Abstract

Systemic thrombolysis remains a guideline-recommended treatment for high-risk pulmonary embolism (PE). 1 Although it is associated with improved survival, it is associated with a 9% risk of major bleeding.1,2 Catheter-directed thrombolysis (CDT) delivers thrombolytic agents directly into the pulmonary arteries with a lower total dose of thrombolytic and potentially a lower risk of bleeding compared with systemic thrombolysis. 3 Therefore, CDT may be an alternative advanced therapeutic option to systemic thrombolysis. However, little data are available describing contemporary trends in utilization of CDT (including CDT with and without ultrasound facilitation) and systemic thrombolysis.
We obtained data from the 2016 to 2018 Nationwide Inpatient Sample (NIS) following the introduction of new billing codes more specific for CDT in October 2015. The NIS represents a sample of 20% of all hospital discharges. We included all adult (⩾ 18 years of age) patients with a diagnosis of acute, nonseptic PE and excluded patients with ischemic stroke, ST elevation myocardial infarction (STEMI), or acute limb ischemia to avoid including patients who received thrombolysis for other indications. We used International Classification of Diseases, Tenth Revision, Procedure Coding System (ICD-10-PCS) codes to identify patients who received systemic thrombolysis or CDT (including isolated CDT and ultrasound-assisted CDT) as previously described. 4 We used ICD-10 Clinical Modification (ICD-10-CM) codes to determine outcomes, including inpatient mortality and bleeding complications. 4 To evaluate semiannual trends (linear on the log-odds scale), logistic regression was used. All statistical analyses were performed by using the weighted values of observations as provided by the NIS to estimate national statistics. 5 The Institutional Review Board at Beth Israel Deaconess Medical Center evaluated this study and deemed it not to qualify as human subjects research.
Among 1,046,410 hospitalizations for acute PE from January 2016 through December 2018, thrombolysis was utilized in 46,675 patients (4.5%). Systemic thrombolysis was used more often than CDT (25,755 [2.5%] vs 20,920 [2.0%], p < 0.001). However, the utilization of CDT increased from 1.4% of all admissions for acute PE in the first half of 2016 to 2.5% in the second half of 2018 (Ptrend < 0.001). During the same period, the use of systemic thrombolysis increased from 2.3% to 2.7% (Ptrend = 0.002) (Figure 1A).
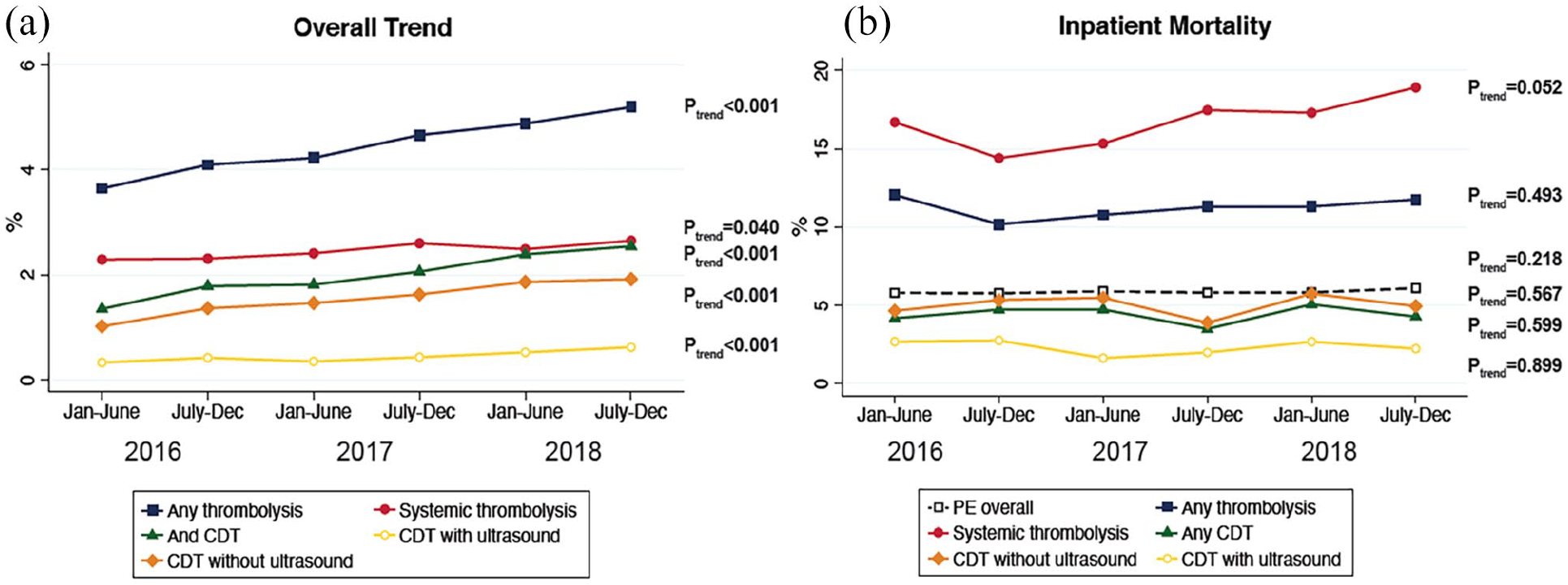
National trends in thrombolytic treatment strategies. (A) Trend in thrombolysis utlization from January 2016 through December 2018 in 6-month intervals. (B) Inpatient mortality from January 2016 through December 2018 in 6-month intervals based on therapy received.
During the study period, the utilization of CDT increased in patients with hypotension (January to July 2016: 1.9% vs July to December 2018: 3.8%, Ptrend = 0.002) or shock (3.2% vs 6.7%, Ptrend < 0.001), but not in patients with cardiac arrest (Ptrend = 0.745) (online supplemental Figure). There was a similar increase in the utilization of CDT over time across all hospital bed sizes and hospital ownerships. In addition, CDT use increased in both teaching hospitals and nonteaching hospitals (Ptrend < 0.001 for all).
There was no change in inpatient mortality in the overall PE population or in patients who received thrombolytic therapy (Figure 1B). There was a temporal increase in the overall rates of any bleeding event (January to July 2016: 10.2% vs July to December 2018: 11.2%, Ptrend < 0.001) and intracranial hemorrhage (0.8% vs 0.9%, Ptrend = 0.009) in the total PE population. However, the frequency of any bleeding event after systemic thrombolysis (13.6% vs 14.4%, Ptrend = 0.654) and CDT (7.9% vs 11.7%, Ptrend = 0.114) remained unchanged. Similarly, the rates of mechanical ventilation and vasopressor use after thrombolysis were unchanged (Ptrend all > 0.05).
The administration of thrombolysis for acute PE increased from 2016 through 2018, primarily driven by a 60% increase in the utilization of CDT. This increase occurred despite a dearth of high-quality data comparing CDT to anticoagulation or systemic thrombolysis. CDT is increasingly being used in patients with hypotension and shock, although these patients only represented 21% of patients receiving catheter-directed, ultrasound-facilitated thrombolysis in the 149-patient, single-armed SEATTLE II trial. 3 Despite the increased use of CDT, there was no change in overall mortality. The increased use of CDT may have been associated with a small increase in bleeding events in all patients with PE seen over the analyzed timeframe, though such a finding is difficult to confirm with this analysis given the limitations of observational, claims-based data. Further randomized studies, such as the planned HI-PEITHO trial and the in-planning PE-TRACT trial, are necessary to determine whether CDT improves short-term (e.g., clinical deterioration) or long-term (e.g., exercise capacity) clinical outcomes to justify its increasing utilization.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X211048544 – Supplemental material for National trends in utilization of thrombolytic therapy for acute pulmonary embolism
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X211048544 for National trends in utilization of thrombolytic therapy for acute pulmonary embolism by Sebastian E Beyer, Colby Shanafelt, Duane S Pinto, Jeffrey L Weinstein, Jay Giri, Akhilesh K Sista, Robert W Yeh, Eric A Secemsky and Brett J Carroll in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Giri has received research support to the institution from St Jude Medical and Recor Medical. He serves on the scientific advisory boards for Astra Zeneca and Philips Medical. Dr Sista has received research support to the NYU department of radiology from Penumbra, Inc. Dr Pinto serves on scientific advisory boards and is a consultant for Abbott Vascular, Boston Scientific, Abiomed, and Medtronic. He is a consultant for Opsens, NuPulseCV, and Teleflex. Dr Yeh reports investigator-initiated research grants from Abbott Vascular, Boston Scientific, and Medtronic, serves on the scientific advisory boards for Abbott Vascular, Boston Scientific, and Medtronic. The remaining authors have no disclosures.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
