Abstract
The US Food and Drug Administration (FDA) has approved multiple systemic vascular endothelial growth factor (VEGF) inhibitors since 2004 to treat various malignancies. Inhibition of the VEGF signaling pathway can result in impairment of vascular wall integrity through medial degeneration and endothelial dysfunction, potentially resulting in arterial (including aortic) aneurysm/dissection. We performed a postmarketing review to evaluate arterial aneurysm/dissection as a potential safety risk for patients with cancer treated with VEGF inhibitors. We searched the FDA Adverse Event Reporting System (FAERS) database and literature for reports of arterial (including aortic) aneurysm/dissection with VEGF inhibitors currently approved by the FDA for a cancer indication. We identified 240 cases of arterial aneurysm/dissection associated with VEGF inhibitors. The median time to onset of an arterial aneurysm/dissection event from the initiation of a VEGF inhibitor was 94 days (range 1–1955 days). Notably, 22% (53/240) of cases reported fatal outcomes related to arterial aneurysm/dissection. We determined the drug–event association as probable in 15 cases that lacked relevant confounding factors for arterial aneurysm/dissection, which is supported by unremarkable computed tomography (CT) findings prior to starting VEGF inhibitor therapy, despite nondrug-associated background arterial aneurysm/dissection generally demonstrating preexisting arterial abnormalities. FAERS and literature case-level evidence suggests that VEGF inhibitors may have contributed to arterial aneurysm/dissection, as a class effect, based on short onset relative to natural history of disease and biologic plausibility. Cardiovascular and oncology healthcare professionals should be aware of this rare, but life-threatening safety risk associated with VEGF inhibitors.
Introduction
Safety data related to arterial (including aortic) aneurysm/dissection in association with systemic vascular endothelial growth factor (VEGF) inhibitors have become a potential pharmacovigilance concern of the US Food and Drug Administration (FDA) based on VEGF inhibitor mechanistic pathways, literature reports, and other databases. Oshima et al. conducted a disproportionality analysis of the Japanese drug regulator Pharmaceuticals and Medical Devices Agency’s (PMDA) adverse event reporting system, and reported an odds ratio of 19.4 (95% CI: 10.2–40.8) for aortic dissection with VEGF inhibitors when adjusting for history of hypertension. 1 These findings suggest arterial aneurysm/dissection is a potential safety signal that warrants further investigation for this class of drugs.
Therapeutics targeting the VEGF signaling pathway are effective for various specified cancer types that are unresectable, metastatic, or with high risk of recurrence. In the early 1970s, the concept of anti-angiogenesis as a potential target for anticancer drugs emerged.2,3 These targeted therapies have significantly improved the prognosis of several solid tumors and hematologic malignancies since FDA’s first approval of a VEGF inhibitor, bevacizumab, in 2004.4–9 Additional anti-VEGF therapies that have subsequently gained FDA approval for a cancer indication include sorafenib, sunitinib, pazopanib, vandetanib, axitinib, ziv-aflibercept, regorafenib, cabozantinib, ponatinib, ramucirumab, and lenvatinib.10,11 Most recently, two bevacizumab biosimilar products, bevacizumab-awwb and bevacizumab-bvzr, were approved by the FDA in 2017 and 2019, respectively.12,13 Online supplemental Table 1 provides a list of the FDA-approved VEGF inhibitors for a cancer indication and the year of their initial US approval from 2004 to 2019. VEGF inhibitor-associated toxicities include cardiovascular events, such as hypertension, thromboembolism, and heart failure.14–16 VEGF exerts acute vasodilating effects on the peripheral vessels. 17 In particular, VEGF receptor 2 stimulates the syntheses of nitric oxide and prostaglandin I2. VEGF inhibitor therapy can result in endothelial dysfunction and suppress healing of damaged endothelium, leading to loss of vascular stability and blood pressure (BP) elevation. 1 Published case reports have described arterial aneurysm/dissection with anti-VEGF therapies, including some patients who were reported as having well-controlled or no preexisting hypertension.17–37 Arterial dissection, particularly aortic dissection, is a relatively uncommon, life-threatening condition that typically presents as an acute catastrophic event. 38 Aortic dissection occurs once per 10,000 patients admitted to the hospital, and approximately 2000 new cases are reported in the US per year. 39 Additionally, aortic dissection was present in 1–3% of all autopsies. 40 Given the limitations of randomized controlled trials, which are often underpowered to detect rare adverse events such as arterial aneurysm/dissection, the objective of this postmarketing safety analysis was to determine if arterial aneurysm/dissection is a VEGF inhibitor-associated class effect in patients with cancer based on case-level evidence known to FDA from the FDA Adverse Event Reporting System (FAERS) and published literature.
Methods
We queried the FAERS database, which is a spontaneous reporting system that contains postmarketing adverse drug event reports submitted to the FDA. Drug manufacturers are required to, and the public, including healthcare professionals and consumers, may report adverse events associated with FDA-regulated products through the MedWatch program. Additional information regarding the FAERS database has been described elsewhere. 41
The FAERS database was searched for postmarketing reports of all arterial aneurysm/dissection events with VEGF inhibitors that are currently approved by the FDA for a cancer indication from the initial US product approval date to December 10, 2019. These reports were retrieved by performing a standardized Medical Dictionary for Regulatory Activities (MedDRA Version 22.1) query in FAERS for reports coded with one or more of the following MedDRA search terms: aortic dilatation (preferred term), aortic infections and inflammations, arterial infections and inflammations (high-level term), and aneurysms and artery dissections (high-level group term).
FAERS reports that were included in this evaluation met the following case definition for the analysis of VEGF inhibitor-associated arterial aneurysm/dissection: a diagnosis of arterial aneurysm/dissection-related events involving any artery of the body (e.g. aorta, intra and extracranial carotid, vertebral, visceral, extremity) reported by a healthcare professional. The World Health Organization-Uppsala Monitoring Centre (WHO-UMC) causality assessment system was applied to determine the likelihood of a causal relationship between the VEGF inhibitor and arterial aneurysm/dissection. 42 A report must have demonstrated a temporal association to be included in the case series, meaning that the arterial aneurysm/dissection must have occurred after exposure to a VEGF inhibitor. We excluded a report from further review and analysis if it did not describe an adverse event of interest, was a duplicate report, provided insufficient information to confirm temporal relationship or diagnosis of the event, was an aggregate summary of safety data from a clinical trial, or the arterial aneurysm/dissection was attributed to an alternative etiology (e.g. tumor invasion, infection).
PubMed and EMBASE databases were also searched for additional cases of arterial aneurysm/dissection with VEGF inhibitors that were not reported in FAERS. The search terms consisted of the product names for the FDA-approved VEGF inhibitors with cancer indications, listed separately and in combination, with ‘dissection’ and ‘aneurysm’. The databases were searched on December 11, 2019, and all English-language case reports and series published in the literature met the same inclusion criteria used for the FAERS cases.
The work was performed under specific federal statutory authority to support public health surveillance; therefore, no approval from an institutional review board or informed consent was required. 43
Results
We identified 239 FAERS cases that met the criteria for inclusion in the case series, and among these, 20 were also reported in the literature.17–19,21–37 Additionally, the literature search retrieved one published case that was not identified in FAERS and met the inclusion criteria. 20 This resulted in a combined FAERS and literature case series of 240 unique cases of arterial aneurysm/dissection events associated with VEGF inhibitors.
Descriptive characteristics of the 240 cases are summarized in Table 1. The patients had a median age of 67 years (range 33–91 years). Overall, 21% (50/240) of cases reported age less than 60 years and 44% (105/240) of cases reported females (Figure 1). The most commonly reported indications for VEGF inhibitor use were colorectal cancer (n = 58) and renal cell carcinoma (n = 49). An arterial aneurysm/dissection event was cited by the reporter as the cause of death in 22% (n = 53) of cases, including 10 cases that reported autopsy findings. In our series, the majority of cases (n = 169) reported arterial aneurysm/dissection events involving the aorta, including aortic dissection (n = 130) and aortic aneurysm (n = 39) with or without rupture. The median time to onset of an arterial aneurysm/dissection event from the initiation of a VEGF inhibitor was 94 days (range 1–1955 days) among the cases that reported this information. Notably, in 73 cases, the time to event onset was within 60 days. In addition, 167 cases reported a diagnostic test to confirm the presence of arterial aneurysm/dissection. Furthermore, a total of 31 cases reported a baseline CT scan or comparable imaging results that did not reveal significant arterial abnormalities prior to initiation of a VEGF inhibitor. We assessed the cases for the presence of potential confounding factors, such as genetic predisposition (e.g. Marfan syndrome, Ehlers–Danlos syndrome), family history of arterial aneurysm/dissection, atherosclerosis, preexisting cardiovascular risk factors (e.g. diabetes, hypercholesterolemia, cigarette smoking, hypertension), presence of baseline aneurysms or inflammatory vasculitis (e.g. aortitis, arteritis), trauma or recent surgery, bicuspid aortic valve and/or aortic coarctation, advanced age, male sex, and concomitant medications associated with arterial aneurysm/dissection (e.g. fluoroquinolones and granulocyte colony-stimulating factors).44–52 After applying the WHO-UMC causality assessment scale to our case series, we determined causality as probable in 15 cases without apparent contributory factors. In these 15 cases, the reported arterial aneurysm/dissection events included aortic dissection (n = 10), aortic aneurysm (n = 1), carotid artery dissection (n = 1), coronary artery dissection (n = 1), intracranial aneurysm (n = 1), and peripheral artery aneurysm (n = 1); six of these cases reported fatal outcomes attributed to arterial aneurysm/dissection. We assessed causality as possible in the remaining 225 cases, which were confounded by relevant comorbidities, concomitant medications, or did not report this information.
Descriptive characteristics of arterial aneurysm/dissection events reported with systemic VEGF inhibitors (n = 240).

Of the 130 cases, there were 46 Stanford type A dissections, 34 Stanford type B dissections, and 50 cases that did not have sufficient information to assign a Stanford category.
Other arterial aneurysm/dissection events occurred in intra and extracranial carotid (n = 29), vertebral (n = 3), visceral (n = 9), extremity (n = 7), not otherwise specified (n = 23) arteries.
Includes 53 fatal cases that reported arterial aneurysm/dissection as the cause of death.
More than 1 risk factor for arterial aneurysm/dissection may have been reported per case.
Hypertension was only considered a confounding factor if the case reported unknown or poor BP control.
Includes cases that reported hypertension as either ‘well controlled’ or having SBP < 160 mmHg and DBP < 100 mmHg.
Other risk factors included aortic insufficiency (n = 1), concomitant granulocyte colony-stimulating factor (n = 3), history of carotid artery laceration (n = 1), left ventricular hypertrophy (n = 1), myocarditis (n = 1), peripheral vascular disease (n = 3), prior radiotherapy (n = 4).
Baseline computed tomography scan or comparable imaging results did not reveal significant arterial abnormalities.
AXI, axitinib; BEV, bevacizumab; BP, blood pressure; CABO, cabozantinib; DBP, diastolic blood pressure; LEN, lenvatinib; N/A, not applicable; PAZ, pazopanib; PON, ponatinib; RAM, ramucirumab; REG, regorafenib; SBP, systolic blood pressure; SOR, sorafenib; SUN, sunitinib; VAN, vandetanib; VEGF, vascular endothelial growth factor; WHO-UMC, World Health Organization-Uppsala Monitoring Centre; ZIV, ziv-aflibercept.
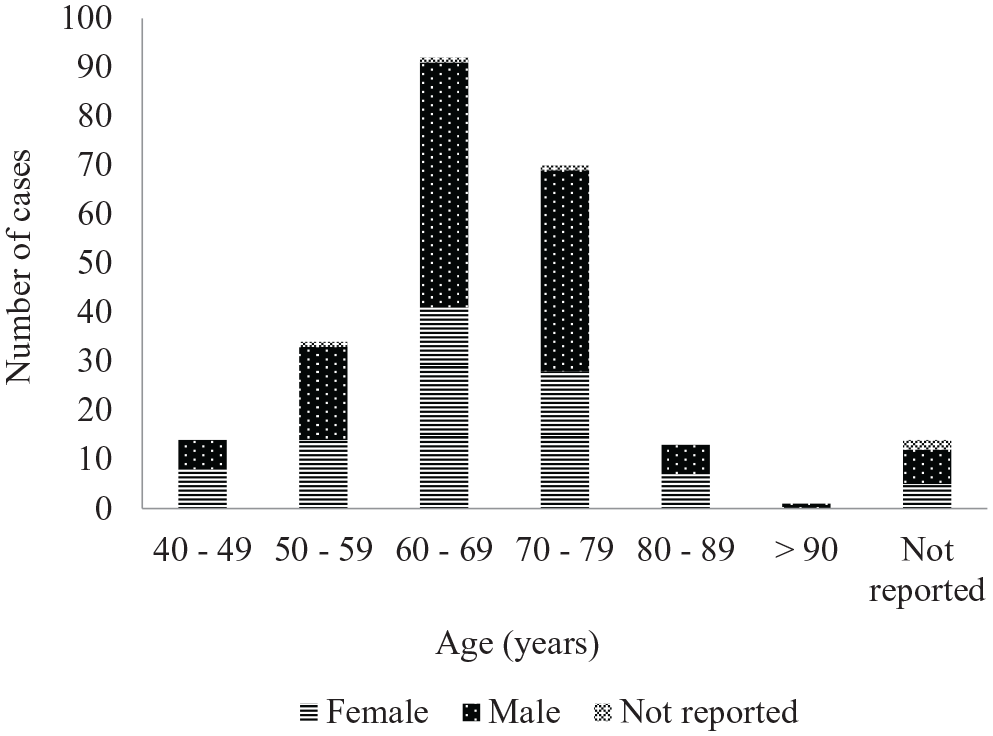
The distribution of cases by age and sex in the US FDA Adverse Event Reporting System (FAERS) and literature case series (N = 240).
Of the 130 cases that reported aortic dissection with or without rupture, 80 of them had sufficient information to be categorized as Stanford type A (n = 46) or Stanford type B (n = 34), of which 19 (41%) and 3 (9%), respectively, died as a result of the dissection. Emergency surgical procedures were performed on 19 of the Stanford type A dissections, with one perioperative death reported. Four Stanford type B repair procedures were performed, and all of them were endoprosthesis. These four repairs reported no dissection-related deaths.
Discussion
Our case series included a total of 240 cases of arterial aneurysm/dissection events reported during treatment with a VEGF inhibitor. The severity of arterial aneurysm/dissection was evidenced by 53 fatal cases attributing death to arterial dissection or ruptured aneurysm, 51 of which involved the aorta; the remaining two cases reported ruptured cerebral aneurysm as the cause of death. Additionally, 44% (n = 105) of the cases reported females, including 46 cases less than 65 years of age. This is unexpected as arterial aneurysm/dissection events are rare among females less than 65 years of age, with an overall incidence rate of less than 2 per 100,000 population. 53 Eight of these 46 cases reported fatal outcomes related to arterial aneurysm/dissection (bevacizumab (n = 6), axitinib (n = 1), sorafenib (n = 1)).
Characteristics of arterial aneurysm/dissection in our case series suggest these are not naturally occurring background events. Overall, we identified 15 probable cases based on the WHO-UMC causality scale that reported no overt confounding factors for arterial aneurysm/dissection, including uncontrolled hypertension. Notably, 31 cases, including 11 probable cases, reported thoracoabdominal CT scan or comparable imaging results that confirmed the absence of baseline arterial aneurysm/dissection immediately preceding initiation of a VEGF inhibitor. In these cases, undetected significant preexisting aortic disease is unlikely given the high sensitivity and specificity of either CT, magnetic resonance angiography, or transesophageal echocardiography of the aorta.54,55 As such, baseline atherosclerotic risk factors, some of which are also shared with malignancy, were considered to be less relevant confounders in these cases. Additionally, in 16 of these 31 cases (including seven probable cases), the time to event onset was within 60 days despite unremarkable baseline imaging results. Unlike the natural progression of aortic disease, these cases suggest the rapid development of aortic aneurysm/dissection within a short timeframe after initiation of anti-VEGF therapy, providing further evidence that arterial aneurysm/dissection may be associated with VEGF inhibitors.56,57 Our findings are consistent with the published Japanese postmarketing database analysis of aortic dissection reported with VEGF inhibitors; the median time to onset was 105 days. 1 Notably, the onset of drug-induced aortic aneurysm/dissection reported with fluoroquinolones occurred within 60 days and has been described to occur as early as in the first 10 days following fluoroquinolone initiation.51,58–60 In our series, 23 cases (including 14 cases of aortic aneurysm/dissection) reported a time to onset within 2 weeks of treatment initiation with a VEGF inhibitor.
The most common underlying risk factor for arterial aneurysm/dissection reported in the case series was hypertension (n = 135). However, hypertension was only considered a confounding factor if the case reported unknown or poor BP control. We defined poorly controlled BP as systolic BP (SBP) ⩾ 160 mmHg or diastolic BP (DBP) ⩾ 100 mmHg during treatment with a VEGF inhibitor; these BP parameters also correspond to grade 3 hypertension for adults according to the National Cancer Institute-Common Terminology Criteria for Adverse Events, Version 5.0. 61 Among 135 cases that reported preexisting or concurrent hypertension, we identified 41 of them as having poorly controlled BP. Notably, two of the 15 probable cases reported preexisting hypertension prior to initiation of a VEGF inhibitor; however, none of these cases reported poorly controlled BP during anti-VEGF treatment. In addition, we identified 75 cases of arterial aneurysm/dissection in patients without preexisting hypertension. Twelve of these cases experienced elevated BP and subsequent arterial aneurysm/dissection within 60 days of treatment initiation with a VEGF inhibitor (sorafenib (n = 3), bevacizumab (n = 3), sunitinib (n = 2), lenvatinib (n = 1), ramucirumab (n = 1), vandetanib (n = 1), ziv-aflibercept (n = 1)). Hypertension is a known adverse drug reaction for all currently FDA-approved VEGF inhibitors with a cancer indication, and the current US prescribing information for these products include general recommendations for monitoring and managing hypertension induced or exacerbated by VEGF inhibitors.62–74 Furthermore, hypertension can occur in up to 80% of patients receiving VEGF inhibitors, and although the kinetics of BP increase with VEGF inhibitors vary, hypertension may be observed as soon as 1 day following initiation of a VEGF inhibitor.75,76 Of note, a prospective study with sorafenib reported that 93% of patients experienced elevated BP by day 6, including some patients who experienced elevated BP within 24 hours of therapy initiation.76,77 Additionally, a retrospective study demonstrated that multiple VEGF inhibitors, including axitinib, cabozantinib, lenvatinib, pazopanib, sorafenib, and sunitinib, were overall associated with mean increases in SBP and DBP of 8.5 mmHg and 6.7 mmHg, respectively. The greatest mean increases in SBP (13.0 mmHg) and DBP (8.4 mmHg) relative to baseline occurred during the first 4 weeks of anti-VEGF treatment. 14
While VEGF inhibitors may contribute to arterial aneurysm/dissection by inducing hypertension, VEGF inhibitor-mediated impairment of vascular wall integrity through medial degeneration and/or endothelial dysfunction may represent another potential mechanism. 17 It is important to note VEGF inhibitors may cause arterial dissection through the inactivation of the phosphatidylinositol-3-kinase-AKT signaling pathway, which may result in decreased expression of the tissue inhibitor of metalloproteinase-1 and overexpression of matrix metalloproteinase 9 (MMP9). 17 MMP9 degenerates collagen tissue and is associated with various vascular diseases, including aortic dissection, due to decreased elasticity of the arterial wall and medial degeneration.17,78,79 This similar biologically plausible mechanism has been described for arterial aneurysm/dissection associated with other drugs, including fluoroquinolones and granulocyte-colony stimulating factors.51,52,58
Vascular repair, when indicated, appeared to have acceptable levels of success in our case series, despite challenges of intervening on patients with advanced malignancies. 80 All the Stanford type B, and all but one of the Stanford type A dissections that were treated surgically and/or endovascularly, reported no procedure-related or dissection-related deaths, although we cannot exclude reporting bias. Nonetheless, this suggests such interventions under appropriate circumstances may have favorable outcomes. No procedure-related antegrade or retrograde propagations were reported among these patients.
We believe a therapeutic class effect is plausible based on the pathophysiologic mechanisms by which VEGF inhibitors induce hypertension and impairment of vascular wall integrity. While the majority of cases were reported with bevacizumab (n = 129) and sunitinib (n = 40), we do not believe this toxicity is worse for these VEGF inhibitors compared to others. These products have been on the US market the longest (over 14 years) among the other FDA-approved VEGF inhibitors with a cancer indication (online supplemental Table 1). Given that the approved number and type of indications vary among the VEGF inhibitors, we recognize differences in product usage pattern also exist, and this may be reflected in reporting volume.
Study limitations and strengths
This analysis is limited by inherent weaknesses of spontaneous reporting systems, such as underreporting of events, variable quality of reported data, including missing data (e.g. concomitant medications and their timing of administration), and the inability to quantify risk from the case reports. We are unable to estimate the incidence of arterial aneurysm/dissection with VEGF inhibitors as spontaneous reporting systems have incomplete capture of outcomes and exposures of interest. However, we consider the number of reported cases of aortic aneurysm/dissection with VEGF inhibitors in our series to be compelling in relation to the background incidence rate of aortic dissection in the general population, which is estimated to be 5 to 30 cases per 1 million people per year. 39 In general, published studies evaluating the prevalence of any type of arterial aneurysm/dissection in patients with cancer are sparse and limited by small sample sizes of different cancer types.81–83 A few retrospective studies have reported varying incidence rates of abdominal aortic aneurysm in patients with lung cancer, ranging from 1% to 11%.82,83 Compared to the general population, the high prevalence of aortic aneurysm observed in patients with lung cancer could be attributed to their many shared risk factors (e.g. advanced age, smoking) and detection bias via increased imaging screening. 83 Although these factors alone are unlikely to account for the relatively short time interval for arterial aneurysm/dissection development after initiation of anti-VEGF therapy.
A major strength of FAERS is the ability to detect rare and serious adverse events under real-world conditions that were not manifested in the clinical development program. In our analysis, the FAERS and literature case-level evidence suggests that VEGF inhibitors may have contributed to arterial aneurysm/dissection events, including patients unlikely to have nondrug-related etiologies, based on temporal association, cases with unremarkable baseline CT scans, and biologic plausibility. This work is an example of the continued importance of pharmacovigilance programs in monitoring marketed drug therapies, as it is improbable that an imbalance in these events would be detected in a typical clinical trial powered around an efficacy endpoint.
Conclusion
To our knowledge, this is the largest published case series of VEGF inhibitors associated with arterial (including aortic) aneurysm/dissection to date. Recently, the manufacturers updated the US prescribing information for all currently FDA-approved VEGF inhibitors with cancer indications with the addition of arterial (including aortic) aneurysm, dissection, and rupture under the Adverse Reactions-Postmarketing Experience section.62–74 Although VEGF inhibitor-associated arterial aneurysm/dissection may be rare, awareness of the potential for this serious adverse reaction should facilitate risk factor reduction, prompt diagnosis, and definitive treatment to avoid fatal vascular outcomes.
Supplemental Material
sj-docx-1-vmj-10.1177_1358863X211006470 – Supplemental material for Arterial aneurysm and dissection with systemic vascular endothelial growth factor inhibitors: A review of cases reported to the FDA Adverse Event Reporting System and published in the literature
Supplemental material, sj-docx-1-vmj-10.1177_1358863X211006470 for Arterial aneurysm and dissection with systemic vascular endothelial growth factor inhibitors: A review of cases reported to the FDA Adverse Event Reporting System and published in the literature by Connie Cheng, Michelle Nadeau Nguyen, Afrouz Nayernama, S Christopher Jones, Michael Brave, Sundeep Agrawal, Laleh Amiri-Kordestani and Daniel Woronow in Vascular Medicine
Footnotes
Disclaimer
The views expressed are those of the authors and not necessarily those of the US Food and Drug Administration.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
