Abstract
This expert consensus statement on the interpretation of peripheral arterial and venous spectral Doppler waveforms was jointly commissioned by the Society for Vascular Medicine (SVM) and the Society for Vascular Ultrasound (SVU). The consensus statement proposes a standardized nomenclature for arterial and venous spectral Doppler waveforms using a framework of key major descriptors and additional modifier terms. These key major descriptors and additional modifier terms are presented alongside representative Doppler waveforms, and nomenclature tables provide context by listing previous alternate terms to be replaced by the new major descriptors and modifiers. Finally, the document reviews Doppler waveform alterations with physiologic changes and disease states, provides optimization techniques for waveform acquisition and display, and provides practical guidance for incorporating the proposed nomenclature into the final interpretation report.
This document has been endorsed by the Society for Vascular Medicine and the Society for Vascular Ultrasound and is copublished in Vascular Medicine and the Journal for Vascular Ultrasound. The contributions are identical except for minor differences in keeping with each journal’s style.
Introduction
Noninvasive spectral Doppler waveform assessment is a principal diagnostic tool used in the diagnosis of arterial and venous disease states. With 200 million people affected by peripheral artery disease worldwide1,2 and > 600,000 hospital admissions yearly for venous thromboembolic disease in the United States,3,4 establishment and adoption of nomenclature for spectral Doppler waveform characterization is essential for communication of disease presence and severity. Overlapping and conflicting Doppler waveform terminologies are considerable,5,6 and awareness of this issue was first published more than a decade ago. 7 Recognizing the need for standardization, the Society for Vascular Medicine (SVM) and the Society for Vascular Ultrasound (SVU) commissioned a writing committee to develop an expert consensus document on the nomenclature for spectral Doppler waveforms. The intent of this expert consensus document is to propose a common nomenclature for the description of arterial and venous Doppler waveforms in order to facilitate improved communication among all practitioners who care for vascular patients. The scope of the document is listed below, and a summary of the major consensus points is listed in Table 1. The writing committee was comprised of sonographer and physician members from SVM and SVU based on their extensive experience in the performance, interpretation, research, and/or teaching contributions to the field of vascular ultrasound. This document was peer reviewed within the SVM and SVU and endorsed by the SVM Board of Trustees and SVU Board of Directors.
Summary of consensus points.

Scope of the consensus document
The purpose of this consensus statement is to:
• Clarify and standardize key definitions and descriptors that are inherent to the analysis of arterial and venous Doppler waveforms.
Review Doppler waveform alterations with physiologic changes and disease states.
• Provide Doppler transducer optimization techniques to enhance the quality and presentation of Doppler spectral waveform and color Doppler data.
• Provide guidance for applying waveform descriptors and modifiers – sonographer’s report and physician’s final interpretation.
History
The initial descriptions of peripheral arterial Doppler waveforms were based on: (1) the audible presence or absence of the systolic and diastolic components of the cardiac cycle; and (2) the display of these flow components relevant to the zero-flow baseline on a recorded strip chart display. These characteristics were the origin of the Doppler waveform descriptors triphasic, biphasic, and monophasic 8 and the historical foundation for classifying blood flow in peripheral arteries.9,10
The triphasic waveform was recognized audibly and displayed as three distinct components of the cardiac cycle: (1) systole; (2) early diastolic flow reversal; and (3) a small forward flow reflective wave in late diastole (Figure 1A). The late diastolic component was absent in a biphasic waveform, leaving only systolic upstroke and early diastolic flow reversal as the audible and displayed cardiac components (Figure 1B). The original description of a monophasic waveform included the observation that the waveform did not cross the zero-flow baseline and was referred to as ‘unidirectional’. 10 Following Doppler technological advancements, the monophasic term was used to describe waveforms that did not retain early diastolic flow reversal or lacked the reflective wave in late diastole. In early publications, monophasic waveforms exhibited only one audible or waveform display component that was likely caused by an arterial obstruction (Figure 1C).
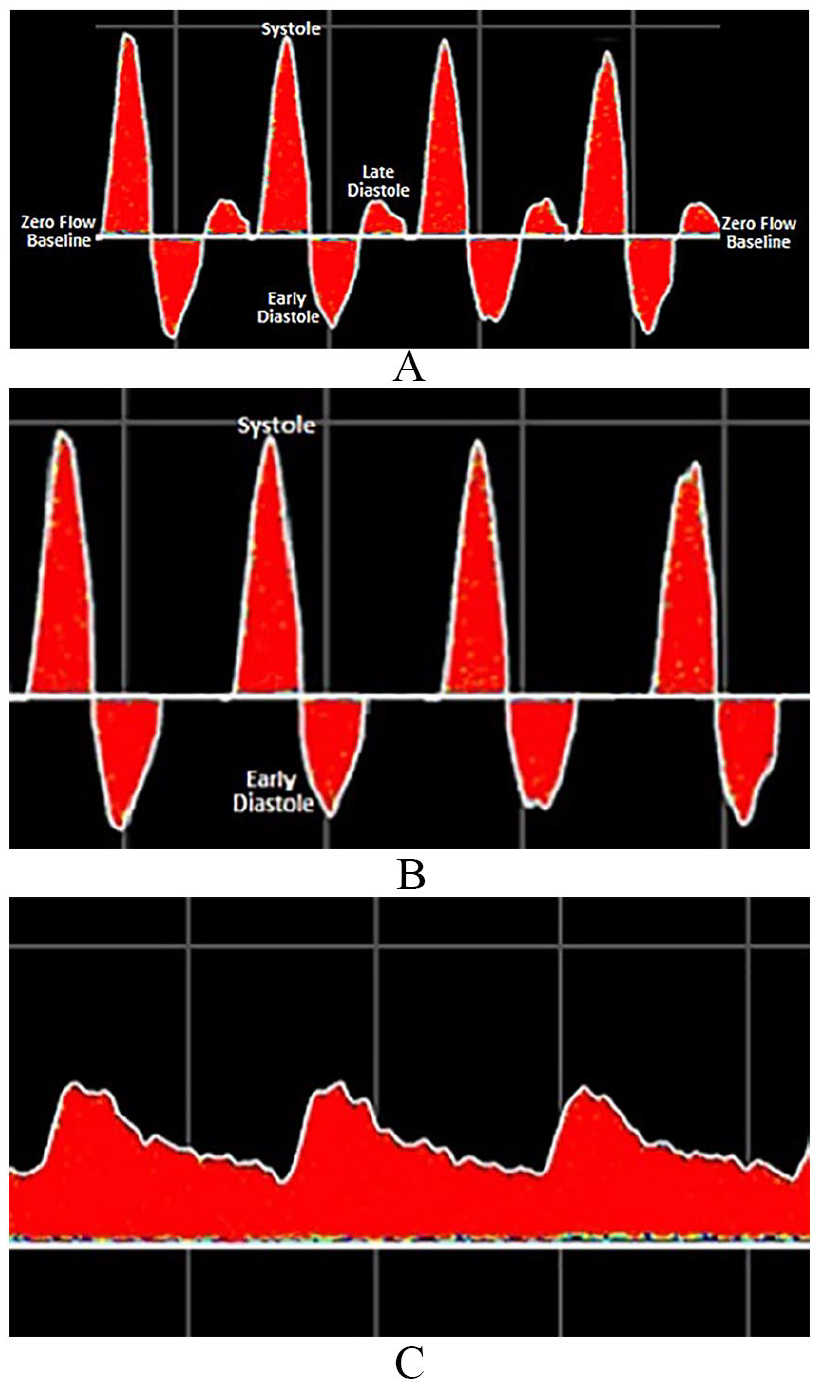
Triphasic (A), biphasic (B), and monophasic (C) Doppler waveforms.
The introduction of duplex ultrasound in the late 1970s was a major shift from indirect physiologic testing and the first significant change in Doppler waveform analysis. Duplex technology enabled the display of the full spectrum of frequencies within the Doppler sample volume. In the early days of duplex waveform analysis, spectral broadening was attributed to the spectral waveform features observed in areas of disturbed or turbulent flow. 11 These characteristics resulted in the addition of waveform descriptors such as ‘laminar’, ‘disturbed’, or ‘turbulent’ (Figure 2).
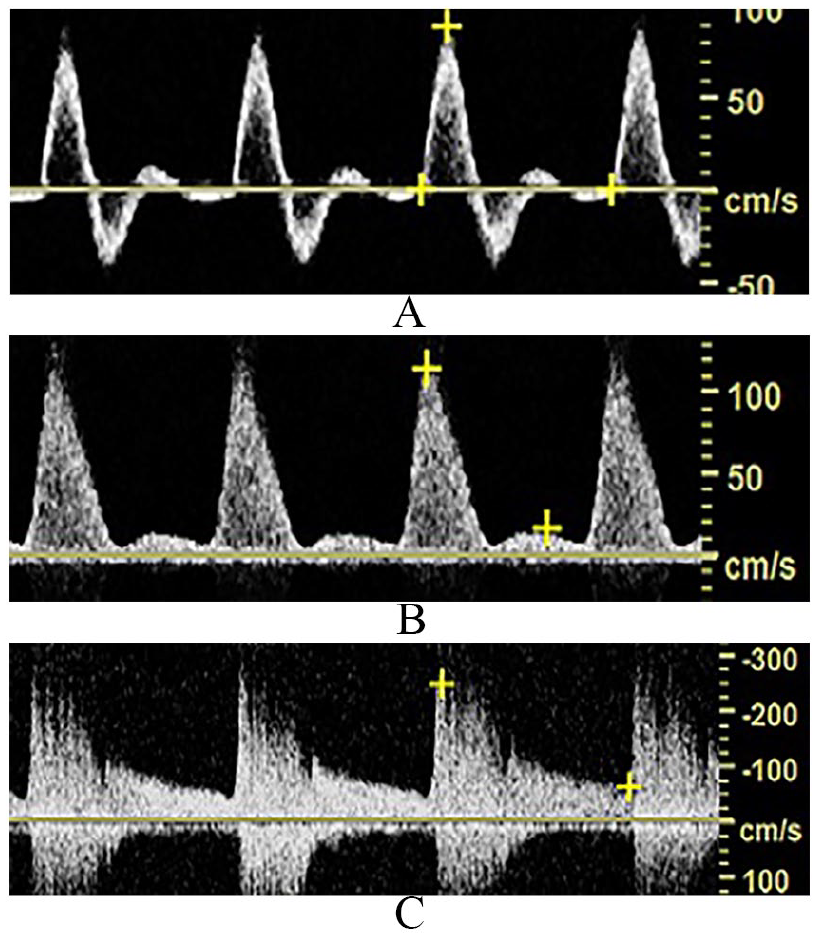
Spectral Doppler waveforms demonstrate laminar (A), disturbed (B), and turbulent (C) flow patterns.
Nature and impact of the problem
The waveform descriptors triphasic, biphasic, and monophasic have been used for more than 50 years, yet standardized application of these terms is not widely evident in the literature (Figure 3).12–14 The term triphasic, depicting three phases including diastolic flow reversal, is the most consistently and commonly used descriptor 5 to characterize normal arterial blood flow; however, triphasic has also been used to describe high-resistive waveforms with two phases 15 and low-resistive waveforms with continuous forward flow throughout diastole. 16 These waveforms have also been described as multiphasic.17,18 Biphasic has been used to characterize both normal19,20 and abnormal arterial flow patterns.15,21 Similar to its triphasic counterpart, the biphasic waveform has been depicted as high resistive with diastolic flow reversal22,23 and low resistive with continuous forward flow throughout diastole.21,24 Monophasic has most commonly been used to describe abnormal waveforms with a single phase found distal to a hemodynamically significant arterial obstruction,18,25 but has also been employed to characterize abnormal blood flow proximal to a significant obstruction and from normal extremities following exercise and exposure to hyperemic conditions or medications21,24,26,27 (Figure 4).
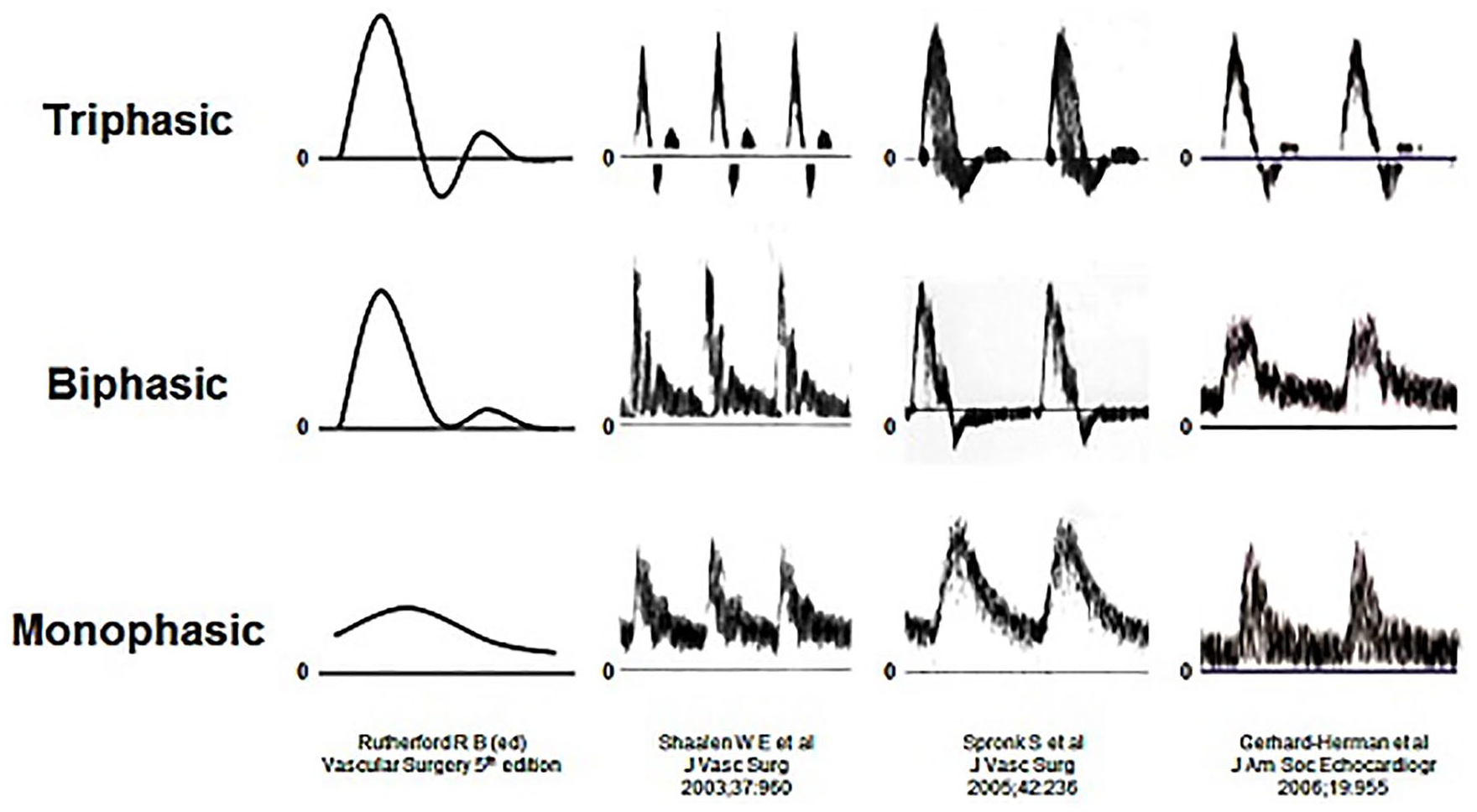
Waveform illustrations from authoritative publications demonstrating the inconsistent characterization of triphasic, biphasic, and monophasic waveform descriptors.
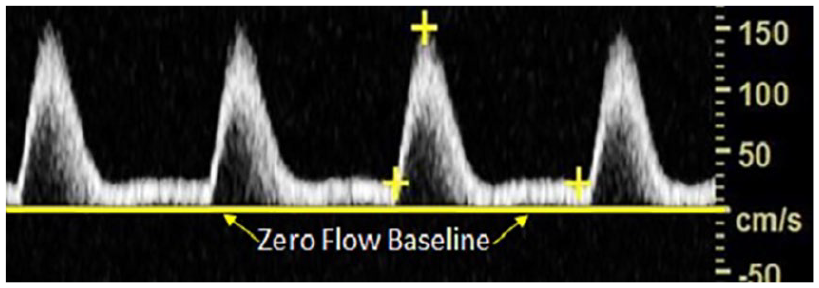
Spectral Doppler illustrates monophasic waveforms.
It should be emphasized that triphasic, biphasic, monophasic, and multiphasic are purely descriptor terminologies and are dependent on how the various terms are defined. In contrast, normal and abnormal are interpretative terms and depend on the specific vessel, vascular bed, pathology, and other factors. Each of the aforementioned waveform descriptors may be normal or abnormal, depending on the clinical setting (e.g. a monophasic waveform is normal in a renal artery and abnormal in a brachial artery).
Additional confusion surrounding Doppler waveforms includes the term phasicity and utilization of a zero-flow baseline. Waveform phasicity has been described as a change in direction and/or velocity. 28 While the zero-flow reference point is crucial to delineating diastolic flow reversal, many publications depict arterial waveforms without a clearly delineated zero-flow reference point. 5
The clinical impact of nonstandardized waveform nomenclature was studied in a survey of nearly 2000 ultrasound professionals. One-fifth of the respondents reported one or more occurrences of repeat arterial Doppler examinations because of lack of understanding of the terminology used to describe Doppler waveforms. 29 Standardization of Doppler waveform nomenclature is vital for the accurate and consistent communication of the results of vascular diagnostic tests and the prevention of inappropriate testing.
Part 1: Nomenclature
Purpose: Clarify and standardize key definitions and descriptors that are inherent to the analysis of arterial and venous Doppler waveforms
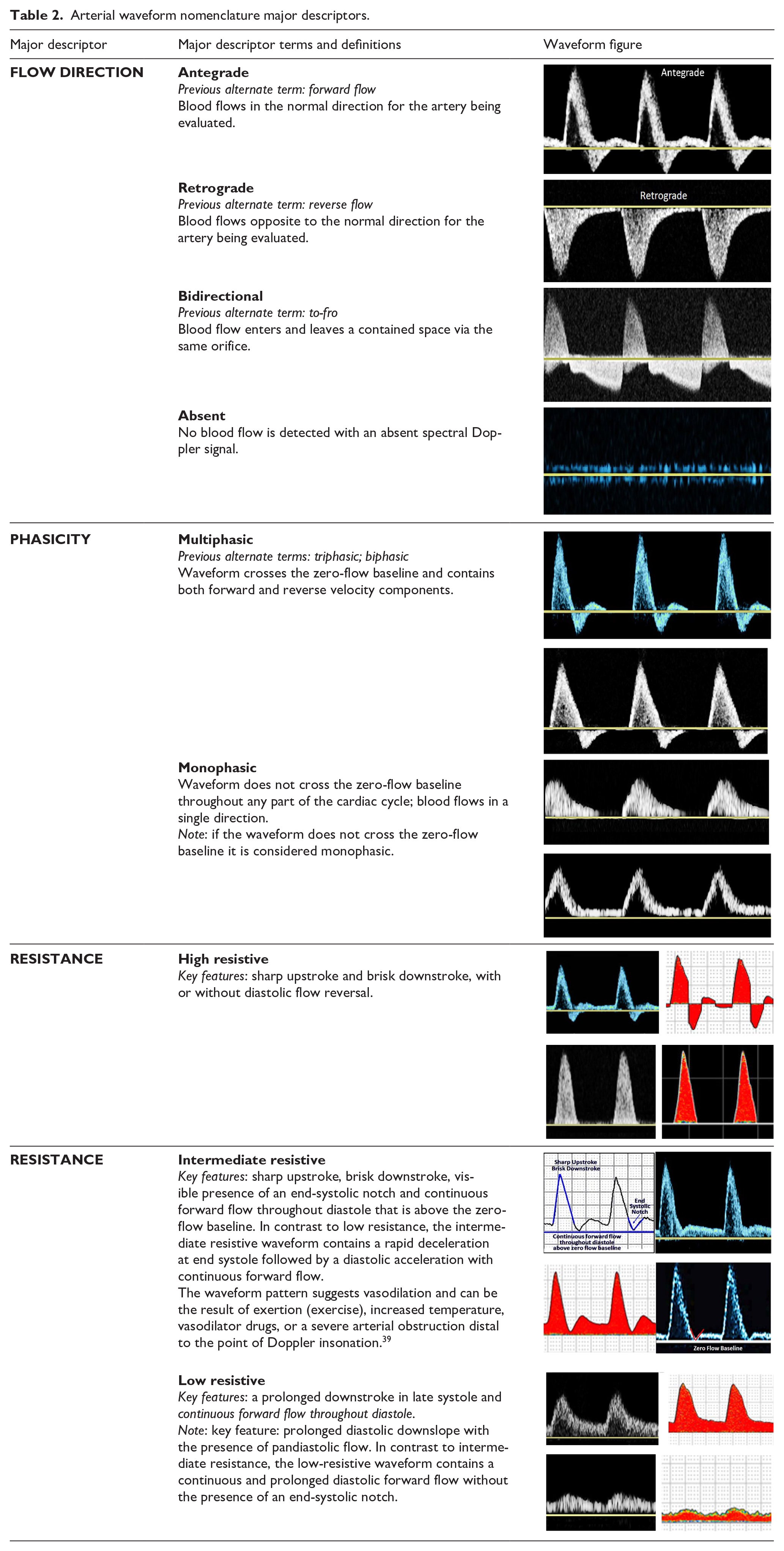
The major descriptor, modifier terms, and their descriptions are listed in Tables 2 and 3.
Arterial waveform nomenclature major descriptors.
Arterial waveform modifier terms.
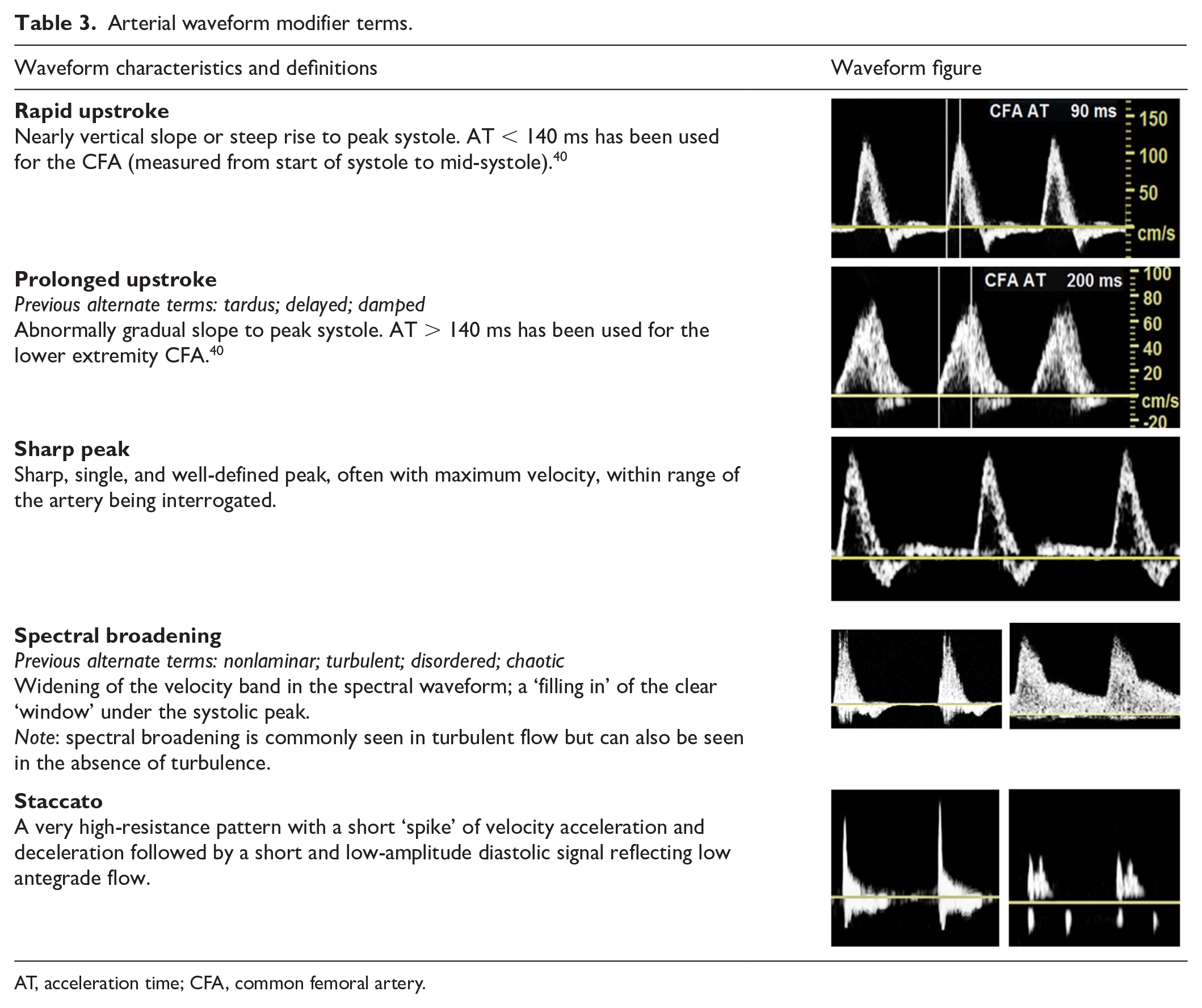
AT, acceleration time; CFA, common femoral artery.

The writing committee recommends that phasicity be described using the terms multiphasic and monophasic. Multiphasic waveforms cross the zero-flow baseline and contain both forward and reverse velocity components. Monophasic waveforms do not cross the zero-flow baseline and reflect blood which flows in a single direction for the duration of the cardiac cycle.
The writing committee recommends that arterial spectral Doppler waveforms be described as demonstrating high, intermediate, or low resistance. High-resistive waveforms have a sharp upstroke and brisk downstroke and may be either multiphasic or monophasic. Low-resistive waveforms contain a prolonged downstroke in late systole with continuous forward flow throughout diastole without an end-systolic notch. Low-resistive waveforms are monophasic. There is a hybrid waveform that is monophasic but has features of both high and low resistivity as it contains both brisk downstroke but also continuous forward flow throughout diastole. This waveform has been variably labeled the ‘biphasic’ waveform in several prior publications 5 and has been the source of much confusion. Thus, the consensus is that waveforms with sharp upstroke, brisk downstroke, and continuous forward flow during diastole above the zero-flow baseline (monophasic) but with the presence of an end-systolic ‘notch’ (representing rapid deceleration during end systole followed by diastolic acceleration) will now be referred to as an intermediate resistive waveform.
As examples, Figures 1A and 1B would be described as multiphasic, high resistive, and Figure 1C would be described as monophasic, low resistive. Figure 2B would be described as monophasic, intermediate resistive. Figure 2C would be described as monophasic, low resistive with spectral broadening. As previously noted, the terms ‘normal’ and ‘abnormal’ are not used in these descriptors as ‘normal’ and ‘abnormal’ are interpretive terms that place the waveform into the text of location and physiologic conditions: a multiphasic, high-resistive waveform may be normal in a resting lower extremity artery but abnormal in an internal carotid artery. The nomenclature presented here serves only as descriptors of the waveforms, thus allowing interpretation of the waveforms to be performed within the appropriate clinical context.
Venous nomenclature
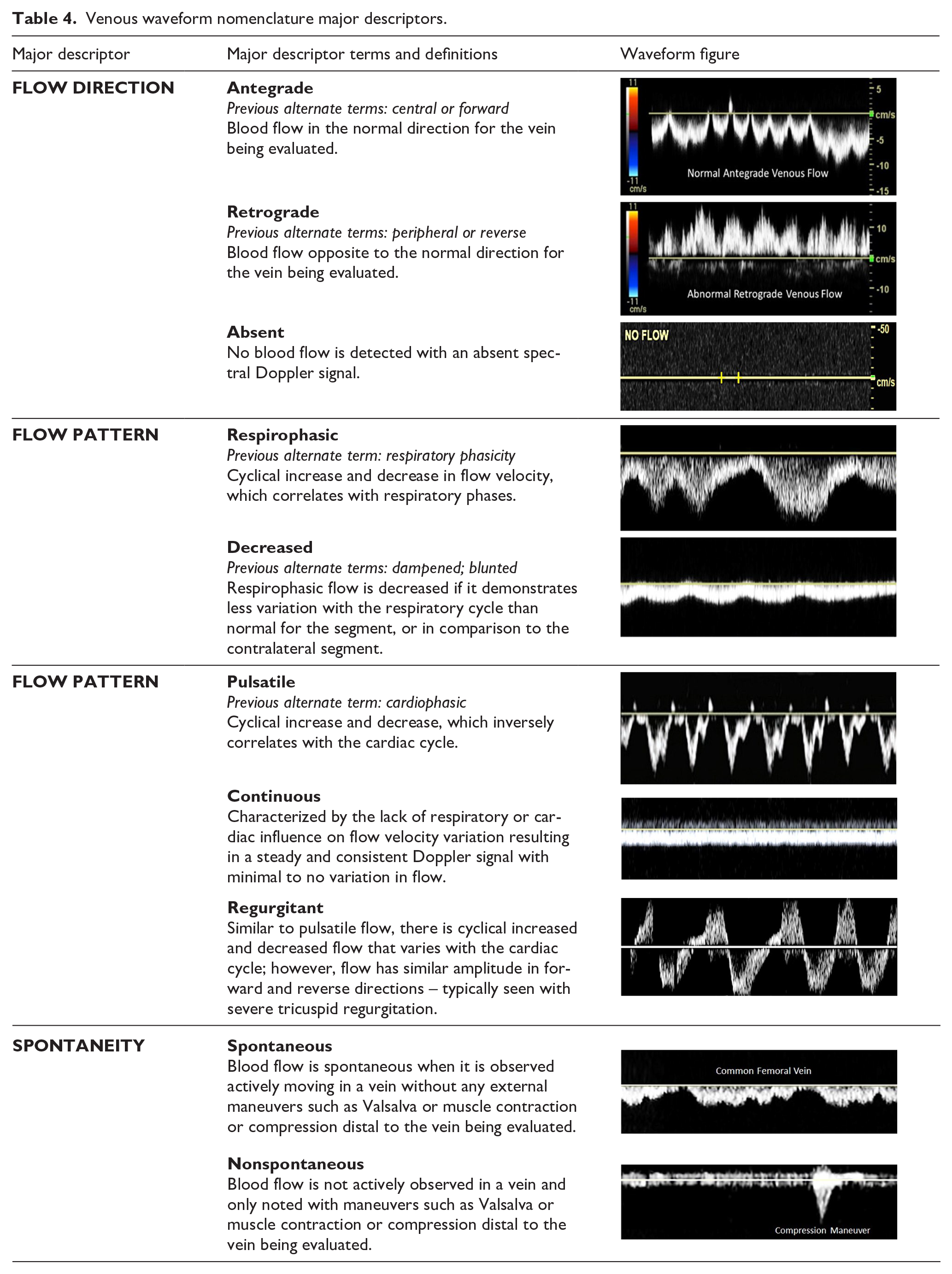
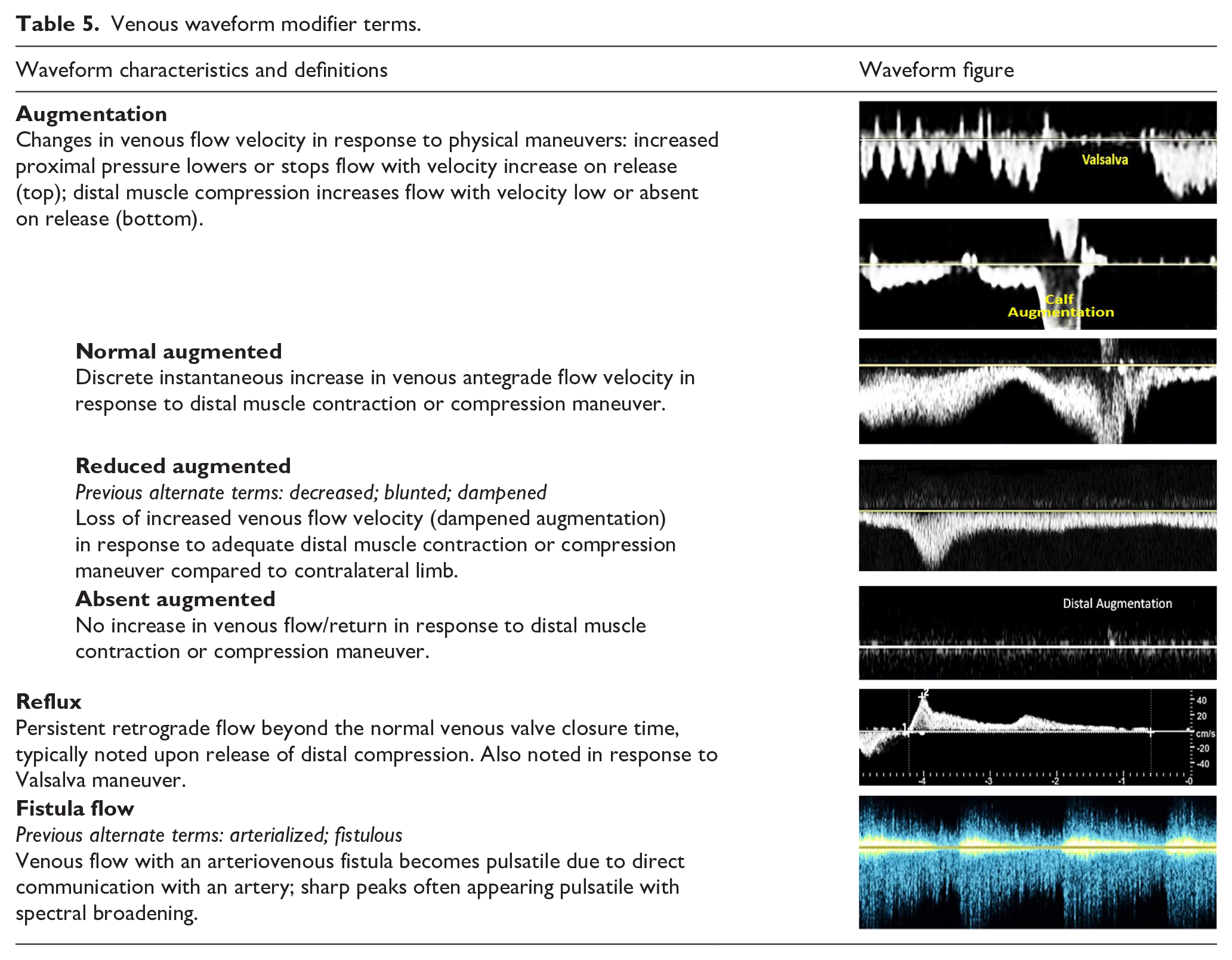
The major descriptors, modifier terms, and their descriptions are listed in Tables 4 and 5.
Venous waveform nomenclature major descriptors.
Venous waveform modifier terms.
Part 2: Doppler waveform alterations with physiologic changes and disease states
Purpose: Review Doppler waveform alterations with physiologic changes and disease states
Overview
Multiple physiologic factors proximally, distally, and at the site being examined contribute to the morphology of the Doppler waveform. Metabolic demands of the tissue bed, changes in pressure, changes in resistance, wave propagation, and wave reflection all impact waveform patterns.
Normal peripheral arterial waveforms
The peripheral arterial circulation supplies the muscular tissues of the upper and lower extremities. Arterial Doppler waveforms may display both antegrade and retrograde waves and are subject to the effects of attenuation and amplification along the vascular tree. Reflection of the pulse wave can also occur at several sites including the aortic bifurcation and aortic branches. 30 The systolic component of an arterial waveform is produced by the contraction of the left ventricle sending a bolus of blood out the ascending aorta, resulting in a rapid increase in volume and velocity.
The spectral display depicts a sharp upstroke or acceleration in an arterial waveform velocity profile from a normal vessel. In the absence of disease, the diastolic component in an arterial waveform reflects the vasoconstriction present in the resting muscular beds. Normal waveforms in a high-resistive bed will display a retrograde (reflected) wave in early diastole. A small antegrade component may be present in mid to late diastole as a result of an antegrade wave generated by proximal compliant large and medium arteries (Table 6).
Physiologic state of normal peripheral arterial waveforms.
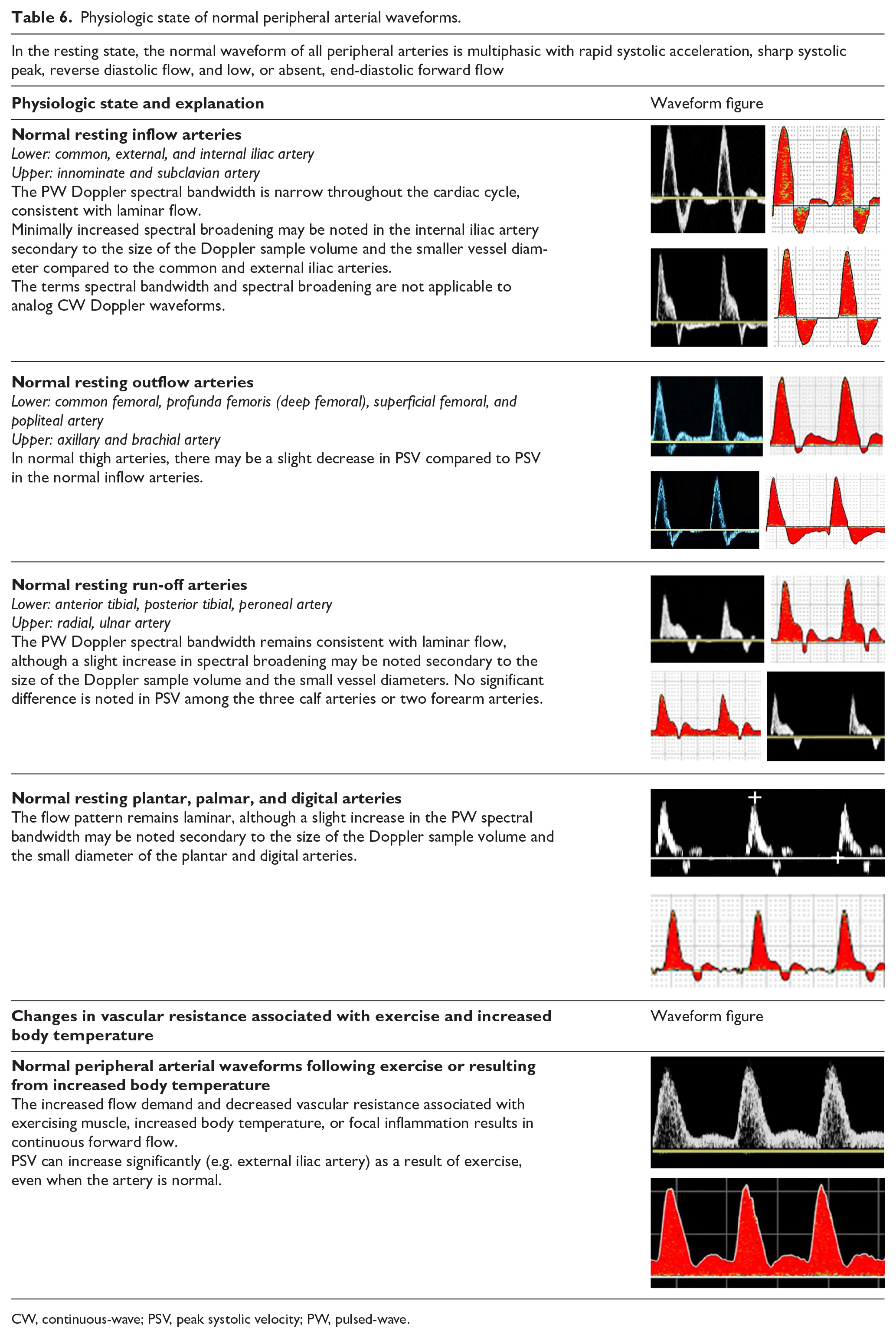
CW, continuous-wave; PSV, peak systolic velocity; PW, pulsed-wave.
Conditions that produce an increased flow to the limb muscles, such as exercise, increased limb temperature, and/or arteriovenous fistula, do so in part by dilating the arterioles in the muscle bed, allowing forward flow throughout diastole. Although the Doppler waveform generally demonstrates a narrow spectral bandwidth, some slight increase in spectral broadening may be noted dependent on the diameter of the arterial segment and the size of the Doppler sample volume (Table 6).
Abnormal peripheral arterial waveforms
The severity of reduction in lumen diameter is reflected in a continuum of changes in the waveform morphology (Table 7). Minimal diameter reduction results in slight disruption to laminar flow without significant increase in peak systolic velocity (PSV). Most notably, early diastolic reverse flow is maintained. Loss of the reverse flow component and transition from a multiphasic to monophasic flow pattern are apparent when the degree of narrowing is sufficient to cause a pressure-flow gradient at the site of stenosis. The severity of arterial compromise is reflected in the continual increase in peak systolic and end-diastolic velocities to a critical value consistent with a pre-occlusive lesion.
Pathophysiologic state of abnormal peripheral arterial waveforms.
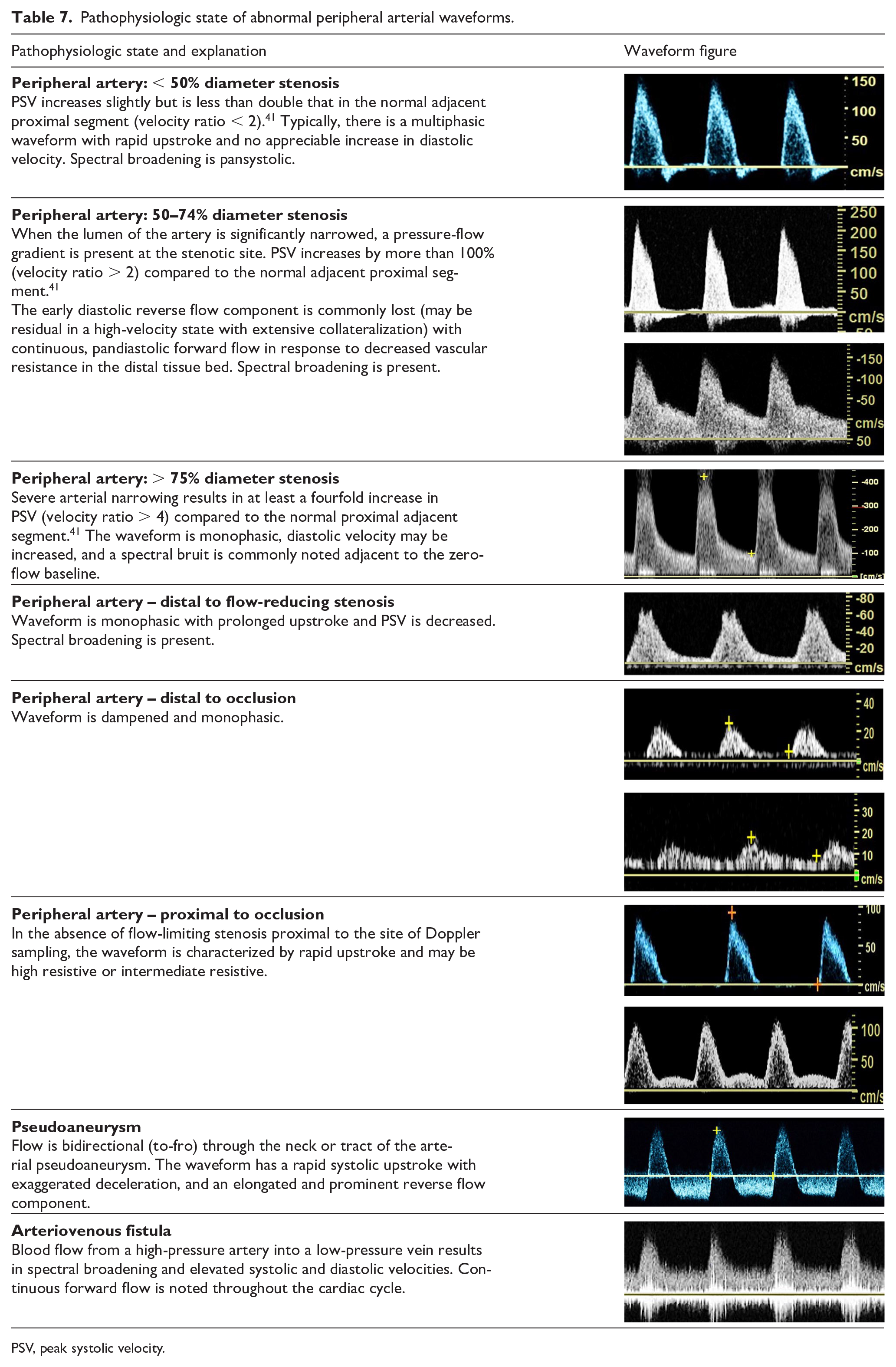
PSV, peak systolic velocity.
The waveform also indicates the location of arterial obstruction. Delayed systolic upstroke suggests flow-limiting disease proximal to the recording site. Distal to a stenosis, ischemia in the tissue bed will result in vasodilation and decreased resistance. Additionally, there is a decrease in distal pressure due to the pressure drop across the stenosis. This pressure drop, along with the lower resistance, results in increased diastolic flow throughout the cardiac cycle distal to a stenosis. Proximal to an occlusion or high-grade stenosis, the resistance will increase. The reflected wave or any antegrade diastolic flow, if normally present, may be reduced or absent and sequential flow-limiting lesions and collateral vessel capacity can affect waveform morphology at a given Doppler sampling location.
Peripheral venous circulation
Overview
Numerous intrinsic and extrinsic factors influence the movement of venous blood. Intrinsic factors include the respiratory cycle, changes in central and segmental venous pressure gradients, vessel compliance, and valve function. Extrinsic factors include gravity, muscle pumps, atmospheric pressure, and external compression of veins, such as may occur from an overlying tumor, artery, mass or fluid collection.
Normal peripheral venous waveforms
Normal venous flow in the larger peripheral and more central veins examined in duplex protocols is spontaneous with low-velocity Doppler waveforms that reflect pressure gradient changes produced by respiratory and cardiac function. Flow velocities are very low in the smaller veins distally in the extremities and may not produce discernable Doppler signals in the resting state. Throughout the periphery, flow velocities vary with respiration due to the changes in intrathoracic and intra-abdominal pressures and are aptly called respirophasic. These patterns of respiratory variations in flow velocity can become suspended, severely dampened, or absent with shallow breathing or breath holding. Peripheral veins that are most distal to the heart, such as calf or forearm veins, demonstrate less spontaneity and respirophasicity compared to the veins closer to the heart. Cardiac filling and contraction also draws and pushes venous flow, with this influence normally stronger in veins closest to the heart termed pulsatile flow31,32 (Table 8).
Physiologic state of normal peripheral venous waveforms.
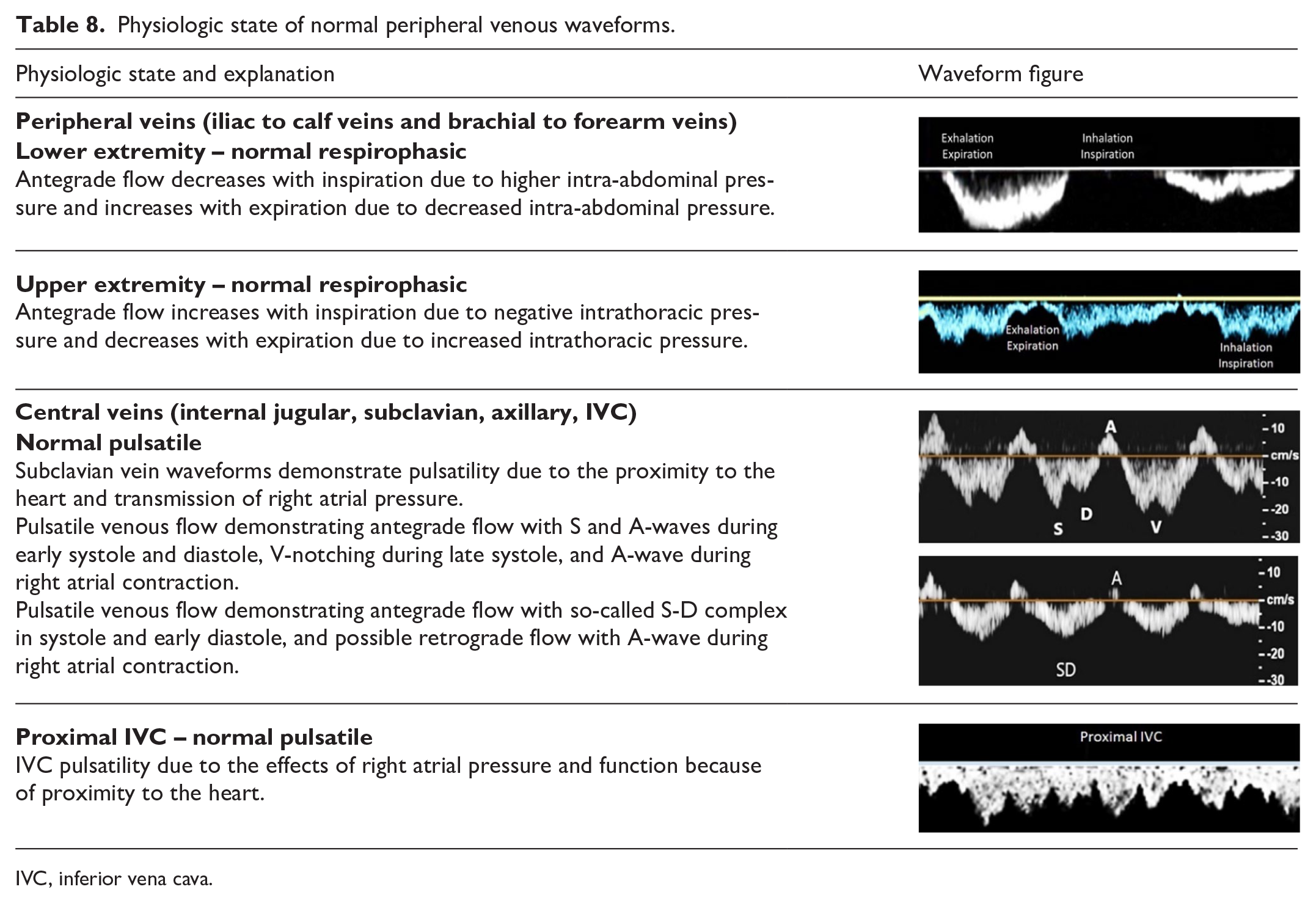
IVC, inferior vena cava.
Abnormal peripheral venous waveforms
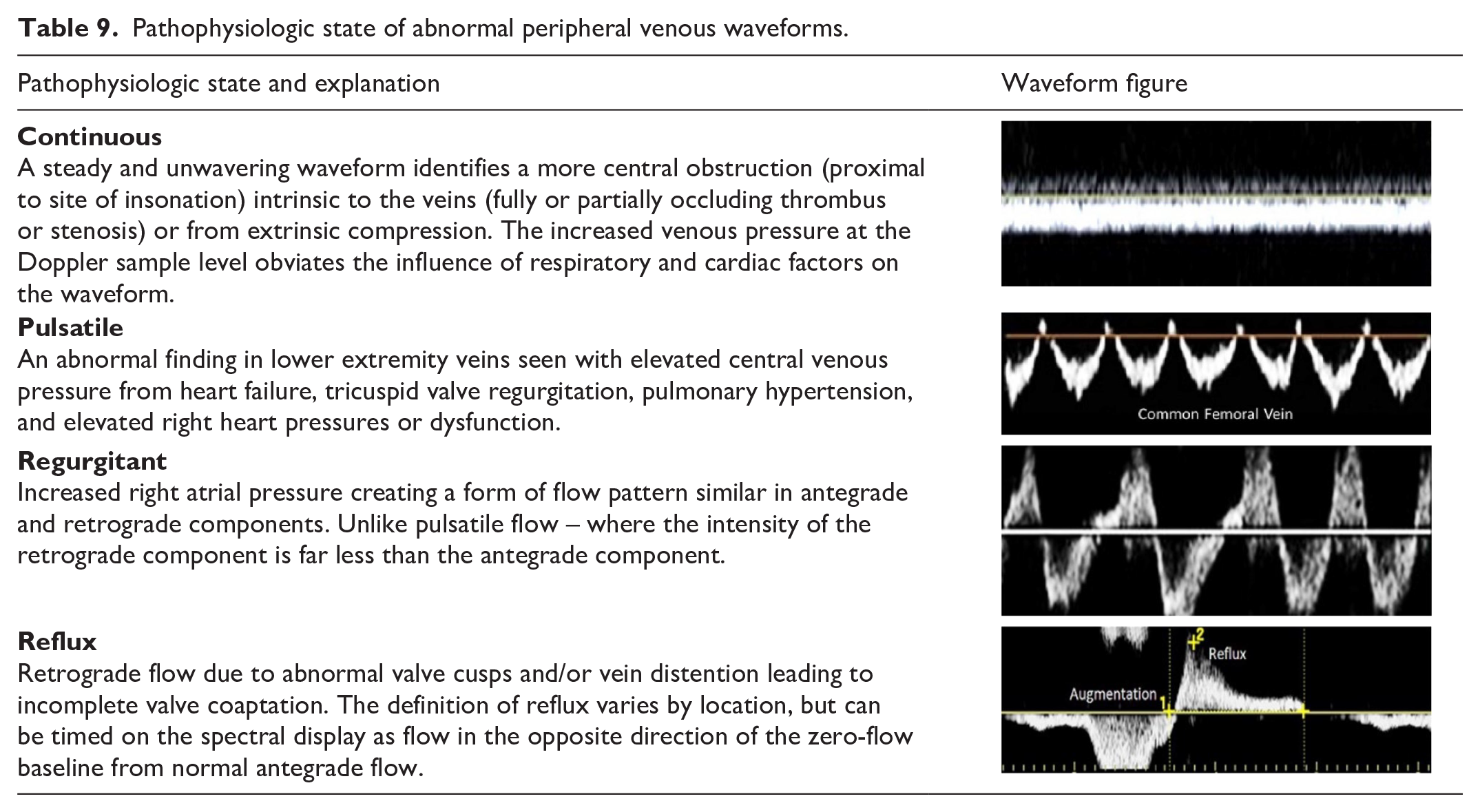
Evaluation for abnormal changes in waveform morphology includes appraisal of flow direction, responses to respiration and cardiac function, and response to physiologic maneuvers. Changes that are the result of central, systemic conditions will be seen bilaterally. Symmetry between left and right-side waveforms is an important aspect of duplex studies requiring comparison of spontaneous signals and response to physical maneuvers.
The normal antegrade flow direction in the venous system may become retrograde when there is valvular incompetence or occlusion in a more central venous segment. Continuous spontaneous venous flow also suggests a more central obstruction (Table 9). While the utilization of augmentation maneuvers remains controversial, advocates maintain it can be helpful in diagnosing a more central obstructive pathology and is discussed in the nomenclature section in greater detail. Although they may be seen with certain pathologies (Table 9), pulsatile waveforms are not normally observed in the lower extremity peripheral veins due to their distance from the heart. However, loss of this pulsatility is abnormal in central upper extremity and abdominal veins. Interpretation of venous waveform morphology is most often done without reporting angle-corrected velocity data, although measuring velocities is essential when evaluating a fistula or a venous stenosis.
Pathophysiologic state of abnormal peripheral venous waveforms.
Cerebrovascular circulation
Overview
The bilateral extracranial cerebral vessels include the common carotid, external carotid, internal carotid, and vertebral arteries. Inflow to these vessels depends on the aortic valve, aortic arch, brachiocephalic, and subclavian arteries. Outflow from these vessels is determined by the status of the basal cerebral arteries (circle of Willis) and the rest of the intracranial cerebral circulation. With normal inflow, waveform features generally reflect the resistance of the distal vascular bed. Proximal occlusive lesions, focal lesions in specific arterial segments, and changes in the resistance of the distal vascular bed all contribute to the changes observed in cerebrovascular flow waveforms.
Normal cerebrovascular waveforms
Normal flow waveforms in the cerebral arteries show a rapid systolic upstroke, reflecting normal proximal vessels and cardiac function, but the characteristics of the diastolic portion of the waveform are determined by the resistance of the distal vascular bed. Because brain tissue normally has a low vascular resistance, a normal internal carotid artery waveform shows a low-resistive pattern with relatively high diastolic velocities and forward flow throughout the cardiac cycle. In contrast, the normal external carotid artery supplies a high-resistive vascular bed (skin, muscle, bone) similar to that of peripheral arteries and displays a typical multiphasic arterial flow pattern. The common carotid artery supplies the internal and external carotid arteries, so the normal common carotid artery waveform contains features of both branch vessels. However, since the internal carotid has a much higher flow rate than the external carotid, the common carotid flow pattern tends to resemble the internal carotid, although typically with less diastolic flow. The normal vertebral artery flow waveform shows a low-resistive pattern like that of the internal carotid artery. As for arterial waveforms in general, the absence of spectral broadening in cerebrovascular waveforms indicates laminar flow. Table 10 illustrates the normal features of flow waveforms in the extracranial cerebral arteries.
Normal cerebrovascular waveforms.
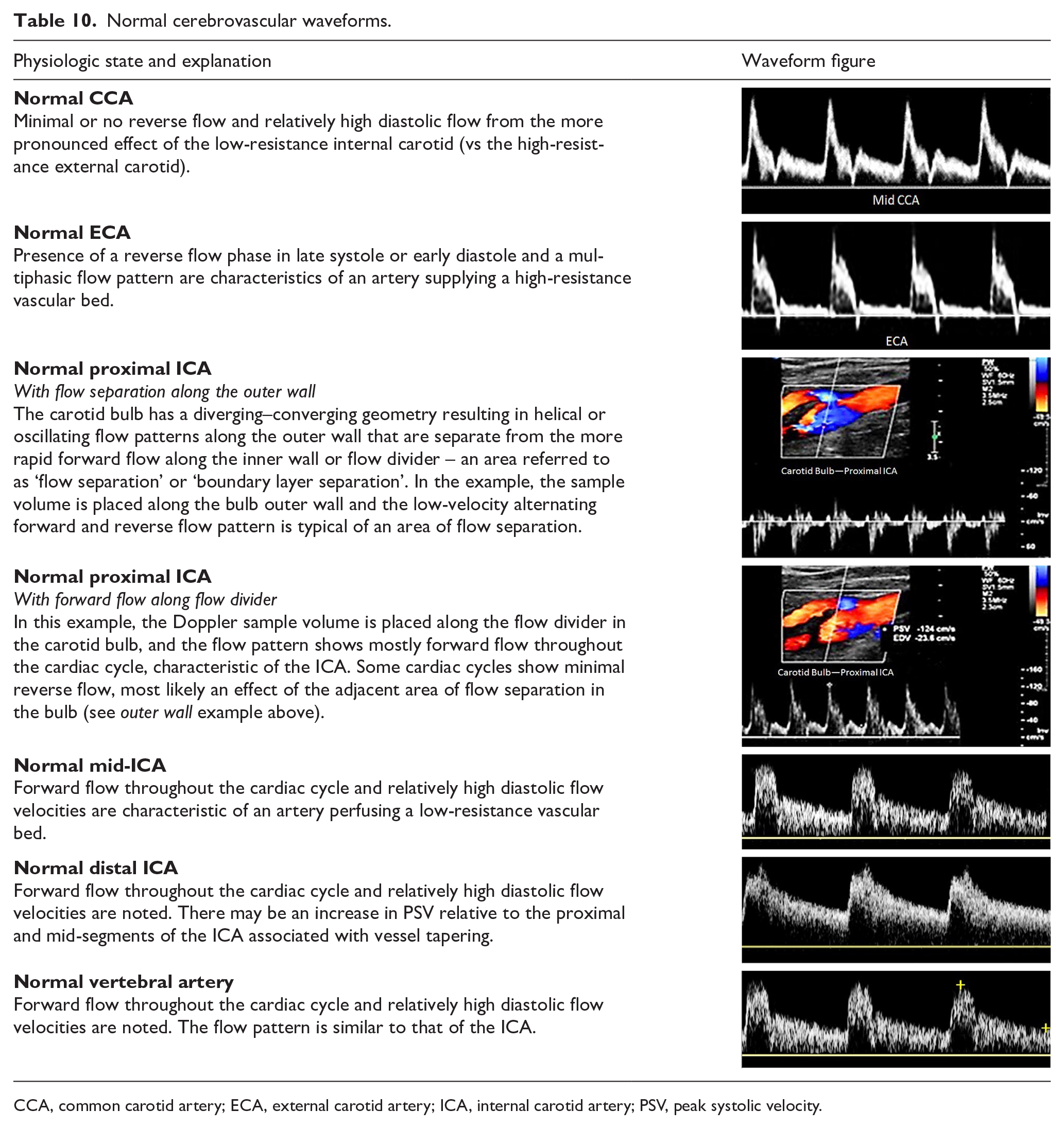
CCA, common carotid artery; ECA, external carotid artery; ICA, internal carotid artery; PSV, peak systolic velocity.
Abnormal cerebrovascular waveforms
As in other segments of the arterial system, significant obstructive lesions involving inflow to the extracranial cerebral arteries result in waveforms with a delayed systolic upstroke, rounded systolic peak, and decreased peak velocities. Focal stenoses produce localized high-velocity jets and post-stenotic turbulence. Distal obstructive lesions cause increased vascular resistance, which appears in the waveform as decreased diastolic flow or reversed flow, features which are particularly noticeable in vessels that normally have a low-resistive flow pattern, such as the internal carotid and vertebral arteries. Table 11 illustrates the features of abnormal flow waveforms in the extracranial cerebral arteries.
Abnormal cerebrovascular waveforms.
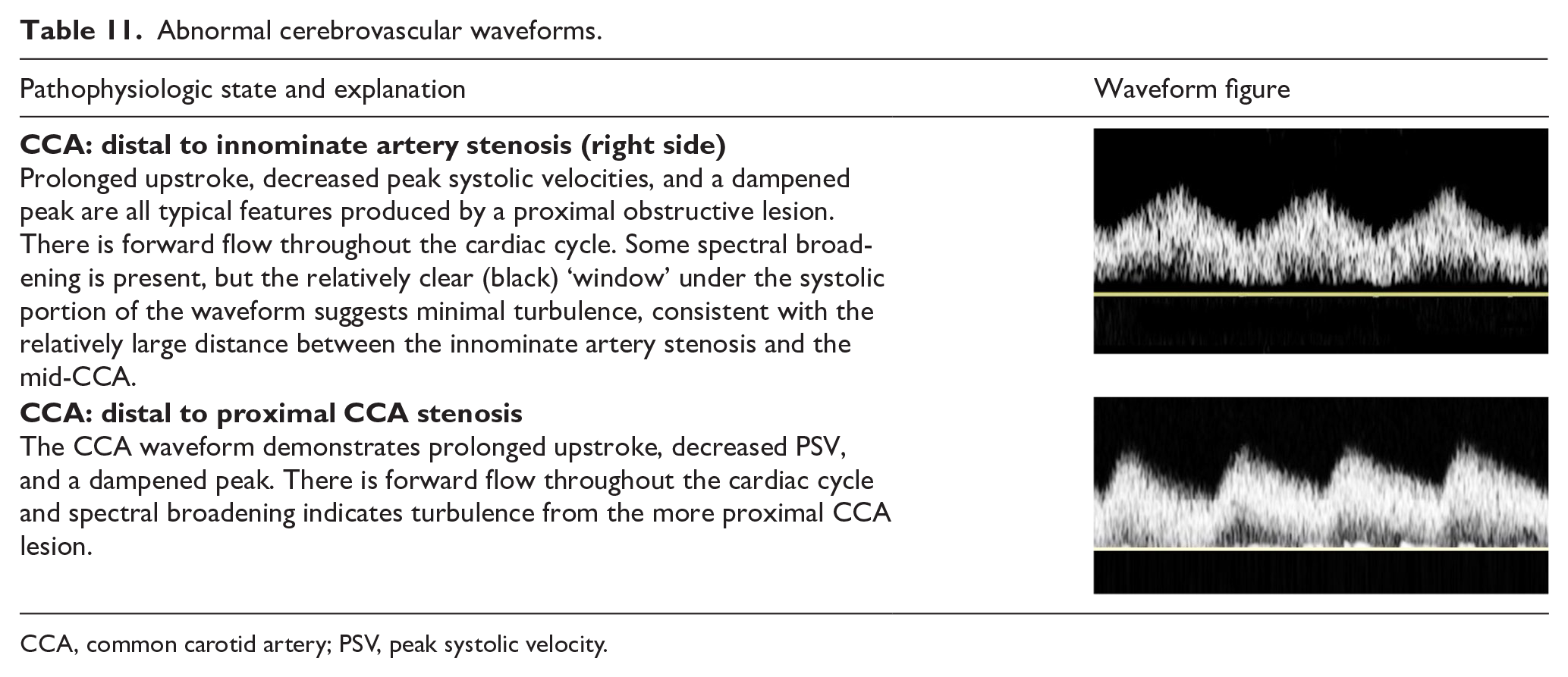
CCA, common carotid artery; PSV, peak systolic velocity.
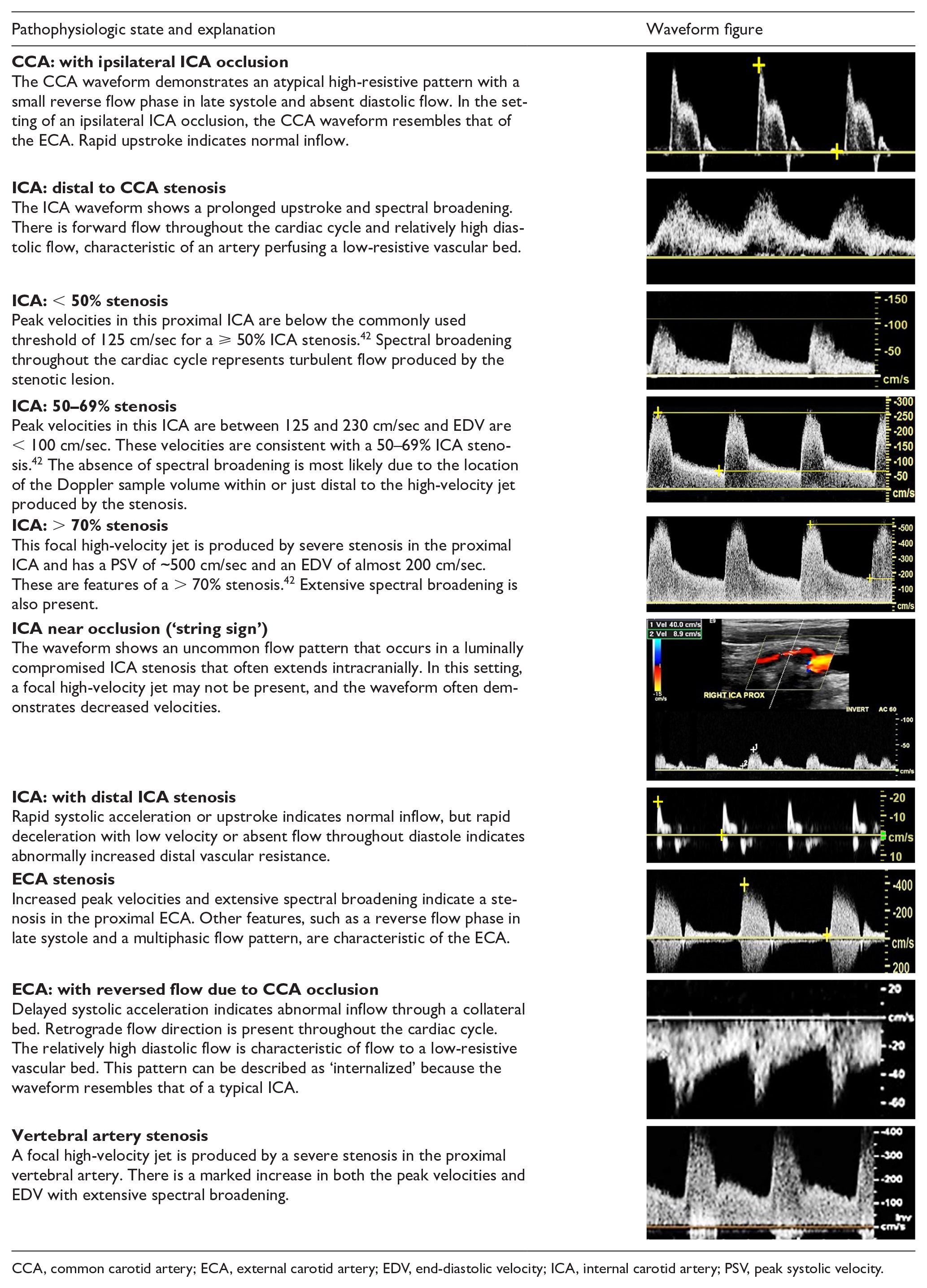
CCA, common carotid artery; ECA, external carotid artery; EDV, end-diastolic velocity; ICA, internal carotid artery; PSV, peak systolic velocity.
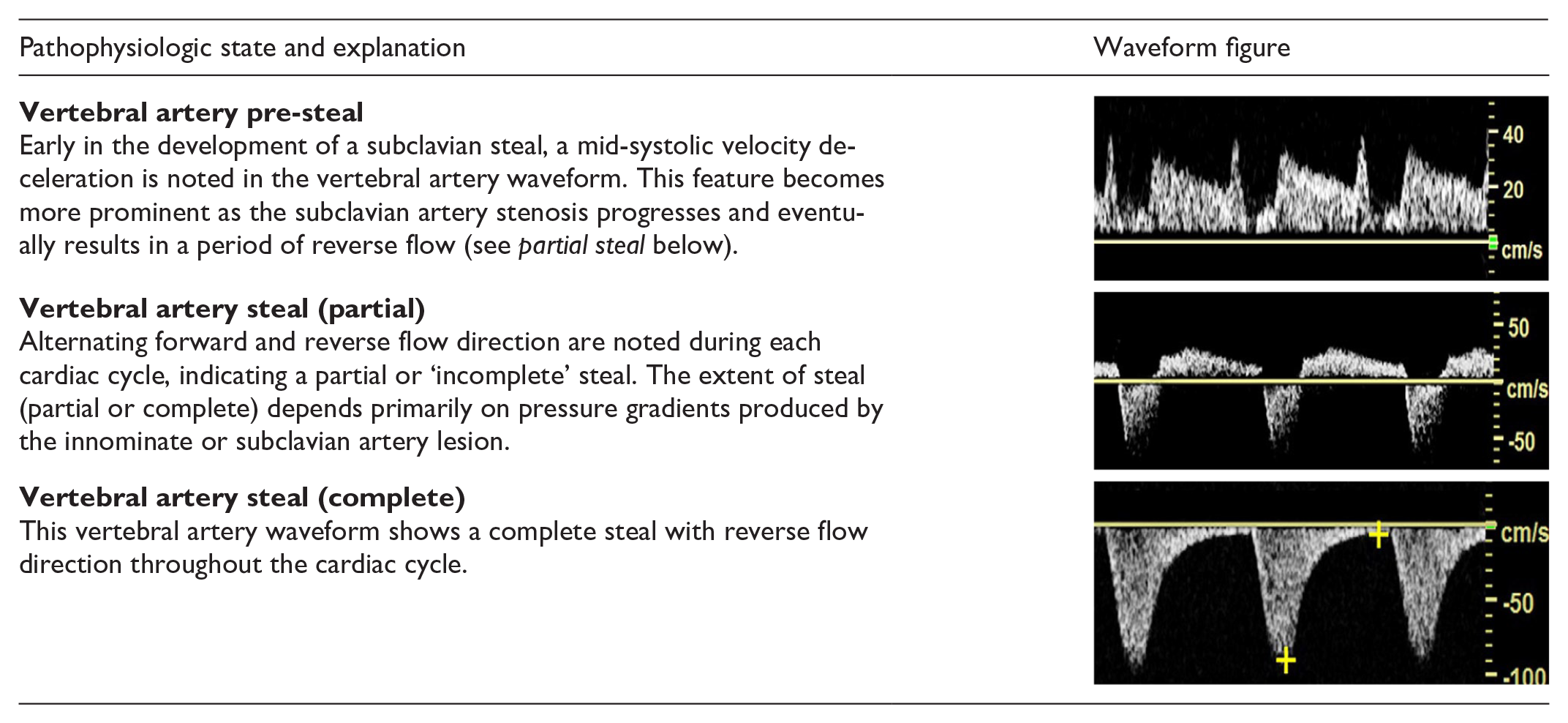
Abnormal carotid Doppler waveforms associated with cardiac disease
There are several cardiac abnormalities which will alter the expected waveform patterns observed in the arteries. Many of the waveform alterations are commonly detected in the carotid arteries, while some can be appreciated as far distally as the femoral arteries. Observed waveform changes may vary depending on the severity of cardiac disease. Table 12 summarizes some of the more frequently encountered waveforms associated with various cardiac disorders.
Abnormal carotid waveforms associated with cardiac disease.
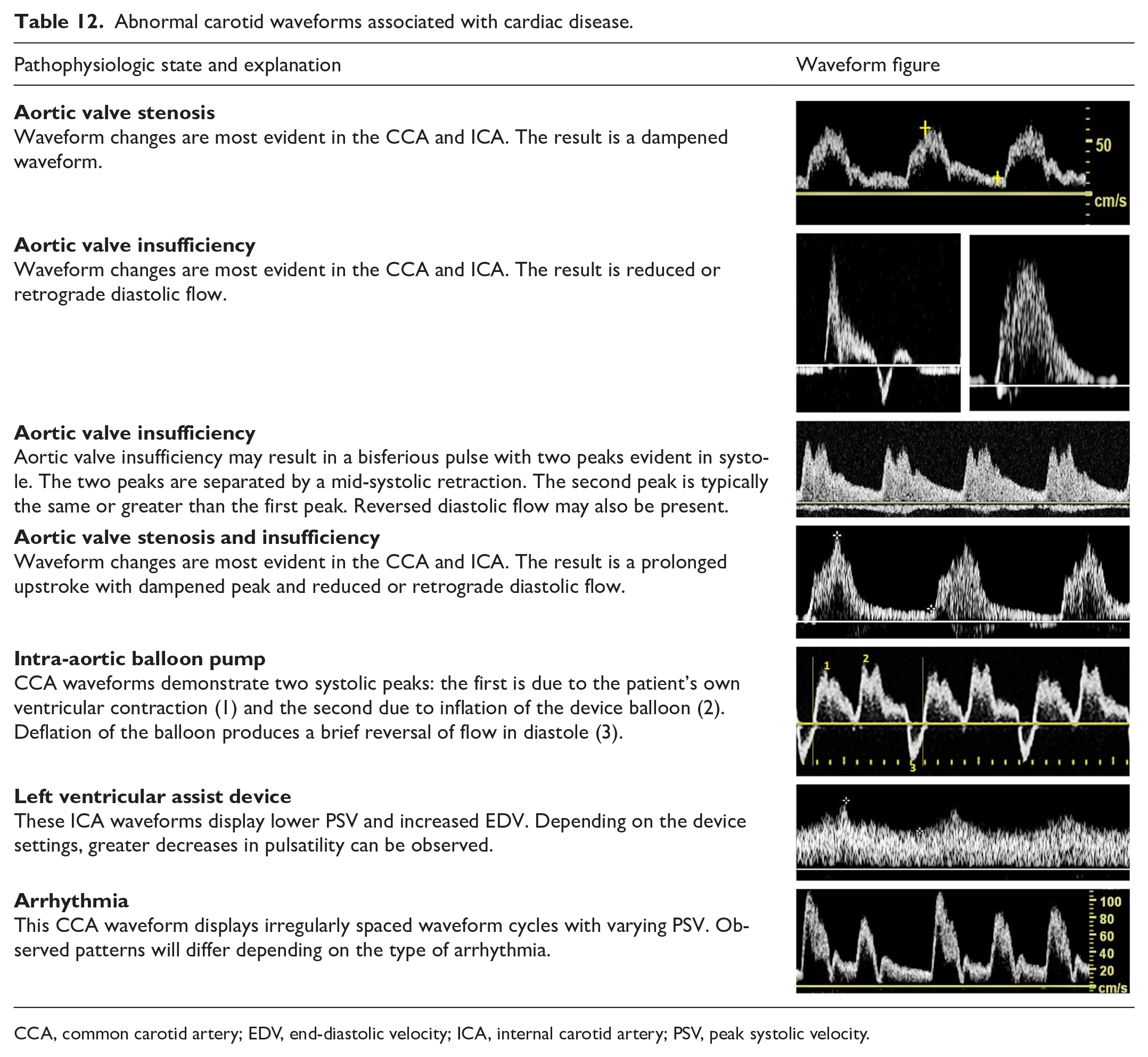
CCA, common carotid artery; EDV, end-diastolic velocity; ICA, internal carotid artery; PSV, peak systolic velocity.
Mesenteric and renal arterial circulation
Overview
Multiple physiologic factors determine the shape of the renal and mesenteric Doppler waveforms, including the metabolic requirements of the end organ and whether the images were obtained in the fasting or postprandial state.
Normal mesenteric arterial waveforms
The celiac artery has major branches (hepatic, splenic, and left gastric), which supply blood flow to the liver, spleen, pancreas, stomach, and superior portion of the small bowel. Because of the high oxygen demands of the organs supplied by the celiac artery and its branches, blood flow is not affected by digestion and peak systolic and end-diastolic velocities are not significantly altered postprandially. 33 In contrast, the superior mesenteric (SMA) and inferior mesenteric (IMA) arteries have multiple branches that carry blood flow to the pancreatic head and the muscular tissues of the small and large intestine. Changes in Doppler waveform morphology in these vessels with fasting and in the postprandial state are shown in Table 13.
Normal mesenteric artery waveforms.

SMA, superior mesenteric artery.
Abnormal mesenteric arterial waveforms
Significant stenosis in the mesenteric vessels is accompanied by changes in the Doppler waveform seen in other vascular beds: elevated peak systolic and diastolic velocities, spectral broadening, and, when severe, a prolonged upstroke may be present (Table 14). Mesenteric artery occlusion should be suspected when flow cannot be detected in an imaged artery after ultrasound system controls for spectral and/or color Doppler have been optimized for slow flow.
Abnormal mesenteric artery waveforms.
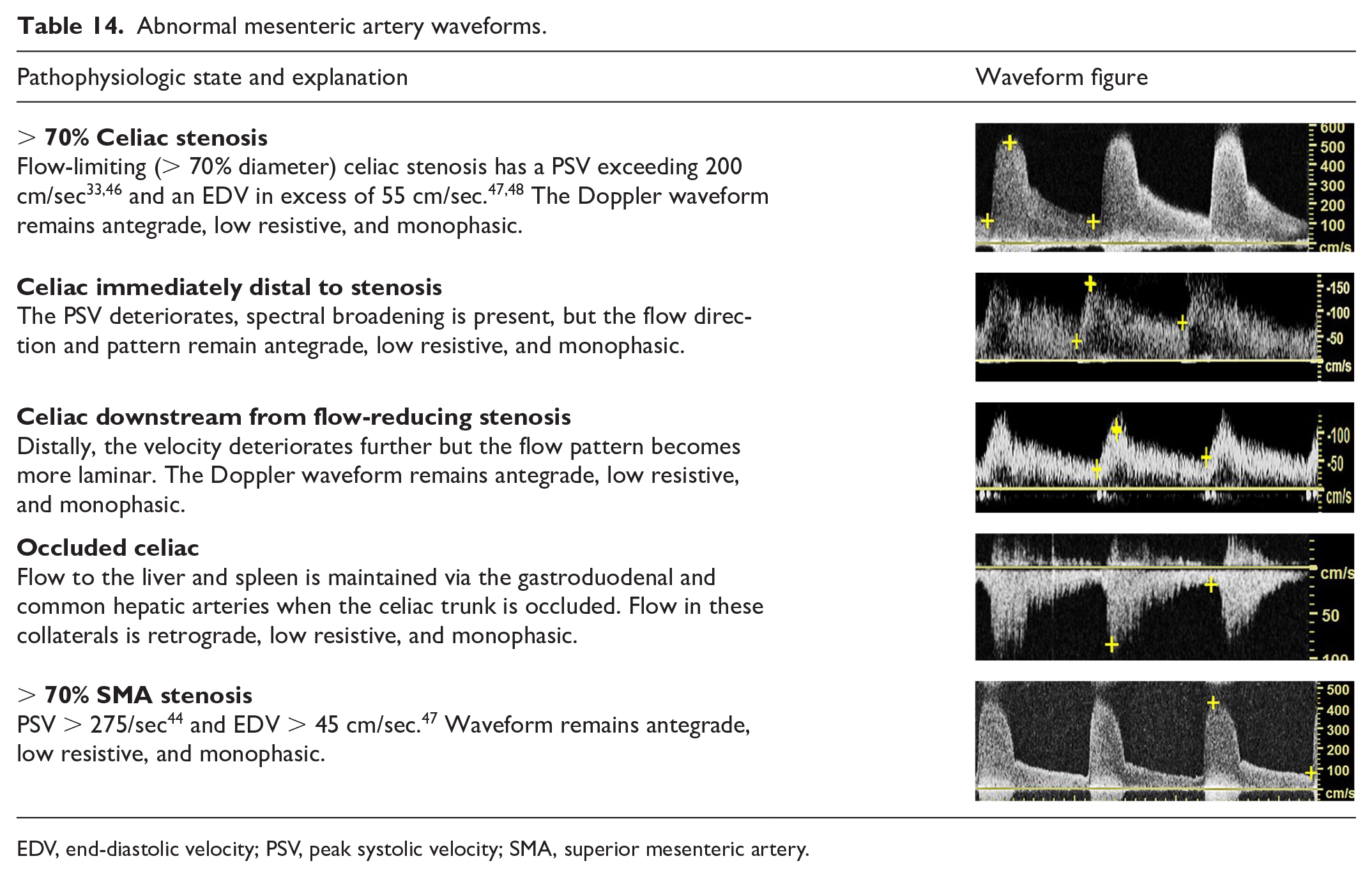
EDV, end-diastolic velocity; PSV, peak systolic velocity; SMA, superior mesenteric artery.
There are few well-validated diagnostic criteria for the inferior mesenteric artery;34,35 however, the Doppler waveform will mimic those of the fasting, postprandial, and stenotic superior mesenteric artery with, most often, lower peak systolic velocities in the fasting and postprandial states.
Normal renal arterial waveforms
The kidneys are high flow demand end-organs, which receive blood from one or more renal arteries. The Doppler waveform will demonstrate a rapid upstroke, sharp peak, and a low-resistive, monophasic waveform consistent with continuous diastolic forward flow (Table 15).
Normal and abnormal renal artery waveforms.
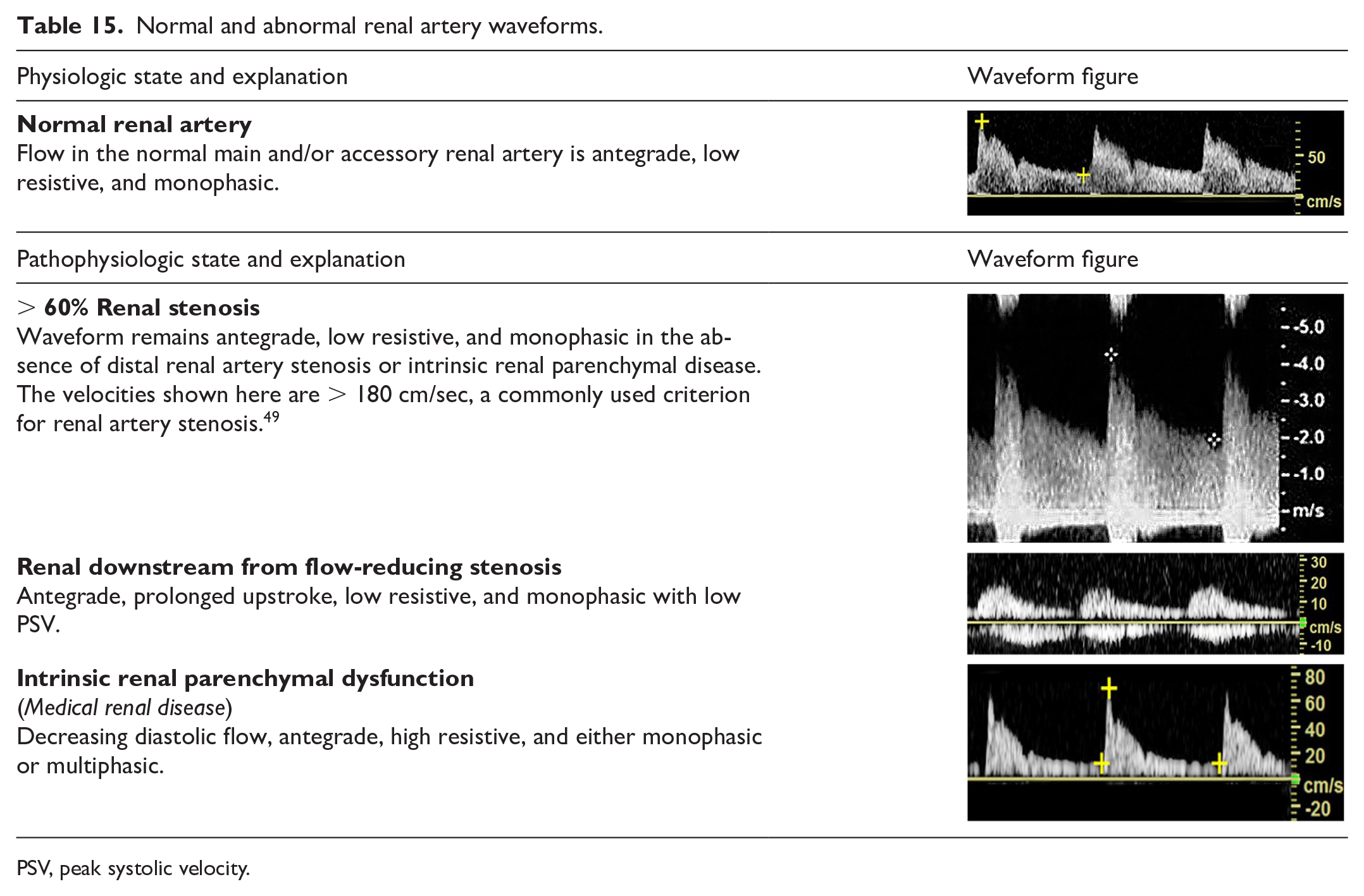
PSV, peak systolic velocity.
Abnormal renal arterial waveforms
Blood flow via adrenal or capsular collaterals may be found in the hilar and parenchymal arteries in patients with renal artery occlusion. The waveform pattern in these vessels will be antegrade, low resistive, dampened, and monophasic (Table 15).
Resistance to arterial inflow to the renal parenchyma may result from a variety of pathologic processes including, but not limited to, interstitial fluid accumulation, hydronephrosis, space-occupying lesions, acute tubular necrosis, and glomerulonephritis.
Mesenteric, renal, hepatic, and portal venous circulation
These veins often will present with unique flow patterns, which are described in detail in Table 16.
Normal mesenteric, renal, hepatic, and portal vein waveforms.
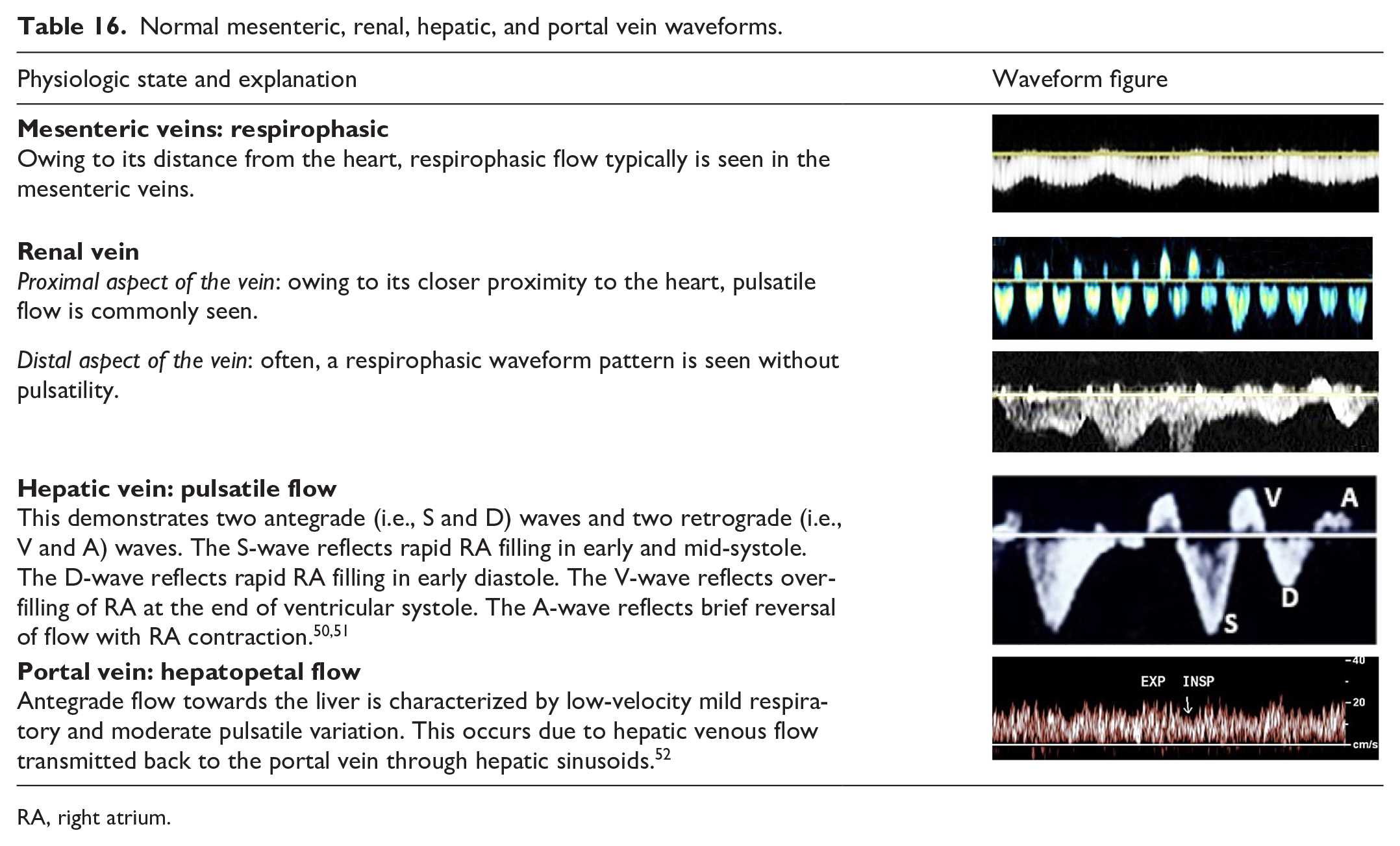
RA, right atrium.
Abnormal hepatic and portal veins
Cardiac and liver abnormalities, including transjugular intrahepatic portosystemic shunts, lead to changes in hepatic and portal vein waveform patterns, as noted in Table 17.
Abnormal mesenteric, renal hepatic, and portal vein waveforms.
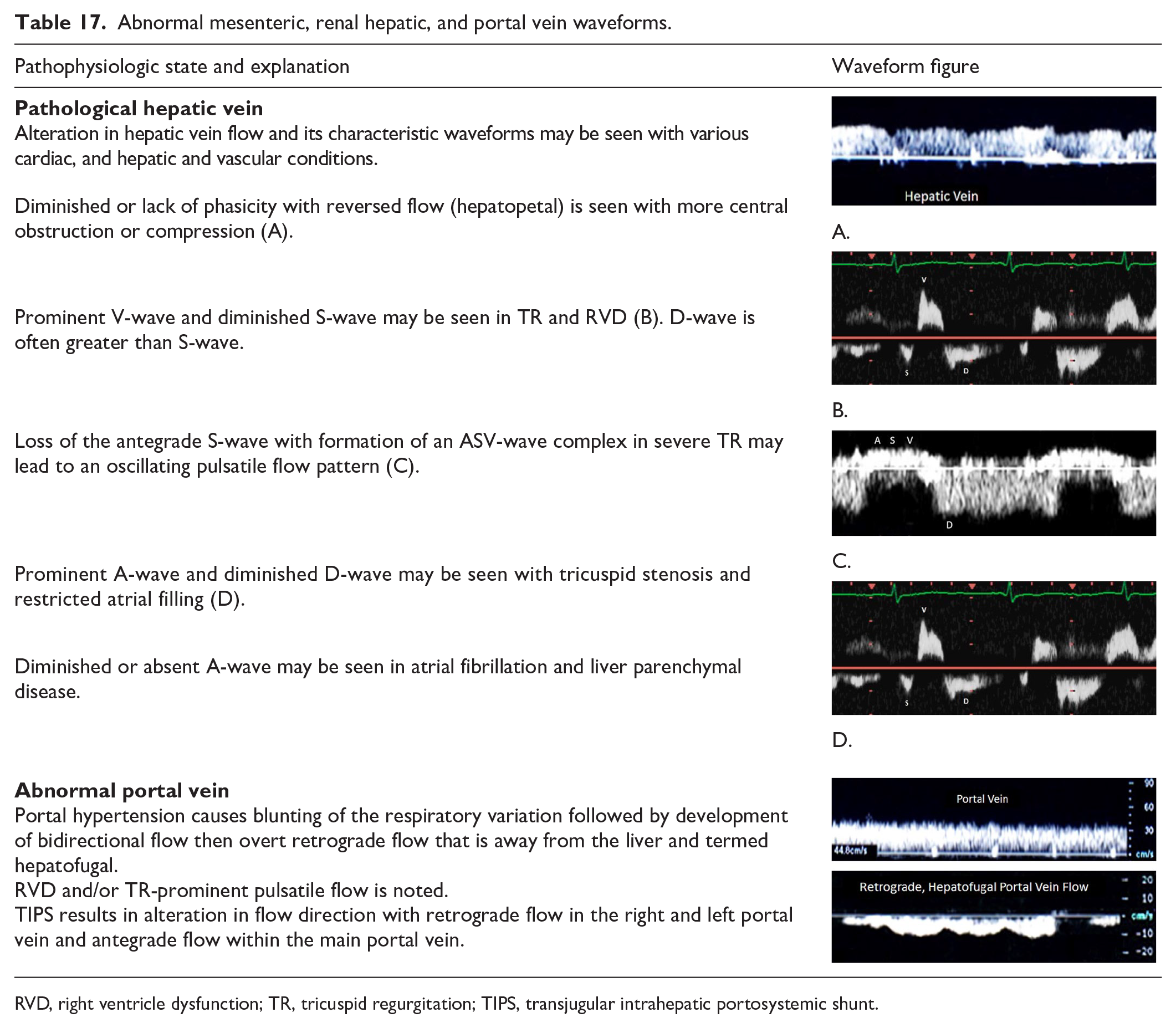
RVD, right ventricle dysfunction; TR, tricuspid regurgitation; TIPS, transjugular intrahepatic portosystemic shunt.
Part 3: Waveform optimization
Purpose: Provide Doppler transducer optimization techniques to enhance the quality and presentation of Doppler spectral waveform and color Doppler data
Waveform optimization is an integral part of Doppler ultrasound and plays a vital role in appropriate creation and interpretation of spectral waveforms and color data. The essential elements for image optimization and potential artifacts that can occur while performing Doppler ultrasound, along with solutions to improve image quality, are presented in Table 18.
Doppler waveform optimization.
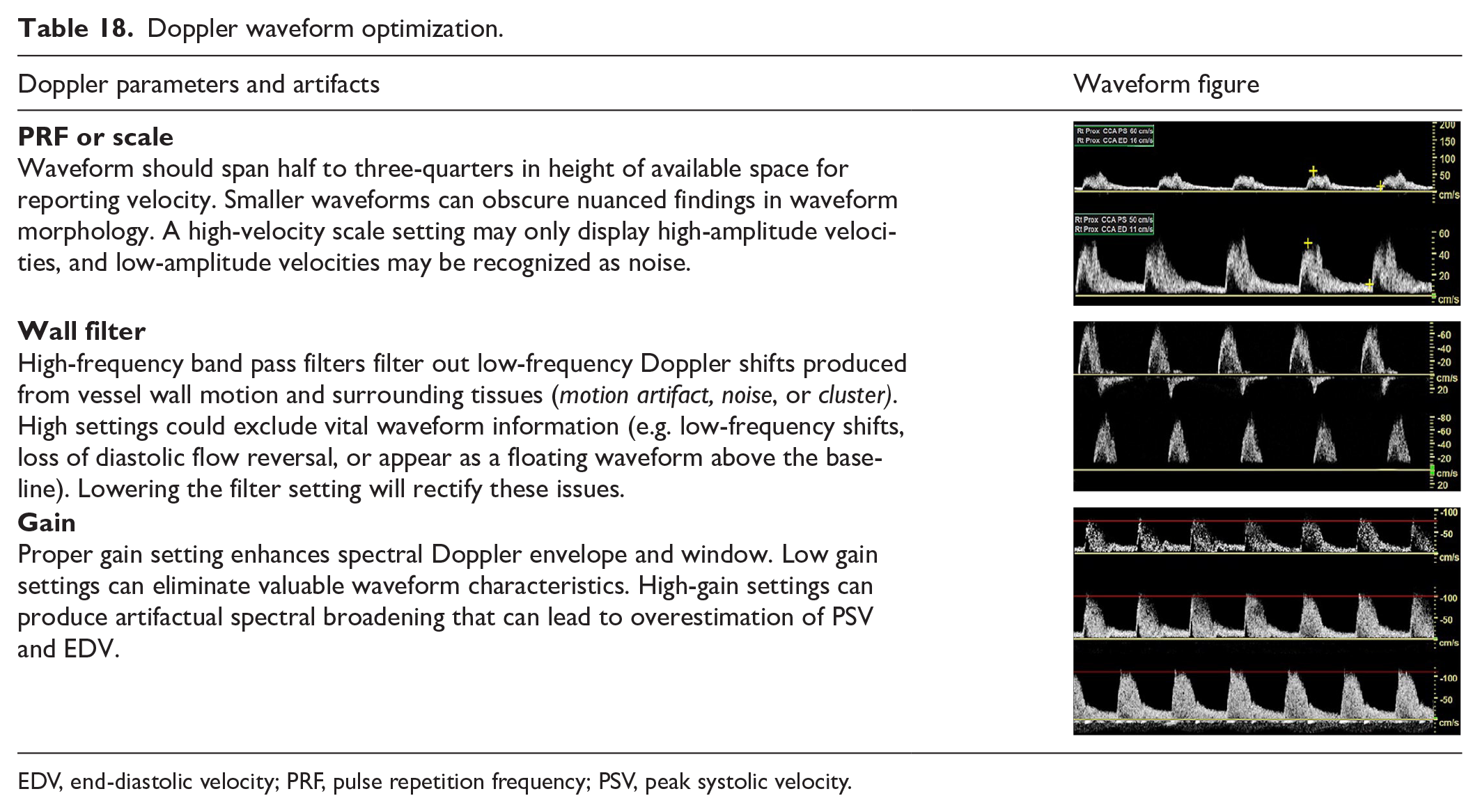
EDV, end-diastolic velocity; PRF, pulse repetition frequency; PSV, peak systolic velocity.

CCA, common carotid artery; CFA, common femoral artery; CW, continuous wave; PRA, profunda artery; PRF, pulse repetition frequency.
Part 4: Interpretation and reporting
Purpose: Provide interpretation and reporting examples to facilitate use of the recommendations outlined in this consensus document
The sonographer will complete a written description of the ultrasound examination, including the study indication, pertinent history, and appropriate images, along with velocity measurements and waveform descriptions. A standardized description of waveforms allows the waveform display to be recreated in words, providing useful information relevant to the clinical indication for the examination.
The conclusion/interpretation is most helpful to the referring physician if it includes not only the diagnostic findings but also the clinical indication and prior relevant examinations.
Illustrative case examples are provided in the supplemental material, which is available online with the article.
Summary
Spectral Doppler waveform assessment has been the principal diagnostic tool for the noninvasive diagnosis of peripheral artery and venous disease since the 1980s.36,37 The absence of uniformly accepted, standardized descriptors for arterial and venous Doppler waveforms has been a long-standing point of controversy 38 and the main impetus for the development of this consensus statement, which is a major step towards clarifying and standardizing the key definitions and descriptors that are inherent to the analysis of arterial and venous Doppler waveforms. All interpreting physicians and sonographers should be able to describe Doppler waveforms, be able to identify the changes which occur with physiologic and disease states, and effectively communicate these waveform characteristics to interpreting physicians so that consistent information is given to referring providers. This consensus statement outlines a simple nomenclature for the description of arterial and venous Doppler waveforms (Table 1), which will hopefully alleviate the confusion regarding Doppler waveform interpretation that has plagued the field for decades.
Supplemental Material
Supplementary_Material – Supplemental material for Interpretation of peripheral arterial and venous Doppler waveforms: A Consensus Statement from the Society for Vascular Medicine and Society for Vascular Ultrasound
Supplemental material, Supplementary_Material for Interpretation of peripheral arterial and venous Doppler waveforms: A Consensus Statement from the Society for Vascular Medicine and Society for Vascular Ultrasound by Esther SH Kim, Aditya M Sharma, Robert Scissons, David Dawson, Robert T Eberhardt, Marie Gerhard-Herman, Joseph P Hughes, Steve Knight, Ann Marie Kupinski, Guillaume Mahe, Marsha Neumyer, Patricia Poe, Rita Shugart, Paul Wennberg, David M Williams and R Eugene Zierler in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Patricia Poe is a consultant for Philips Ultrasound. No other authors have any potential conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
