Abstract
This study retrospectively compared the outcomes of patients who received ultrasound facilitated catheter-directed thrombolysis (UFCDT) versus systemically administered ‘half-dose’ thrombolysis (HDT) in 97 patients with PE. The outcomes assessed included changes in baseline pulmonary artery systolic pressure (PASP), right ventricle/left ventricle ratio (RV/LV), cost and duration of hospitalization, death, bleeding, and recurrent venous thromboembolism in the short and intermediate term follow-up. Analyses were performed using a covariance adjustment propensity score approach to address baseline differences between groups in variables associated with PASP and RV/LV, covarying baseline scores. The baseline mean ± SE PASP dropped from 49.3 ± 1.1 to 32.5 ± 0.3 mmHg at 36 hours in the HDT group, and from 50.6 ± 1.2 to 35.1 ± 0.4 mmHg in the UFCDT group; group × time interaction p-value = 0.007. Corresponding drops in the RV/LV were from a baseline of 1.26 ± 0.05 to 1.07 ± 0.01 in the HDT group and from 1.30 ± 0.05 to 1.14 ± 0.01 in the UFCDT group at 36 hours; group × time interaction p-value = 0.269. Statistically significant decreases were noted in PASP and RV/LV for both the HDT and UFCDT at 36 hours and follow-up. PASP through follow-up was significantly lower in the HDT than the UFCDT group. Likewise, RV/LV was lower in the HDT group. The duration and cost of hospitalization were lower in the HDT group (6.2 ± 1.4 days vs 1.9 ± 0.3 days, p < 0.001; US$12,000 ± $3000 vs $74,000 ± $6000, p < 0.001). We conclude that both UFCDT and HDT lead to rapid reduction of PASP and RV/LV, whereas HDT leads to a lower duration and cost of hospitalization.
Keywords
Introduction
Standard or full-dose systemic thrombolysis has been recommended for massive pulmonary embolism (PE) or submassive PE with markers of myocardial injury.1,2 There has been resistance in the use of thrombolysis among practitioners for fear of intracranial hemorrhage (ICH), which has been around 2–3% in PE. 3 The focus of many studies has been to accept this complication rate and identify subgroups of patients in whom the benefits of thrombolysis would outweigh the risks. 4 It has been recently shown that patients with both massive and submassive PE respond favorably to peripherally administered low-dose thrombolysis with equal efficacy to standard thrombolysis while eliminating or reducing bleeding, including ICH.5–9
In a small, single-center study, no bleeding occurred when using systemically administered half-dose thrombolysis (HDT) in conjunction with a reduction of parenteral anticoagulation. 5 Ultrasound facilitated catheter-directed thrombolysis (UFCDT) with low-dose tissue plasminogen activator (tPA) has led to very low ICH but a major bleeding rate of as high as 10–11%.10,11 This modality has been proven effective in the lungs as well as in other vascular beds. As compared to HDT, UFCDT uses less tPA, yet is labor intensive and requires expensive catheters. In up to 87% of the cases reported thus far, two catheters had to be used: one in each lung.10,11 From both clinical effectiveness and a cost-value standpoint, one may question the true necessity for placement of an infusion catheter in the lungs as the lungs are the only organs in which all venous flow converges. They receive the entire cardiac output. Thus, from a pathophysiological standpoint, the potential benefit of delivery of the thrombolytic agent via a catheter versus through the patient’s veins has not been proven. The role of ultrasound to contribute to an additional clinical improvement by fragmentation of fibrin strands above and beyond the effect of thrombolysis is also questionable. Consequently, we hypothesized that HDT will be comparable to UFCDT. The current study, therefore, sought to compare the safety and efficacy outcomes of patients with massive and submassive PE who were treated with HDT versus UFCDT.
Methods
Study design
This study was a retrospective analysis of 97 patients who underwent UFCDT or HDT for massive and submassive PE. From April 2013 through April 2016, 47 patients with PE were treated with UFCDT using the EKOS catheter (EKOS, Bothel, WA, USA). They were treated at two facilities of the Banner Health System in Mesa, Arizona by seven operators. The operators were all interventional cardiologists. These patients were compared to the first 50 patients who were matched for sex and age from a pool of 256 patients who were treated with HDT during the same period. Consecutive patients from the pool of HDT patients who were the same sex and within 5 years of age of the UFCDT patients were chosen after review of their records. The study was approved by the Banner Health Institutional Review Board. Written informed consent was not required due to the retrospective nature of the study.
Study end points
Notwithstanding the retrospective nature of the study, safety and efficacy end points were defined a priori. Safety outcomes consisted of death, major bleeding, recurrence of venous thromboembolism (VTE), and transfusion of packed red blood cells as a result of bleeding. These outcomes were measured at 30 days and at follow-up. For the HDT group, the follow-up was prescheduled whereas for the UFCDT group electronic medical records were utilized to obtain the information. Prespecified data were utilized to collect follow-up information. There was no loss of follow-up in the HDT group. Clinical data were incomplete in eight UFCDT patients. These patients received a questionnaire via US mail and all responded, thereby completing the follow-up process. The administrative staff at Arizona Cardiovascular Consultants completed the data collection forms. The GUSTO (Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries) trial bleeding criteria were used to define bleeding. Bleeding was said to be present if there was GUSTO severe/life-threatening or moderate bleeding. Severe/life-threatening bleeding was present if there was either intracranial hemorrhage or bleeding that caused hemodynamic compromise and required intervention. Moderate bleeding was defined as any bleeding requiring blood transfusion but without hemodynamic compromise.
Efficacy outcomes consisted of changes in the right ventricle/left ventricle ratio (RV/LV) and in PA Systolic Pressure (PASP) by echocardiography within 36 hours after completion of thrombolysis and at follow-up. The range of subsequent echocardiography was 10–36 hours. No patient (except the two who died) was excluded from analysis. The clinical outcomes were reviewed and discussed among the investigators. A consensus decision was required to adjudicate the results. There was no imaging core laboratory. The measurements in the imaging reports were accepted as accurate. If an outcome parameter in a study was missing, that study was retrieved and the said parameter measured by the cardiologist of the group. There was no blinding process.
An effort was made to evaluate utilization outcomes as well. These consisted of costs related to PE, the duration of intensive care unit (ICU) admission, and hospitalization.
Massive and submassive PE was defined according to the American Heart Association definitions. 2 In brief, massive PE was defined as presence of syncope, systemic arterial hypotension, cardiogenic shock, or resuscitated cardiac arrest. Submassive PE was defined as evidence of RV dysfunction in the presence of a normotensive status.
Previous VTE was defined as objectively documented PE or deep vein thrombosis (DVT) within the preceding 20 years but excluding the current presentation. Active cancer was defined as presence of any cancer for which the patient was receiving or had declined to receive treatment at the time of presentation. Thrombophilic state was presence of any known or newly identified hypercoagulable condition (protein C or S deficiency, factor V Leiden, prothrombin gene 20210, antiphospholipid antibodies). D-dimer and troponin I were considered elevated if they were over three times the upper limits of normal values. Diagnosis of comorbid conditions was based on having the prespecified diseases on admission. Heart disease included patients with coronary artery or valvular disease of at least moderate intensity. Lung disease consisted of those with restrictive or obstructive disease who were receiving treatment. Renal disease was considered to be present if the creatinine clearance was < 50 mL/min or the serum creatinine was over 1.7 g/dL.
Patients in the UFCDT group received treatment within 6 hours to 4 days after admission. The procedures were usually performed during regular workdays and rarely on holidays and after hours. In the HDT group, treatment was given 0.5 ± 0.3 hours after admission or consultation request. Echocardiography was performed on admission, within 36 hours, and between 8 and 12 months after the index hospitalization. Diagnosis of PE was made by computerized tomography angiography of the pulmonary arteries in 96% of the patients and by high probability ventilation-perfusion scan in the remainder. In the UFCDT group, access was obtained through the right common femoral or internal jugular vein. A 5 or 6 French sheath was inserted. The pulmonary artery was engaged using a pigtail catheter and pulmonary angiography performed in at least two planes. Although during this time PASPs were obtained, echocardiographic measurements were used for the calculation of pressures. The site of thrombus was identified and the EKOS catheter was placed within that area. The infusion zones of catheters used were 6–12 cm in length
All patients were admitted to the ICU. The catheters were removed 15–26 hours after insertion.
Thrombolytic regimen
In both groups, tPA was the only thrombolytic agent utilized. In the UFCDT group, 42 patients (89%) received two catheters with a tPA dose given at 24 ± 2 mg in 15 hours. In the 11% who had one catheter, the dose was 22 ± 1 mg given over 24 hours. In the HDT group, patients received the low-dose tPA protocol previously reported. 5 In brief, 10 mg of tPA was given over 1 minute, followed by 40 mg in 2 hours, through a peripheral venous access site. A modified dose of heparin was utilized in this group as described below.
Anticoagulation regimen
In the UFCDT group, the anticoagulation was variable. On admission, full-dose heparin was given to all patients. The dose consisted of 80 units of unfractionated heparin/kg per hour followed by 18 units/kg as maintenance dose. A total of 40 patients (85%) received enoxaparin following tPA administration and before oral anticoagulation was initiated for 1–5 days. During UFCDT, no heparin was given. In the UFCDT group, warfarin was used as the oral anticoagulant in 15 patients (32%), rivaroxaban in 14 (30%), and apixaban in 18 (38%) patients. Full loading and maintenance doses of a direct oral anticoagulant (DOAC) were given to UFCDT group.
In the HDT group, heparin was the only parenteral anticoagulant. It was given for 24 hours only and subsequently discontinued. The loading dose was at 70 mg/kg and did not exceed 6000 units. The maintenance dose was started at 8–10 units/kg per hour. Partial thromboplastin time (PTT) was maintained between 50 and 80 seconds. No heparin bolus was given for lower PTT values. Approximately 15 minutes after termination of heparin, the maintenance dose of a DOAC was initiated. Apixaban was given to 27 patients (54%), rivaroxaban to 16 (32%) and edoxaban to 7 patients (14%). The mean follow-up was 38 ± 4 months.
Cost analysis
A simple analysis of cost was made by computing the total charges which were presumed to be directly related to the treatment of PE. The itemized bill generated for the duration of hospitalization for each patient was reviewed. The charges related directly to PE were identified and their cumulative sum computed. If the patient’s admission was solely due to PE, the entire hospital charge was attributed to PE. If PE was incidental to other reasons, only those charges related to PE were considered. These included the charges made for tPA, catheters, anticoagulants, ICU and general floor admission, and imaging studies. The results were rounded to the nearest $1000. All values related to cost are expressed in US Dollars.
Statistical analysis
Summary statistics are reported as means (SDs) for continuous variables and counts (percentages) for categorical variables. Baseline differences between groups were evaluated using independent-samples t-tests or Mann–Whitney tests for continuous variables, and chi-squared or Fisher’s exact tests for categorical variables, as appropriate. Outcome analyses were performed using a regression (covariance) adjustment propensity score approach to address baseline differences between groups in variables associated with PASP and RV/LV under a generalized estimating equations model, covarying baseline scores. Assuming no unmeasured confounders, conditioning on the propensity score allows an unbiased estimate of the average treatment effect at that value of the propensity score. Changes in PASP and RV/LV over time were evaluated using a generalized estimating equations approach with autoregressive (AR1) working correlation matrices. The interaction between group and time, as well as the main effects of both group and time were evaluated. Bonferroni-corrected pairwise comparisons were used to localize differences. Results of both the propensity-adjusted analysis and the unadjusted analysis are provided. A p-value of < 0.05 was considered statistically significant. Statistical analyses were performed with SPSS, Version 24 (IBM Corp., Armonk, NY, USA).
Results
The patients’ demographic and clinical characteristics by group are shown in Table 1. Table 2 provides the results of the propensity-adjusted analysis. The c-statistic for this propensity score was 0.84, indicating good ability of the propensity score model to predict treatment status. For PASP, while controlling for baseline scores, the interaction between group and time was statistically significant, p = 0.007. PASP was lower in the HDT group than in the UFCDT group, both at 36 hours (p < 0.001) and at follow-up (p = 0.003). For RV/LV, controlling for baseline scores, the interaction between group and time was not statistically significant, p = 0.269. RV/LV was lower in the HDT group than in the UFCDT group, at both 36 hours (p < 0.001) and follow-up (p < 0.001). Results of the unadjusted analysis are shown in Table 3 for comparison.
Clinical characteristics of study groups.
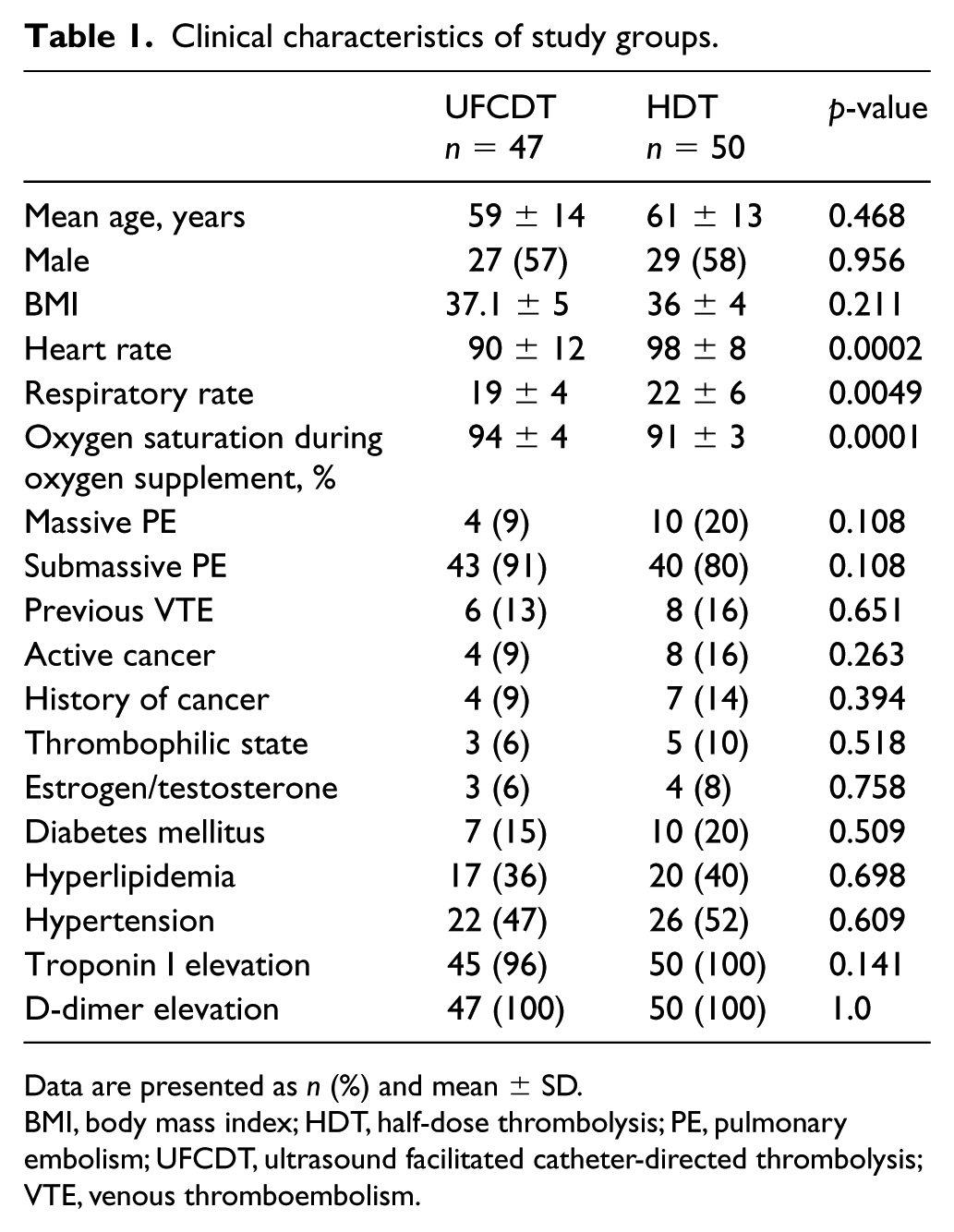
Data are presented as n (%) and mean ± SD.
BMI, body mass index; HDT, half-dose thrombolysis; PE, pulmonary embolism; UFCDT, ultrasound facilitated catheter-directed thrombolysis; VTE, venous thromboembolism.
Propensity-adjusted values for PASP and RV/LV by group over time.
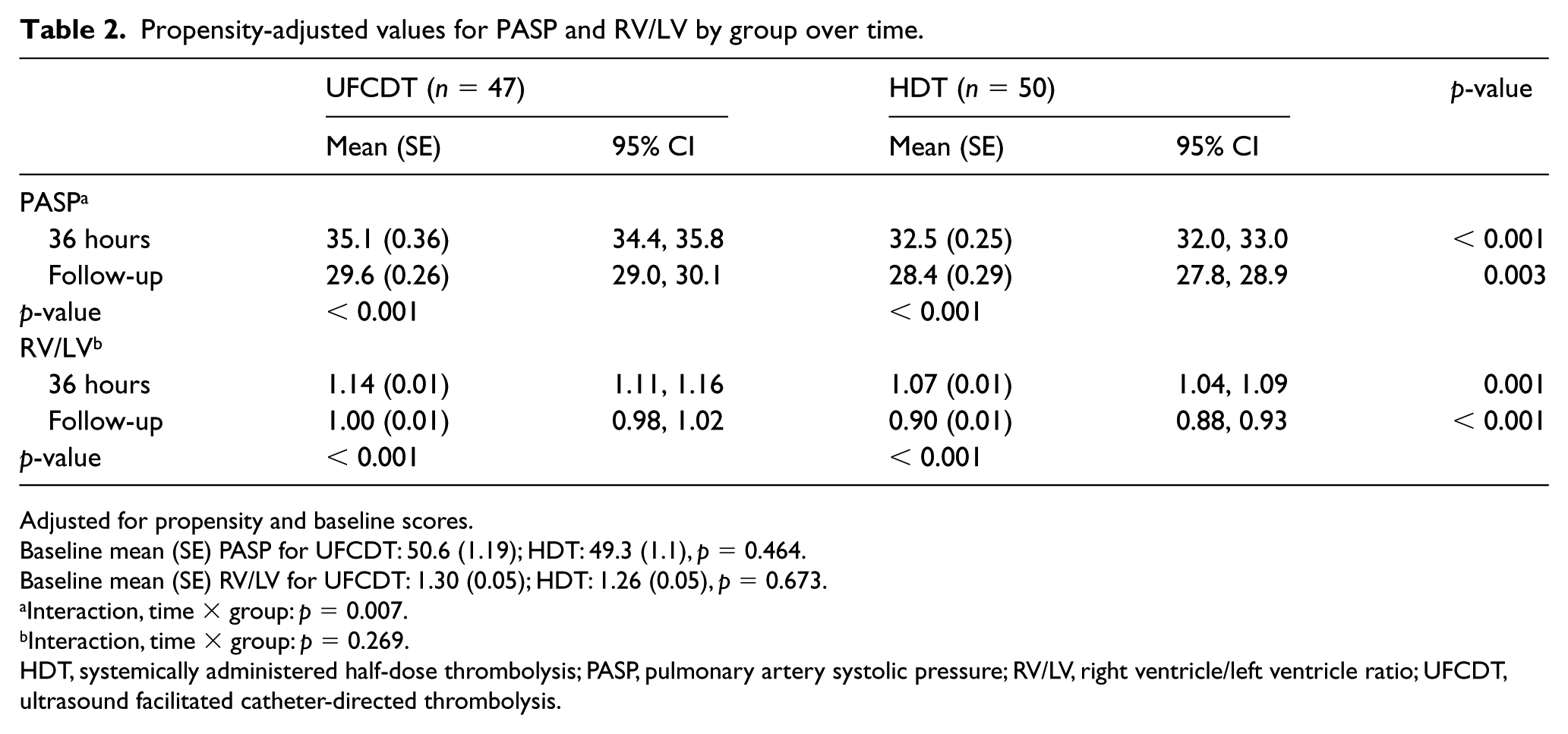
Adjusted for propensity and baseline scores.
Baseline mean (SE) PASP for UFCDT: 50.6 (1.19); HDT: 49.3 (1.1), p = 0.464.
Baseline mean (SE) RV/LV for UFCDT: 1.30 (0.05); HDT: 1.26 (0.05), p = 0.673.
Interaction, time × group: p = 0.007.
Interaction, time × group: p = 0.269.
HDT, systemically administered half-dose thrombolysis; PASP, pulmonary artery systolic pressure; RV/LV, right ventricle/left ventricle ratio; UFCDT, ultrasound facilitated catheter-directed thrombolysis.
Unadjusted values for PASP and RV/LV by group over time.
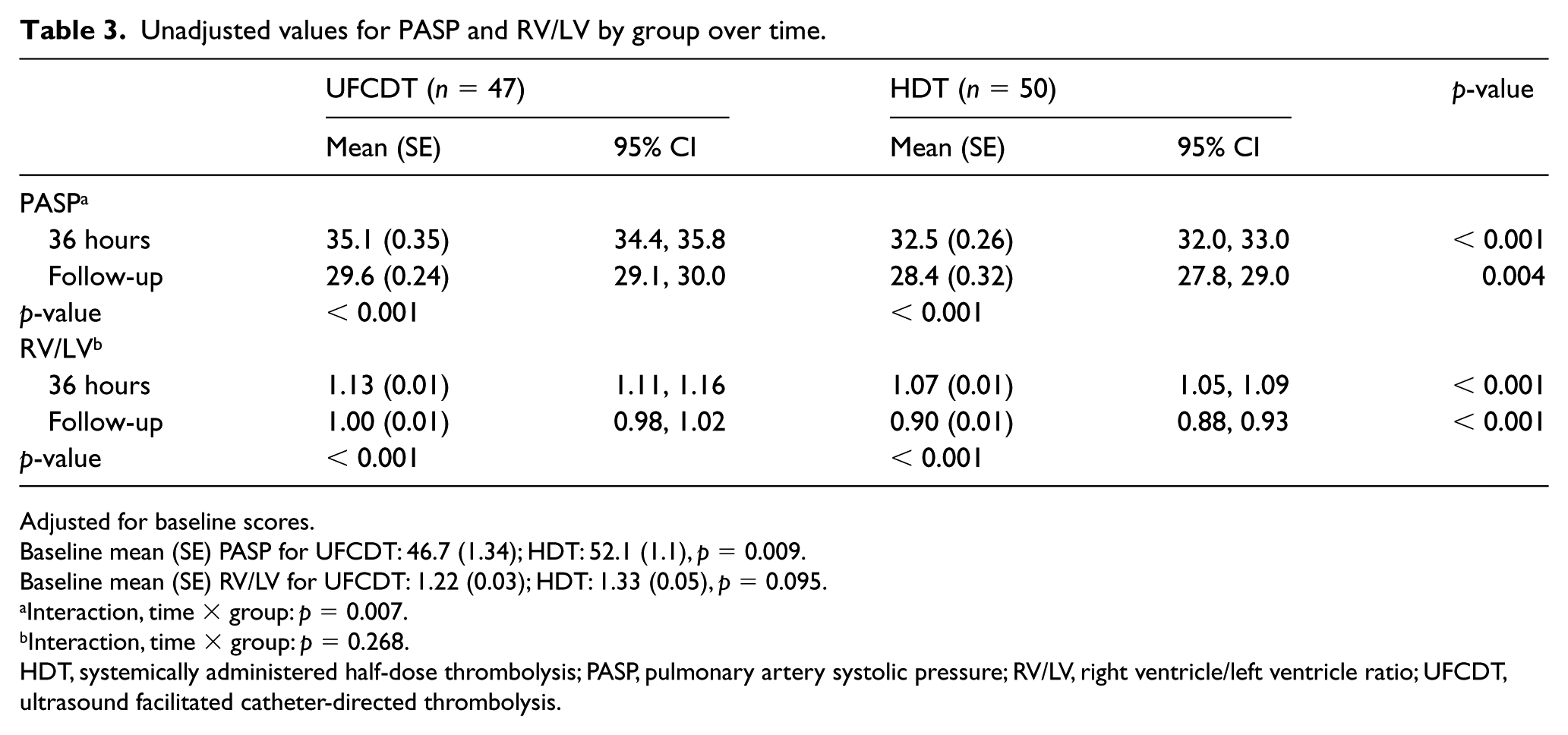
Adjusted for baseline scores.
Baseline mean (SE) PASP for UFCDT: 46.7 (1.34); HDT: 52.1 (1.1), p = 0.009.
Baseline mean (SE) RV/LV for UFCDT: 1.22 (0.03); HDT: 1.33 (0.05), p = 0.095.
Interaction, time × group: p = 0.007.
Interaction, time × group: p = 0.268.
HDT, systemically administered half-dose thrombolysis; PASP, pulmonary artery systolic pressure; RV/LV, right ventricle/left ventricle ratio; UFCDT, ultrasound facilitated catheter-directed thrombolysis.
All patients in the UFCDT group required ICU admission whereas only 12 patients (24%) from the HDT group were admitted to the ICU. The duration of hospitalization was 1.9 ± 0.3 days in the HDT group versus 6.2 ± 1.4 in the UFCDT group. Two people in the UFCDT group died due to procedure-related complications. One death was due to bleeding after discharge and at home, which had resulted from unrecognized exsanguination from the access site, and the other death was during the procedure. Hematoma developed in two other patients in the UFCDT group. Transfusion was required in three patients in the UFCDT group and no patients in the HDT group. There were no bleeding complications in the HDT group. At follow-up, 75% of the HDT group and 68% of the UFCDT group were still on anticoagulation. There were no VTE-related deaths at follow-up in either group.
The outcome of adverse events within the first 30 days is shown in Table 4. For each individual component there was no significant difference between the two groups.
Safety outcomes at 30 days, drug dose and hospitalization duration between the two groups.
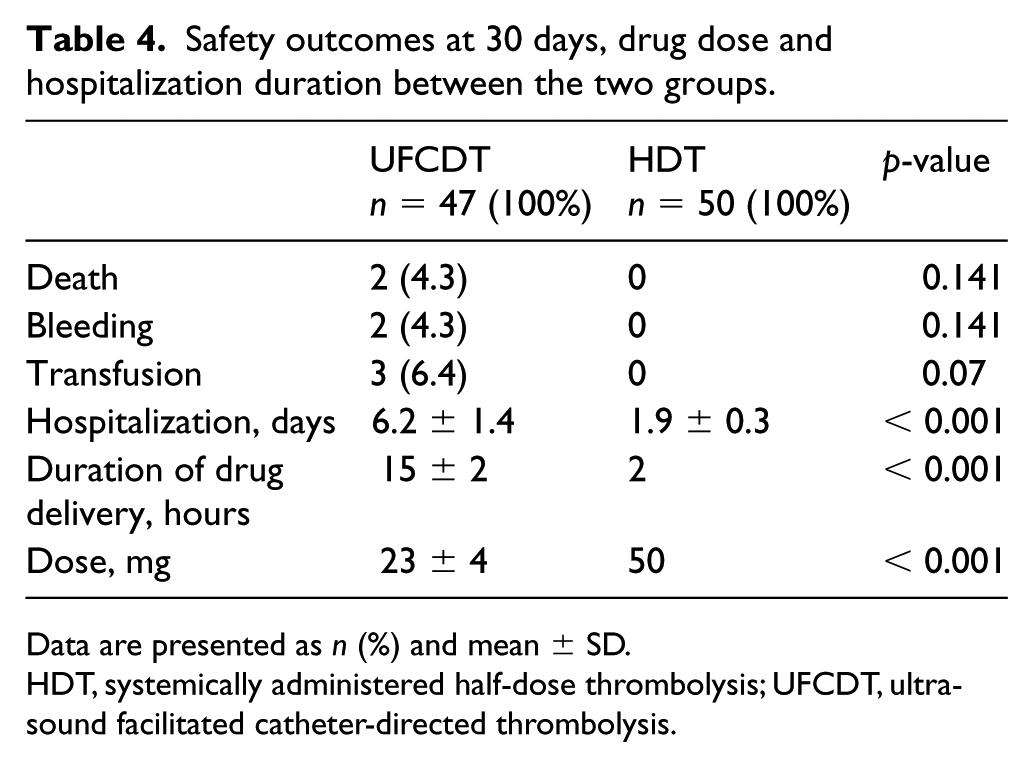
Data are presented as n (%) and mean ± SD.
HDT, systemically administered half-dose thrombolysis; UFCDT, ultrasound facilitated catheter-directed thrombolysis.
The mean dose of tPA infusion was 23 ± 4 mg in the UFCDT group and 50 mg in the HDT group. The average duration of infusion in the UFCDT group was 15 ± 2 hours and 2 hours in the HDT group. Two patients in the UFCDT group versus one patient in the HDT group had recurrent PE at follow-up. They had been all off anticoagulation. Two patients received repeat HDT.
Using the simple approach described in the ‘Cost analysis’ section, the mean charges for treatment in the HDT group (after adjustment for insurance plans) were $12,000 ± $3000 and in the UFCDT group were $74,000 ± $6000. In most patients, this difference was driven by the ICU stay and cost of the catheters. The breakdown of charges included the following: ICU bed = $7200/night; telemetry bed = $3200/night; imaging = $3100; laboratory = $800; tPA = $2500; UFCDT catheter = $8000/each; miscellaneous = $3000. The total charges were approximately twice as much as adjusted charges. The duration of hospitalization was significantly less in the HDT group post-thrombolysis: parenteral anticoagulation was limited to only 24 hours with subsequent switching to a DOAC, early ambulation, and fewer ICU admissions.
Discussion
The results indicate that both UFCDT and HDT lead to a substantial reduction in PASP and RV/LV within a short period of time. This reduction is relatively rapid and the benefits persist into the intermediate follow-up. This finding is consistent with previous literature.5–11 Lungs are exquisitely sensitive to thrombolysis. Even markedly lower doses of tPA, in the order of 0.5–2 mg, have been shown to alleviate symptoms and improve hemodynamics in acute pulmonary emboli. 12 Such efficacy is based on the simple physiologic principle that the lungs receive the entire cardiac output, allowing all of the thrombolytic agent to enter and re-enter the lungs causing multiple ‘hits’ and in multiple cycles for as long as the molecules of the thrombolytic agent are present and active.
The individual end points of death, bleeding, and transfusion were not different between the two groups. The sample size was not enough to evaluate for these individual end points.
In the SEATTLE II study, major bleeding occurred in 10% of the patients who had received UFCDT for massive and submassive PE. 10 In the ULTIMA trial, no significantly increased bleeding rate occurred between patients receiving anticoagulation alone versus UFCDT. 11 In our study, bleeding complications occurred only in the UFCDT group and none was seen in the HDT group. However, this reduction was not statistically significant. It may have been due to modification of the heparin dosing in the HDT group. Failure to reduce the dose of concomitant anticoagulation can potentially increase the risk of bleeding. Almost all studies that have reported relatively high bleeding rates had uniformly added thrombolysis to standard dose anticoagulation regimens.3,4,10,11 It stands to reason that when thrombolysis is added to a standard anticoagulation regimen, the risk increases. To reduce the risk, the dose of one or both treatment components should be reduced. 5 The frequency of bleeding was reduced in the OPTALYSE PE Trial by reduction of the tPA dose. 13 Follow-up analysis of risk factors leading to bleeding was identified as massive PE and recurrent venous puncture attempts. 14
Whether or not UFCDT is superior to thrombolysis without ultrasound facilitation is a debatable issue. There are limited data which have compared the two modalities. Most have been retrospective analyses and, given lack of power, variations in methodology, and limited study population, no definitive conclusion can be derived on the beneficial role of ultrasound.15–18
In one study, a statistically significant increase in overall and ICU length of stay was noted in patients receiving UFCDT without additional benefit as compared to patients who had received passive catheter-directed thrombolysis. 16
A randomized controlled trial investigated the effectiveness of ultrasound energy in resolving thrombosis in iliofemoral DVT. There was no statistically significant difference in the efficacy and safety end points between patients receiving UFCDT versus passive catheter-directed thrombolysis. 17 However, pulmonary arteries are distinctly different from the peripheral venous bed with respect to secretion of venoactive substances, and hence direct comparison between the two modalities could not be made.
Another study found no advantage of UFCDT in terms of efficacy and safety outcomes versus passive catheter-directed thrombolysis. Fluoroscopy and procedure times were significantly higher in the UFCDT arm. 18
A recent retrospective analysis of 699 PE patients treated with HDT was compared with 3069 patients receiving full-dose thrombolysis. 19 All patients were in the ICU before thrombolysis was administered. Without propensity risk adjustment, in-hospital mortality was 21% in the full-dose group compared with 12% in the HDT group (p < 0.01). With propensity scoring, mortality and rates of major bleeding were found to be similar. However, treatment escalation was required more frequently (53.8% vs 41.4%; p < 0.01) in the HDT group. The patients in both groups received thrombolysis within up to 2 days following admission to the ICU. 19 Earlier administration of thrombolysis could have improved the outcome. There was no indication that the patients in the HDT group received a lower concomitant dose of heparin, as we used in our study. This difference may have accounted for the absence of a significant reduction in the rate of bleeding in the HDT group. Furthermore, how and in what time period the 50 mg dose of tPA was administered was unclear.
In our study, the cost of treatment was approximately four times higher in the UFCDT group. This was chiefly driven by the cost of the catheters, and ICU admission. The use of warfarin in 31% of the UFCDT group and keeping the patients hospitalized to achieve a therapeutic international normalized ratio (INR), were additional contributing factors.
The duration of hospitalization in the HDT group was less than one-third of that of the UFCDT group. In the HDT group, DOACs were uniformly used and the duration of parenteral anticoagulation was limited to 24 hours. This approach led to early transition to oral anticoagulation and hence early discharge.
At follow-up, indefinite anticoagulation was maintained in 75% of the HDT group versus 69% of the UFCDT group, accounting for the low recurrence rate of VTE in both groups.
The frequency of massive PE was higher in the HDT group as the treating physicians treated both massive and submassive PE in the same fashion, whereas in the UFCDT group the operators preferred more stable patients to perform intervention on. This created a bias in favor of the UFCDT group. With propensity score adjustment, baseline PASPs, but not the RV/LVs, were found to be significantly higher in the HDT group. Additionally, the reduction in PASP and RV/LV with time was found to be significantly lower in the HDT group.
Limitations of the study
This study was retrospective and consequently did not have the rigor of a prospective randomized trial. There was a far larger variation in the anticoagulation regimen and use of new oral anticoagulants in the UFCDT group. Additionally, the time to administration of thrombolytic therapy was substantially longer in that group. Therefore, some of the beneficial effects in the HDT group may have been driven by these differences alone, notwithstanding adjustment by propensity scoring. There were more operators with variable expertise in the UFCDT group. Similarly, the choice and duration of anticoagulation were variable and at times not optimal. Close to one-third of the patients received warfarin, which according to current guidelines is regarded as a second-tier therapy for VTE. The major reason for this choice was driven by the misguided belief that, since reversal agents were available for warfarin (and only for one of the four available DOACs), the former was ‘safer’. In the HDT group, however, there was no such variation, as the same protocol was applied to every patient and all patients received a DOAC. Adherence to the HDT protocol, as opposed to no definitive protocol in the UFCDT group, may have altered the outcomes in favor of the HDT group.
The cost analysis provided was simple and could have missed other costs associated with PE treatment. The analysis was based on our review of itemized hospital charges of what we believed to be charges definitively related to PE. It was therefore an evaluation of the charges rather than the costs. It did not show the actual negotiated rates or real-world values. We may have missed other associated charges and hence the values computed are probably an underestimate of the true charges. Nonetheless, the same approach was applied to both groups and provided some insight as to cost differences consistent with predictable expectations. These values, however, should be interpreted with caution given the limitations noted above.
Conclusion
The results indicate that both UFCDT and HDT lead to rapid reduction of PASP and RV/LV. The individual components of death, bleeding, and transfusion at 30 days were not different between the groups. The cost of treatment, duration of hospitalization, and ICU stay were significantly lower in the HDT group. However, these results are preliminary and derived from a relatively small study population. Ideally, randomized prospective trials with adequate power and a larger number of patients would be required to confirm our findings. If so confirmed, at a national level, the simplicity of HDT can be protocolized and incorporated in any community hospital, thereby allowing for widespread adoption. This is in contrast to UFCDT, which is labor intensive, requires an advanced infrastructure, and usually requires centers with catheterization laboratory capabilities.
Footnotes
Acknowledgements
The authors would like to acknowledge the assistance of Jenny Maxson and Robert Flynn for the administrative work related to the approval of the study protocol.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
