Abstract
Peripheral artery disease (PAD) is caused by atherosclerosis and associated with an increased risk of leg amputation, cardiovascular disease, and death. A healthy diet has been shown to reduce the risk of cardiovascular events, but relationships between diet, fiber intake, and incidence of PAD are virtually unknown. The aim was to investigate the long-term impact of diet on the development of PAD among 26,010 middle-aged individuals in the prospective Malmö Diet and Cancer study (MDCS). Data on dietary intake were collected through a 7-day food diary combined with a food questionnaire and a 1-hour interview. Adherence to a recommended intake of six dietary components – saturated fat, polyunsaturated fat, fish and shellfish, fiber, fruit and vegetables, and sucrose – was scored (sum 0–6 points) to assess a diet quality index, adjusting for potential confounders. Cox regression analysis was used to estimate associations between diet variables and PAD incidence expressed in hazard ratios (HR) with 95% CI. During a median follow-up of 21.7 years, 1122 participants developed PAD. Diet score was associated with a reduced risk of PAD in multivariable analysis (p = 0.03). When mutually adjusting for all dietary variables, only adherence to recommended levels of fiber intake was associated with a reduced risk of incident PAD (HR 0.84; 95% CI 0.72–0.99). In this prospective, population-based study including 26,010 participants with over 20 years of follow-up, a healthy diet, especially a high intake of fiber, was associated with a reduced risk of PAD. Primary prevention programs directed against PAD should therefore include a fiber recommendation.
Introduction
Peripheral artery disease (PAD) is primarily caused by atherosclerosis in the arteries distal to the aortic bifurcation as a consequence of hemodynamic disturbances and hypercholesterolemia, causing endothelial dysfunction. 1 PAD affects quality of life and is associated with a higher risk of limb amputation 2 and death, 3 as well as myocardial infarction and stroke. There are several well-recognized risk factors for PAD, such as age, smoking, hypertension, hyperlipidemia, physical inactivity, and diabetes mellitus. 4
The risk of developing cardiovascular disease appears to be reduced by maintaining a high-quality diet. 5 A Mediterranean diet, including a high intake of fruit, vegetables and nuts, and a low intake of saturated fat and meat, has been shown to be associated with a decreased risk of ischemic stroke and myocardial infarction. 6 Fiber intake has consistently been associated with a lower risk of all-cause mortality. 7 However, investigations of associations between dietary habits, especially fiber intake, and incidence of PAD during long-term follow-up of middle-aged adults are limited. In a community-based, prospective study conducted in the United States, it was reported that a higher intake of meat was associated with a higher risk of PAD. 8
The aim of this study was to evaluate if diet quality and fiber intake influences the risk of PAD development in the population-based prospective Malmö Diet and Cancer study (MDCS) of middle-aged individuals free from cardiovascular disease and diabetes mellitus at baseline.
Method
Study population and data collection
Baseline investigations in the prospective MDCS were carried out between 1991 and 1996. The participants had to live in Malmö, Sweden, and have a good knowledge of Swedish. Men participating in the study were born between 1923 and 1945, and women were born between 1923 and 1950. A total of 30,447 participants were recruited at the start of the study, of whom 28,098 underwent diet assessment and anthropometric measurements, and completed an extensive questionnaire. More detailed descriptions of the recruitment and data collection process can be found elsewhere.9,10 Participants with prevalent PAD, prevalent abdominal aortic aneurysm (AAA), prevalent cardiovascular disease (prior myocardial infarction or stroke), or diabetes mellitus (with a higher likelihood to change dietary habits after diagnosis) were excluded from the study population at baseline. A total study population of 26,010 participants (Figure 1) was followed up until December 31, 2016. The study was ethically approved by the Regional Ethical Review Board in Lund, Sweden (Dnr §LU5190) and all participants gave informed consent at baseline. The study protocol followed the ethical guidelines outlined in the 1975 Declaration of Helsinki.
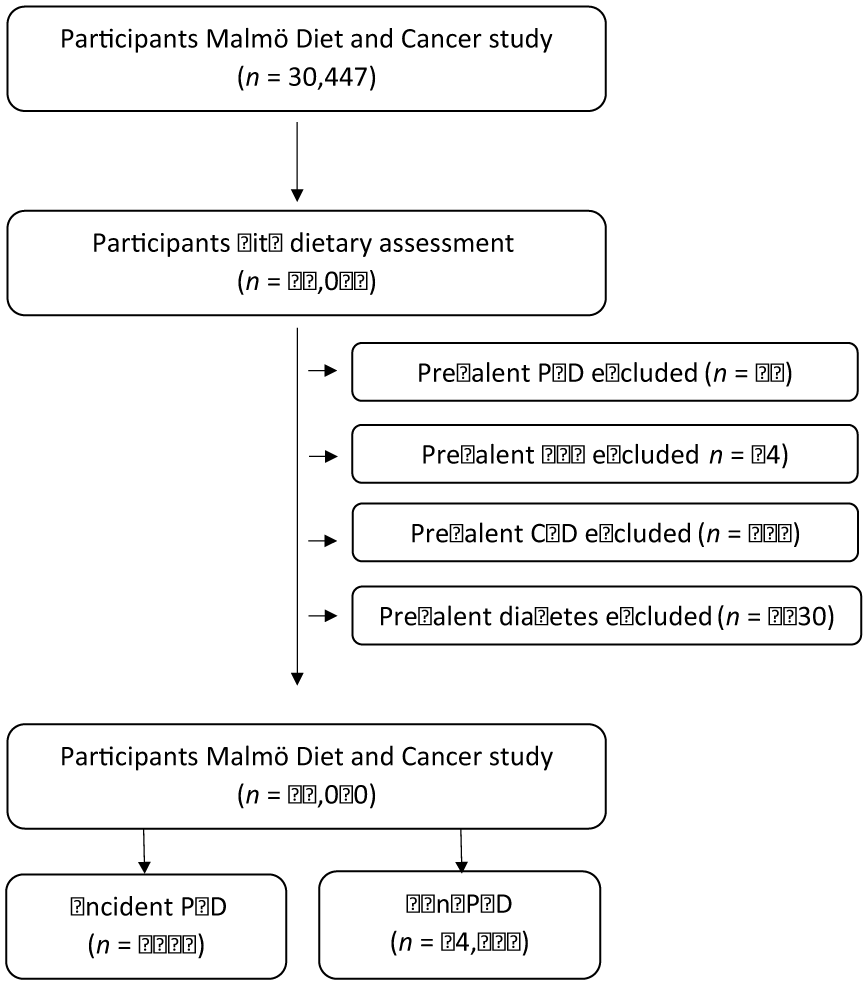
Descriptive flow diagram of study participants, dietary data, and exclusions.
Diet assessment method and construction of the diet quality index
Dietary information was obtained with a modified diet history method specifically designed for the MDCS. 11 A 7-day food diary recorded food intake at lunch and evening meals and the intake of cold beverages. This diary was combined with a 168-item food frequency questionnaire covering foods regularly consumed during the past year, taking frequency and portion-size of foods into account. 11 This was combined with a 1-hour interview where detailed data on portion sizes and cooking practices of the foods collected in the food diary were gathered. During the interview, information was checked so there was no overlap between the food diary and the questionnaire. The dietary assessment methods for measuring nutrient intake showed good ranking validity when compared to a reference method. 12 A diet quality index was developed to assess the quality of diet based on recommended dietary intake in the Swedish Nutrition Recommendations. 13 Firstly, candidate nutritional components had to be available within the MDCS cohort. Salt intake and trans-fatty acids were excluded because information on these dietary factors was lacking or of poor validity. Secondly, dietary components considered most important in assuring overall diet quality (i.e. food groups and macronutrients) in relation to chronic disease were primarily considered. Thirdly, dietary component inter-correlation was investigated in order to assess the mutual independence between components. Six dietary components were selected considering these three aspects: saturated fat, polyunsaturated fat, fish and shellfish, fiber, fruit and vegetables, and sucrose. Recommendations for each of the dietary components were outlined in the Swedish nutrition recommendations 13 and the Swedish dietary guidelines, 13 and one point was given for every achieved recommendation. Since only 2% of the study population had an intake of saturated fat in accordance with the recommendations, the cut-off points had to be adjusted. Cut-offs for reaching the recommendations were therefore: saturated fat ⩽ 14 energy (E) %, polyunsaturated fat 5–10 E%, fish and shellfish ⩾ 300 g/week, dietary fiber ⩾ 2.4 g/megajoule (MJ), fruit and vegetables (fruit juices excluded) ⩾ 400 g/day, and sucrose ⩽ 10 E%. Adherence to recommended levels was summed in a scoring system with a maximum score of six points. The diet quality index was categorized in three groups: low (0–1 points), medium (2–4 points), and high (5–6 points). Maintaining a high score on the diet quality index has been shown to decrease mortality.5,14
Endpoint ascertainment
Registries logging first diagnosis of PAD or surgical procedures for PAD were linked with the participant’s personal number. Registries included were the Cause of Death Register and the Inpatient and Outpatient Registers. All registered deaths in Sweden and causes of death are registered in the Cause of Death Register. The Inpatient Register includes information on dates of admission and discharge as well as diagnostic and procedural codes from all hospitalizations in Sweden. The Cause of Death Register includes diagnoses of underlying and contributing causes of death from death certificates. In both registers, diagnoses are coded using a Swedish revision of the International Classification of Diseases (ICD), Revision 8 (443.90; 443.99; 440.20; 445,00; 445,98; 445,99), Revision 9 (443X; 440C), and Revision 10 (I73.9 [all sub-codes except I73.9A]; I70.2 [all sub-codes]). Codes for embolism to the lower extremity were excluded (444.20, 444; I74.3, respectively). Surgical procedures are coded using a Swedish classification system (Op6): 8884; 8885; 8815; 8816; 8817; 8818; 8886; 8887; 8825; 8826; 8827; 8828; 8861; 8862; 8865; 8866; Klassifikation av kirurgiska åtgärder ( KKÅ) 97: PDH 10; PDH 20; PDH 21; PDH 22; PDH 24; PDH 30; PDH 35; PDP 10; PDP 30; PDQ 10; PDQ 30; PDU 82; PDU 83; PDU 84; PDU 99; PEF (all sub-codes); PEH (all sub-codes); PEN (all sub-codes); PEQ (all sub-codes); PEU 82; PEU 83; PEU 84; PEU 99; PFH (all sub-codes); PFN (all sub-codes); PFP (all sub-codes); PFQ (all sub-codes); PFU 82; PFU 83; PFU 84; PFU 99; PGH 20; PGH 21; PGH 22; PGH 23; PGH 30; PGH 31; PGH 40.
Other variables
The civic registration numbers for each participant were used to determine age and sex. Nurses registered the height, weight and blood pressure of all participants, and by using the formula kg/m2 a body mass index (BMI) was calculated. Systolic blood pressure ⩾ 140 mmHg or diastolic blood pressure ⩾ 90 mmHg or antihypertensive drug treatment was defined as hypertension. Information on lifestyle factors was collected through a self-reported questionnaire. Smoking habits were categorized as never, former, or current smoker. A smoking score was defined by stratifying time since smoking cessation in tertiles. The score ranged from 0 to 4: 0 = never smokers; 1 = no smoking for 25–51 years; 2 = no smoking for 12–24 years; 3 = no smoking for 1–11 years; 4 = currently smoking. In a subgroup of individuals in the present study, this smoking score was highly associated with pack-years smoked. 15 Education level was divided in five categories based on the highest education level attained: less than 9 years, elementary school (9–10 years), upper secondary school (11–13 years), university without a degree, and university degree. Leisure-time physical activity was determined by using a questionnaire and expressed as metabolic equivalent of task (MET) hours per week and categorized in five groups. MET hours were based on level of intensity and time spent on 17 different activities. Alcohol consumption was divided into six groups, where individuals who had not consumed alcohol in the last year were classified as zero-consumers. The remaining group was stratified into gender-specific quintiles according to their self-reported consumption noted in the food diary. Total energy intake was reported as kcal/day from diet and supplements, including alcohol. The term ‘dietary assessment method’ refers to the change in coding practice after 1 September 1994 and the term ‘season’ refers to the time of the year when the data collection took place. 11 Non-adequate reporters of energy intake were identified by comparing their reported energy intake with their total energy expenditure. Individuals with a reported energy intake above or below the 95% CI for total energy expenditure were categorized as ‘mis-reporters’. Individuals answering ‘yes’ to the questionnaire item ‘Have you substantially changed your dietary habits in the past?’ were classified as ‘dietary-changers’. Patients with diabetes mellitus were excluded at baseline and mis-reporters and dietary-changers were excluded in a sensitivity analysis. There was no reassessment of dietary intake over time.
Validation of PAD diagnosis
A total of 100 patients with a diagnosis of PAD were randomly selected for the validation procedure using patient record data. Among 100 patients, 69 had critical limb ischemia, 13 had acute limb ischemia, 15 had intermittent claudication, and one had asymptomatic PAD. Of the 13 patients with acute limb ischemia, 12 had acute thrombotic occlusion and one had an embolic occlusion. Two patients had venous insufficiency and were thus misdiagnosed. The diagnosis of PAD could therefore be confirmed in 98% of cases and symptomatic PAD in 97%.
Statistical analysis
Baseline characteristics for sex, age, BMI, diet and lifestyle variables were presented as mean and SD for continuous variables, and as total count and percentage for categorical variables. Hazard ratios (HR) with 95% CI were calculated for these variables by using the Cox proportional hazards regression model. The time scale was defined as years of follow up. Calculations of HR for diet variables were adjusted for age, sex, diet assessment method, season, and total energy intake. The calculations of HR for lifestyle factors, hypertension, and medication were adjusted for age and sex. HR was calculated for the diet quality index in all categories (low, medium, and high) and for adherence or non-adherence to each dietary recommendation. These variables were adjusted for age, sex, total energy intake, diet assessment method, and season in the basic model. In the multivariable model, alcohol consumption, physical activity, smoking status, education level, and BMI were added. The six dietary components included in the mutually adjusted model were adjusted for the variables in the multivariable model, as well as for the other five diet quality index components. Statistical analyses were carried out in IBM SPSS, version 25 (IBM Corp., Armonk, NY, USA) and the chosen level of statistical significance was p < 0.05.
Results
Demographic baseline characteristics
During a median 21.7 years (IQR 18.7–23.4) of follow up, 1122 out of 26,010 (4.3%) patients were diagnosed with PAD. The baseline characteristics of diet variables and lifestyle factors in those who later developed PAD and those who did not are presented in Table 1. Older people, male participants, and smokers ran an increased risk for the development of PAD. Individuals with a moderate alcohol consumption had a lower incidence of PAD (Table 1).
Association of baseline characteristics and incident PAD.
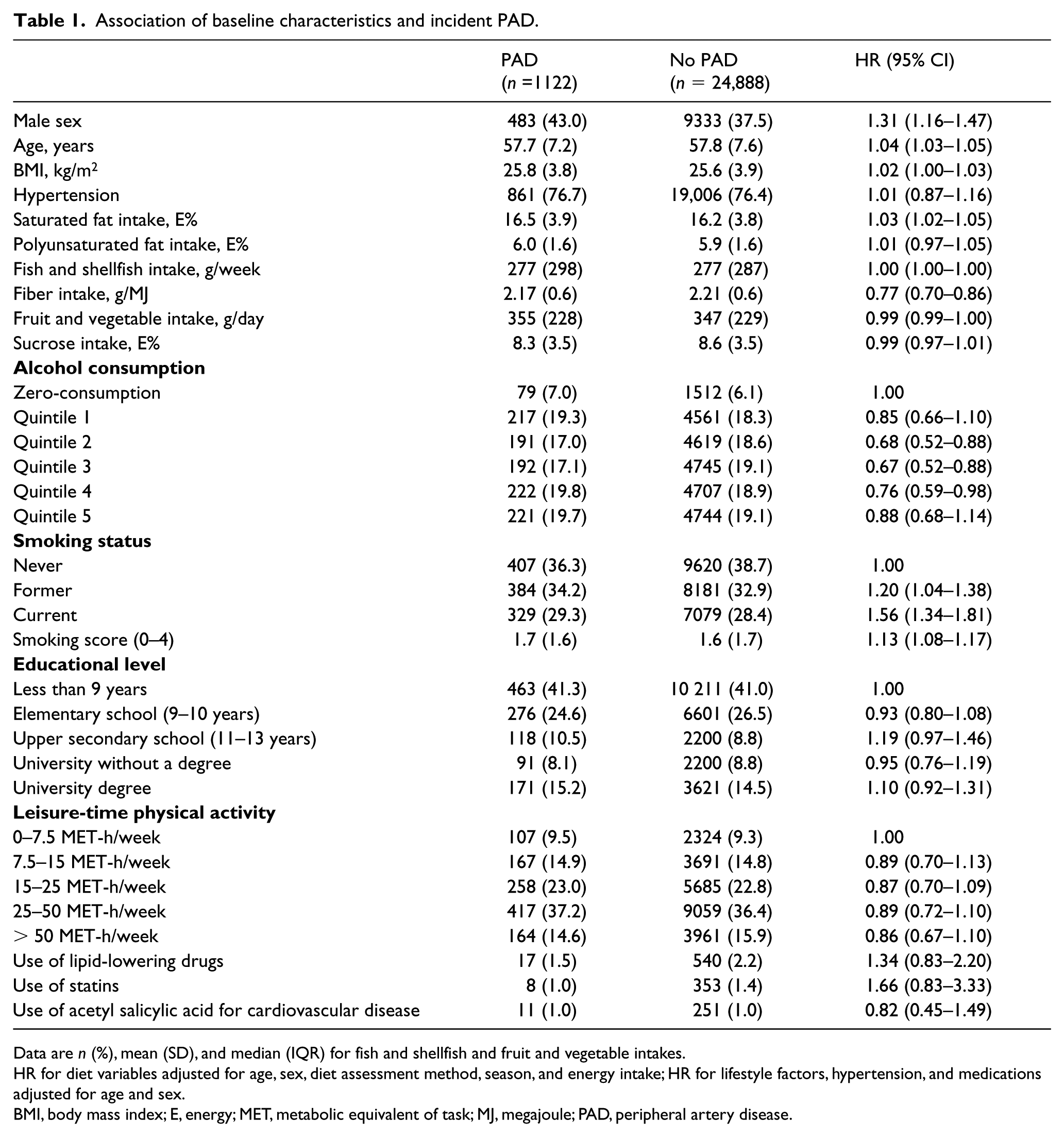
Data are n (%), mean (SD), and median (IQR) for fish and shellfish and fruit and vegetable intakes.
HR for diet variables adjusted for age, sex, diet assessment method, season, and energy intake; HR for lifestyle factors, hypertension, and medications adjusted for age and sex.
BMI, body mass index; E, energy; MET, metabolic equivalent of task; MJ, megajoule; PAD, peripheral artery disease.
Diet quality index and PAD
The diet quality index was associated with a lower risk for PAD development (p = 0.009) in the basic model. In the multivariable model adjusting for multiple potential confounders, this association was attenuated (p = 0.06) (Table 2). A 1-point increase in dietary score was associated with a 14% decreased risk of PAD in the basic model (HR 0.86; 95% CI 0.77–0.96), whereas in the multivariable model this association was not significant (HR 0.90; 95% CI 0.80–1.01). When entering the diet score (0–6 points) in the multivariable model as a continuous variable, instead of stratified in tertiles, a decreased risk for PAD development was demonstrated (p = 0.03).
Association of diet quality index and incident PAD.
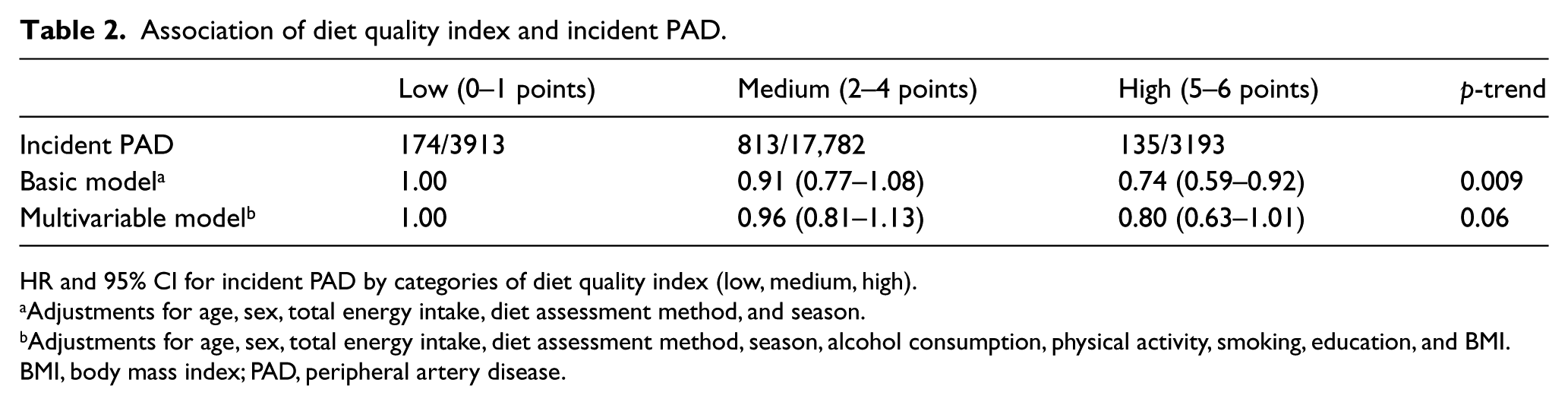
HR and 95% CI for incident PAD by categories of diet quality index (low, medium, high).
Adjustments for age, sex, total energy intake, diet assessment method, and season.
Adjustments for age, sex, total energy intake, diet assessment method, season, alcohol consumption, physical activity, smoking, education, and BMI.
BMI, body mass index; PAD, peripheral artery disease.
Adherence to components of diet quality recommendations and PAD risk
Individuals who adhered to the recommended intakes of fiber (HR 0.79; 95% CI 0.70–0.91) and fruit and vegetables (HR 0.82; 95% CI 0.72–0.93) had a significantly lower risk of PAD in the basic model (Table 3). This significantly decreased risk remained in both the multivariable model (HR 0.83; 95% CI 0.72–0.95) and the mutually adjusted multivariable model (HR 0.84; 95% CI 0.72–0.99) regarding adherence to recommendations on fiber intake. The risk also remained significantly decreased for individuals adhering to the recommended intake of fruit and vegetables in the multivariable model (HR 0.85; 95% CI 0.74–0.96), but not in the mutually adjusted multivariable model (HR 0.91; 95% CI 0.78–1.05).
Association of adherence to diet quality index components and incident PAD.
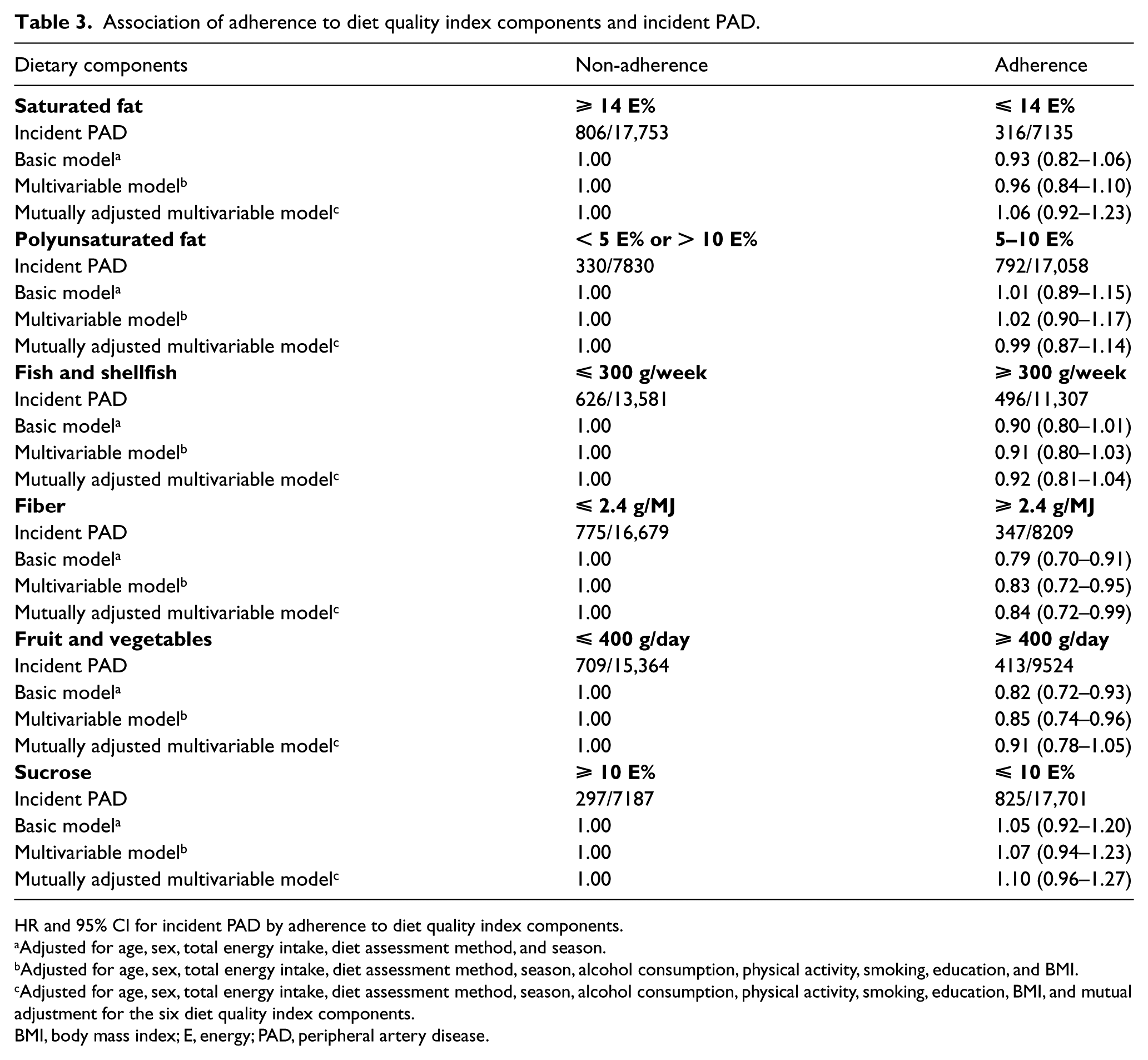
HR and 95% CI for incident PAD by adherence to diet quality index components.
Adjusted for age, sex, total energy intake, diet assessment method, and season.
Adjusted for age, sex, total energy intake, diet assessment method, season, alcohol consumption, physical activity, smoking, education, and BMI.
Adjusted for age, sex, total energy intake, diet assessment method, season, alcohol consumption, physical activity, smoking, education, BMI, and mutual adjustment for the six diet quality index components.
BMI, body mass index; E, energy; PAD, peripheral artery disease.
Sensitivity analysis
A sensitivity analysis was performed after removing mis-reporters (n = 4799) and dietary-changers (n = 5623). The association between the diet quality index and incident PAD was virtually unchanged. The association between the six dietary components and incident PAD showed that the association of adherence to fiber intake and reduction of PAD risk persisted, and that fish and shellfish reduced the risk of incident PAD (HR 0.84; 95% CI 0.72–0.97) in the mutually adjusted multivariable model (online supplemental Table 1).
Discussion
The main finding in this primary prevention, population-based, prospective cohort study on non-diabetic individuals with a median follow-up of 21.7 years, was an inverse association between adherence to a healthy diet and incident PAD. A decreased risk for PAD was especially found among participants complying with recommendations on fiber intake. Additionally, a lower risk for PAD was also observed among individuals adhering to recommended levels of fruit and vegetables, but this association was attenuated when adjusting for the other dietary variables in the index. These results show that an individual’s diet has an impact on long-term incidence of PAD.
A high diet quality index, defined as a continuous variable, was also associated with a lower risk of PAD. Similar findings have previously been found when examining the association between the diet quality index and cardiovascular disease risk, defined as myocardial infarction and stroke, in the same cohort. 5 Since myocardial infarction, stroke, and PAD all share the same pathogenic mechanisms, 16 evidence points towards a healthy diet and nutritional components playing a major role in the atherosclerotic process irrespectively of its anatomical localization. A healthy lifestyle including adherence to a Mediterranean diet, moderate alcohol consumption, physical activity, and avoidance of smoking, was reported to reduce the risk of PAD in another prospective cohort with high-risk individuals despite a much shorter median follow-up time of only 4.8 years. 17 Additionally, a randomized controlled trial has shown that individuals who adhered to a Mediterranean diet had a lower rate of incident PAD compared to a control group assigned to counseling on a low-fat diet. 18
Adherence to the recommendation for dietary fiber intake was shown to reduce the risk of PAD in the present study. No previous study has exclusively examined the potential role of dietary fiber intake in the pathogenesis of PAD. Whole-grain consumption was not found to be associated with reduced risk of PAD in a prospective cohort study on 14,082 individuals in the US, out of whom 1569 developed PAD during a mean follow-up of 19.9 years. 8 A possible explanation contributing to this lack of association might be that, although whole grains contain fiber, other foods are also important fiber sources. Fiber intake was not analyzed specifically in that study. In addition, the low fiber and whole-grain intake in the US population decreased the possibility to observe associations. A decreased risk of PAD in men with a higher intake of folate, a vitamin often found in fiber-rich products, was reported in another prospective cohort study 19 in which 46,036 men were followed for 12 years. This finding may be associated with intake of supplemental folate from multivitamin medicaments. Compared to the lowest quintile of intake of folate, those in the highest quintile had a higher intake of fiber. 19
The protective role of dietary fiber may be explained by several mechanisms. Soluble fiber prolongs the emptying of the stomach by forming a gel, which is thought to lower postprandial blood glucose and lipid increase, both of which are risk factors for atherosclerosis. Fiber also distends the stomach resulting in hormone secretion that increases satiation, thereby reducing food intake and weight, as well as improving glucose metabolism. By decreasing body weight, fiber intake also reduces blood pressure. Furthermore, the rate of bile acid excretion has been shown to be increased by dietary fiber, lowering total and low-density lipoprotein cholesterol. Cholesterol synthesis is also inhibited by production of short-chain fatty acids from fiber by bacteria in the gut. Since hyperglycemia, hyperlipidemia, and hypertension are all risk factors for the development of PAD and other cardiovascular diseases,20,21 this might offer a possible explanation for the protective effects of fiber intake. Another plausible explanation, however, could be that individuals with a higher intake of fiber also might have a higher intake of other healthy nutrient components and a healthier lifestyle in general, featuring physical activity and no tobacco use. 22
Adherence to recommended levels of fruit and vegetable intake was associated with a significantly decreased risk of PAD in the basic and multivariable models in the present study, before adjusting for dietary fiber. In contrast, no relation between consumption of fruit and vegetables and incident PAD could be demonstrated in the ARIC (Atherosclerosis Risk in Communities) study. 8 The different results may partly be explained by the different diet assessment methods. The ARIC study used a food frequency questionnaire, whereas the current study measured nutrient intake in grams. The defensive mechanism of a high-quality diet on PAD development may be due to a reduction of endothelial dysfunction and improved blood flow and oxygen perfusion in atherosclerotic areas of the vessel. 23 Vitamin C, present in fruit and vegetables, is shown to restore endothelial function, 24 further contributing to protection from atherosclerosis and PAD.
High intakes of fruit and vegetables were both found to be associated with a reduced risk of AAA in the same cohort during a shorter follow-up time. 25 In contrast to the results when risk of AAA development was assessed, 25 diet score and adherence to recommended levels of fiber were associated with decreased PAD risk in the present study. This discrepancy might be due to a type 2 statistical error in the study on AAA, since the number of AAA individuals were fewer and follow-up time was shorter than for PAD individuals. 25 Nevertheless, whereas AAA and PAD shared some similar risk factors such as age, male sex, current smoking, and hypertension in the two cohort studies, it should be taken into account that development of diabetes mellitus among the study participants throughout the study period 26 is likely to be associated with incident PAD, but not with incident AAA. In fact, diabetes mellitus is a protective factor reducing the incidence of AAA. 27 This difference in clinical risk factor profiles justifies that PAD and AAA are viewed as different diseases with different pathophysiologies, which therefore should be analyzed and discussed separately.
In the present study, PAD events were defined by using hospital registers, thus identifying only participants with symptomatic disease. It would therefore have been desirable to have measured ankle–brachial index (ABI) at study entry, allowing identification of asymptomatic PAD as well. Re-invitation of surviving study participants for measurement of ABI would also have been interesting to increase the total number of PAD cases, perform subgroup analysis of factors associated with symptomatic and asymptomatic incident PAD, and increase power in the overall statistical analysis. Furthermore, iteration of lifestyle and dietary measurements might have yielded more accurate data and enabled stronger conclusions. For instance, smoking prevalence has decreased in the general population. 28 It is, however, unlikely that the middle-aged study participants markedly changed their diet in their later stage of life. 29 In addition, individuals who might have changed their diet due to other diseases, such as diabetes, AAA and cardiovascular disease, were excluded at study entry. However, no censoring or adjustments in analysis for incident cardiovascular disease or incident diabetes mellitus was performed. Food intake was self-reported and the accuracy of the results in both this, and other similar studies, depend on the individual’s honesty about his or her diet. This could have resulted in misclassified estimations. Therefore, it was considered a methodological strength and justified to perform sensitivity analysis after removing mis-reporters and dietary-changers, and it was indeed found that compliance with the recommendation on fish and shellfish intake was associated with a 16% decreased risk for incident PAD, whereas there was no significant decreased risk in the statistical models including all 26,010 study participants. Even though there was a loss in statistical power by removing a significant number of individuals defined as mis-reporters and dietary-changers (10,422), it was only possible to show a significant decreased risk in the sensitivity analysis. Nevertheless, the dietary assessment methods used have been shown to have high validity.12,30 Several strengths of the present study need to be emphasized – the large sample size, the high rate of cumulative incidence of PAD, and the follow-up of over 20 years – must be considered as strong methodological advantages, which were prerequisites to follow these middle-aged participants into older age, where the incidence of PAD peaks.31,32 Furthermore, the validity of the PAD diagnosis was high, with confirmation of the diagnosis in 98% of the cases.
Conclusion
In conclusion, a higher intake of fiber was associated with a reduced risk of symptomatic PAD development in the present study. Primary prevention programs directed against PAD should therefore include the fiber intake recommendation of at least 2.4 g of fiber per total energy intake measured in megajoules.
Supplemental Material
10.1177_1358863X19867393_Supplementary_table – Supplemental material for Healthy diet and fiber intake are associated with decreased risk of incident symptomatic peripheral artery disease – A prospective cohort study
Supplemental material, 10.1177_1358863X19867393_Supplementary_table for Healthy diet and fiber intake are associated with decreased risk of incident symptomatic peripheral artery disease – A prospective cohort study by Andrea Kulezic, Sara Bergwall, Shahab Fatemi, Emily Sonestedt, Moncef Zarrouk, Anders Gottsäter and Stefan Acosta in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
