Abstract
Patients with critical limb ischemia have nonhealing wounds and/or ischemic rest pain and are at high risk for amputation and mortality. Accurate evaluation of foot perfusion should help avoid unnecessary amputation, guide revascularization strategies, and offer efficient surveillance for patency. Our aim is to review current modalities of assessing foot perfusion in the context of the practical clinical management of patients with critical limb ischemia.
Keywords
Continuing medical education (CME) credits and maintenance of certification (MOC) points are available for reading this article This opportunity is made possible through a joint partnership with University of Virginia School of Medicine (UVASOM). For instructions, please visit the Society for Vascular Medicine’s website at
Accreditation statement
The University of Virginia School of Medicine (UVASOM) is accredited by the ACCME to provide continuing medical education for physicians. UVASOM designates this journal-based CME activity for a maximum of one AMA PRA Category 1 Credit™. Participants should claim only the credit commensurate with the extent of their participation in the activity. UVASOM, as an accredited provider, awards 1 hour of participation (consistent with the one AMA PRA Category 1 Credit™) to participants who successfully complete this educational activity. Successful completion of this CME activity, which includes participation in the evaluation component, enables the participant to earn up to one MOC II point in the American Board of Internal Medicine’s (ABIM) Maintenance of Certification (MOC) program. It is the CME provider’s responsibility to submit participant completion information to ACCME for the purpose of granting ABIM MOC II credit. UVASOM maintains a record of participation for 6 years.
Disclosures
The faculty, staff, and planning committee of the University of Virginia Office of Continuing Medical Education have no financial affiliations to disclose. The CME planning committee disclosed the following: Heather Gornik receives intellectual property rights and equity from FlexLife Health; Aditya Sharma is supported by National Institutes of Health, AstraZeneca, and Vascular Medcure; and Valerie Clark has no financial affiliations to disclose. Kevin Rogers has been granted 10 FDA-approved pressure wires by Phillips to use in the assessment of limb perfusion (separate project). He has no direct financial compensation from this company. Miguel Montero-Baker has stock in Profusa, Inc. The other authors have no financial relationships or interests with a proprietary entity that produces healthcare goods or services related to this article.
Introduction
Critical limb ischemia (CLI) is the most severe form of atherosclerotic peripheral artery disease (PAD). 1 Patients with CLI have nonhealing wounds and/or ischemic rest pain and are at high risk for amputation and mortality.2,3 Better understanding of foot perfusion should enable our ability to avoid these adverse outcomes in patients with CLI.
The ideal test for foot perfusion would be inexpensive, readily available, reproducible, and improve the clinician’s ability to predict outcomes. Additionally, the ideal test might provide perfusion data specific to the area of the foot with a wound. 4 Improving the ability to evaluate foot perfusion would theoretically benefit patients with CLI and providers by: (1) assisting with the etiology of a nonhealing foot wound; (2) identifying patients with poor perfusion in the angiosome of interest who might benefit from revascularization; (3) identifying patients with seemingly adequate perfusion who may not require revascularization; (4) selecting a target vessel for revascularization; (5) providing insight when revascularization is sufficient; and (6) facilitating surveillance for patency. 4 Accurate assessment of foot perfusion should help to avoid unnecessary amputation and to direct revascularization attempts to patients with wounds due to ischemia.
However, current modalities of foot perfusion have limitations. For example, arterial noncompressibility makes the ankle–brachial index (ABI) non-meaningful in a significant minority of patients with CLI, and transcutaneous oximetry has a high coefficient of variation.5–9 Intra-procedural measures of foot perfusion are attractive to the interventionalist to direct the target and extent of revascularization, and measures such as ‘angiographic blush’ have been reported to correlate with wound healing. 10 However, ‘angiographic blush’ is somewhat subjective and dependent on technique. Two-dimensional (2D) angiographic perfusion imaging is an intra-procedural method that attempts to provide objective angiographic data of perfusion but is also technique-dependent and is yet to be validated.
Another limitation of certain modalities, such as the ABI, is the inability to provide information regarding regional foot perfusion. The ‘angiosome hypothesis’ asserts that establishing direct, in-line flow to the area of the limb with an ischemic wound provides superior wound healing rather than pursuing indirect, global revascularization of the limb. 11 The evidence for the angiosome hypothesis is limited, supported by studies that are small and observational, but the angiosome hypothesis is biologically plausible and almost impossible to test in a randomized fashion. Regardless, the ability to assess regional foot perfusion is an attractive attribute of a test.
Our aim is to review current modalities of assessing foot perfusion in the context of the practical clinical management of patients with CLI with the goal of improving limb outcomes.
Modalities for the assessment of foot perfusion
Various modalities for assessing foot perfusion are listed in Table 1. The ideal test for foot perfusion is discussed above.
Summary of foot perfusion modalities.
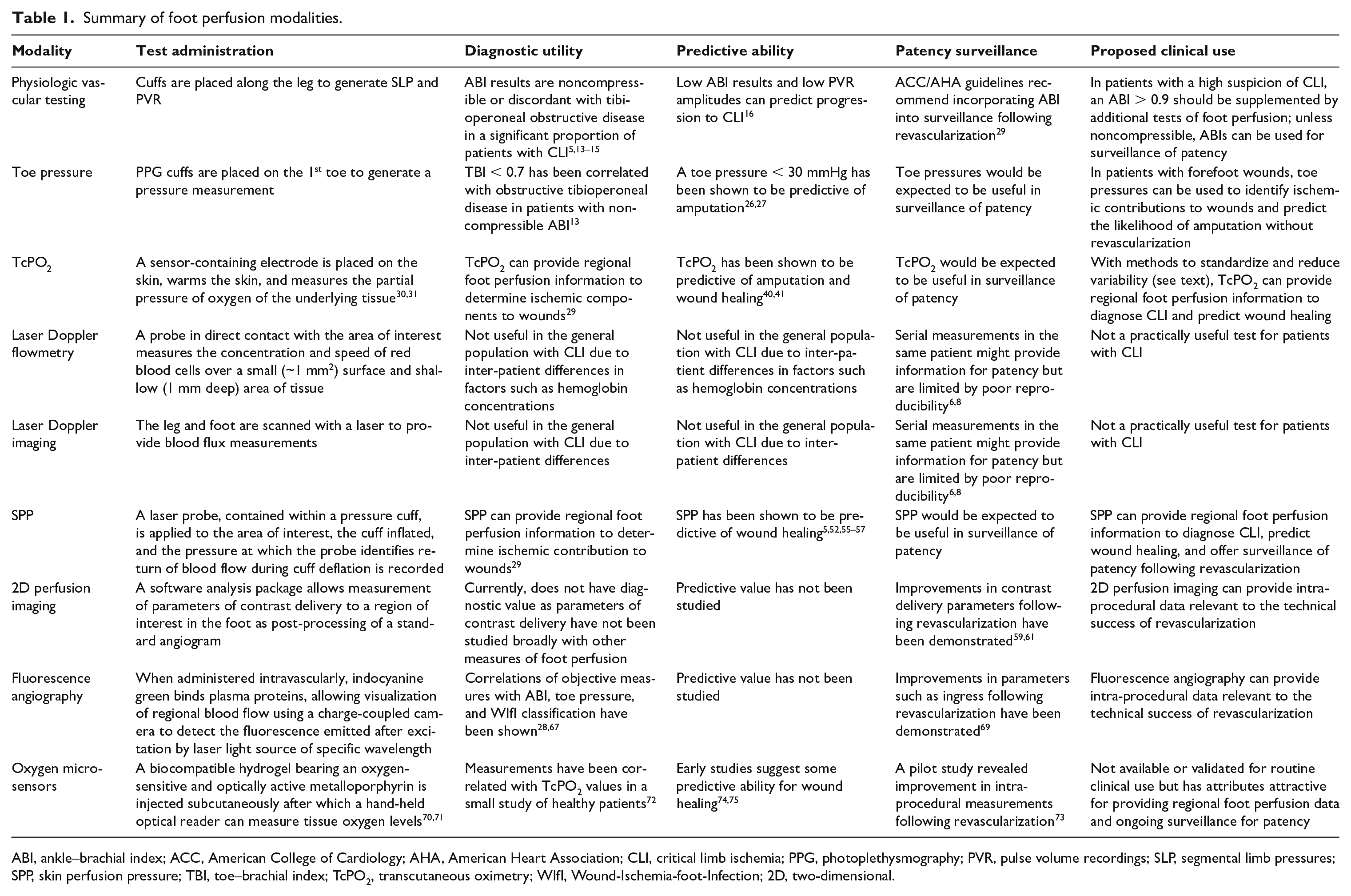
ABI, ankle–brachial index; ACC, American College of Cardiology; AHA, American Heart Association; CLI, critical limb ischemia; PPG, photoplethysmography; PVR, pulse volume recordings; SLP, segmental limb pressures; SPP, skin perfusion pressure; TBI, toe–brachial index; TcPO2, transcutaneous oximetry; WIfI, Wound-Ischemia-foot-Infection; 2D, two-dimensional.
Physiologic vascular testing
A traditional physiologic vascular study with segmental limb pressures and pulse volume recordings is central in assessing and diagnosing the PAD patient. The ankle–brachial index (ABI) is a gold standard in the diagnosis of PAD in the general population and has predictive value for cardiovascular events independent of traditional risk factors. 12 However, there are limitations to the ABI as a stand-alone test for the CLI patient. For example, arterial noncompressibility makes the ABI non-meaningful in a significant minority of this patient population 5 and limits the ability of ABI results to identify obstructive tibioperoneal disease. In a study of 125 limbs with ABI values greater than 1.4, 74% of tibioperoneal arteries were occluded or stenotic by catheter-based angiography. 13 In a separate study, among 89 patients with rest pain or tissue loss who had compressible ABIs and underwent angiography, ABI values did not statistically differ across subgroups with zero, one, two, or three patent tibioperoneal arteries, suggesting that noninvasive ankle pressures do not correlate with anatomic severity of obstructive PAD below the knee. 14 Moreover, among 10,756 patients thought to have CLI who underwent revascularization in a multicenter registry, ABI was measured in only 56% of patients. 15 When measured, the ABI was noncompressible in 11% of patients and normal or borderline (between 0.9 and 1.4) in 21% of patients. These results suggest that vascular testing is underutilized in CLI patients, that ABI measurements are nondiagnostic or falsely reassuring in almost a third of CLI patients, and that there is a disconnect between clinical diagnosis and ABI results. The ABI may be a better assessment of calf perfusion, relevant to the claudicant, rather than an accurate test for regional foot perfusion. 4
Pulse volume recordings complement the ABI in the traditional vascular diagnostic study. In a retrospective study of 1500 patients undergoing vascular studies at a single center, 23 patients had a pulse volume recording amplitude of 3 mm or less, an ABI of 0.35 or less, and were asymptomatic. Over a mean follow-up of 45 months, 48% of these asymptomatic patients developed CLI. 16
Toe pressures and toe–brachial indices (TBI) can offer incremental information to ABIs. Toe pressures are less subject to the noncompressibility that is often seen in patients with severe arterial medial calcinosis. 17 In normal physiology, toe pressures are typically 20–30 mmHg lower than ankle pressures; thus, normal TBIs range from 0.7 to 0.8.
There are currently two methods to measure toe pressures: photoplethysmography (PPG) and laser Doppler (LD). There are minor differences in the sensitivity of the two techniques, such as laser Doppler’s intrinsic ability to measure lower toe pressures as it does not require pulsatile flow. 18 Studies have previously demonstrated adequate reproducibility of each modality; however, there is variability when comparing the two techniques.19–21 Therefore, it is suggested that the same modality to measure toe pressures be used for serial measurements in a single patient.
Absolute toe pressures of ⩽ 30 mmHg in the presence of rest pain or ⩽ 50 mmHg in the presence of ulceration are traditional cut-offs for the diagnosis of CLI. 22 In clinical studies, absolute toe pressures have been correlated to a likelihood of wound healing. For non-diabetic patients, a toe pressure of ⩾ 30 mmHg predicts successful wound healing. 23 However, in diabetic patients, a toe pressure ⩾ 45–50 mmHg may be required for successful wound healing.24,25 There are limited data describing the relationship of toe pressures with clinical outcomes, such as cardiovascular mortality and amputation. For example, in a retrospective study of symptomatic PAD patients referred for angiography, a toe pressure ⩽ 30 mmHg was associated with increased amputation and death while adjusting for ABI (hazard ratio (HR) 2.13, 95% CI 1.52–2.98; HR 2.46, 95% CI 1.38–4.40, respectively). 26 A retrospective study of 566 patients with CLI analyzed the predictive value of toe pressures for major amputation by categorizing patients into medical and revascularization groups. A toe pressure of ⩽ 30 mmHg predicted amputation in the medical group (OR 5.0, 95% CI 2.0–12.4) but not in the revascularization group. 27 In clinical practice, the likelihood of wound healing may better follow a probability spectrum of toe pressures rather than discreet cut-offs applied to all patients. That is, a diabetic with a large, infected wound requires more perfusion (reflected by a higher toe pressure) than a non-diabetic with a small, sterile ulcer.4,28
The measurement of toe pressures is noninvasive and relatively inexpensive with widespread availability due to low cost and reproducibility. 21 There are limitations to the use of toe pressures in assessing foot perfusion, including prior amputation of the first toe, extreme calcification causing noncompressibility, vasoconstriction of digital arteries (which may be avoided by obtaining toe pressures in a warm room), 29 and perhaps accuracy of the toe pressure in representing perfusion to the heel.
Physiologic vascular testing should be a first-line test in patients with CLI. Pulse volume recordings and toe pressures should be standard components of a vascular diagnostic study to detect ischemia as a cause or contributor of a foot wound, particularly if the ABI result is borderline or normal (0.9–1.4). 29
Transcutaneous oximetry
Transcutaneous oximetry (TcPO2) is a modality that measures oxygen diffusion from the capillary beds to the epidermal layer of the skin.6,8 A sensor-containing electrode is placed on the skin, warms the surrounding skin leading to local hyperthermia, and measures the partial pressure of oxygen in the underlying tissue.30,31
Absolute TcPO2 values or normalized TcPO2 indices can be used clinically. An absolute TcPO2 value greater than 50 mmHg in the foot is normal. 7 A transcutaneous regional perfusion index (TcRPI), or normalized TcPO2, can be calculated by dividing the TcPO2 value in the limb by the TcPO2 of the chest. 32 Also, during TcPO2 measurement, 100% oxygen can be administered in such cases where the TcPO2 value should rise to greater than 100 mmHg, whereas less than 30 mmHg is consistent with PAD.6–8 This ‘oxygen challenge’ might distinguish low pCO2 values due to PAD versus barriers to diffusion, which can include edema, inflammation leading to increased oxygen consumption, vasoconstriction from cold exposure or dehydration, and calluses. For example, a TcPO2 value less than 30 mmHg in ambient air and greater than 100 mmHg on 100% oxygen suggests adequate arterial inflow but the presence of a local barrier to oxygen diffusion.7,33
Clinically, TcPO2 can assess the severity of PAD, need for revascularization, potential wound healing, and response to revascularization.6,9,30,31,34–39 Data suggest TcPO2 is superior to other physiologic vascular studies for predicting wound healing, especially in those with diabetes or renal failure.40,41 In a retrospective study comparing laser Doppler velocimetry (LDV), Doppler ankle pressure (DAP), and TcPO2 in predicting healing of ischemic forefoot ulcerations and amputations, TcPO2 was superior to LDV and DAP, with a sensitivity and specificity of 100% and 88% for TcPO2 compared to 79% and 96% for LDV and 75% and 26% for DAP, respectively. Similarly, accuracy was 95% with TcPO2 versus 87% for LDV and 52% for DAP. 41 In a prospective study comparing TcPO2, arterial segmental pressure (ASP), and arterial segmental indices (ASI), 204 wounds in non-diabetics, diabetics, and those with chronic renal disease were evaluated. TcPO2 had better predictive value for wound healing in all three populations compared to ASI and ASP. 40 In individuals with diabetes and chronic lower extremity ulcers, Pecoraro et al. found that higher peri-wound TcPO2 was associated with higher rates of healing and re-epithelialization, while a peri-wound TcPO2 of less than 20 mmHg was associated with a 39-fold increased risk of early healing failure. 39 Studies show that TcPO2 values greater than 40 mmHg correlate with spontaneous wound healing.8,31,36,40,42 TcPO2 is also associated with amputation risk, such that a TcPO2 less than or equal to 20 mmHg has been associated with an increased risk of amputation. 38
TcPO2 can be predictive of wound healing at the site of amputation. In a retrospective observational study of 307 patients undergoing minor foot amputation, TcPO2 had an area under the operating characteristic curve of 0.75 (95% CI 0.70–0.81) for discriminating what amputation sites healed at 3 months, with an optimal cut-off of 38 mmHg. 37 After revascularization, TcPO2 results may not reach maximal levels for 3–4 weeks, which can guide timing of additional debridement or minor amputations.8,43
TcPO2 can predictably identify individuals with nonhealing ischemic wounds who may benefit from hyperbaric oxygen therapy (HBOT).36,44,45 For example, studies have found that an increase in the TcPO2 value by greater than or equal to 10 mmHg when breathing 100% oxygen identified individuals who may benefit from HBOT to improve wound healing. 46 In a pressurized chamber, TcPO2 measurements during HBOT were able to predict the success or failure of wound healing with a reliability of 74% using a cut-off value of 200 mmHg. 36 In addition, a TcPO2 value at sea-level of less than 15 mmHg combined with an in-chamber TcPO2 value of less than 400 mmHg predicted failure of HBOT therapy with a reliability of 75.8% and a positive predictive value of 73.3%. 36
There are several limitations to using transcutaneous oximetry as a marker for severity for arterial disease. TcPO2 is a marker of skin perfusion and, as a result, can be falsely low or high in various scenarios. Barriers to diffusion can lead to falsely low TcPO2 values, while a paradoxical increase in TcPO2 values can be seen in individuals with diabetes mellitus because of increased metabolic demands of the tissue and arteriolar shunting in the microcirculation.6–9 Peri-wound TcPO2 measurements are likely higher than the oxygenation of the wound itself. 47 The electrodes used to measure TcPO2 values cover a small area and, as such, multiple readings may be needed to obtain clinically relevant information.6,8 Leg positioning can also play a role, with decreasing TcPO2 values as the leg is further elevated.48,49 Finally, temperature can also impact the values, with studies showing that a 1°C reduction in temperature results in a 2–3% lower TcPO2 reading. 7 As such, the reliability of TcPO2 values is controversial. 31 In 54 patients with PAD, the reproducibility of a single TcPO2 measurement was moderately reproducible with an intraclass correlation coefficient ranging from 0.62 to 0.98 and with a repeatability coefficient of 30 mmHg (i.e. there is a 95% probability that repeat measurements will differ by < 30 mmHg). 50 The substantial variability in serial TcPO2 measurements may be the most significant limitation to this modality’s use in routine clinical practice.
By taking multiple readings, utilizing a normalized TcPO2, and/or performing the oxygen challenge, some of these limitations can be minimized.6–8,32,33
Despite the limitations, TcPO2 can be used to predict the success of wound healing with HBOT, 36 wound healing in individuals with diabetes 39 or after revascularization, 37 and identify those individuals who are at increased risk for amputations. 38 Particular benefits of TcPO2 include that it is a noninvasive, bedside tool with no radiation exposure, making it a safe method to provide information regarding arterial perfusion. 6
Laser Doppler flowmetry and imaging and skin perfusion pressure (SPP)
Laser Doppler techniques for assessing foot perfusion have been described since the 1980s. 51 Laser Doppler flowmetry involves a probe in direct contact with the area of interest that measures the concentration and speed of red blood cells over a small (~1 mm2) surface and shallow (1 mm deep) area of tissue. Since hemoglobin concentrations and vasculature in the area of interest differ among patients, this modality is limited to serial measurements in a particular patient, rather than providing information generalizable to a patient population with CLI. Even within the same patient, serial measurements are limited by motion artifact and temperature changes.6,8 Laser Doppler imaging can assess a larger area of interest by scanning the leg and foot with a laser to provide blood flux measurements. To minimize variability for meaningful measurements in a single patient, the laser wavelength, speed of scanning, and height of the laser to the tissue of interest must be consistent.6,8 Such limitations have precluded these laser Doppler techniques from being widely used for assessing foot perfusion in patients with CLI.
The measurement of SPP was described as early as 1967 as a method for assessing foot perfusion. 52 Radioisotope clearance methods, photoplethysmography, and laser Doppler techniques have been used to measure SPP. Radioisotope clearance and laser Doppler, but not photoplethysmography, have been shown to have good correlation. 53 Given the ease and practicality of laser Doppler compared to Xenon washout, laser Doppler is the preferred modality to measure SPP. 54 Commercially available devices incorporate a laser probe within a pressure cuff. The probe is applied to the area of interest, the cuff inflated, and the pressure at which the laser probe identifies return of blood flow during cuff deflation can then be recorded. SPP measurements have been shown to have statistically significant correlations with ABI, toe pressure, and TcPO2 values in 211 patients with PAD. 52
SPP cut-offs between 30 and 40 mmHg have been shown to have predictive value. In 94 limbs with tissue loss managed without revascularization, a SPP cut-off of 40 mmHg had a sensitivity of 72% and a specificity of 88% for wound healing. 52 In this report, SPP was also an independent predictor of wound healing when adjusting for ankle pressure, toe pressure, and TcPO2. In a prospective study of 29 limbs with ulcers or gangrene managed without revascularization, a SPP cut-off of 30 mmHg had an accuracy of 79% (p < 0.002) in predicting wound healing. 5 In a retrospective study of 47 patients with CLI managed conservatively, a SPP cut-off of 35 mmHg was optimal for discriminating wound healing. 55 In 19 patients undergoing toe amputation, a SPP cut-off of 30 mmHg provided an accuracy of 71% for healing the amputation site. 56 In 53 consecutive patients with CLI managed conservatively, a SPP cut-off of 40 mmHg had an accuracy of 81% for predicting wound healing. The dichotomous SPP variable of greater than 40 mmHg was also an independent predictor of wound healing when adjusted for variables including age, sex, diabetes, and infection (odds ratio (OR) 14.2, 95% CI 3.6–55.8). 57 Finally, in a prospective study of 100 patients with nonhealing wounds, a SPP cut-off of 30 mmHg predicted wound healing 87% of the time and was superior to TcPO2 which predicted wound healing accurately only in 64% of the cohort (p < 0.0002). 58
SPP obtained with laser Doppler is noninvasive and time-efficient. This modality also can measure hemodynamics at a wound edge, perhaps providing more reliable assessment of perfusion at the wound site than toe pressure. There are some data on the ability of SPP to predict wound healing that suggest it is superior to ABI and TcPO2, albeit the evidence base consists of a limited number of small studies and may be subject to publication bias.5,58
Two-dimensional angiographic perfusion imaging (Figure 1)
Two-dimensional perfusion imaging is a software analysis package that allows measurement of parameters of contrast delivery to a region of interest in the foot. It requires no additional radiation, intra-procedural time, or contrast. Data for this modality exist but are sparse. For example, in a study of 89 patients with CLI, 2D perfusion imaging was performed successfully in 90% of patients undergoing angiography. In the majority of the patients, an increase in perfusion as assessed by this modality was observed after revascularization. 59 In another study of 24 patients undergoing lower extremity angiography, ABI correlated well to time-to-peak and peak density ratios – two parameters obtained with 2D angiographic perfusion imaging. 60 Finally, in a study of 21 patients undergoing digital subtraction angiography prior to lower extremity revascularization, an improvement in 2D angiographic perfusion parameters was observed following revascularization. 61
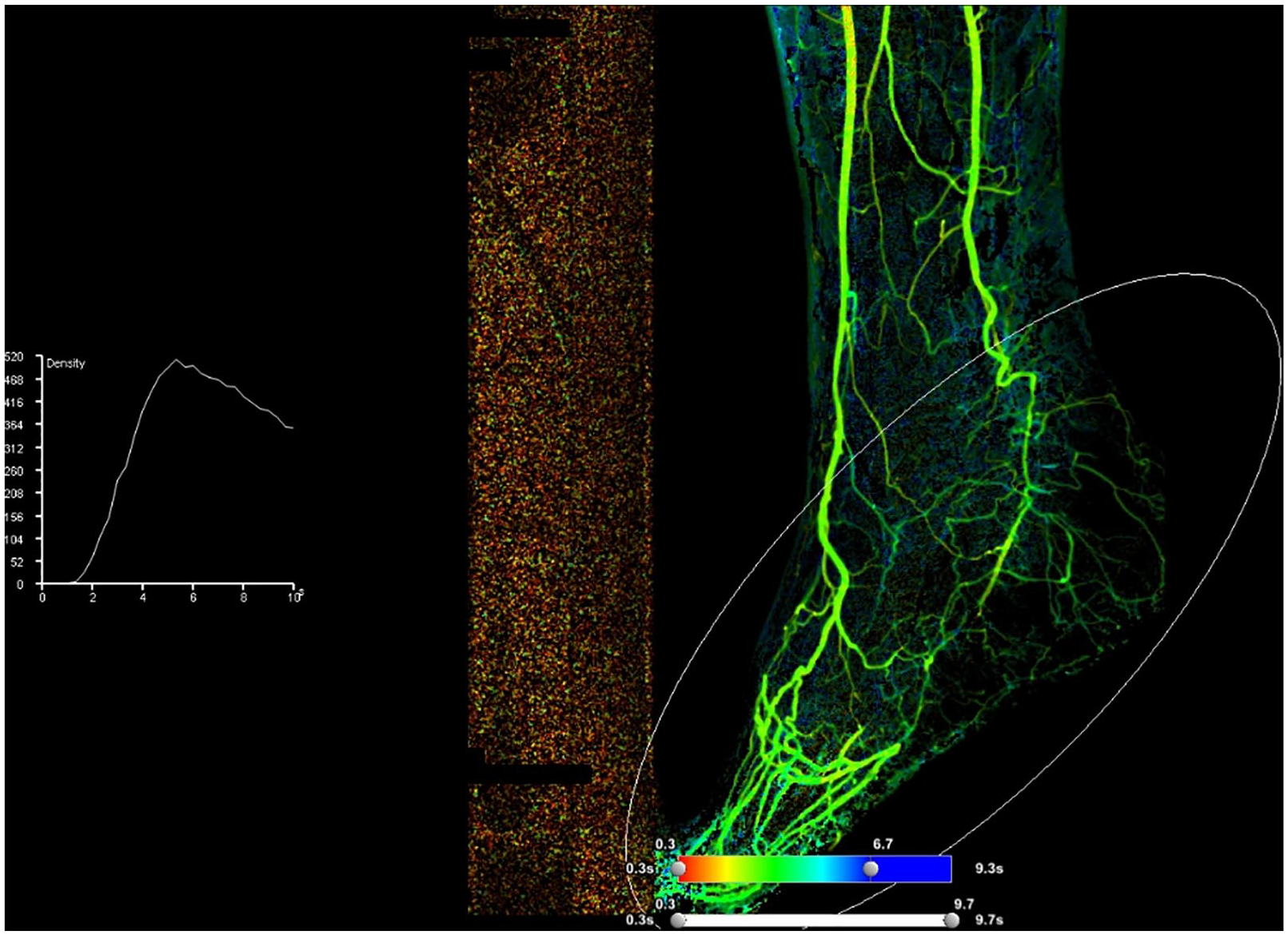
Two-dimensional angiographic perfusion. Right: Color map of the foot generated from a standard catheter-based angiogram. Left: Time density curve representing the time to peak contrast density in the foot. The average time to peak contrast density for this patient was 4.0 seconds.
Potential benefits of 2D perfusion imaging is that it is practical, as it can be incorporated into a standard angiogram. It also is ‘angiosome specific’, providing information of regional foot perfusion, and theoretically could guide revascularization decisions in real time. Limitations currently are that there is no evidence on longitudinal outcomes associated with 2D perfusion parameters. In addition, this modality is not standardized. That is, the technique of performing an angiogram to obtain 2D perfusion parameters would need to be the same across operators and institutions so that the data generated would be generalizable to patient care.
Indocyanine green angiography
Indocyanine green (ICG) is a water-soluble contrast agent that was originally approved by the US Food and Drug Administration (FDA) in 1959. When administered intravascularly, it becomes quickly bound to plasma proteins, where it can be used to visualize local-regional blood flow using a charge-coupled camera to detect the fluorescence emitted after excitation by a laser light source of a specific wavelength. While its use in areas of multiple fields of surgery has been described for decades, it has seen somewhat of a renaissance in recent years as an adjunct tool for evaluation of perfusion and viability in bowel, 62 skin flaps, 63 and CLI with tissue loss. 64
The implementation of ICG angiography (ICGA) for evaluating perfusion can largely be thought of in two discrete uses: subjective and objective data. 65 It is important to understand the difference in these uses when utilizing the technology, as implementing it for one purpose may restrict or preclude its use for the other. To obtain the objective fluorescence intensity curves to assess foot perfusion in the patient with CLI, a single area of the foot is analyzed and there must be virtually no movement of either the foot or camera once recording begins (Figure 2). However, looking in this single view may limit subjective visualization of regional perfusion variations in the entire foot that might be seen at different angles.
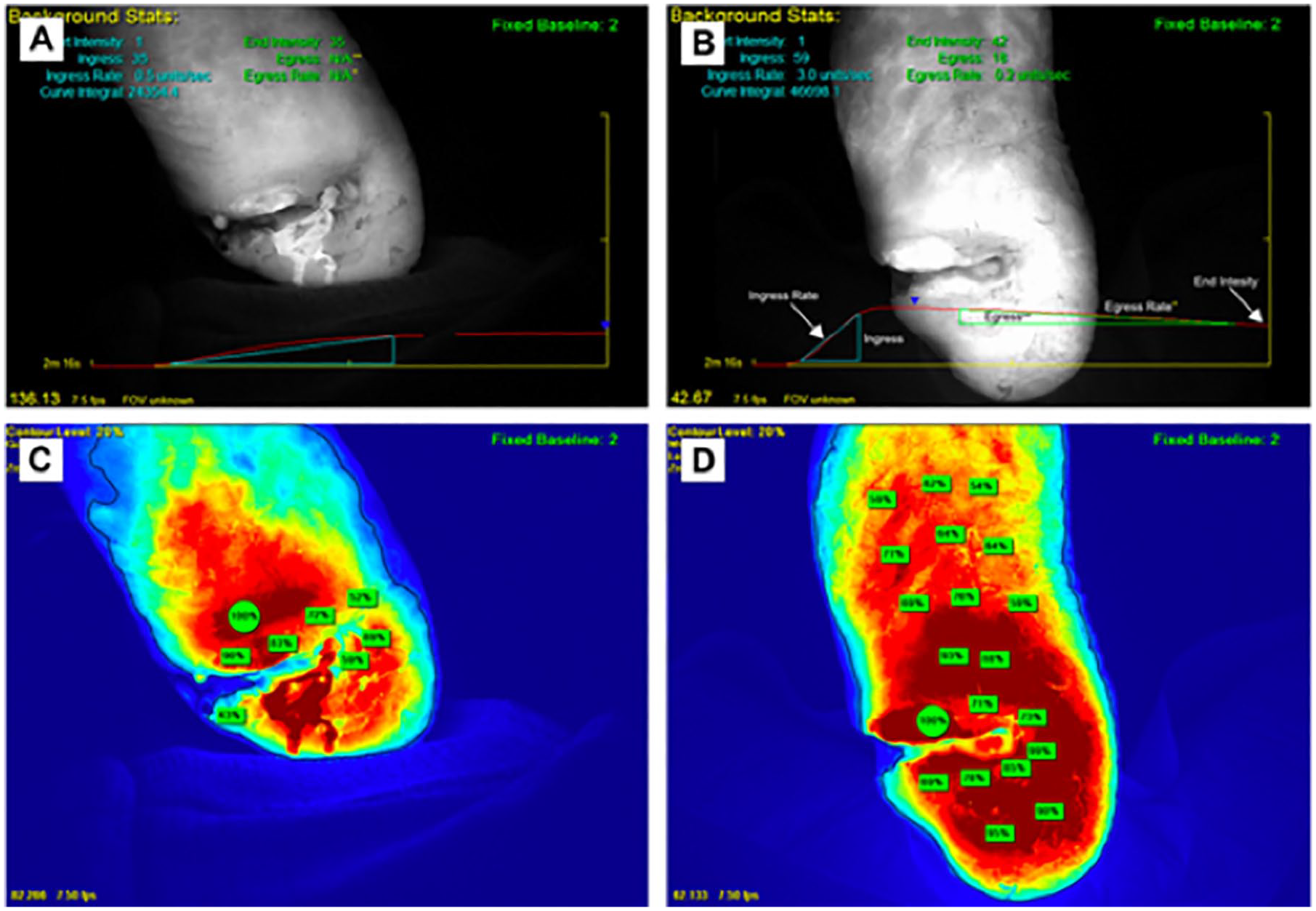
ICGA images in a patient with a nonhealing heel wound. (A) ICGA grayscale image pre-intervention with ingress-egress curve. (B) ICGA grayscale image post-intervention with a sixfold increase in ingress rate and appearance of egress. (C) ICGA heat-map pre-intervention. (D) ICGA heat-map post-intervention.
Subjective evaluation of ICGA images is what is largely utilized in the plastic surgery and general surgery literature to give additional information beyond the ‘eyeball test’ of whether local tissue perfusion is adequate to avoid subsequent ischemic necrosis. 66 In the evaluation of foot wounds, subjective evaluation has likewise been shown to identify areas of localized poor perfusion that corresponded with areas of ischemic failure.
Of objective data gathered from ICGA, ingress (the increase in fluorescence intensity from baseline) and ingress rate (the increase in intensity over the time to maximum intensity) have shown some correlation with modalities such as ABI and toe pressure. 67 Furthermore, they have been shown to detect severe ischemia, based on the Wound-Ischemia-foot-Infection (WIfI) classification, with reasonable accuracy.28,68 The parameter of time to half-maximal intensity has also been used by some groups as another measurement to detect response to revascularization. 69
Limitations of ICGA include the need for intravenous access, a contraindication in those with previously documented iodinated contrast allergies due to concern of cross-reactivity, the need for specialized equipment, and an inability to assess perfusion deeper than 5 mm. 4 There also may be different threshold values using systems from different manufacturers, and the test variability across different environmental factors such as temperature, ambient light and timeframe following revascularization have not been thoroughly evaluated. This modality does, however, appear to show promise for providing incremental information to the clinician treating patients with CLI.
Oxygen Microsensors
A recently developed approach to monitoring foot perfusion uses injectable oxygen microsensors to directly monitor oxygen in the subcutaneous tissue in vivo. This concept involves the subcutaneous injection of a small, soft, biocompatible hydrogel bearing an oxygen-sensitive and optically active metalloporphyrin. After initial injection of the microsensor, a hand-held optical reader is placed over the injection location to measure extravascular tissue oxygen levels. The chemistry underlying the oxygen-sensing measurement is the well-understood phosphorescence quenching reaction. 70 The novelty of the approach is the use of a hydrogel-based microsensor that mitigates the foreign body response 71 and allows for continuous noninvasive oxygen measurements for months after sensor placement. A recent healthy volunteer study provided validation of the ability of oxygen microsensors to reliably detect changes in tissue oxygen confirmed by TcPO2, with data over 3 months after sensor injection. 72
Microsensors provide a localized measurement of dissolved oxygen levels in the microenvironment within and immediately around the hydrogel. Multiple microsensors can be measured simultaneously by multiple optical readers, allowing data to be reported either in terms of oxygen levels per location, or as a ratio that is obtained when a practitioner uses one of the reader locations as a reference.
A first-in-human clinical study provided a proof-of-concept for the use of subcutaneous microsensors to monitor tissue oxygen in ischemic limbs of patients with CLI. 73 That pilot study reported data from 10 CLI subjects undergoing endovascular revascularization, showing that on average tissue oxygen in the affected foot increased after the procedure. An ongoing post-market Registry study (Oxygen Monitoring Near Ischemic Areas, OMNIA) is characterizing how oxygen measurements performed continuously during revascularization, and assessed at follow-up visits, compare with standard clinical hemodynamics and wound healing. Preliminary findings74,75 show that increases in oxygen levels assessed during endovascular revascularization procedures were associated with wound healing 3 months following the procedure. While these studies show potential, additional evidence will be required to establish an accepted treatment algorithm that uses direct tissue oxygen measurements to guide clinical decisions.
Injectable subcutaneous oxygen microsensors have been commercialized (LumeeTM Oxygen Platform; Profusa Inc., South San Francisco, CA, USA) and have obtained the European CE Mark approval for measuring oxygen in subcutaneous tissue; the device has not yet been approved for use in the United States.
Localized continuous measurements of tissue oxygen are of interest for clinical CLI management. Because injectable microsensors directly sense oxygen in the native interstitial fluid, they may provide sensitivity to both arterial insufficiency and microvascular impairment, making it a relevant parameter for treatment planning and long-term monitoring. Additional applications for injectable oxygen microsensors include prediction of wound healing, detection of microvascular disease in diabetic patients, and assessment of ischemia/reperfusion injury.
Advanced modalities
Additional modalities exist that are exciting for the assessment of limb perfusion and include positron emission tomography, contrast echocardiography, and magnetic resonance imaging.6,8 To date, these modalities focus on perfusion to skeletal muscle of the calf rather than specifically focusing on blood flow to the foot, which has relatively little skeletal muscle. Though these advanced modalities may hold promise in the future for the assessment of foot perfusion, they are currently limited by time, expense, lack of availability, and need for expertise. Regardless, encouragement in improving the assessment of limb perfusion with these modalities is warranted.
Discussion
Patients with CLI have poor limb outcomes. Providers managing patients with CLI and/or nonhealing foot wounds often encounter the following questions: (1) Is ischemia contributing to the inability to heal? (2) Should this patient be revascularized? (3) Which vessel should be revascularized? (4) Is the intra-procedural revascularization result sufficient to improve the chance of wound healing? (5) Does there remain patency in the revascularized limb? Enhancing the ability to assess foot perfusion should answer these questions, empower providers to better care for patients with CLI, and improve limb outcomes for patients with nonhealing wounds. 4
The ideal test for foot perfusion would be inexpensive, readily available, reproducible, and improve the clinician’s ability to predict outcomes. Additionally, the ideal test might provide perfusion data specific to the area of the foot with a wound. The perfect test for assessing regional foot perfusion does not currently exist. ABIs are frequently either noncompressible, incongruent with tibioperoneal anatomy, or discordant with the clinical presentation of a patient with CLI.13,14 Additional tests to supplement the ABI – such as toe pressures, TcPO2, and SPP – are supported by guideline recommendations for the care of patients suspected to have CLI. 29 However, toe pressures do not provide regional perfusion assessment of the entire foot, and TcPO2 suffers from a high coefficient of variability.31,50 ICGA is an attractive intra-procedural modality to assess perfusion but is limited by availability and the required technical expertise. 2D perfusion angiography is also an attractive intra-procedural assessment of foot perfusion but is yet to be standardized and vetted for long-term limb outcomes. Oxygen microsensors, while also understudied, provide characteristics of an ideal assessment of foot perfusion (Table 1).
It is likely that the optimal tool for predicting limb outcomes will involve prediction models that incorporate clinical and perfusion variables from multiple modalities to provide a probability of wound healing that could guide clinical decision making. Clinical prediction models for CLI have been investigated but have not incorporated regional foot perfusion modalities.76,77 For example, in the study population of PREVENT III (patients with CLI who underwent infrainguinal bypass), a prediction model was developed for risk-stratifying these subjects with CLI for amputation-free survival. ABI, but not other measures of limb perfusion, were considered in the model. 78 The clinical variables of need for dialysis, age over 75 years, hematocrit value, presence of tissue loss, and presence of CAD were found to be predictive in multivariable analyses and were used to generate a risk score that stratified patients into 1-year risk categories, with the lowest risk group having a 1-year amputation-free survival of 33% and the highest risk group having a 1-year amputation-free survival of 93%. We believe that incorporating a more detailed foot perfusion assessment could provide even better risk stratification and, more importantly, could provide modifiable risk markers. That is, refining foot perfusion assessment strategies could better provide indications for revascularization, guide revascularization targets, and offer modalities for surveillance following foot revascularization – all of which would potentially improve limb-specific outcomes.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Rogers has been granted 10 FDA-approved pressure wires by Philips to use in the assessment of limb perfusion (separate project). He has no direct financial compensation from this company. Dr Montero-Baker has stock in Profusa, Inc. Drs. Braun, Biswas, and Morrison have no disclosures relevant to industry.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
