Abstract
High-risk pulmonary embolism (PE) requires hemodynamic and respiratory support along with reperfusion strategies. Recently updated European guidelines assign a low class of recommendation to extracorporeal membrane oxygenation (ECMO) for high-risk PE. This systematic review assessed clinical outcomes after ECMO in high-risk PE. We searched electronic databases including PubMed, Embase and Web of Science from January 2000 to April 2020. Efficacy outcomes included in-hospital survival with good neurological outcome and survival at follow-up. Safety outcomes included lower limb ischemia and hemorrhagic and ischemic stroke. Where possible (absence of high heterogeneity), meta-analyses of outcomes were undertaken using a random-effects model. We included 16 uncontrolled case-series (533 participants). In-hospital survival with good neurological outcome ranged between 50% and 95% while overall survival at follow-up ranged from 35% to 95%, both with a major degree of heterogeneity (I2 > 70%). The prevalence of lower limb ischemia was 8% (95% CI 3% to 15%). The prevalence of stroke (either hemorrhagic or ischemic) was 11% (95% CI 3% to 23%), with notable heterogeneity (I² = 63.35%). Based on currently available literature, it is not possible to draw definite conclusions on the usefulness of ECMO for high-risk PE. Prospective, multicenter, large-scale studies or nationwide registries are needed to best define the role of ECMO for high-risk PE.
Keywords
Introduction
Pulmonary embolism (PE) is the third most frequent cardiovascular disease representing a major public health concern. 1 The clinical presentation of PE is very broad, spanning from asymptomatic cases discovered incidentally to severe PE with arterial hypotension, cardiogenic shock or even cardiac arrest. 2 The latter situation, defined as high-risk PE, requires an expedited multidisciplinary approach including hemodynamic and respiratory support along with pharmacological, surgical or interventional reperfusion strategies. 3 Despite an increased use of extracorporeal membrane oxygenation (ECMO) in the last decade, 4 recently updated European guidelines assign a low class of recommendation to this temporary mechanical circulatory support in the management of high-risk PE. 3
To best define the role of ECMO in this specific subgroup of critically ill patients, we aimed to systematically review published literature reporting clinical outcomes after ECMO for high-risk PE.
Methods
We reported this review according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 5 We registered a protocol for this review in PROSPERO (CRD42019136282).
Eligibility criteria
Our population of interest was represented by adult patients (age > 18 years) with confirmed diagnosis of PE complicated by cardiogenic shock or refractory cardiac arrest. The intervention of interest was the implantation of veno-arterial ECMO. This means veno-venous ECMO was excluded.
We included studies of any design and any type except letters, editorials, commentaries, and conference abstracts. We excluded original articles that did not focus specifically on outcomes following ECMO used for PE complicated by cardiac arrest or cardiogenic shock. As a result, we excluded articles with broader scope, which only reported the outcomes after ECMO used for PE as part of subgroup or multivariate analyses. This was decided since these articles did not report sufficient information regarding patients’ characteristics. We also excluded original articles with fewer than 10 participants. Comparators could be any strategy with maximal optimal medical treatment without ECMO.
The efficacy outcomes were in-hospital survival with good neurological outcome, survival at follow-up, and successful ECMO weaning. In-hospital survival with good neurological outcome was defined as survival with a cerebral performance category (CPC) score of 1–2 on a 5-point scale. 6 The safety outcomes were lower limb ischemia, dialysis, hemorrhagic and ischemic stroke, and groin bleeding requiring surgical revision. Other outcomes were economic outcomes (costs of strategy, cost-effectiveness of ECMO vs no ECMO), duration of ECMO support (days), and predictors of survival outcomes.
Search strategy and study selection
English language studies were searched in electronic databases (PubMed [including Medline], Embase [Ovid], Cochrane Library [Wiley], Web of Science [Clarivate Analytics], and Scopus [Elsevier]) in February 2019 for relevant literature published from January 2000. This time frame was purposely chosen to analyze the results according to modern ECMO technology and more advanced expertise in the management of high-risk PE. We used a combination of free-text and thesaurus terms for the concepts relevant to the topic (see online Supplemental Material 1). Search equations were defined by an experienced information specialist (CG). Gray literature was also considered through the analysis of references found in Google with a query by exact thematic expressions. The ClinicalTrials.gov and PROSPERO databases were also queried to ensure the value of a systematic review. An automated alert regarding new publications becoming available during the course of the review was created and an update was done on September 15, 2019. A final update was done covering the period up to April 2020.
Three independent reviewers (AMe, MP, XA) screened all identified bibliographic records for title/abstract and then for full text. Disagreements were discussed and resolved through consensus. The study flow and reasons for exclusion at the full-text level were documented in the PRISMA flow chart (Figure 1).
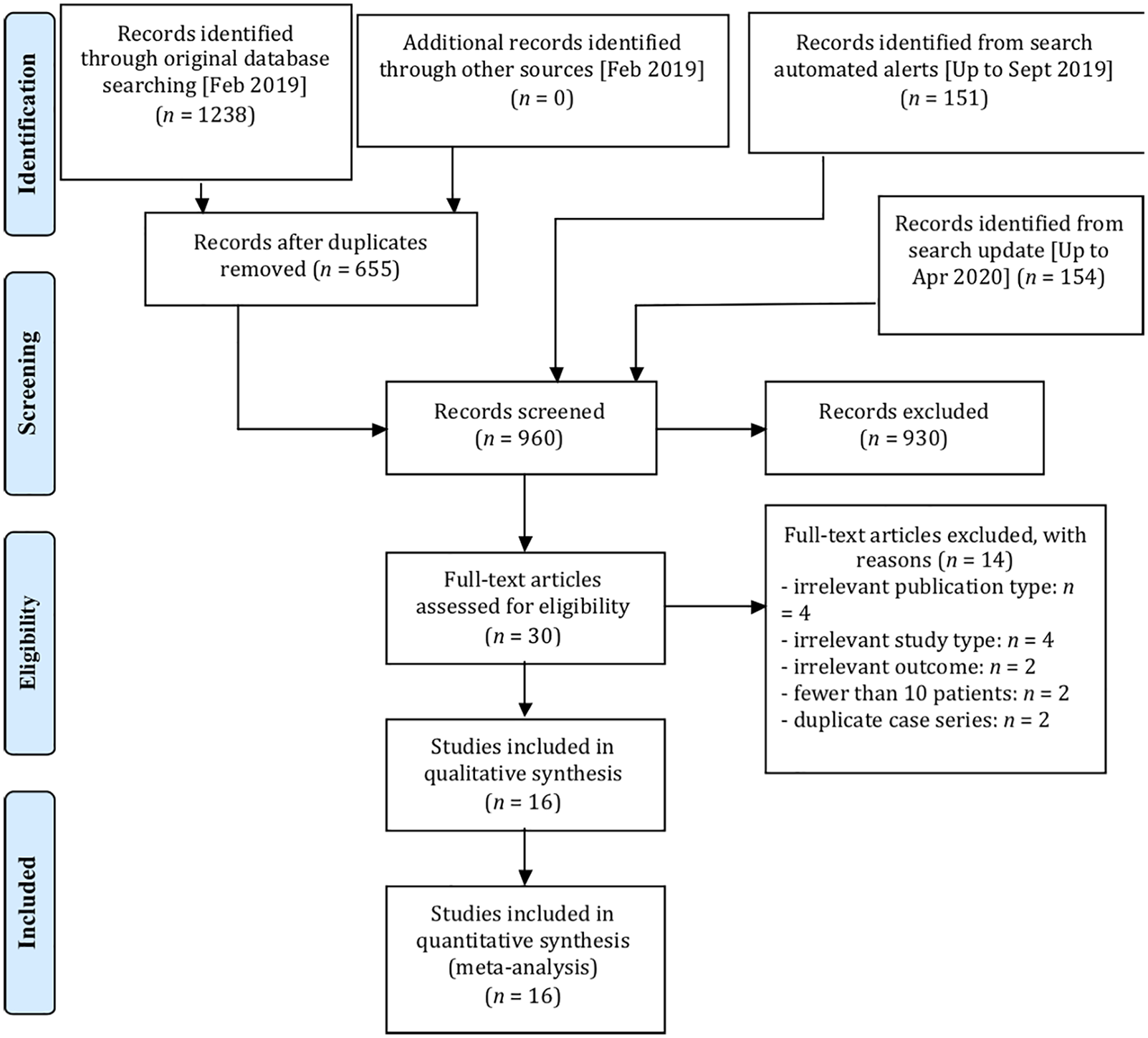
PRISMA flow chart.
Data extraction and quality assessment
One reviewer (MP) extracted relevant data using an a priori defined pre-piloted extraction sheet and completed data extraction sheets were cross-checked by another reviewer (AMe). In addition to the outcomes of interest, we extracted relevant information regarding the baseline characteristics of patients, type of PE treatment (thrombolysis, catheter-directed therapy, surgical embolectomy, ECMO alone) and period of follow-up.
Quality assessment was undertaken by two reviewers (AMa and XA) and cross-checked between the reviewers. Any disagreements between reviewers were resolved through a discussion. We assessed study quality using checklists published by the US National Institutes of Health according to each identified study design. 7 We rated the overall quality of studies as poor, fair or good, accounting for each study’s limitations as emphasized by the items (nine in total) within the checklists. More precisely, the decision to rate as poor, fair or good was made by consensus of two reviewers (AMa and XA) based on the number of YES answers provided on each item (a YES answer denoting higher quality). An article had to have eight to nine items with YES answers to be rated good quality, five to seven items with YES answers to be rated fair quality, and fewer than five items with YES answers to be rated poor quality (see online Supplemental Material 2).
Data synthesis and analysis
The review results were organized in texts and in Table 1, which tabulated characteristics, results for included primary studies, and study quality.
Summary table of the included studies.
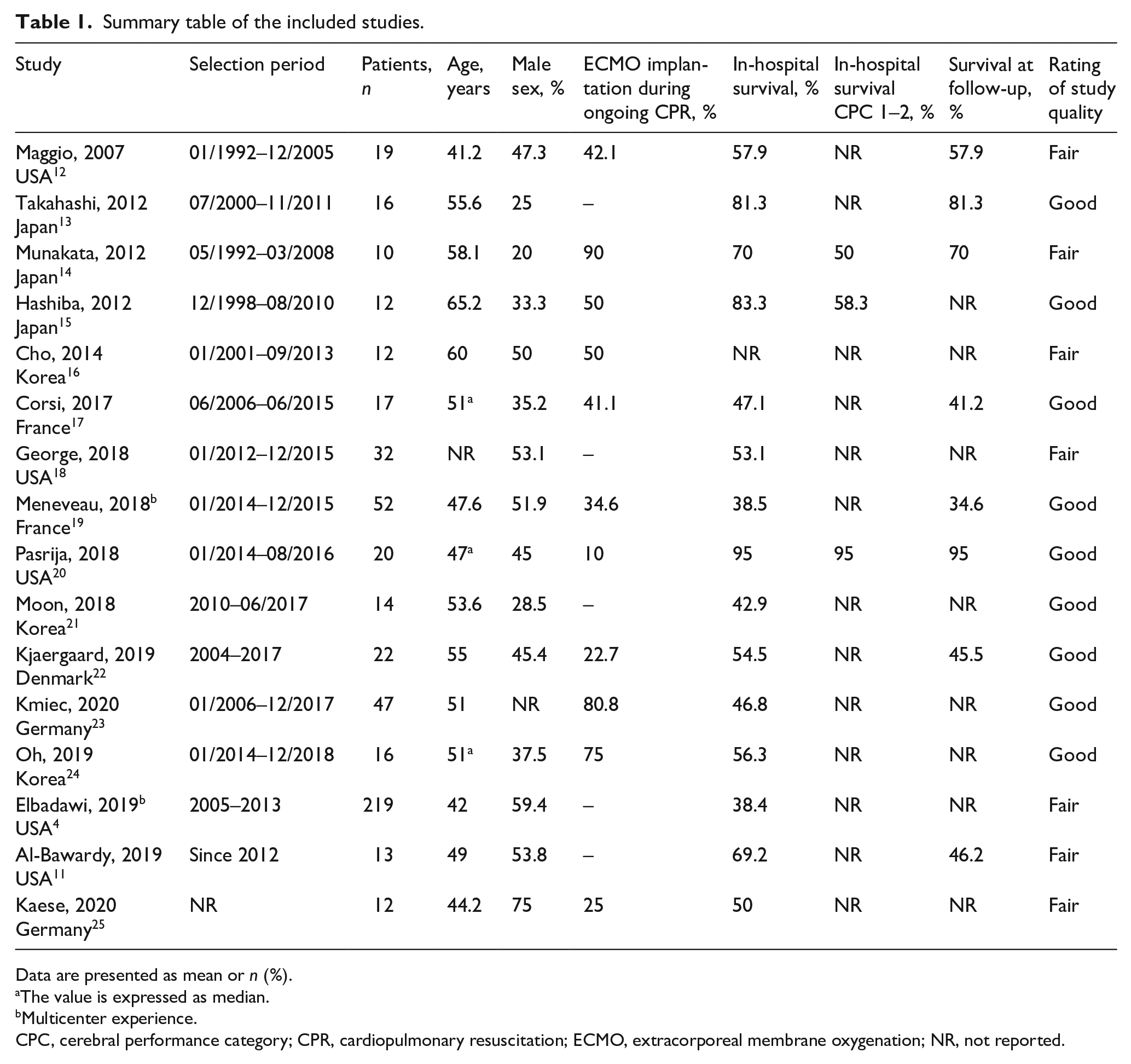
Data are presented as mean or n (%).
The value is expressed as median.
Multicenter experience.
CPC, cerebral performance category; CPR, cardiopulmonary resuscitation; ECMO, extracorporeal membrane oxygenation; NR, not reported.
We built 95% CIs of the estimates (for both efficacy and safety outcomes) using the Wilson method. 8 We attempted to calculate a pooled estimate for proportions with a random-effects model to account for the likely heterogeneity across studies using the metaprop command on Stata 16.0 (StataCorp LLC, College Station, TX, USA). 9 In all analyses, we assessed the heterogeneity of findings using the Cochran’s Q test (p-value threshold of 0.05) and calculating the I2 statistic. In the event of substantial heterogeneity (defined as I2 > 70%), we chose not to report any pooled estimates and explored reasons of heterogeneity using random-effects meta-regressions and subgroup analyses accounting for variables with confounding potential. Based on available data (i.e. those reported consistently across studies), we explored reasons of heterogeneity for the variables age, proportion of patients with PE complicated with cardiac arrest, and proportion of patients who had subsequent surgical embolectomy by doing meta-regressions, which were undertaken when at least 10 studies could be included in the analyses. Last, we attempted to detect small-study bias using funnel plot and undertook the Egger’s test when there was the same minimal number of studies. 10
Results
Of 960 records identified and screened at title/abstract level, 30 were examined for full text, of which 16 studies enrolling a total of 533 participants were included (Figure 1).4,11–25
Characteristics of included studies
Eleven (68.7%) of the 16 included studies were published in 2017–2020.4,11,17–25 Eleven were conducted in Asia (n = 6)13–16,21,24 or in the USA (n = 5),4,11,12,18,20 the remaining five being from European centers.17,19,22,23,25 The majority of articles (87.5%) described single-center experiences, which explains the small sample sizes (from 10 to 47) across these studies.11–18,20–25 Peripheral ECMO was predominantly used (97.3% of cases) over central ECMO. In the majority of studies, the enrolled population were heterogeneous with regards to high-risk PE (those who had PE complicated by cardiac arrest as opposed to those who had cardiogenic shock). The same was observed with regards to subsequent first or second-line treatment (thrombolysis, catheter-directed embolectomy or surgical embolectomy).
In 11 studies, there was no comparator group (uncontrolled case-series design).4,11–14,17,18,20,22,24,25 In three studies, authors compared the outcomes after ECMO with a group of patients who did not receive ECMO (non-randomized controlled study design).16,19,21 However, the comparability of both groups was deemed questionable because of the small sample size of comparator groups in two studies (fewer than 10 patients),16,21 which was potentially not sufficient to identify true differences in patients groups, or because of insufficient description of the comparator group. 19
For these reasons, quality assessment of the 16 included studies was made using the tool specific to case-series. 7 For those three studies with a comparator group, we only took the ECMO group in subsequent sections of the article. Nine studies were rated as being of good quality,13,15,17,19–24 while seven were rated as being of fair quality.4,10,12,14,16,18,25 The main limitations across the studies were the presence of apparent differences across the enrolled population (with regards to duration of cardiopulmonary resuscitation, age, PE complicated by cardiac arrest or cardiogenic shock, and subsequent therapy received after ECMO) or the absence of attempts made by authors to identify clinical predictors of survival. Full quality assessment of studies is reported in the online Supplemental Material 2. Overall, by nature, these studies were associated with a low level of evidence.
Efficacy outcomes
In-hospital survival with good neurological outcome ranged between 50% and 95%, with high heterogeneity (I2 = 79.9%; three studies), which could not be explained by apparent reasons based on patients’ characteristics. In-hospital survival was reported in 15 studies and ranged between 38.4% and 95%, again with a considerable degree of heterogeneity (I2 = 73.7%); none of the meta-regressions were successful in explaining these between-studies differences (see online Supplemental Material 3). We found no funnel plot asymmetry, which therefore does not suggest a risk of small-study bias (see online Supplemental Material 4).
The same observations were made for overall survival at follow-up (range 34.6% to 95%; I2 = 79.8%; eight studies). The proportion of successful ECMO weaning ranged between 36.5% and 100% (10 studies). Data available were not sufficient to allow statistical explorations of reasons of heterogeneity, which was very high (I2 = 79.1%). For each outcome, forest plots (with or without pooled estimates depending on degree of heterogeneity) are reported in Figure 2, while relevant meta-regression plots are reported in online Supplementary Material 3.
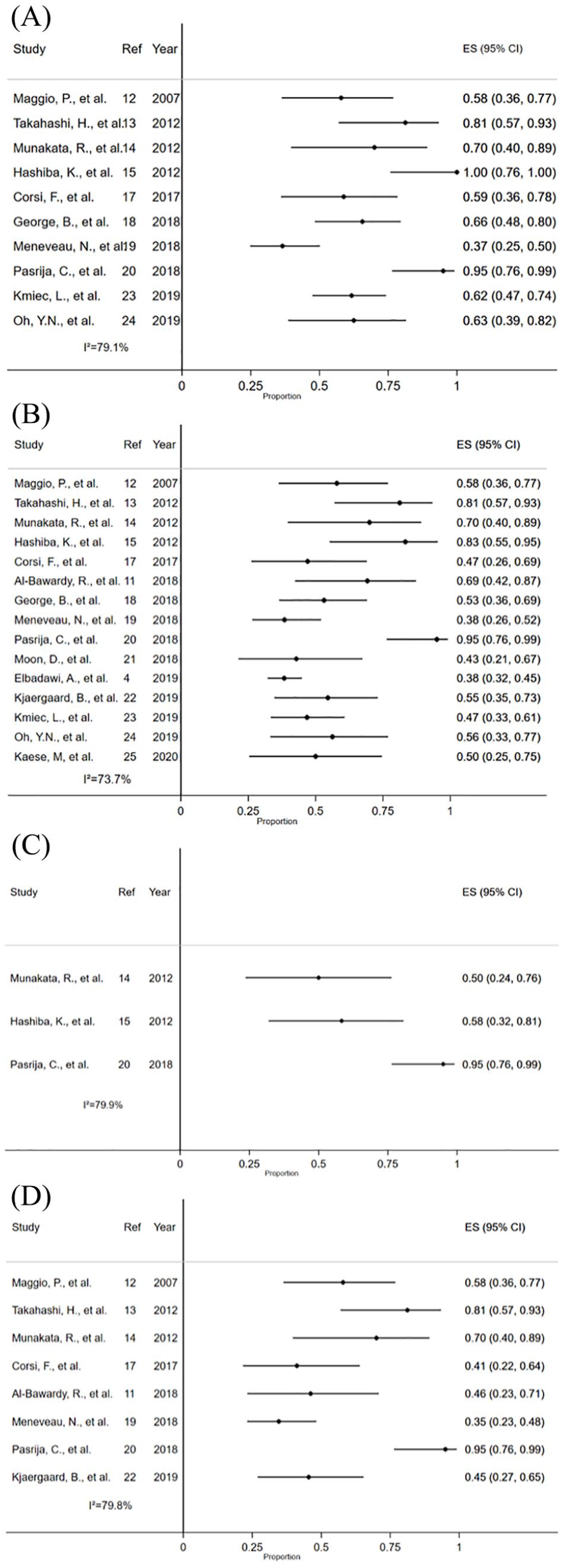
Forest plot of the efficacy outcomes: (A) successful ECMO weaning; (B) in-hospital survival; (C) in-hospital survival with good neurological outcome; and (D) survival at follow-up.
Safety outcomes
The prevalence of lower limb ischemia ranged from 6.3% to 14.3%, with a pooled estimate of 8% (95% CI 3% to 15%; I2 = 0%; four studies). Only one study detailed the revascularization strategy adopted (lower limb ischemia in two patients, one each underwent lower limb or toe amputation). 17 In three studies that reported results on patients who required dialysis, the prevalence was very heterogeneous (I2 = 83.6%) with no apparent reasons, ranging from 20% to 76.5%. Only one study reported specifically the occurrence of ischemic stroke 17 and a different one the occurrence of hemorrhagic stroke. 18 Based on five studies, the prevalence of stroke (either hemorrhagic or ischemic) ranged from 3.1% to 35.7% with a pooled estimate of 11% (95% CI 3% to 23%) and a notable degree of heterogeneity (I2 = 63.3%). Figure 3 depicts the forest plot for safety outcomes. Only one study reported the onset of groin bleeding requiring surgical revision in one out of 10 patients (10%). 14
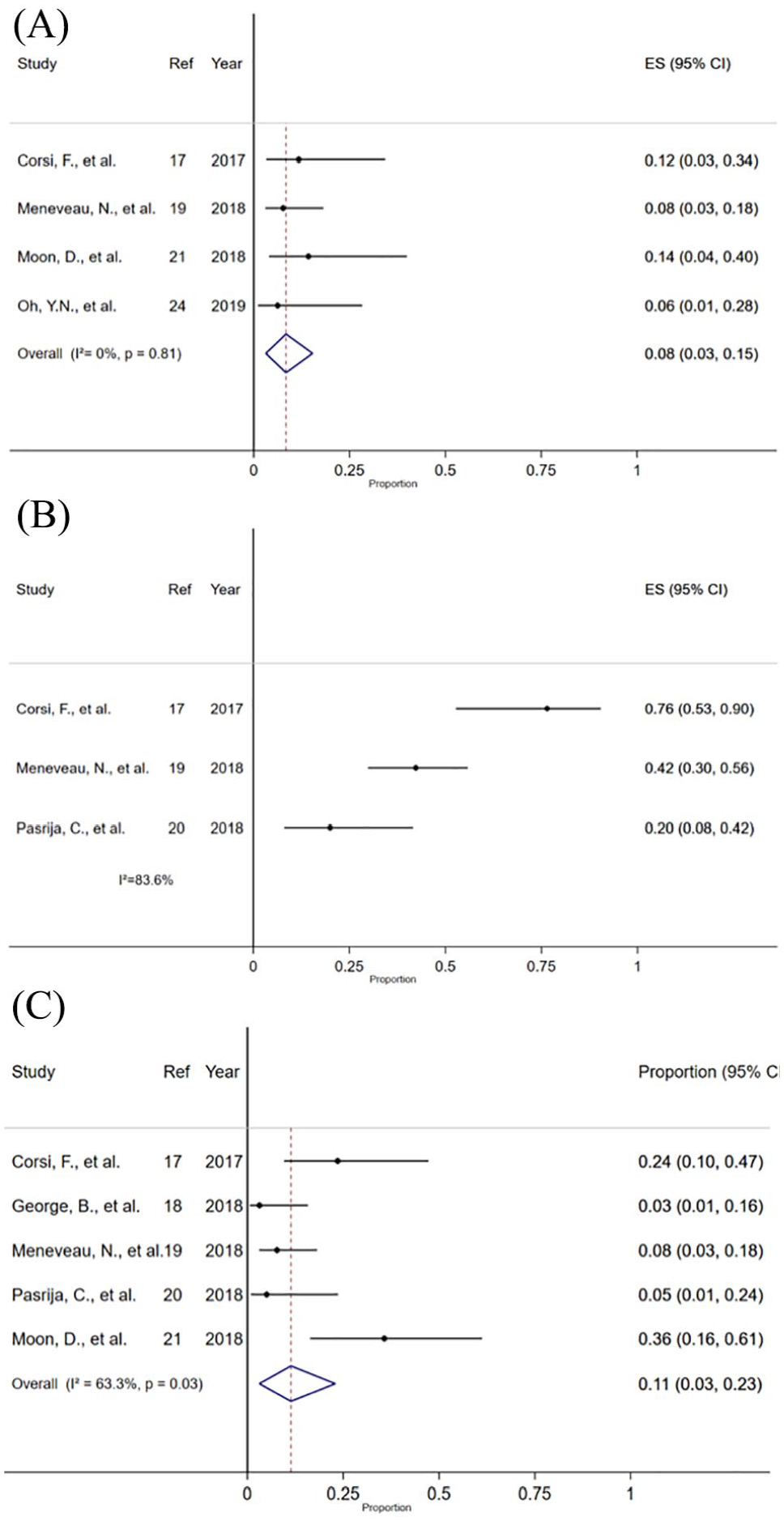
Forest plot of the safety outcomes: (A) lower limb ischemia; (B) dialysis; and (C) stroke (ischemic and hemorrhagic).
Other outcomes
We found no studies that reported the cost or cost-effectiveness of ECMO. The duration of ECMO support ranged between 1.0 and 5.5 days. Two studies reported analyses of factors associated with survival outcomes.4,16 Hypertension and cardiopulmonary resuscitation duration > 15 minutes were associated with poorer survival, 16 while other factors identified were older age, female sex, congestive heart failure, chronic pulmonary disease, and obesity. 4
Discussion
Although epidemiological data found decreasing case fatality rates over time, PE still represents a leading cause of morbidity and mortality worldwide. 26 Patients with high-risk PE display mortality rates ranging from 25% in cases of refractory cardiogenic shock to 65% in the presence of cardiac arrest requiring cardiopulmonary resuscitation.27,28
Nevertheless, growing evidence suggests that an aggressive program, including ECMO, may improve survival of high-risk PE, 29 the overall experience with ECMO for high-risk PE is sparse. In the absence of a published systematic review, we have comprehensively screened and assessed the published literature reporting the outcomes of ECMO for high-risk PE. The major finding was that in-hospital survival was reported at between 38% and 95%, displaying a high heterogeneity across the studies.4,11–15,17–25 In articles providing detailed information, in-hospital survival with good neurological outcome was between 50% and 95%.14,15,20 Different reasons could explain this variability. First, the majority (13 out of 15) of the articles reporting in-hospital mortality are single-center case series and selection bias could not be formally excluded. The small sample size (range: 10–47 patients) of these articles could at least partially explain this extreme variability in published in-hospital mortality. Indeed, the higher mortality observed here stem from two multi-institutional experiences analyzing real-world data.4,19 Prospective, multicenter, nationwide registries should be strongly encouraged to overcome these intrinsic limitations and offer practical information to best define the role of ECMO in the management of high-risk PE. Moreover, the choice of the reperfusion strategy in combination with ECMO could have an impact on the overall survival of patients with high-risk PE. The decision-making algorithm should be done on a case-by-case basis and often depends on the expertise of each institution. Systemic thrombolysis during ECMO support should be avoided owing to a prohibitive risk of lethal hemorrhagic complications (in particular intracranial bleeding).12,20 Anticoagulation alone proved successful in six out of 11 survivors in a preliminary experience of 19 ECMO for high-risk PE. 12 In the modern era, anticoagulation-induced clot dissolution and spontaneous fibrinolysis allowed for a 47% long-term survival of 17 ECMO-treated patients with life-threatening massive PE. 17 However, the role of ECMO as a stand-alone technique with anticoagulation, as well as the benefit of additional mechanical clot-removal therapies, is controversial and needs further investigation. 3 Although surgical pulmonary embolectomy has historically been considered as the treatment of last resort for high-risk PE,30–32 more recent analyses showed improved outcomes.13,20 These experiences shared an aggressive approach as a ‘bridge to surgery’ through prompt preoperative ECMO implantation in order to improve the hemodynamic status and end-organ functions, and to perform surgical embolectomy in better conditions. A large, multicenter series including 52 patients found a significant survival advantage in the group treated with ECMO in combination with surgical embolectomy as compared to the groups with ECMO as a stand-alone treatment or after failed thrombolysis. 19 As suggested by Pasrija et al., an integrated approach to ECMO for high-risk PE seems a reasonable solution, with an initial period (5 days) of mechanical support with anticoagulation alone waiting for end-organ function recovery followed by surgical or percutaneous embolectomy only in case of right ventricular dysfunction with persistent pulmonary thrombus. 20 When pulmonary embolectomy could not be performed owing to the critical conditions of high-risk patients with PE, percutaneous catheter-directed treatments may represent an alternative in such cases. George et al. reported promising results with a 73% in-hospital survival in patients who received concomitant catheter-directed thrombolysis and ECMO support in a retrospective comparative series. 18 Whatever the reperfusion strategy, data reported here suggest that survival at follow-up was quite similar to in-hospital survival, showing that acute-phase survivors display satisfactory intermediate-term survival.11–14,17,19,20,22 Finally, the concept of a pulmonary embolism response team (PERT) appeared recently in the USA in an effort to provide a multidisciplinary approach to high-risk PE.33,34 As clearly stated by the latest European Society of Cardiology guidelines, the exact composition and operating mode of a PERT are not fixed, 3 probably contributing further to these extremely variable outcomes of high-risk patients with PE managed with ECMO. Future efforts should be addressed to a better identification and standardization of local PERTs, following the concept of the Heart Team in the setting of valvular heart and coronary artery disease.35,36
Despite its ease of implantation, management and decannulation, ECMO displays various complications. 37 The articles included in this report allowed for an analysis of lower limb ischemia, renal injury requiring renal replacement therapy, and neurologic complications. Lower limb ischemia is a serious complication of peripheral ECMO, whose physiopathology is multifactorial. 38 We found a prevalence of lower limb ischemia of 8% (95% CI 3% to 15%) using data from four studies.17,19,21,24 Conversely, Bonicolini et al. reported in a narrative review including 28 studies an incidence of limb ischemia in peripheral femoro-femoral ECMO ranging from 10% to 70%. 38 The authors explained this highly variable incidence as a consequence of the extreme difference across the studies in terms of baseline characteristics, ECMO indications, cannulation techniques, limb ischemia definition, detection tools, distal perfusion cannula modalities, and timing of insertion. The analysis of renal injury requiring renal replacement therapy and neurologic complications found an extreme heterogeneity across the studies. Consequently, the interpretation of such results could potentially lead to erroneous conclusions; hence these results should be viewed cautiously.
Limitations
Our study presents several limitations. Despite our efforts to use a systematic search approach, the potential for publication bias cannot be excluded as we may have missed some unpublished relevant studies with findings not favorable to ECMO. Similarly, we have not included findings from articles with broad scope, which may have only reported the outcomes after ECMO used for PE as part of subgroup or multivariate analyses. Consequently, the main limitation of the present review is the limited data it relied upon. Moreover, the lack of baseline patients’ characteristics, granularity on ECMO timing, and comparison among centers regarding expertise in intensive care units as well as surgical care for patients with PE could undermine the statistical power of our analysis, limiting the possibility to better understand the heterogeneity across studies. Another limitation is that we have undertaken a meta-regression to test the influence of age (using mean age reported in each study) on in-hospital survival. Age being a type of participant characteristic that can vary substantially within studies, and can only be summarized at the study level, it is possible our meta-regression could not identify properly the effect of age on in-hospital survival (ecologic bias). 39 Last, we have estimated the heterogeneity of our results by the statistic I2, which may be biased in meta-analyses with a small number of studies. 40
Conclusion
Exhaustive and generalizable conclusions on the usefulness of ECMO for high-risk PE are difficult to obtain from the available studies in the literature. In order to best define the role of ECMO in the setting of high-risk PE, we recommend future research under the form of prospective, multi-institutional, large-scale studies or nationwide registries.
Supplemental Material
5._Supplemental_Material_final_clean_copy – Supplemental material for Efficacy and safety of extracorporeal membrane oxygenation for high-risk pulmonary embolism: A systematic review and meta-analysis
Supplemental material, 5._Supplemental_Material_final_clean_copy for Efficacy and safety of extracorporeal membrane oxygenation for high-risk pulmonary embolism: A systematic review and meta-analysis by Matteo Pozzi, Augustin Metge, Anthony Martelin, Caroline Giroudon, Justine Lanier Demma, Catherine Koffel, William Fornier, Pascal Chiari, Jean Luc Fellahi, Jean Francois Obadia and Xavier Armoiry in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
