Abstract
Retinal vein occlusion (RVO) is a common retinal vascular lesion, and a leading cause of visual impairment. Patients with RVO have an increased risk for cardiovascular disease and share multiple common risk factors. In this study, we investigated the endothelial function and arterial stiffness of patients with RVO compared to healthy-control (CL) subjects. We enrolled 40 consecutive patients with RVO and 40 CL subjects. RVO was diagnosed by an ophthalmologist, endothelial function was evaluated by flow mediated dilation (FMD) in the brachial artery, and carotid-femoral pulse wave velocity (PWV) and augmentation index (AIx) of the radial artery were measured to evaluate arterial stiffness and reflected waves, respectively. No significant differences were detected between the studied groups in sex, age, presence of hypertension or dyslipidemia, body mass index, systolic and diastolic blood pressure levels, total cholesterol levels, and smoking habits (p > 0.05 for all). However, patients with RVO had impaired FMD (p = 0.002) and increased PWV (p = 0.004), even after adjustment for several confounders. Both FMD and PWV were also significantly and independently associated with the development of RVO. Furthermore, a significant and positive correlation between PWV and systolic blood pressure existed only in the CL group. Therefore, we have shown that RVO is associated with significant endothelial dysfunction and increased arterial stiffness. Our results strengthen the vascular theory, according to which, systemic endothelial dysfunction and arteriosclerosis play a significant role in the pathogenesis of RVO.
Keywords
Introduction
Retinal vein occlusion (RVO) is the second most common retinal vascular lesion, being a significant cause of unilateral visual impairment and blindness.1,2 Based on the location of the vein lesion, RVOs are divided into central (CRVO) and branch (BRVO) retinal vein occlusion. The occlusion in CRVO is located in the central retinal vein in the lamina cribrosa and/or posterior to it, whereas in BRVO the occlusion occurs in any branch of the central retinal vein, especially at the arteriovenous crossing.
Several systemic and ocular risk factors have been identified as potent contributors in the development of RVO. Classic risk factors include advanced age, hypertension, hyperlipidemia, and diabetes mellitus (DM).3–8 Moreover, smoking, elevated body mass index (BMI), black race, oral intake of the contraceptive pill, glaucoma, and thrombophilic risk factors have been associated with RVO.3–10
Although multiple mechanisms have been suggested, the exact pathogenesis of RVO has yet to be elucidated. It has been proposed that BRVO is mainly caused by the compression of the branch vein at the arteriovenous crossing by the branch retinal artery, 11 while CRVO is possibly caused by a disturbance of circulation at the trunk of the central retinal vein near the lamina cribrosa. 2 Other possible mechanisms include hemodynamic changes (venous stasis), degenerative changes of the vessel wall, and blood hypercoagulability (Virchow’s triad). 12
Patients with RVO have an increased risk of cardiovascular diseases (CADs), sharing also several common risk factors.13,14 The exact mechanism connecting them remains unclear. Endothelial dysfunction and impaired arterial wall properties are key factors of cardiovascular pathophysiology.15–18 Furthermore, it has been demonstrated that both of them are associated with impaired ocular circulation, and more specifically with diseases, such as diabetic retinopathy,19,20 glaucoma,21,22 and sarcoid uveitis. 23 However, a few data exist in the literature concerning their possible pathogenetic role in the development of RVO.12,24,25 Therefore, we aimed to determine the existence of a direct and significant association among RVO development, atherosclerosis, endothelial function, and arterial wall properties, examining whether they are implicated in RVO pathogenesis.
Methods
Study design and population
In this case-control study, we included 40 consecutive naive and previously untreated patients with RVO (either CRVO or BRVO) and 40 consecutive healthy-control subjects (CL). All the participants were of Caucasian origin. Patients with RVO were recruited from the 1st Department of Ophthalmology of the Athens Medical School, Greece. The CL population consisted of healthy subjects who referred to the outpatient cardiology department of the 1st Department of Cardiology of the Athens Medical School, Greece, for preventive cardiology examination. None of the CL subjects had a history of CAD, systemic or other inflammatory diseases or ocular diseases. All subjects detected with peripheral artery disease (PAD) had asymptomatic lower extremity artery disease.
The ophthalmological examination was performed by the same experienced ophthalmologist, while the vascular function was evaluated by the same observer who was unaware of the participants’ disease status. Every participant refrained from caffeine, alcohol, smoking, and any food for 12 hours before the vascular examination.
Demographic and clinical data, including age, sex, BMI, smoking habits, hypertension, and hypercholesterolemia were collected. Hypertension was diagnosed in all subjects who had antihypertensive medication or had systolic blood pressure (SBP) ⩾ 140 mmHg or diastolic blood pressure (DBP) ⩾ 90 mmHg.
To identify as many as possible straightforward associations between RVO, arteriosclerosis, and arterial vascular parameters, we excluded any potential confounders. Therefore, subjects suffering from DM (or who had pre-diabetes), based on the diagnostic criteria of the American Diabetes Association, 26 and/or were under regular use of antidiabetic medication, were excluded from the study. Furthermore, patients suffering from systemic diseases, severe renal insufficiency (glomerular filtration rate < 30.0 mL/min, calculated with Modification of Diet in Renal Disease [MDRD] formula), 27 known or suspected neoplasm, hematological malignancies, immunosuppression, recent (within the previous 6 months) surgery, pregnancy or lactation, severe valvular disease, atrial fibrillation, chronic heart failure, and a history of coronary artery disease, myocardial infarction or stroke were excluded from the study. Moreover, patients suffering from glaucoma or any other retinal disorder, such as hypertensive retinopathy or age-related macular degeneration, were not included in the study.
The study was performed according to the Helsinki Declaration and was approved by the Institutional Ethics Committee. Written informed consent was obtained by every participant in the study.
Assessment of retinal vein occlusion
All participants underwent a complete ophthalmological examination, which included measurement of best-corrected visual acuity (Standard Snellen chart), slit lamp biomicroscopy, tonometry (Goldman applanation), and funduscopy after pupil dilation. RVO was identified by the presence of one or more of the following clinical features in the fundus: tortuous and dilated retinal vein vessels, flame and/or dot and blot hemorrhages, cotton-wool spots, hard exudates, and macular edema. Fluorescein angiography and optical coherence tomography (OCT) scans (Spectralis HRA + OCT; Heidelberg Engineering, Heidelberg, Germany) were performed on all the patients in order to confirm the diagnosis and to quantitatively evaluate the retinal thickness, respectively.
Evaluation of endothelial function
Endothelial function was evaluated by estimating the flow mediated dilation (FMD) in the brachial artery, as it has been described previously. 28 Briefly, after a 10-minute rest, the participants’ right brachial artery was scanned in longitudinal section, 5 cm above the antecubital fossa using a linear array ultrasound transducer. A pneumatic cuff placed distal to the ultrasound probe was then inflated to supra-systolic pressure on the forearm for 5 minutes to induce reactive hyperemia. After release of the ischemia cuff, the brachial artery diameter was measured every 15 seconds for 2 minutes. FMD was defined by the following formula: FMD(%) = [(maximum artery lumen diameter after cuff release – artery lumen diameter at baseline) / artery lumen diameter at baseline] × 100. The repeatability of the technique in our institution (determined according to the Bland-Altman method) was 5.0%, suggesting good repeatability.
Evaluation of aortic elastic properties
Carotid-femoral pulse wave velocity (PWV), a well-established index of aortic stiffness, was calculated from measurements of pulse transit time and the distance traveled between two recording sites (PWV = distance in meters / transit time in seconds) by using a well-validated noninvasive device (SphygmoCor, AtCor Medical, Sydney, Australia), as previously described.29,30
Measurement of wave reflection indexes
The augmentation index (AIx) of the central (aortic) pressure waveform was measured as an index of wave reflection. AIx is a composite measure of the magnitude of wave reflection and arterial stiffness, which affects the timing of wave reflection. Because AIx is influenced by changes in heart rate, it was also corrected accordingly (corrected for a steady heart rate of 75 beats per minute – AI75). AI75 was measured with a validated, commercially available system (SphygmoCor, AtCor Medical) that uses the principle of applanation tonometry and appropriate acquisition and analysis software for noninvasive recording and analysis of the arterial pulse, as previously described. 31 Waveforms of radial pressure were calibrated according to sphygmomanometric SBP and DBP measured in the brachial artery.
Statistical analysis
All variables were tested for normal distribution of the data. Normally distributed data were expressed as mean ± SD, and skewed variables as median with first and third quartiles. The comparisons of mean values between groups of normally and not skewed continuous variables were performed with Student’s t-test and Mann–Whitney test, respectively, while a chi-squared test was used to test for group differences between categorical variables. The correlations between continuous variables were analyzed by the Pearson’s correlation test. Regression analysis was used to test for differences in FMD, PWV, and AIx between study groups after adjustment for possible confounders (age, sex, BMI, presence of hypertension, dyslipidemia, and smoking habits). Statistical significance was indicated by p < 0.05. All statistical calculations were performed using SPSS software, version 20.0 (IBM Corp., Armonk, NY, USA).
Results
The demographic and clinical characteristics of the patients and the CL population are shown in Table 1. No significant differences existed between the studied groups in mean age (68 ± 11 years vs 69 ± 6 years, p = 0.80) and sex status (48% male vs 50% male, p = 0.85), as well as in BMI, frequency of hypertension, dyslipidemia, and peripheral artery disease, smoking habits, and the levels of SBP, DBP, and total cholesterol (p > 0.05 for all).
Demographic and clinical characteristics of the participants.
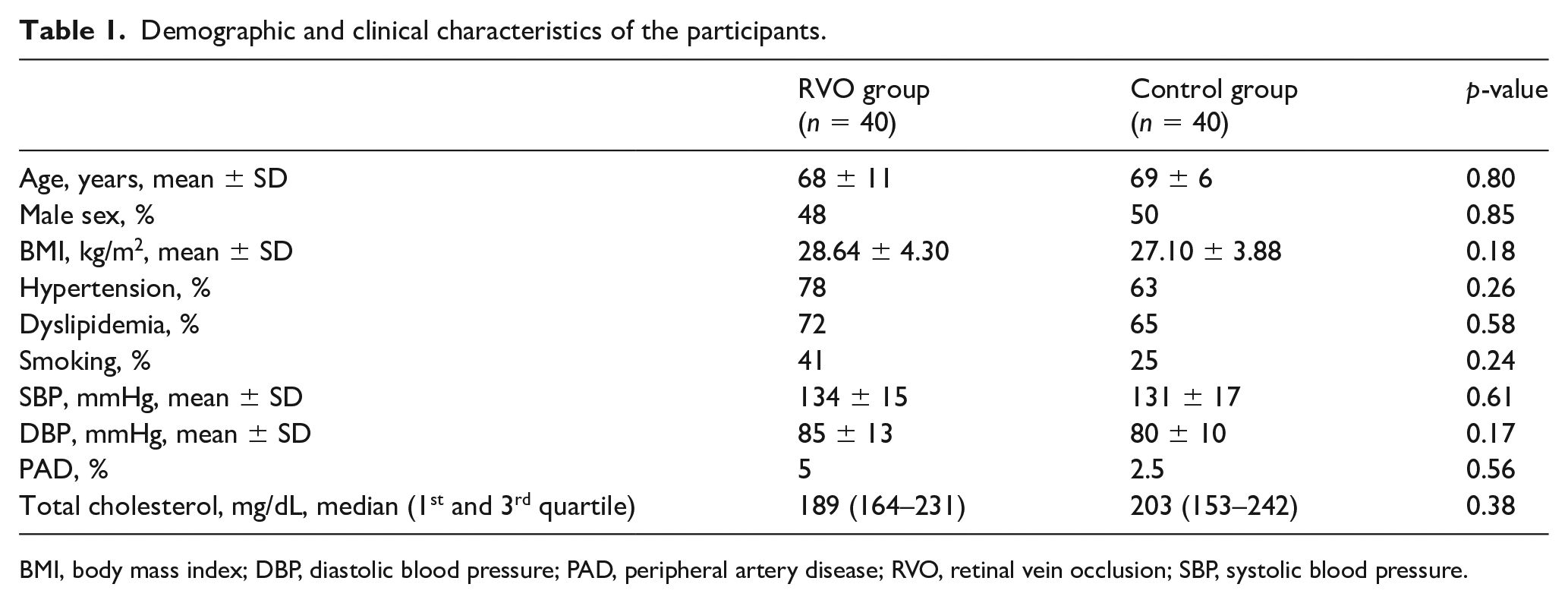
BMI, body mass index; DBP, diastolic blood pressure; PAD, peripheral artery disease; RVO, retinal vein occlusion; SBP, systolic blood pressure.
The patients with RVO, compared to CL subjects, had impaired FMD values (3.56 ± 0.81% vs 6.80 ± 2.79%, p < 0.001), increased PWV (11.21 ± 2.41 m/sec vs 8.66 ± 1.50 m/sec, p < 0.001), and elevated AIx (30.21 ± 7.45% vs 23.80 ± 7.58%, p = 0.01) (Figure 1).
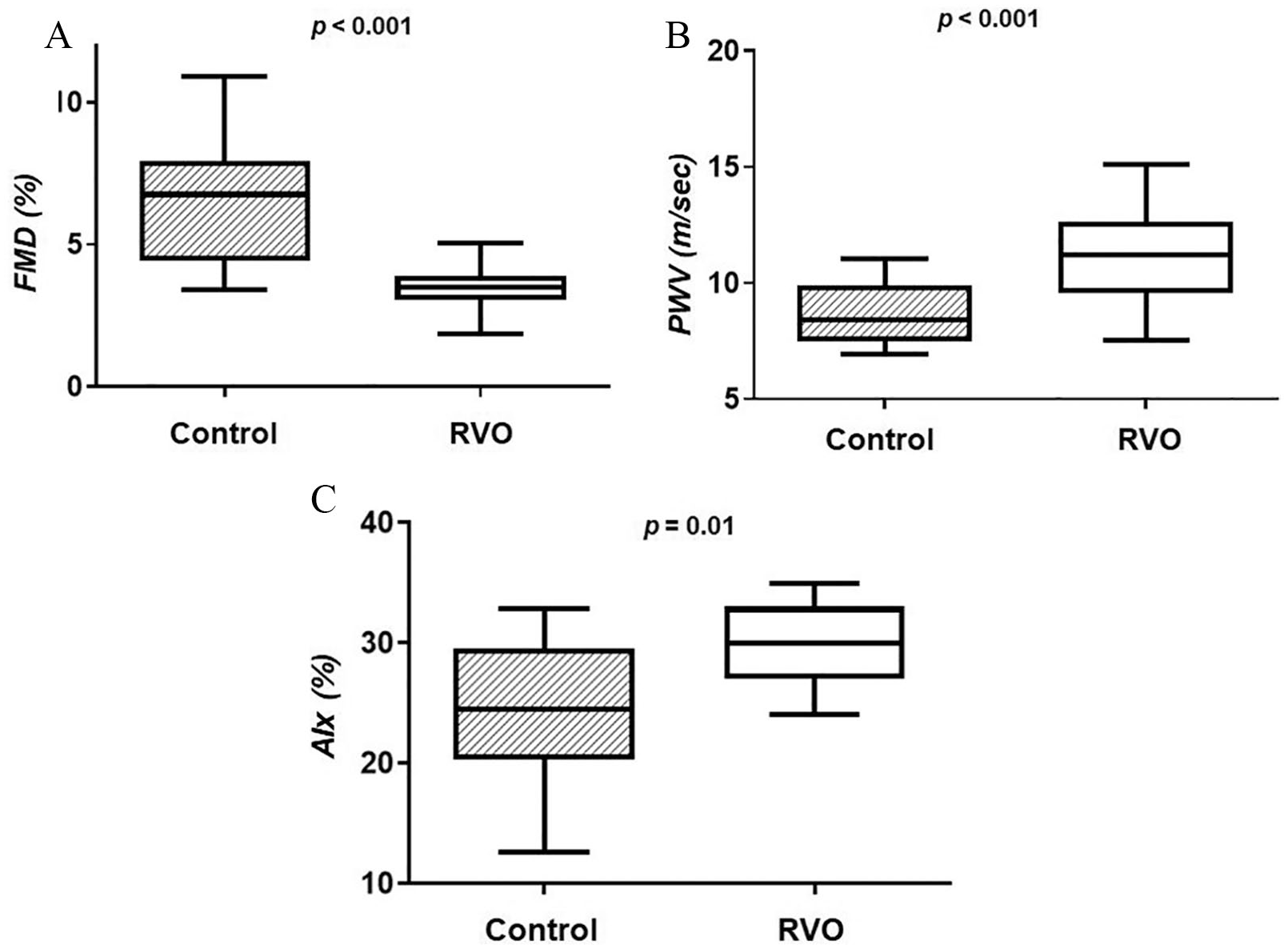
Box plots representing the differences in FMD (A), PWV (B), and AIx (C) between control subjects and patients with RVO.
Interestingly, both FMD and PWV were significantly impaired in patients with RVO compared to CL subjects, even after adjustment for multiple known confounders, such as age, sex, BMI, presence of hypertension, dyslipidemia, and smoking habits (p = 0.002 and p = 0.004, respectively) (Table 2).
Differences in FMD, PWV, and AIx between studied categories after adjustment for demographics and cardiovascular risk factors.
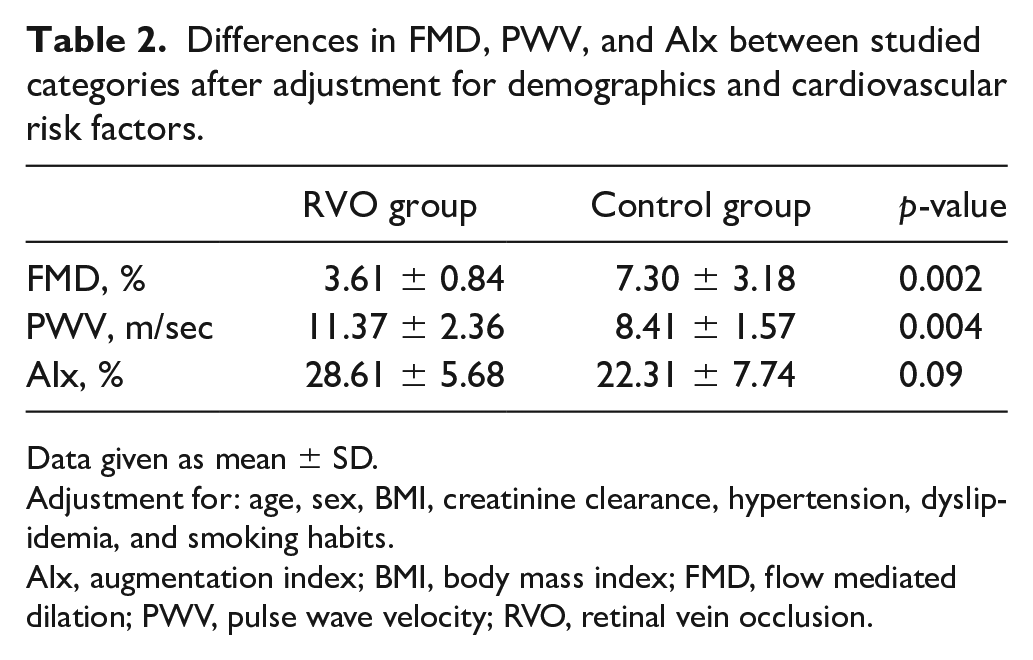
Data given as mean ± SD.
Adjustment for: age, sex, BMI, creatinine clearance, hypertension, dyslipidemia, and smoking habits.
AIx, augmentation index; BMI, body mass index; FMD, flow mediated dilation; PWV, pulse wave velocity; RVO, retinal vein occlusion.
In order to detect which arterial parameter had the highest predictive value for the development of RVO, we applied a logistic regression model. Although univariate analysis revealed that both FMD and PWV were significantly associated with RVO (Table 3), impaired FMD was better associated with the development of RVO (odds ratio (OR) 0.41, 95% CI 0.21–0.80, p = 0.008). Furthermore, to exclude the impact of several covariates, such as age, sex, hypertension, dyslipidemia, BMI, and smoking habits, we applied a multinomial regression analysis. As shown in Table 4, impaired FMD was independently associated with increased odds of RVO development (OR 0.24, 95% CI 0.08–0.81, p = 0.02) and that for every 1% increase in FMD there is an anticipate 75% decrease in the odds of RVO. Also, a multinomial regression analysis with confounders such as age, sex, hypertension, dyslipidemia, BMI, and smoking habits revealed that higher PWV was also independently associated with elevated odds of RVO development (OR 4.52, 95% CI 1.18–17.39, p = 0.04).
Results of the univariate regression analysis of the association of RVO development (dependent variable) with FMD, PWV, and AIx.
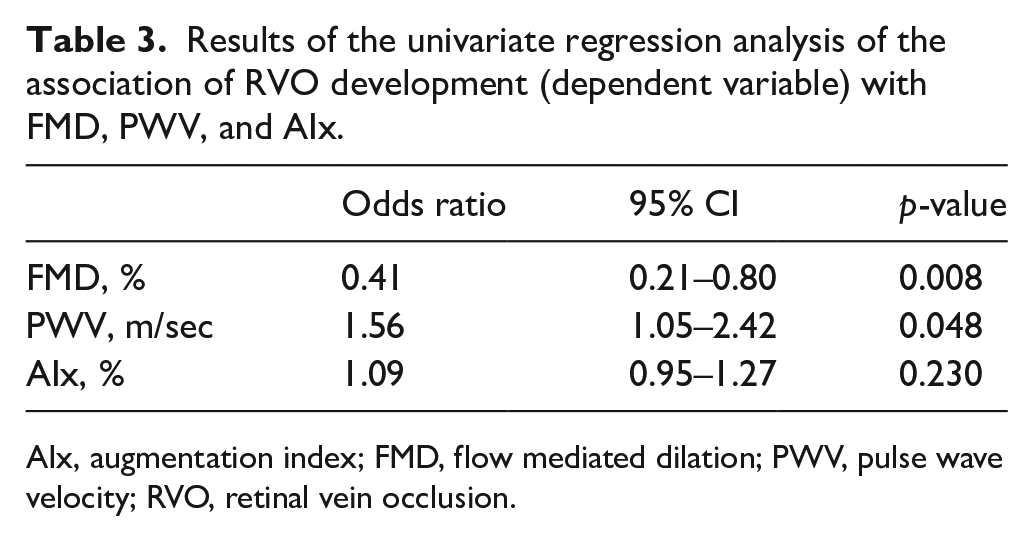
AIx, augmentation index; FMD, flow mediated dilation; PWV, pulse wave velocity; RVO, retinal vein occlusion.
Logistic regression analysis for the odds of RVO after adjustment for several covariates.
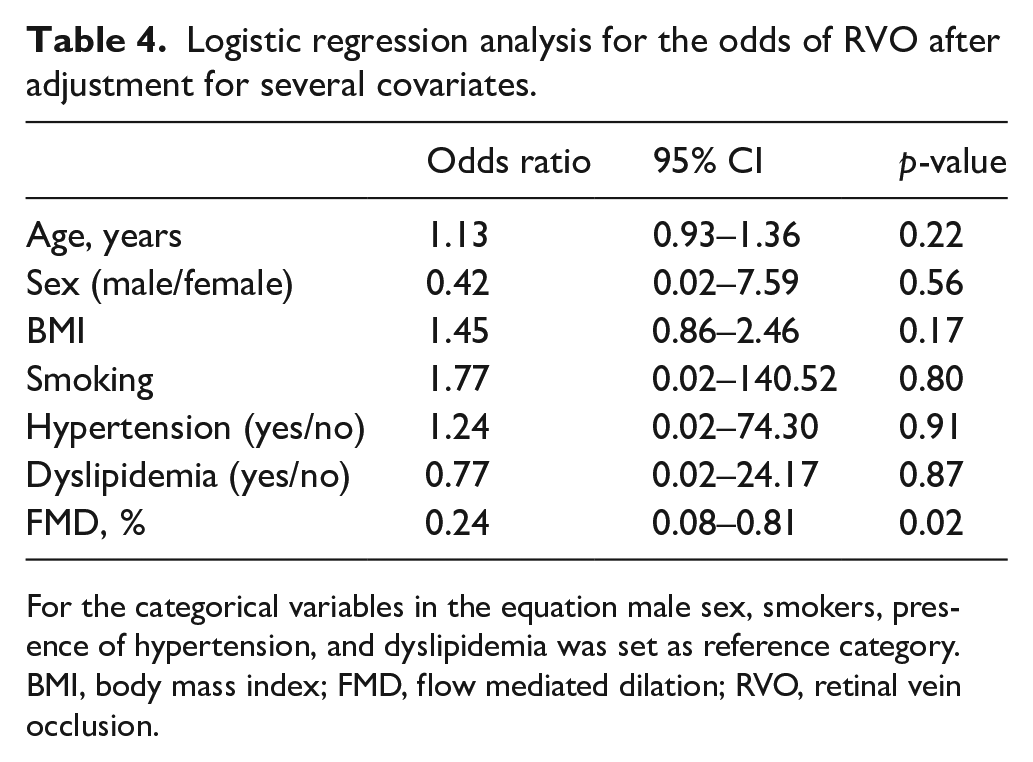
For the categorical variables in the equation male sex, smokers, presence of hypertension, and dyslipidemia was set as reference category.
BMI, body mass index; FMD, flow mediated dilation; RVO, retinal vein occlusion.
Finally, among the patients with BRVO, no significant correlation was detected between PWV and SBP (r = −0.104, p = 0.74), while a significant and positive correlation existed in the control group (r = 0.372, p = 0.02).
Discussion
In the present study, we found that vascular peripheral endothelial function and arterial stiffness are impaired in patients with RVO, compared to a CL population. For the first time in the literature, we documented that the deterioration of both endothelial function and arterial wall properties could be identified as novel independent risk factors for the development of RVO, since a significant and independent association with RVO was detected, regardless of the impact of multiple confounders.
Endothelial function and retinal vein occlusion
Endothelium plays an important role in the vascular function by secreting multiple substances that regulate vascular tone and structure, platelet activity, coagulation factors, vascular inflammation, cell migration, and proliferation.32,33 Endothelial dysfunction is characterized by reduced nitric oxide (NO) bioavailability, coexists with altered vascular reactivity, precedes the development of overt atherosclerosis,30,34 and is associated with the development of CAD and a prothrombotic or hypercoagulable state.35,36
Concerning the underlying mechanism linking RVO development with arterial impairment, it has been suggested that RVO is the consequence of the damage that occurs to the vein vascular endothelium and intima media. More specifically, turbulent blood flow and greater shear stress, because of the mechanical obstruction of the vein through the rigid artery at the arteriovenous crossing (BRVO) or in the lamina cribrosa (CRVO), result in endothelial cell damage and cell proliferation.37,38
As we mentioned before, systemic endothelial dysfunction is implicated in the pathogenesis of several retinal vascular disorders, including sarcoid uveitis, 23 glaucoma,21,22 and diabetic retinopathy,19,20 being a key factor for the development of the latter. 20 However, it still remains unclear whether and to which extend impaired FMD could reflect the local endothelial dysfunction in retinal microvasculature.
Our findings suggested that systemic endothelial dysfunction is strongly and independently associated with RVO, highlighting the close relationship of retinal microvascular abnormalities with systemic atherosclerosis. Furthermore, the logistic regression analysis excluded the impact of various covariates in the association between FMD and RVO, demonstrating that the detected differences were attributed to the presence of RVO. Therefore, we hypothesized that systemic endothelial dysfunction could be identified as an independent risk factor for the onset of RVO.
Our findings confirm a previous study by Tanano et al. 24 They documented the existence of a significantly deteriorated endothelial function in patients with BRVO, excluding though from their studied population patients with CRVO and smokers. In contrast to them, we demonstrated that the proposed association is present regardless of the type of RVO, and we also showed that this association is not impeded by the participants’ smoking habits.
Arterial stiffness and retinal vein occlusion
We also documented an increase in arterial stiffness in patients with RVO, suggesting an important role for systemic arterial stiffness in the disease onset.
Central retinal artery and vein lie together in the same adventitial sheath, 38 while at arteriovenous crossings common adventitial sheath is also shared by branch retinal arteries and veins. It has been proposed that the more rigid arterial wall compresses the vein, increasing thus the likelihood of vein occlusion, a risk which is more prominent in cases of arteriosclerotic thickening of the arterial wall. 38 Consistent with the aforementioned theory were the findings by Frangieh et al., who histopathologically demonstrated that in almost all the examined cases with BRVO, moderate to severe sclerosis was present at the crossing arterioles. 39 Moreover, it has been recently postulated that atherosclerotic arteries may produce increased endothelin-1, resulting in stimulating venous vasoconstriction, elevating thus the risk of venous occlusion. 40
It is well established that carotid-femoral PWV efficiently and non-invasively evaluates the aortic arterial stiffness.41,42 Furthermore, arterial wall properties (structure and geometry) and SBP are the key factors that influence the arterial stiffness and subsequently the PWV levels. 25 When structural impairment is present, the arterial stiffness is less dependent on SBP. 43 In our study, we did not detect a significant correlation between blood pressure and PWV in patients with RVO, whereas in the CL population PWV correlated positively and significantly with the levels of SBP. These results suggest that the higher PWV levels in patients with RVO could be attributed to the structural impairment of the arterial wall, a condition which further elevates the risk of CADs.
Similar findings were also found by Kaderli et al., 11 Nakazato et al., 25 and Chen et al., 44 who showed that increased arterial stiffness was present in patients suffering from BRVO. All the previous studies examined the existence of this association only in patients with BRVO. Moreover, the previous studies neither evaluated the arterial stiffness by measuring the carotid-femoral PWV (the gold standard index of the aortic-systemic arterial stiffness) nor excluded the influence of several covariates in the detected association. On the contrary, we showed that elevated PWV is significantly associated with RVO, regardless of the impact of multiple confounders.
Furthermore, impaired PWV has been recognized as an index of CAD and as an independent predictor of future cardiovascular risk.45,46 Our results highlight the systemic nature of vascular disease in patients with RVO, proposing that PWV may be a key screening tool for the risk of CAD in patients with RVO. However, more prospective studies are needed in order to determine the exact role of arterial stiffness in the development of RVO and to reveal the value of PWV in predicting cardiovascular morbidity and mortality in patients with RVO.
Study limitations
Despite the interesting findings of this study, there are some inherent limitations. The relatively small sample size did not allow examination in a univariate or multivariate analysis the impact of further confounders, such as the impact of PAD or the use of antihypertensive agents. Moreover, the stringent exclusion criteria for patients in this study limit the generalizability of our findings.
Conclusion
In our study, we documented that patients with RVO have impaired endothelial function and increased arterial stiffness, providing further insights in the pathophysiology of the disease. These findings merit further investigation to strengthen the vascular theory, according to which systemic endothelial dysfunction and arteriosclerosis is linked to RVO development.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
