefit of statins and PCSK9 inhibitors, patients with PAD continue to be undertreated with lipid-lowering therapies. A study published in 2020 by Reynolds et al. examined statin utilization in 11,059 patients with severe PAD, including some with CLI, from 2002 to 2015. 6 This study demonstrated that although there was an increase in statin utilization from 50% in 2002 to 66% 2015, statin therapy in patients with PAD remains suboptimal. In addition, a study by Berger and Ladabo published in 2017 demonstrated an underuse of medical therapy and lifestyle counseling in patients with PAD, with statin therapy only utilized in 33% of patients. 53 To highlight the variability in utilization rates, a meta-analysis of eight studies examined the rate of statin usage in 67,915 patients with PAD and found high variability of statin usage between sites, ranging from 11% to 79%. 54
In addition to the underutilization of statin prescriptions by providers, there are also patient-related challenges to incorporate lipid-lowering strategies into the management of PAD. The main side effects of statins that usually lead to patient noncompliance are myalgia and muscle cramps reported in 5–10% of patients. 55 In addition, other rare but serious side effects include rhabdomyolysis, which is estimated to affect two cases/10,000 person-years. While the FOURIER and ODYSSEY OUTCOMES trials reported no major unacceptable side effects, including injection site reactions (3.4%) and muscle symptoms (3.8%), PCSK9 inhibitors are expensive and therefore not available to all patients with PAD.15,16 Despite the potential side effects and barriers, statins and PCSK9 inhibitors have demonstrated a net benefit effect in lowering LDL-C and improving outcomes in patients with PAD.
Limitations
Much of the evidence on lipid-lowering agents in patients with PAD has been abstracted from trials that mainly enrolled patients with CAD. Consequently, many of the studies in this review had smaller sample sizes and therefore were not completely powered to evaluate for a clear benefit in patients with PAD.
Conclusions
Current professional society guidelines recommend statin therapy for all individuals with PAD.8,9 In current literature, statins have been associated with improved mortality, MACE, MALE, and symptomatic outcomes in these patients. In addition, PCSK9 inhibitors are also emerging as a viable additional lipid-lowering therapy. However, despite current guideline recommendations and evidence to support the benefit of statins and PCSK9 inhibitors, patients with PAD are consistently undertreated due to both prescriber and patient-related factors. We hope that this review will increase awareness of the net benefit of lipid-lowering therapies. We call for more research to study the benefits of lipid-lowering therapies, focusing on mainly PAD populations.
Footnotes
Acknowledgements
The authors would like to thank the Minneapolis Heart Institute Foundation for their support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
