Abstract
Segmental arterial mediolysis (SAM) is a rare but serious nonatherosclerotic, noninflammatory vasculopathy of unknown etiology that often results in dissection, aneurysm, occlusion, or stenosis of, primarily, the abdominal arteries. Current literature lacks consensus on diagnostic criteria and management options for SAM. This review summarizes 143 cases and aims to advance appropriate recognition and management of SAM. Literature review of all relevant SAM case studies from 2005 to 2018 yielded 126 individual SAM cases from 66 reports. We identified 17 additional SAM cases from our center, bringing our analysis to 143 patients. Patients with SAM were most commonly men (68%) in their 60s. Hypertension (43%), tobacco use (12%), and hyperlipidemia (12%) were common comorbidities. Abdominal pain (80%) and intraabdominal bleeding (50%) were the most common presenting symptoms. Computed tomography was the most frequently used imaging method (78%), and histology was available in 44% of cases. The most commonly affected vessels were the superior mesenteric (53%), hepatic (45%), celiac (36%), renal (26%), and splenic (25%) arteries with aneurysm (76%), dissection (61%), and arterial rupture (46%). Treatments included coil embolization (28%), abdominal organ surgery (24%), open arterial repair (21%), and medical management (20%). Case-specific treatment modalities yielded symptom relief in the vast majority (91%) of patients, with a mortality rate of 7%.
Keywords
Continuing medical education (CME) credits and maintenance of certification (MOC) points are available for reading this article. This opportunity is made possible through a joint partnership with University of Virginia School of Medicine (UVASOM). For instructions, please visit the Society for Vascular Medicine’s website at
The University of Virginia School of Medicine (UVASOM) is accredited by the ACCME to provide continuing medical education for physicians. UVASOM designates this journal-based CME activity for a maximum of one AMA PRA Category 1 Credit™. Participants should claim only the credit commensurate with the extent of their participation in the activity. UVASOM, as an accredited provider, awards 1 hour of participation (consistent with the one AMA PRA Category 1 Credit™) to participants who successfully complete this educational activity. Successful completion of this CME activity, which includes participation in the evaluation component, enables the participant to earn up to one MOC II point in the American Board of Internal Medicine’s (ABIM) Maintenance of Certification (MOC) program. It is the CME provider’s responsibility to submit participant completion information to ACCME for the purpose of granting ABIM MOC II credit. UVASOM maintains a record of participation for 6 years.
The faculty, staff and planning committee of the University of Virginia Office of Continuing Medical Education have no financial affiliations to disclose. The CME planning committee disclosed the following: Heather Gornik is supported by CVR Global and receives intellectual property rights from Summit Doppler Systems, Inc. and intellectual property rights and stock/ownership from FlexLife Health. Aditya Sharma is supported by National Institute of Health Sciences, AstraZeneca, and Biomet Biologics. Geoff Barnes is supported by Pfizer/Bristol-Myers Squibb (BMS), Blue Cross Blue Shield of Michigan, and the National Heart, Lung, and Blood Institute and serves as a consultant for Pfizer/BMS, Janssen, and Portola. Valerie Clark has no financial affiliations to disclose. The authors disclosed no financial relationship or interest with any proprietary entity producing healthcare goods or services.
Introduction
Segmental arterial mediolysis (SAM) is a rare but serious nonatherosclerotic, noninflammatory vasculopathy of un-known etiology. It is characterized by lysis of the medial layer of the arterial wall, often resulting in dissection, aneurysm, occlusion, or stenosis.1,2 SAM most commonly affects the abdominal aortic branches, such as the celiac, mesenteric, and/or renal arteries with occasional carotid, cerebral, and coronary artery involvement. 3 Depending on the involved artery and underlying pathology (dissection with stenosis versus rupture), patients with SAM may present with chronic abdominal pain, acute mesenteric ischemia, or even hemorrhagic shock. 4 Previous case studies have reported high rates of emergent presentation related to arterial rupture and hemorrhage in approximately two-thirds of patients, with a mortality rate reaching up to 50%.1,5 Literature addressing the diagnosis, management, and prognosis of SAM is lacking, with conflicting data addressing its morbidity and mortality.
Ever since its original description by RE Slavin and JC Gonzalez-Vitale in 1976, case reports continue to comprise the majority of SAM-related literature. 6 With a low incidence in the United States of around 1 in 100,000 per year, many centers lack the experience to appropriately identify and manage patients with SAM. 7 Based on the limited literature, there is no current consensus on common criteria or best practices to diagnose SAM. While common presenting symptoms include abdominal pain, nausea, and vomiting, diagnostic imaging and/or histology is necessary to help diagnose SAM and exclude other similar arteriopathies. While histology remains the gold standard method when available to confirm the diagnosis of SAM, the increasing quality of noninvasive imaging modalities, such as computed tomography (CT) and magnetic resonance (MR) angiograms, has led to a shift towards greater use of imaging modalities over histologic sampling.8,9 There are also current discrepancies on anatomic involvement, and the use of inflammatory markers, autoimmune serologies, and genetic testing, required to diagnose SAM.2,10 Furthermore, there is considerable overlap between SAM and other similar arteriopathies (e.g. FMD, localized vasculitis of the gastrointestinal (GI) tract (LVGT), isolated dissection, and aneurysm), calling for the need to standardize the diagnostic criteria for SAM and its mimics.
To address the current gaps in the literature regarding the diagnosis, management, and prognosis of SAM, we present a detailed analysis of published case studies and systematic reviews of SAM between 2005 and 2018. Based on our inclusion and exclusion criteria, we identified 126 patients diagnosed with SAM based on 66 different publications. We specifically sought to summarize reported diagnostic strategies and definitions, vessel pathology and involvement, and management strategies. By summarizing current literature and adding our center’s experience with 17 cases, we provide up-to-date data to help providers identify and manage SAM.
Methods
Chart review at our center
Charts of 1416 patients, age 18 through 60 years, who presented at Abbott Northwestern Hospital, Minneapolis, MN between January 2000 and March 2017, were retrospectively reviewed for the diagnosis of nonatherosclerotic abdominal vasculopathy including arterial stenosis, wall thickening, dissection, aneurysm, inflammation or pseudoaneurysm involving the renal, celiac, hepatic, splenic, gastric, or mesenteric arteries. Patients were included for review if they were confirmed to have any of these findings with no imaging evidence of atherosclerosis involving the artery of interest. We elected to focus on patients < 60 years of age based on the demographic data currently published on SAM, which indicate the majority of patients will be included in our selected age range (18–60 years). Since we wanted to clearly exclude pathology from atherosclerosis, this age range best fit our criteria. The Allina Institutional Review Board approved this study. Informed consent for research review of the medical record was in accordance with Minnesota state law.
Diagnosis
SAM was considered if an imaging study revealed evidence of dissection and/or aneurysm with or without organ infarction in multiple mesenteric and/or renal arteries with exclusion of FMD, LVGT, connective tissue disorder, or other vasculopathies, and no significant concurrent arterial wall thickening (< 3 mm) or elevation of inflammatory markers (erythrocyte sedimentation rate (ESR) < 20 mm/h and C-reactive protein (CRP) < 5 mg/dL).11,12 One patient also had a pathological finding consistent with SAM, including lysis of the outer arterial media resulting in separation of the media from the adventitia leading to dissecting pseudoaneurysms.3,4,13 To exclude isolated idiopathic or trauma-related lesions, we required multiple vessel involvement to diagnose SAM. The presence of more severe wall thickening and/or elevated inflammatory markers are more indicative of LVGT or polyarteritis nodosa (PAN), and therefore were used as exclusionary markers. All patients noted to have median arcuate ligament syndrome (MALS) were excluded from our review. From this review, 17 patients met SAM inclusion criteria and are incorporated into our literature review (143 patients in total) (Table 1).
Chart review: patient characteristics (n = 17 men).
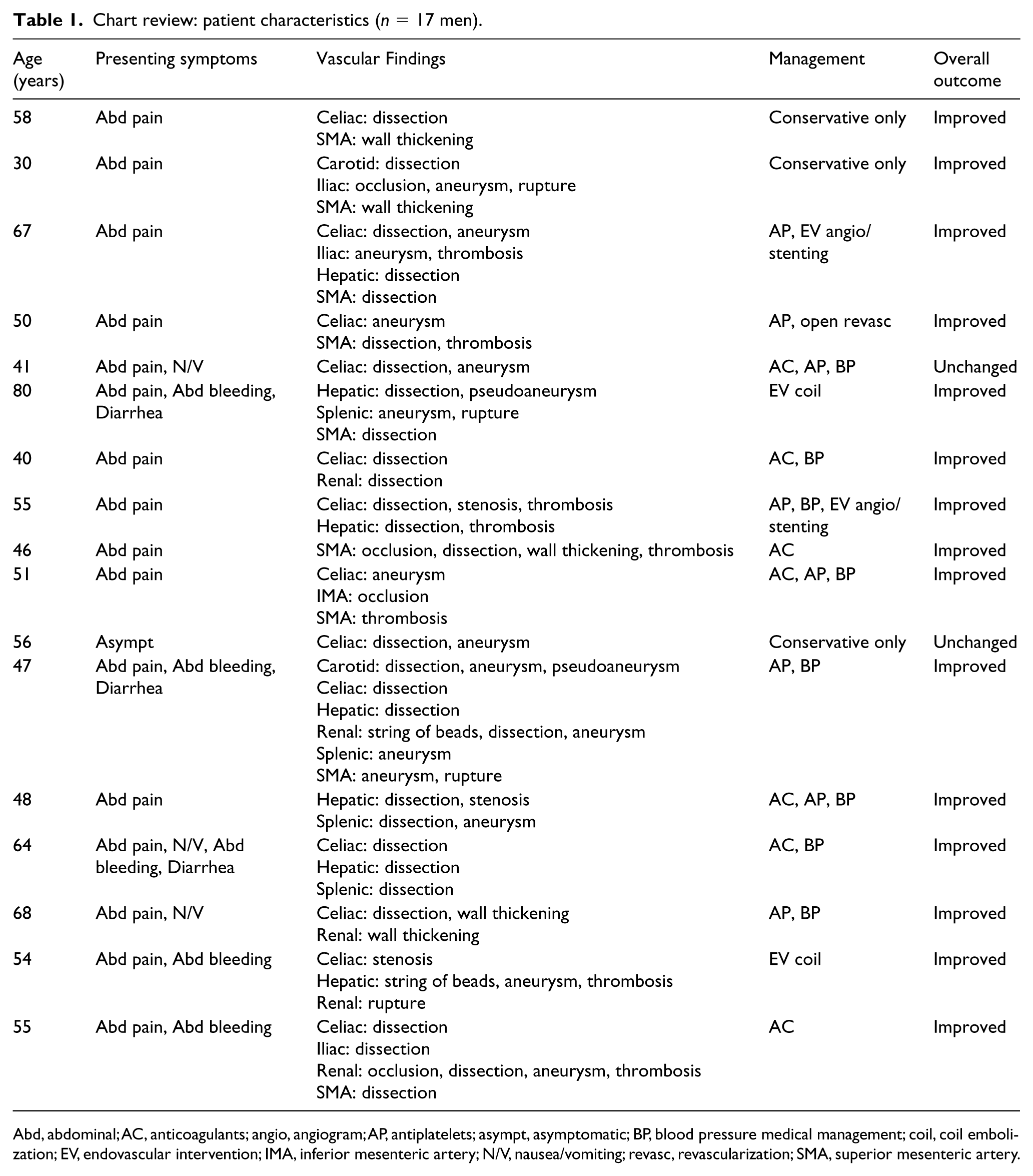
Abd, abdominal; AC, anticoagulants; angio, angiogram; AP, antiplatelets; asympt, asymptomatic; BP, blood pressure medical management; coil, coil embolization; EV, endovascular intervention; IMA, inferior mesenteric artery; N/V, nausea/vomiting; revasc, revascularization; SMA, superior mesenteric artery.
Differential diagnoses
All cases were reviewed by a vascular medicine specialist to confirm final diagnoses according to the diagnostic criteria listed below. The diagnostic criteria were either based on consensus committees or expert opinion.
Prior to the publication of the new criteria proposed by the ‘First International Consensus on the diagnosis and management of fibromuscular dysplasia’, we considered the diagnosis of FMD if an imaging study revealed stenosis, occlusion, dissection, or aneurysm in the renal and/or extracranial cerebrovascular arteries with concurrent ‘string of beads’ appearance (multifocal FMD) or circumferential stenosis (focal FMD) in the setting of no aortic involvement and normal inflammatory markers.14,15
The diagnosis of focal single-organ vasculitis (SOV) is always presumptive and requires exclusion of systemic illness at the time of diagnosis as well as throughout the period of continued care. The diagnosis of SOV, including LVGT, requires no additional source of vascular inflammation at both the time of diagnosis and over a 6-month follow-up period. 16 Localized vasculitis of the GI tract was considered in patients with recent history or presence of acquired GI manifestations (including abdominal pain, nausea or vomiting, diarrhea, weight loss, melena) and a histopathological evidence of vasculitis in a GI specimen; or high-probability angiographic findings (smooth segmental narrowing, dilatation, occlusion or aneurysms affecting one or more GI arteries) with significant wall thickening (⩾ 3 mm) and the absence of vessel changes consistent with atherosclerosis, FMD or SAM). 17 Inflammatory markers can be normal or elevated.
Vascular Ehlers–Danlos syndrome (type IV) was considered if the patient met Villefranche criteria and was confirmed by genetic testing.18,19
The diagnosis of Loeys–Dietz syndrome (LDS) was considered if a patient had hypertelorism, a bifid uvula and/or cleft palate and generalized arterial tortuosity with widespread vascular aneurysm and dissection. 20 Diagnosis was confirmed by genetic testing to reveal heterozygous mutations in the genes encoding transforming growth factor beta receptors 1 or 2, SMAD family member 3, or transforming growth factor beta 2 or 3, all of which result in production of a less functional protein and are responsible for a different LDS subtype. 21
There is no consensus on one diagnostic criteria for either SAM or LVGT. The diagnostic criteria we used for SAM and LVGT were mostly consistent with the majority of previous reports. However, based on our experience with SAM, we elected to include patients that have more than one vessel involvement. Patients with only one vessel involvement were grouped under isolated aneurysm or dissection. The criteria used for inflammatory markers in SAM and LVGT were based on our experience since some patients with SAM-related dissection usually present with elevated markers, and not all patients with LVGT would have elevated ones.
Literature review
Literature was systematically reviewed to identify all SAM publications by searching public PubMed databases for all full-text articles published in English between January 1 2005 and July 6 2018. Key search terms included ‘segmental arterial mediolysis’ and ‘segmental mediolytic arteriopathy’. We elected to review cases starting from 2005 because SAM was becoming better recognized and more reliably diagnosed.
Inclusion/exclusion criteria
Publications were eligible for inclusion if they: reported at least one case of SAM, reported patient demographics (at least age and sex), reported vessels and pathology involved, and reported on diagnostic strategy (imaging or labs or histology or symptoms). Case reports and case series of patients with proven or probable SAM were eligible for inclusion.
Publications were excluded if they: did not fulfill the above criteria, were published outside of our search range, or were not available in English. A total of 126 unique cases were identified from 66 different case reports and case studies (Supplementary Table 1).4,5,8–10,13,22–81
Study definitions and data extraction
Presenting symptoms were defined based on specific reports. Incidental findings were classified as asymptomatic. Cerebrovascular symptoms included headache, vision changes, loss or altered consciousness, or intracranial bleeding. An ESR greater than 15 mm/h (for males < 50 years of age), greater than 20 mm/h (for males > 50 and females < 50 years of age), greater than 30 mm/h (for females > 50 years of age) OR a study that reported ESR as ‘elevated’ was considered elevated. ESR that fell below the above range or were reported as ‘normal’ or ‘unremarkable’ were considered normal. A CRP level greater than 0.50 mg/dL or a study that reported CRP as ‘elevated’ was considered elevated. Tachycardia (⩾ 100 bpm), bradycardia (⩽ 60 bpm), elevated blood pressure (⩾ 130 mmHg/80 mmHg), and low blood pressure (⩽ 90 mmHg/60 mmHg) were defined based on the American Heart Association guidelines. Studies that reported patients as presenting ‘hypertensive’ or ‘elevated blood pressure’ or ‘high blood pressure’ were also classified as elevated blood pressure. Studies that reported patients as presenting ‘hypotensive’ or with ‘low blood pressure’ were also classified as low blood pressure. Hemoglobin was classified as normal if it fell within Mayo Clinic definitions: 13.5–17.5 g/dL (males), 12.0–15.5 g/dL (females), and classified as low if values were below these ranges or if patients were transfused with red blood cells (RBC) at presentation. Hematocrit was classified as normal if it fell within Mayo Clinic definitions: 38.8–50% (males), 34.9–44.5% (females), and classified as low if values were below these ranges or if patients were transfused with RBCs at presentation.
A string of beads radiographic finding was defined if a study specifically reported ‘beads’, ‘beading’, or ‘string of beads’.
Follow-up was defined individually as the most recent hospital encounter or clinic visit after initial presentation and discharge. At follow-up, symptoms were classified as improved if they were eased or alleviated, and as worsened if they occurred again or new related symptoms were present. Four reviewers independently extracted the above prespecified data from literature that fulfilled eligibility criteria. Questions of classification were reviewed with the guidance of a vascular medicine specialist.
Statistical methods
Descriptive statistics are displayed as median and 25th and 75th percentiles for continuous variables; number and percentage with characteristic are given for categorical variables. Formal analyses were not done given the small sample sizes within groups. Data were summarized using Excel 2016 (Microsoft). Linear regressions, one-way ANOVA, and chi-squared contingency analyses with two-way Fischer’s exact tests were developed in Prism 8.1.1 (GraphPad Software, La Jolla, CA, USA).
Results
Chart review
Seventeen patients with SAM were identified from our center’s chart review. Demographic and case data are summarized for each patient in Table 1.
Study demographics, presenting symptoms, vital signs, and imaging
Reported patient demographics and comorbidities are summarized in Table 2. The median age of the total 143 study participants was 55 years and 67.8% were men. Hypertension, tobacco abuse, hyperlipidemia, and diabetes mellitus were reported in 42.7%, 11.9%, 11.9%, and 1.4% of the patients, respectively. Overall, the most common symptom at presentation was abdominal pain (79.7%), followed by intraabdominal bleeding (49.7%). Less commonly reported presenting symptoms included: nausea/vomiting (16.1%), cerebrovascular symptoms (11.9%), melena/hematochezia (5.6%), diarrhea (4.9%), and shock (4.2%). Seven patients (4.9%) were asymptomatic. Patients presented with higher rates of elevated (42.4%) than low (25.4%) blood pressure. Around half of patients had low hemoglobin and/or hematocrit at presentation. At baseline, ESR was measured in 33 patients (23.1%), of whom 33.3% had an elevated ESR. Among the 51 (35.6%) patients with a CRP measurement at baseline, the majority (68.6%) had elevated levels. Computed tomography (CT/CTA) was the most frequently used imaging method (77.6%), followed by histology (44.1%), catheter-based angiogram (CBA) (35.0%), laparotomy (32.9%), magnetic resonance angiography (MRA) (9.1%), and duplex ultrasound (DUS) (9.1%). Other imaging methods were used in 9.8% of cases.
Patient presentation (n = 143) a .
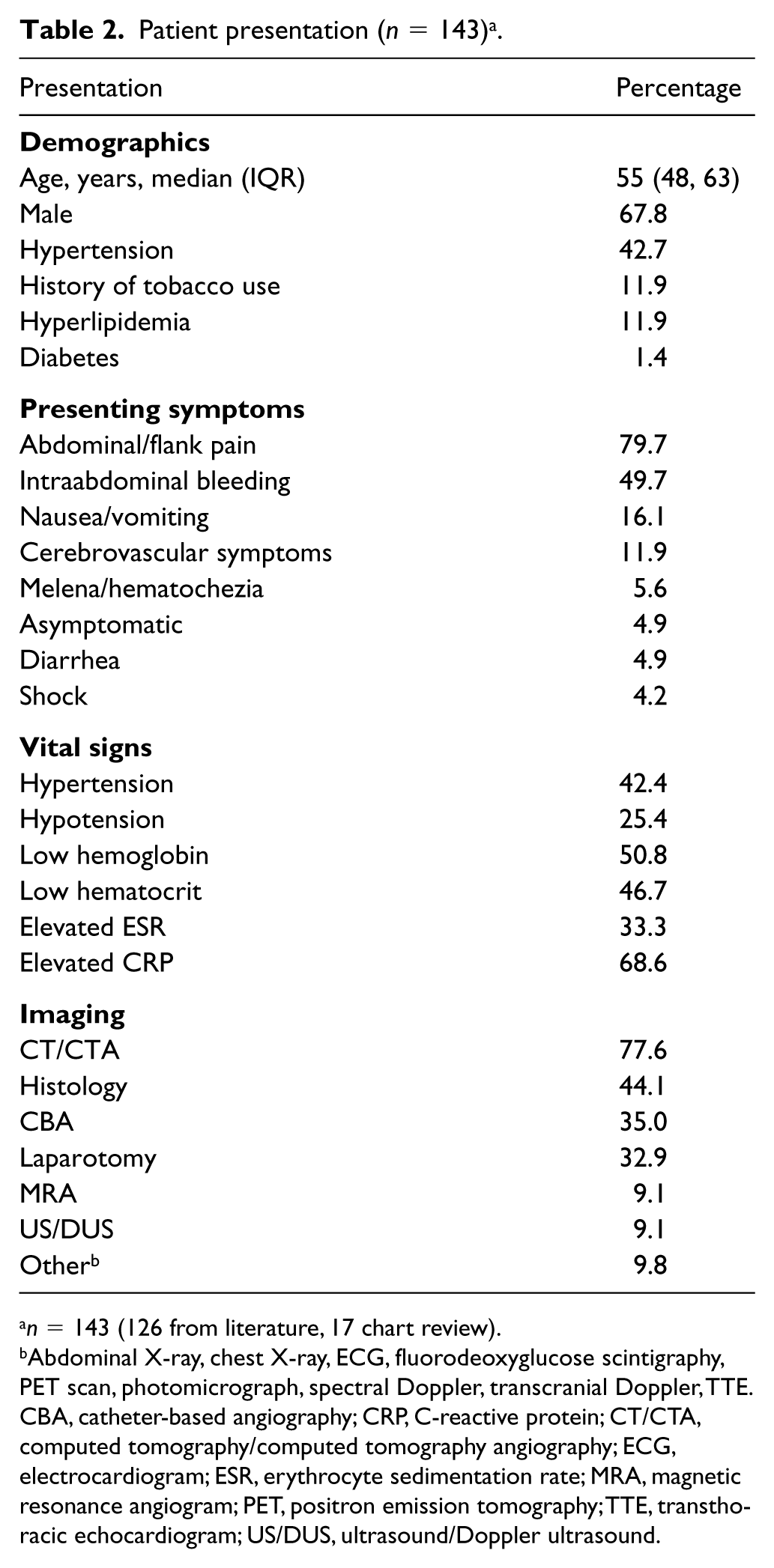
n = 143 (126 from literature, 17 chart review).
Abdominal X-ray, chest X-ray, ECG, fluorodeoxyglucose scintigraphy, PET scan, photomicrograph, spectral Doppler, transcranial Doppler, TTE.
CBA, catheter-based angiography; CRP, C-reactive protein; CT/CTA, computed tomography/computed tomography angiography; ECG, electrocardiogram; ESR, erythrocyte sedimentation rate; MRA, magnetic resonance angiogram; PET, positron emission tomography; TTE, transthoracic echocardiogram; US/DUS, ultrasound/Doppler ultrasound.
Involvement, treatments, and outcomes
Arteries most commonly affected were the SMA (53.1%), hepatic (44.8%), celiac (35.7%), renal (25.9%), and splenic (24.5%). Cerebrovascular involvement was reported in 13.3% and inferior mesenteric artery (IMA) involvement in 10.5% of patients. Over half of patients (62.2%) reported involvement of multiple arteries (Table 3). The most common pathologic findings were arterial aneurysm (76.2%), dissection (60.8%), and rupture (45.5%). Other pathologies reported were arterial stenosis (18.9%), occlusion (16.9%), ‘string of beads’ (14.7%), thrombosis (14.7%), pseudoaneurysm (11.9%), and wall thickening (7.7%).The full breakdown of pathologic and anatomic distributions is outlined in Figure 1. The most common diagnostic strategy used for the 143 cases was CT/CTA as the primary determinant of SAM (59.4%). Histology was the second most employed diagnostic method (44.1%) followed by CBA (29.4%), and laboratory diagnostic tests (e.g. genetic testing, CRP, ESR, ANCA (antineutrophil cytoplasmic antibodies), CBC (complete blood count), CMP (comprehensive metabolic panel)) (10.5%). Eight patients were diagnosed with SAM during autopsy (5.6%).
Involvement, treatments, and outcomes.
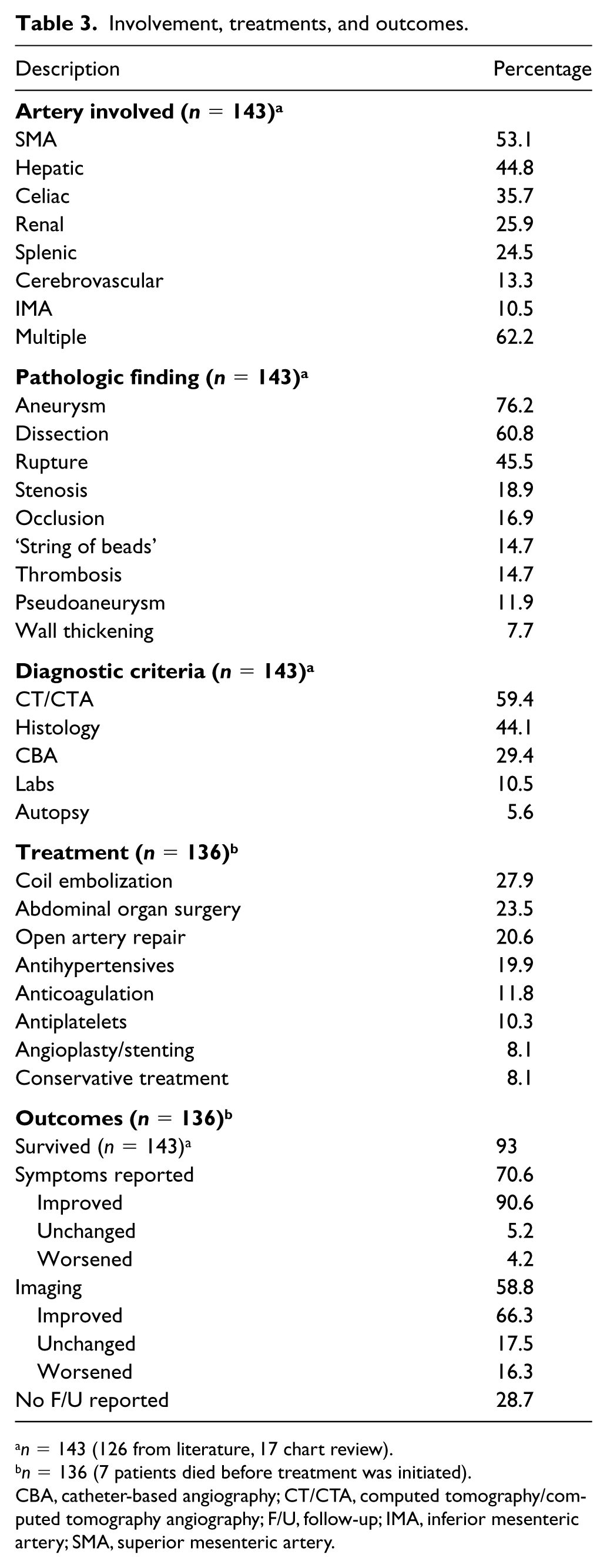
n = 143 (126 from literature, 17 chart review).
n = 136 (7 patients died before treatment was initiated).
CBA, catheter-based angiography; CT/CTA, computed tomography/computed tomography angiography; F/U, follow-up; IMA, inferior mesenteric artery; SMA, superior mesenteric artery.
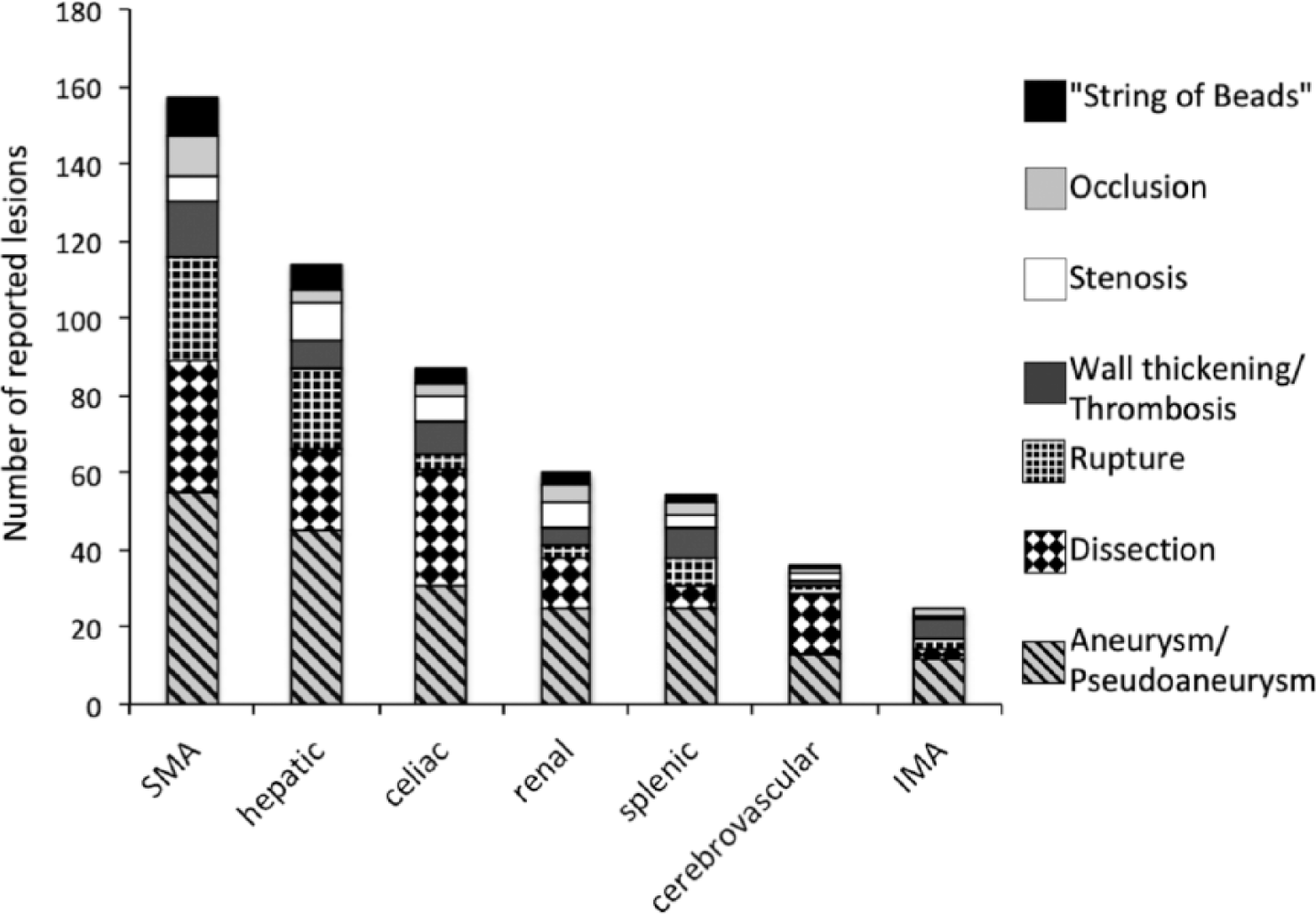
Location and type of 539 reported lesions from 143 SAM patients.
A considerable proportion of cases of SAM with histology available had undergone laparotomy (66%) and open surgery (45%), allowing for collection of tissue biopsies. Patients without available tissue biopsies were more commonly male (76%) and did not present as emergently with abdominal bleed (34%) as did those with available histology (69%) (Table 4). Fourteen patients were reported to have string of beads, but did not have histology, making these probable but not definitive cases of SAM.
Comparing SAM cases with and without histological confirmation.
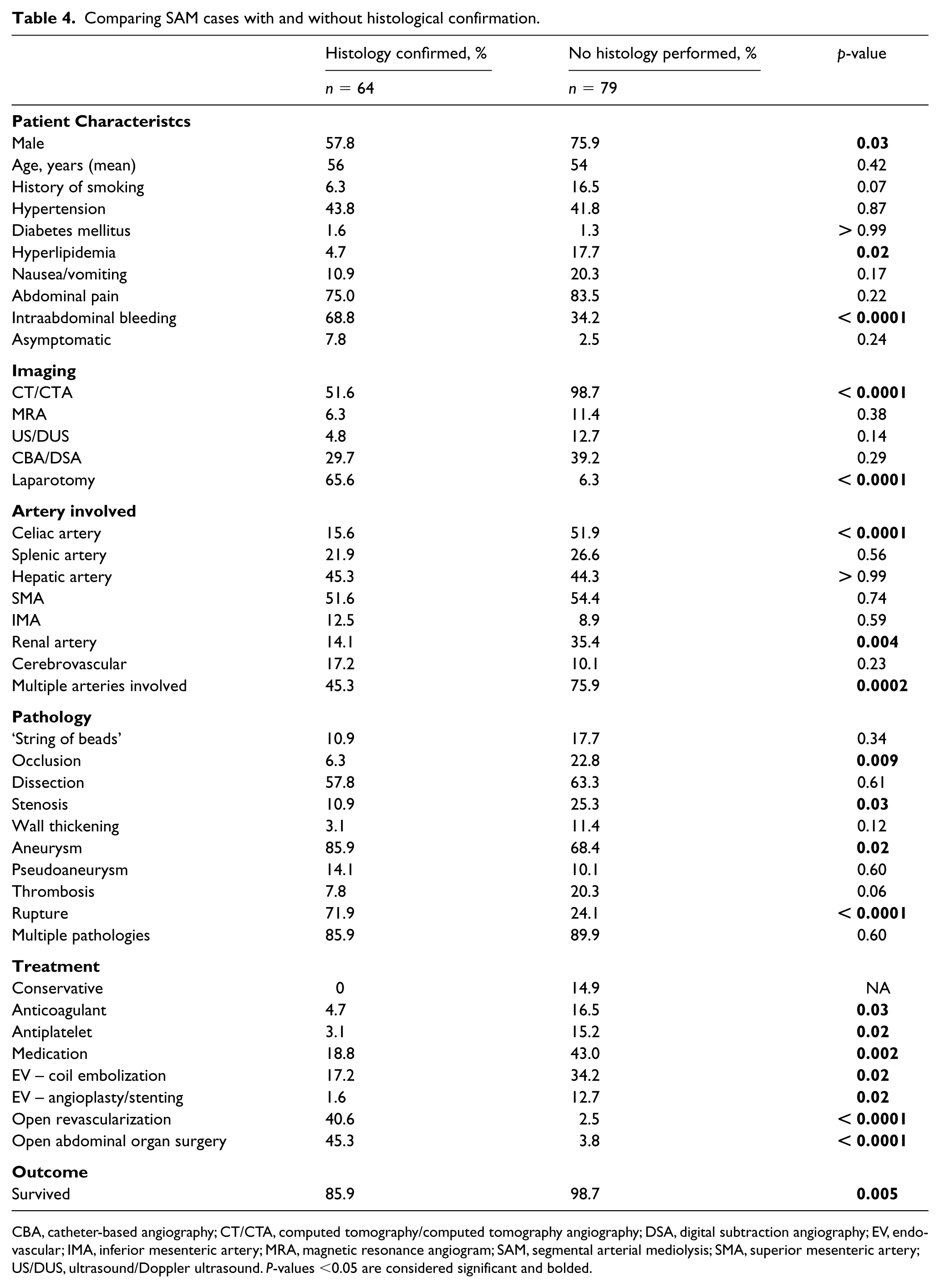
CBA, catheter-based angiography; CT/CTA, computed tomography/computed tomography angiography; DSA, digital subtraction angiography; EV, endovascular; IMA, inferior mesenteric artery; MRA, magnetic resonance angiogram; SAM, segmental arterial mediolysis; SMA, superior mesenteric artery; US/DUS, ultrasound/Doppler ultrasound. P-values <0.05 are considered significant and bolded.
Coil embolization was the most commonly reported treatment modality (27.9%), followed by abdominal organ surgery (23.5%) (including hemi/colectomy n = 13, gastrectomy n = 6, splenectomy n = 5, pancreatectomy n = 5, nephrectomy n = 2, ileocecectomy n = 1, cholecystectomy n = 1, duodenectomy n = 1), and open arterial repair (20.6%). The most common medical managements reported were antihypertensive (19.9%), anticoagulation (11.8%), and antiplatelet agents (10.3%). Angioplasty or stenting was also used in 11 patients (8.1%). Conservative treatment only, including pain control and observation, was implemented in 8.1% of the total patient population.
The majority (90.6%) of patients with a reported final outcome and/or follow-up symptoms (n = 96) experienced improvement by the conclusion of the case report. Of the 80 patients who underwent follow-up imaging, 66.3% showed improvement, 17.5% remained unchanged, and 16.3% worsened.
Discussion
We identified a total of 143 patients with SAM comprised of 126 published cases and 17 cases diagnosed at our center. Prior extensive literature reviews by Shenouda et al. in 2014 and by Kim et al. in 2016, had reported 85 and 101 cases of SAM, respectively.1,22 Based on our inclusion and exclusion criteria, only some of the patients from these two reviews were eligible to be considered in our study. Furthermore, we report 83 additional cases that were not part of either review. Additionally, in 2018, Naidu et al. from the Mayo Clinic published a large single-center report that included 111 SAM cases. 2 These data were not incorporated into our literature review and analysis since they lacked specific patient demographics that could have affected our collective data analysis. Naidu et al. reported renal arteries to be the most commonly affected vessels (47%). The majority of their patients received medical management (47% antiplatelets, 46% anticoagulants, 36% beta-blockers, 33% angiotensin-converting enzyme inhibitors) that resulted in no mortality by the end of their report. Furthermore, 80% of their patients with available follow-up studies were stable or had disease regression. 2
Epidemiology
Based on previous reports, the predicted incidence of SAM in the United States of one in 100,000 per year is likely an underestimate. 7 Furthermore, previous reports had used different diagnostic criteria making it difficult to predict an accurate disease incidence. Understanding key patient demographics can help alert physicians to common phenotypes of patients with SAM. Our review demonstrates a slight predominance of males, accounting for 67.8% of patients, with a median age of 55 at presentation. However, disease manifestation has been reported in a wide age range (25–88 years). Of note, including only patients aged 18–60 years from our center (17 patients) could have affected the age range for the whole series and could have led to a lower age range overall.4,26 Hypertension (43%), history of tobacco use (12%), hyperlipidemia (12%), and diabetes (1.4%) are common comorbidities. The coexistence of SAM and hypertension is not surprising, as hypertension is considered a vascular wall stressor. Although more data are needed, blood pressure control may be appropriate to reduce the risk of SAM-related morbidity and mortality. More research is also needed to further clarify the correlation between cigarette smoking and SAM outcomes.
Clinical presentation
Abdominal pain is well established as the most common presenting symptom of SAM. In our review, 80% of patients reported abdominal pain at presentation, which is slightly higher than the rates by other reviews (66–74%).1,2,22 Surprisingly, close to half of the patients presented with abdominal bleeding, indicating that many patients with SAM are first diagnosed after a serious vascular event. While Shenouda et al. reported 29% of their 85 patients presented with hemorrhagic shock, 1 previous reports have not tracked abdominal bleeding at presentation. In our review, 12% of patients presented with cerebral symptoms (bleeding, headache, aphasia, decreased consciousness), which is very consistent with other reports of 14% and 12%.1,22 Thus, early diagnosis of SAM is crucially important to allow appropriate and timely management in order to reduce disease-related morbidity and mortality.
Our analysis showed the SMA to be the most affected vessel (53%) followed by the hepatic (45%), celiac (36%), renal (26%), splenic (25%), and cerebral (13%) arteries. Consistent with other reviews, the least commonly involved abdominal artery was the IMA and its branches (11%).1,2 We report a lower rate of renal artery involvement (26%) compared to Naidu et al. (47%), 2 which could be representative of our review’s improved diagnostic differentiation between SAM and FMD. When renal arteries are involved, it can be difficult to distinguish FMD from SAM. 11 Additionally, because many subjects did not undergo head/neck imaging unless presenting with suggestive symptoms, the reported involvement may not adequately capture the concurrent involvement of cerebral vessels. Although not all reports indicated involvement of multiple vessels, 88% of our review patients had either involvement of multiple arteries (62%) or had multiple types of lesions in the same vessel (26%). We recommend reserving SAM diagnosis for patients with one or more pathologies involving multiple arteries or multiple pathologies involving one artery in order to reliably differentiate SAM from isolated aneurysms or dissections.
Diagnostic imaging and laboratory analyses
There are no established consensus-based diagnostic criteria for SAM. Our review reports CT/CTA as the primary tool used to diagnose SAM and exclude mimics (59%). Histology was the second most employed diagnostic method (44%) followed by CBA (29%), and laboratory diagnostic tests (e.g. genetic testing, CRP, ESR, ANCA, CBC, CMP) (11%). While the diagnosis of SAM can certainly be confirmed via histology, the increasing quality and availability of CTA and MRA imaging has helped reduce the need for invasive arterial biopsy, which is often not feasible.8,9 Our literature review of cases over 12 years observed a reduction of using histologic samples, likely because of safety concerns and challenges with obtaining a biopsy from an inflamed or dissected artery, and noticed an increase in using noninvasive imaging (CTA or MRA) as the primary diagnostic tool (Figure 2). Our report concurs with others, that catheter-based angiography is reserved for critical cases with urgent presentation, as 54% of our patients who underwent CBA had presented with abdominal bleeding.1,9 When feasible and safe, we still recommend obtaining a tissue biopsy to exclude similar arteriopathies, such as fibromuscular dysplasia (FMD).
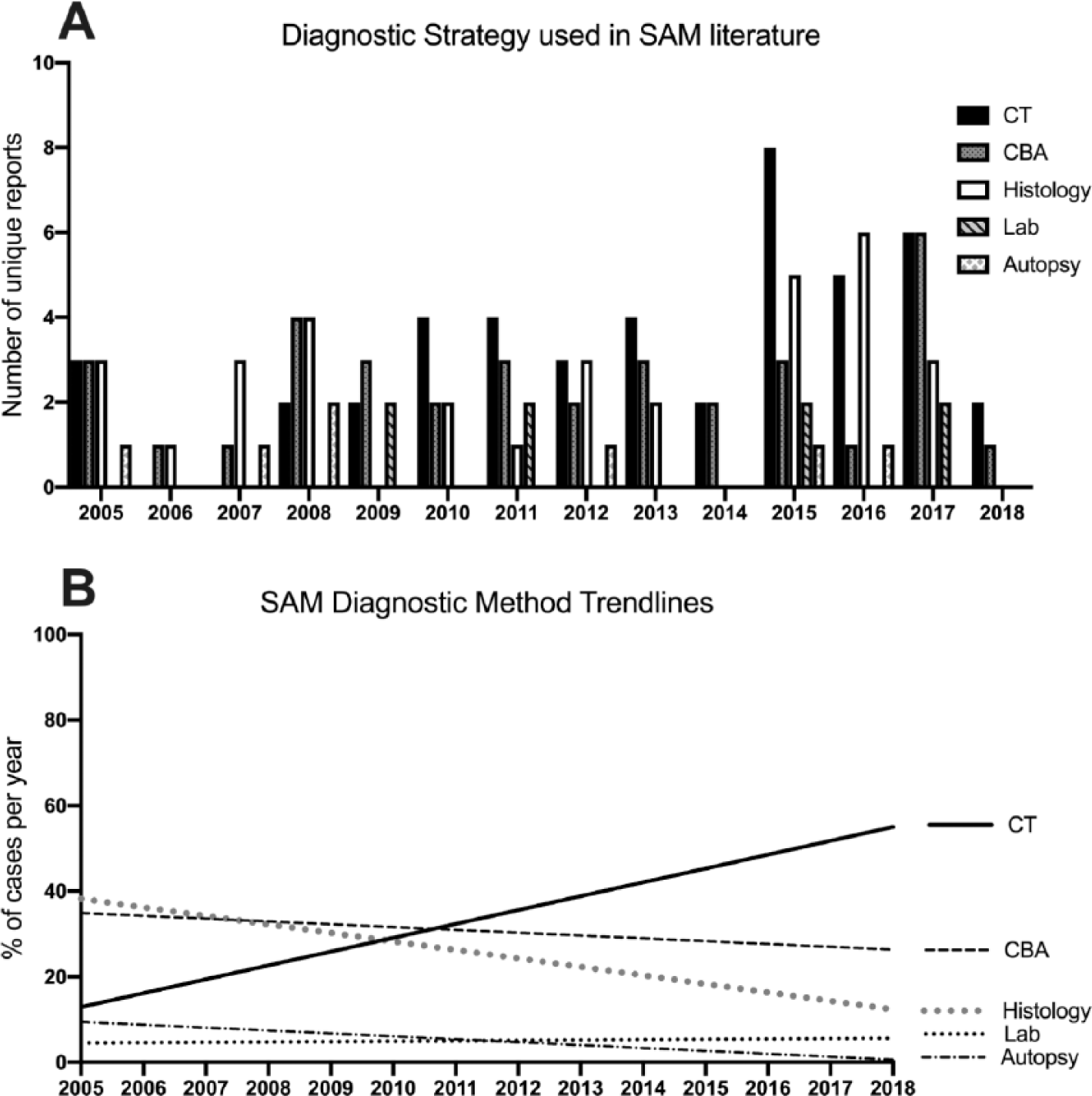
Reported methods for diagnosing SAM. (A) A total of 121 cases were identified from the 66 case reports4,5,8–10,13,22–81 with unique diagnostic strategies. For publications detailing multiple patients, a case was counted as unique if the diagnostic strategy was specifically based on that patient’s presentation and was considered redundant and only recorded once if the same strategy was applied to multiple patients in the report. The coding of the diagnostic strategy used for each report is outlined in Supplemental Table 1. (B) Linear regressions of the yearly percent of cases using each strategy show the increase in using CT as a diagnostic tool and the decrease in CBA and histology.
There is particular interest and need to distinguish SAM from FMD. Although ‘string of beads’ is one of the most notable radiographic findings in multifocal FMD, it has also been reported in other mimicking vasculopathies.12,82 –84 Twenty-one cases included in our meta-analysis reported a beading appearance visualized on CT or MRA. Similarly, 28% of 111 patients with SAM from a single-center analysis were reported to have a beading pattern in at least one artery. 2 While ‘string of beads’ is more commonly reported in FMD, dissections are most often seen with SAM. When only 19.7% of patients in the FMD US Registry had arterial dissection, 60.8% of our SAM patients had this finding. 85 The ‘First International Consensus on the diagnosis and management of fibromuscular dysplasia’ recently modified the diagnosis of FMD to an idiopathic, segmental, nonatherosclerotic and noninflammatory disease of the musculature of arterial walls, leading to stenosis of small and medium-sized arteries. 15 Additionally, the recent FMD consensus established that after diagnosing focal or multifocal FMD in one vessel, the presence of aneurysms, dissections, or tortuosity in all other vascular beds would be considered multivessel involved FMD. 15 Based on updated definitions of FMD, it is very difficult to distinguish SAM from FMD in cases with string of beads in addition to other vascular pathologies affecting multiple arteries without tissue histology. Seven of the 21 cases in our review that reported to have ‘string of beads’ or ‘beaded’ radiographic findings, were also confirmed to have SAM based on histological sampling. The remaining 14 patients that reported involvement in a different artery than the one with the ‘string of beads’ finding could have met the new criteria for FMD, making it impossible to accurately confirm SAM diagnosis without tissue biopsy. However, the majority of these 14 patients were male and had a more dramatic presentation with additional arterial dissection or rupture, making SAM the likely diagnosis.
Although the only definitive way to distinguish SAM cases with a beading appearance from multifocal FMD is by tissue biopsy, certain demographics, symptom characteristics, lab and imaging findings, as well as disease course, may help with the differential diagnostic process. 15 Table 5 provides comparison between SAM and FMD which may help with the differential diagnostic process, especially when tissue biopsy is not available.4,14,15,86 –88
Distinguishing characteristics of SAM and FMD.
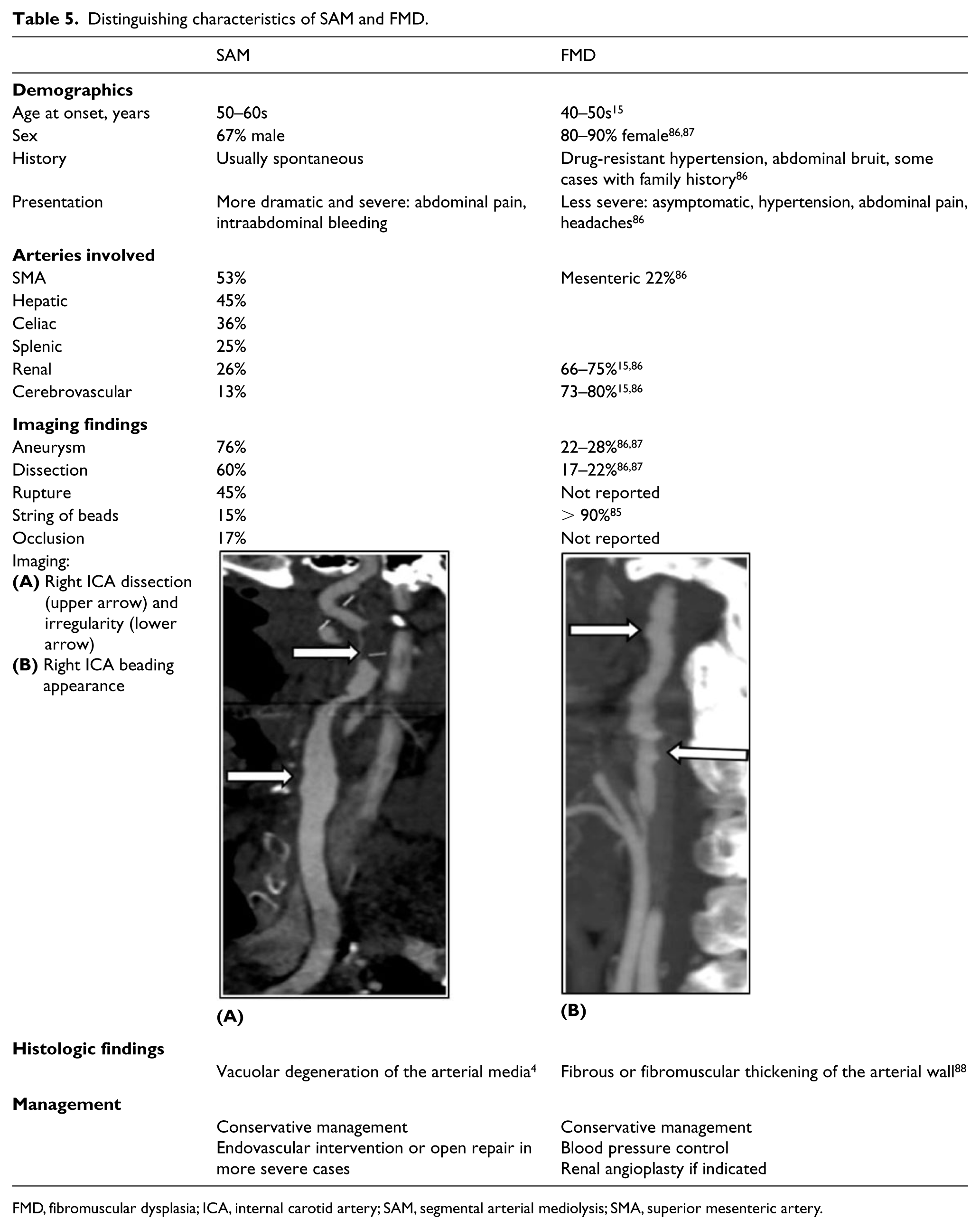
FMD, fibromuscular dysplasia; ICA, internal carotid artery; SAM, segmental arterial mediolysis; SMA, superior mesenteric artery.
A significant proportion of SAM cases who reported histologic findings had undergone laparotomy (65%) and open surgery (44%), allowing for sampling of tissue biopsies. Most subjects without available tissue biopsies were male (76%) and did not present as emergently with abdominal bleed (35%) as did those with available histology (68%). Furthermore, the celiac and renal arteries were significantly more commonly involved in cases without available histology. The ‘string of beads’ finding was reported at comparable rates (11% with vs 18% without histology). The significant differences in treatment and outcomes among the two groups is best explained by the more severe and emergent presentation in patients with available tissue biopsy. Given the fact that biopsy was mainly available in cases requiring open intervention, it is hard to assume that patients without available histology do not represent true cases of SAM. Table 4 outlines the differences in presentation, pathology, and treatment for patients with or without reported histology findings.
We briefly share a typical case of SAM from our institution of a 47-year-old male who presented with sudden onset abdominal pain with nausea and vomiting. CTA revealed hematoma, stenosis, dissection, pseudoaneurysm, aneurysm, and infarct involving various cerebral and abdominal arteries (Figure 3) concerning for arteriopathy, such as SAM. The patient required laparotomy, ileocecectomy, end ileostomy, and abdominal washout of 2 liters of hemoperitoneum, which allowed for histological sampling confirming SAM. Genetic testing excluded mimicking connective tissue disorders. 21
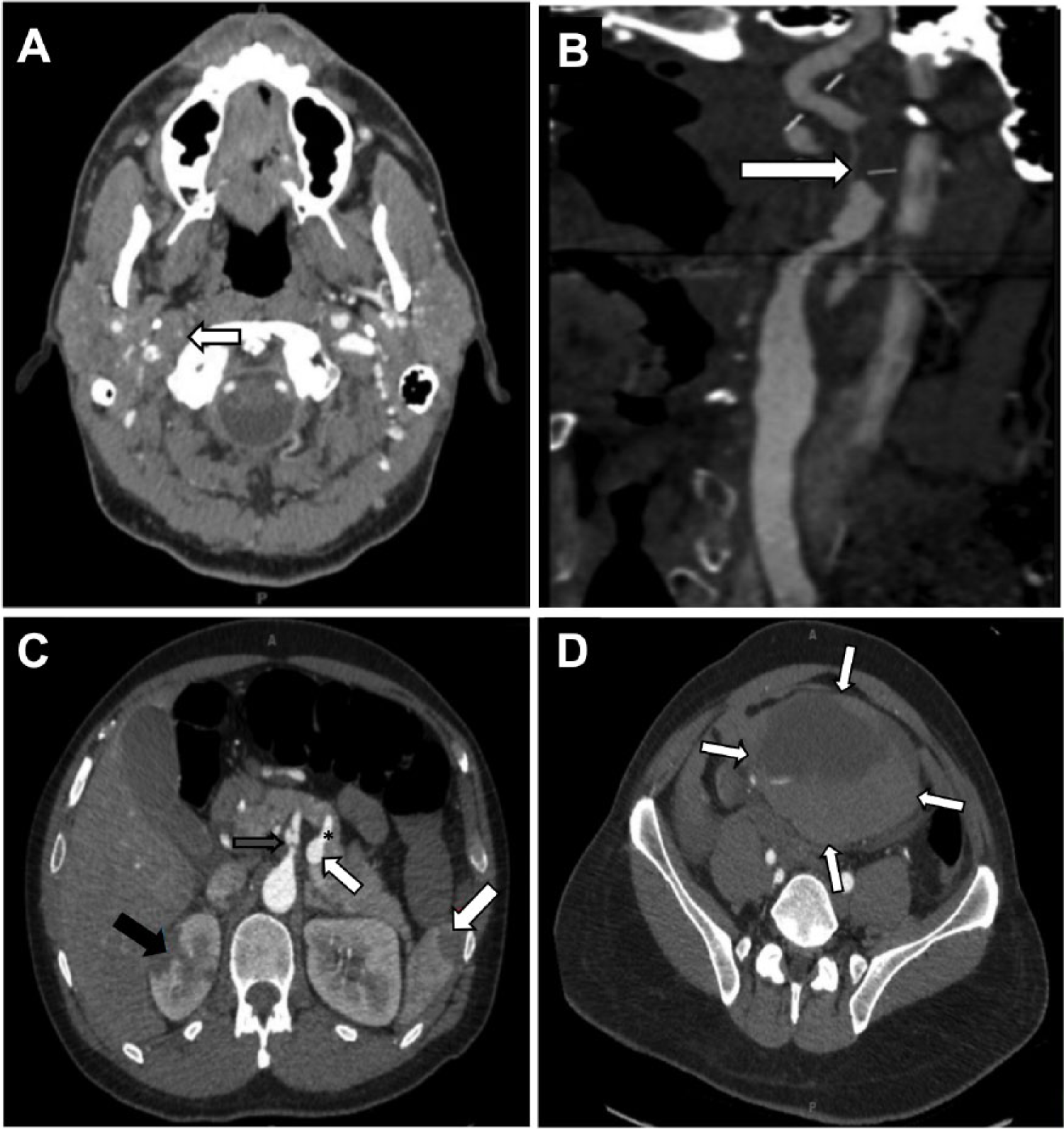
(A) Head and neck CT angiogram transverse image revealed right ICA dissection with severe stenosis (arrow) and (B) same CT study, sagittal image showed right ICA dissection with critical stenosis (arrow). (C) Abdomen and pelvic CT angiogram transverse image with contrast revealed celiac artery dissection with pseudoaneurysm (gray arrow), splenic artery aneurysm (small white arrow with asterisk), right renal infarct (black arrow), and splenic infarct (large white arrow) and (D) expanding intraabdominal hematoma measuring 11.6 × 13.1 × 8.2 cm (arrows).
Based on the individual presentations and pretext probability, CBC, creatinine, liver function, CRP, ESR, ANCA, antinuclear antibodies, and genetic testing should be considered. While most centers, including Naidu et al. and our own, have previously used elevated inflammatory biomarkers as exclusion criteria for diagnosis, our review has noted that of those receiving laboratory workup, 33% had elevated ESR and 69% had elevated CRP, which can be due to concurrent dissection and/or intraabdominal bleeding. 2 Further research is required to determine the role of inflammatory markers in the diagnosis of SAM and in differentiating it from other inflammatory mimics like PAN or LVGT.
Management and outcomes
The two most common interventional strategies were coil embolization (28%) and abdominal organ surgery (24%). Despite being the most commonly used treatment modality in our review, coil embolization was used less often than previously reported (33–79%).1,22 Based on the high rate of emergent presentations in our review, abdominal organ surgeries were performed to manage arterial-related complications. The next most commonly used management modalities were open arterial repair (21%), blood pressure control (20%), anticoagulation (12%), and antiplatelet therapies (11%). Angioplasty/stenting was the least common management approach, used in 8% of patients. Past reports showed a higher mortality rate (9%) with open than with endovascular repair (0%), indicating fewer complications with the latter approach. 1 Finally, we report a lower rate of conservative approach (8.1%) compared to previous reports (19%). 22 This is likely explained by under-reporting of conservative management while focusing on interventional strategies. 22 Based on increased disease awareness and the availability of better imaging qualities, we expect that more SAM cases will be diagnosed and managed with conservative rather than invasive approaches.
We report an excellent (93%) survival rate for patients with SAM, where 91% experienced symptom improvement and/or relief by the end of the report. Shenouda et al. reported a mortality rate of 26% in 2014. Similarly, Kim et al. reported a mortality rate of 22%.1,22 A more recent report by Naidu et al. in 2018 reported 0% mortality during their follow-up period. 2 This can be explained by improved sensitivity and specificity of the diagnostic criteria used to distinguish SAM from more fatal diseases such as connective tissue or vasculitis disorders.
Our diagnostic approach for patients presenting with abdominal vasculopathies is summarized in Figure 4. Based on our experience with SAM, we recommend conservative therapy that includes blood pressure control, bowel rest, blood count monitoring, and pain control in most patients. We offer endovascular over open repair to unstable patients with abdominal emergencies caused by arterial rupture. Based on our current understanding of SAM progression, our center recommends thin-slice CTA imaging at 1, 3, 6, and 12 months to exclude disease progression or serious complications, such as enlarging pseudoaneurysms and/or expanding hematoma. Further surveillance is based on the artery involved and severity of the initial presentation. We believe that our diagnostic criteria are appropriate to diagnose SAM and exclude mimicking pathologies. More research is required to reach consensus on diagnostic criteria and management approach.
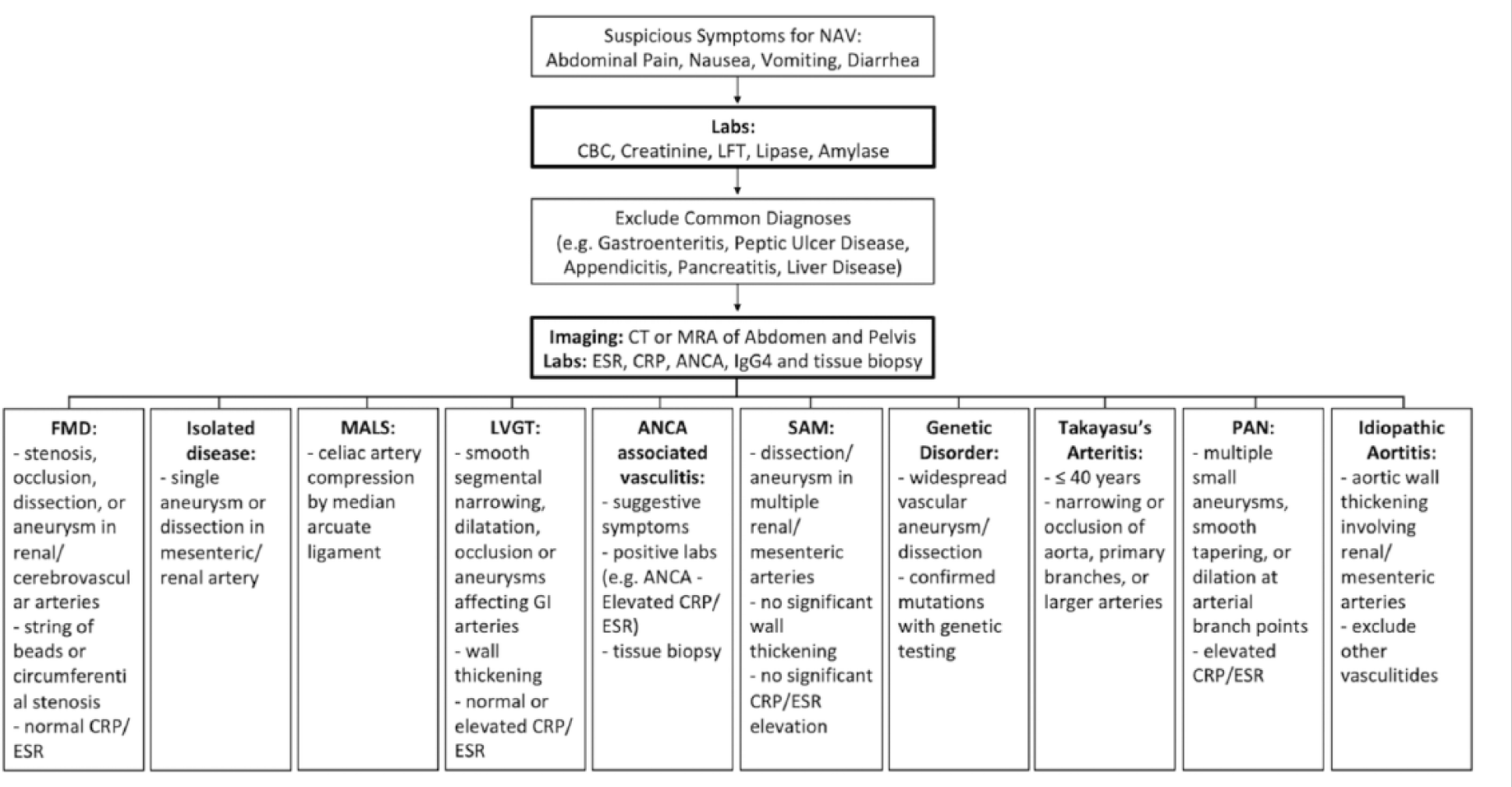
Methods of differentially diagnosing nonantherosclerotic abdominal vasculopathies.
Study limitations
This is a retrospective review of case studies reported by different medical centers which used different diagnostic criteria and reported different variables related to SAM. To best mitigate these discrepancies, we use the word ‘reported’ to refer to any given variable. As a meta-analysis reviewing papers by other authors, we are unable to completely confirm accurate diagnosis of SAM in all cases included. We trust the credibility of the authors and reviewers involved in these publications to have appropriately distinguished SAM from FMD and other mimics. Furthermore, the new criteria used at the 2019 first international consensus on FMD diagnosis has expanded the required arterial involvement for the diagnosis of FMD to include any single or multiple medium artery, making it very hard to distinguish SAM from FMD in situations when there are multiple arterial pathologies involving multiple medium size arteries. 15 Therefore, a few of the cases with proposed SAM diagnosis in our series, particularly the 14 cases with string of beads and no histology, could have been considered to have FMD based on the new diagnostic criteria. Additionally, case reports are biased towards reporting rare and unique SAM cases with complications. Therefore, the circulating literature may be skewed towards presenting more severe cases that require intervention. Conversely, fatal SAM cases may be underreported which can result in an underestimated mortality rate.
This review is also limited by the lack of family history and relevant genetic study results. As our experience with genetic disorders continues to grow based on evolving genetic testing, we expect a more standardized diagnostic approach to include genetic counseling and testing based on availability. We also note that limiting the age range of our center’s review to 18–60 years could have resulted in an underestimation of the sample size.
Conclusion
Segmental arterial mediolysis is a rare disorder that affects middle-aged adults and may, rarely, lead to major complications including death. More research is needed to standardize diagnostic criteria and establish consensus on appropriate management. We present our center’s recommendation for appropriate diagnosis and management of SAM favoring a conservative strategy.
Supplemental Material
10.1177_1358863X19873410_Supplemental_Table – Supplemental material for Segmental arterial mediolysis (SAM): Systematic review and analysis of 143 cases
Supplemental material, 10.1177_1358863X19873410_Supplemental_Table for Segmental arterial mediolysis (SAM): Systematic review and analysis of 143 cases by Nedaa Skeik, Sydney L Olson, Gopika Hari and Mary L Pavia in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
