Abstract

Keywords
The central premise, and promise, of anticoagulation clinics is that they can provide safer and more reliable therapeutic anticoagulation through protocolized drug monitoring and dosing, and thereby minimize the risk of both de novo thromboembolic and hemorrhagic events. Anticoagulated patients are particularly at risk for gastrointestinal (GI) hemorrhage. It is well known that anticoagulants, whether warfarin or direct oral anticoagulants, nearly double the risk of GI hemorrhage. 1 In addition, these patients are often elderly and/or treated with antiplatelet therapy (APT), multiplying hemorrhage risk even further.1,2 Two simple strategies could potentially reduce anticoagulated patients’ risk of upper GI hemorrhage: (1) deprescribing of unnecessary APT 3 and (2) prescribing proton pump inhibitors (PPI gastroprotection) to patients at high risk for upper GI hemorrhage, a practice which reduces the odds of this adverse event by nearly three-quarters and is supported by a multi-society expert panel. 4 In this study, we sought to estimate the proportion of patients referred to a network of anticoagulation clinics who might benefit from either of these practices.
We performed a retrospective analysis of patients newly referred to any of six anticoagulation clinics throughout the state of Michigan that participate in a Blue Cross Blue Shield-sponsored quality improvement consortium, the Michigan Anticoagulation Quality Improvement Initiative (MAQI 2 ), between January 2011 and May 2018. The clinics do not routinely discontinue APT or systematically address the use of PPI gastroprotection. Clinic staff obtain a structured medical and medication history when patients enroll in clinic, which is subsequently abstracted into a registry. For all index visits, we extracted data on demographics, comorbidities, indications for anticoagulation, and other risk factors for upper GI hemorrhage, including APT use, glucocorticoids, non-steroidal anti-inflammatory drugs (NSAIDs), and COX-2 inhibitors. We examined prescription of PPIs in ‘high-risk’ patients according to the 2008 multi-society recommendations (concomitant warfarin + APT) and in standard-risk patients (all other patients). As a sensitivity analysis, we also examined rates of H2 blocker prescription, although this drug class provides inferior gastroprotection and is not recommended. Dosage and frequency of APT and PPI medications were not available in the database. We counted the following as appropriate indications for APT: history of coronary artery disease (CAD) or myocardial infarction (MI), peripheral artery disease, percutaneous intervention, coronary artery bypass graft, cerebrovascular accident or transient ischemic attack, or antiphospholipid antibody syndrome.
During the study period, 6907 patients initiated warfarin therapy and enrolled in the MAQI 2 registry. The mean age of patients was 65.6 years (SD 15.3), 51.6% were male, 81.0% were Caucasian, 15.3% were African American, and 3.6% reported another race. The two most common indications for anticoagulation were stroke prevention in atrial fibrillation (46.0%) and venous thromboembolism (40.7%). In addition to warfarin, 39.2% of patients used aspirin alone, 2.0% used a thienopyridine alone, and 3.8% used dual-APT. In total, 45.0% of patients were considered high risk for upper GI hemorrhage based on combined use of warfarin and APT (Figure 1). APT was the most prevalent risk factor for upper GI hemorrhage, followed by oral glucocorticoid prescription (8.3%), NSAIDs (3.5%), and COX-2 inhibitors (0.5%). Characteristics of the study sample can be found in the online Supplement 1. PPIs were prescribed to 31.7% of patients overall, and 34.0% of high-risk patients. Considering H2 blockers, in addition to PPIs, 41.0% of high-risk patients were on a gastroprotective medication. Among the 45.0% of patients treated with APT, there was no identifiable indication for APT in 44.6% of them (or 20.0% of the entire patient sample). In all, 36.0% of the entire sample could benefit from one or both of the two strategies to minimize the risk of upper GI hemorrhage.
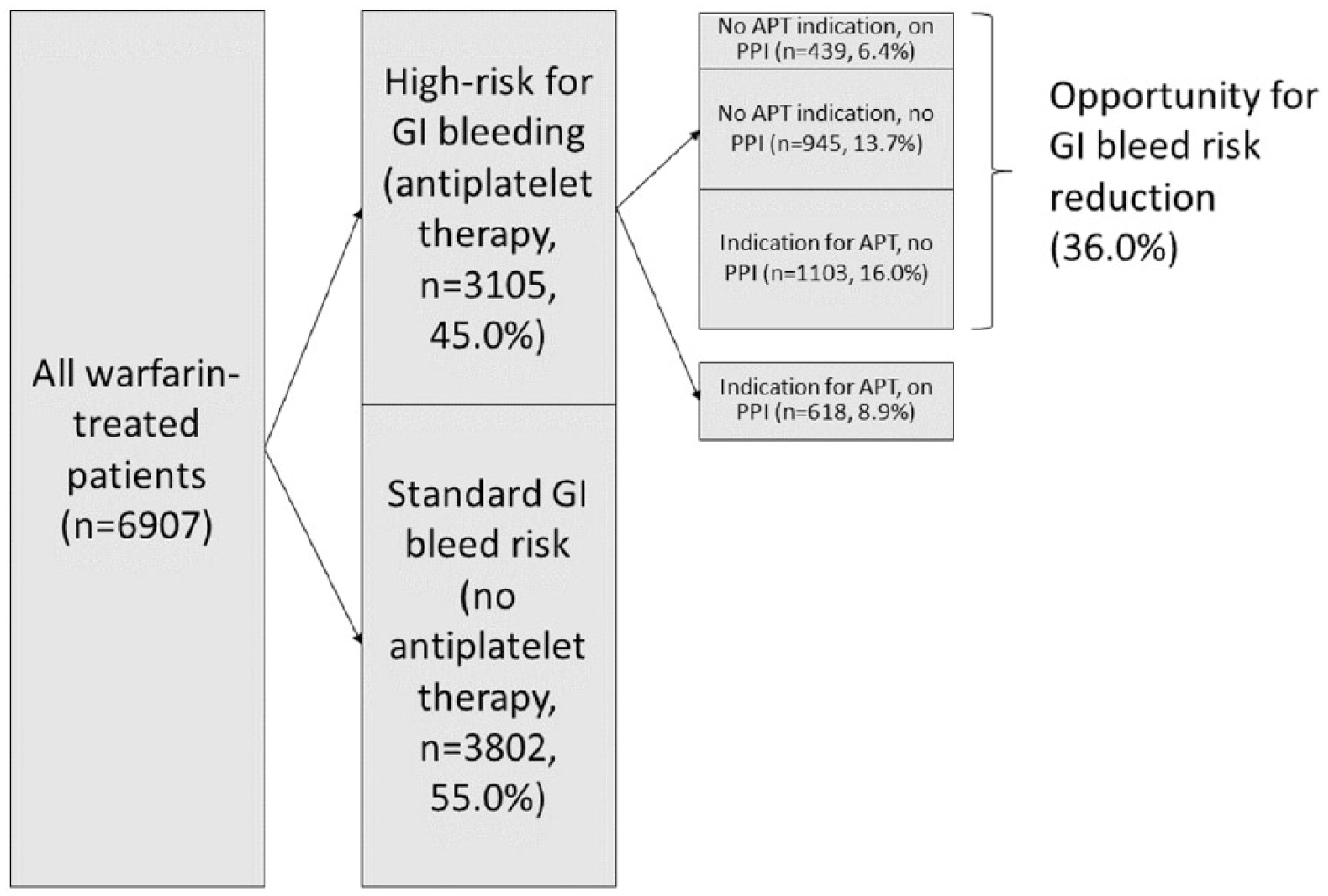
Prevalence of patients at high risk for upper GI hemorrhage, and of patients with opportunities for GI hemorrhage risk reduction based on unnecessary APT or failure to use appropriate PPI gastroprotection.
In conclusion, our findings show that almost half of patients followed in a network of anticoagulation clinics are treated with APT, conferring a high risk of upper GI hemorrhage. Among these high-risk patients, we found this risk could be greatly reduced in 80% by either discontinuing the APT when no identifiable indication exists, or by initiating PPI gastroprotection. In the past, anticoagulation clinics have generally restricted their activities to laboratory monitoring and warfarin dose adjustment. However, they are well positioned and well advised to play a more proactive and holistic role, including helping patients minimize the risk of serious GI hemorrhage, 5 which can often be fatal or disabling, especially in older patients. 2 To our knowledge, this is the first time modifiable risk factors for upper GI hemorrhage have been investigated in this population. Study limitations include inability to detect undocumented use of over the counter aspirin, NSAIDs, and PPIs, no data on dosing of PPIs, and no data on medication adherence. These limitations notwithstanding, our data highlight an important opportunity to prevent gastrointestinal morbidity among patients on warfarin therapy, and should serve as a call to action to develop and implement new treatment protocols in anticoagulation clinics.
Supplemental Material
10.1177_1358863X18815971_Supplemental_Material – Supplemental material for Missed opportunities to prevent upper GI hemorrhage: The experience of the Michigan Anticoagulation Quality Improvement Initiative
Supplemental material, 10.1177_1358863X18815971_Supplemental_Material for Missed opportunities to prevent upper GI hemorrhage: The experience of the Michigan Anticoagulation Quality Improvement Initiative by Jacob E Kurlander, Xiaokui Gu, James M Scheiman, Brian Haymart, Eva Kline-Rogers, Sameer D Saini, Scott Kaatz, James B Froehlich, Caroline R Richardson and Geoffrey D Barnes in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors have the following conflicts of interest: Jim Froehlich serves as a consultant for Janssen Pharmaceuticals, Merck, and Novartis, has received grant/research support from Blue Cross Blue Shield of Michigan, Fibromuscular Dysplasia Society of America, and serves on an advisory committee for Boehringer Ingelheim and Pfizer. Eva Kline Rogers is a member of Anticoagulation Forum Board of Directors and serves on the Steering Committee for QUANTUM-AF. Geoffrey Barnes serves as a consultant for Pfizer/Bristol-Myers Squibb, Janssen, and Portola, and has received grant funding from Blue Cross Blue Shield of Michigan,NHLBI, and Pfizer/Bristol-Myers Squibb. Scott Kaatz serves as a consultant for Janssen, Pfizer, Portola, Roche, Bristol Myer Squibb, and has received institutional research funding from Janssen.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The MAQI 2 consortium is supported by Blue Cross Blue Shield of Michigan. The research of Dr Kurlander and Dr Saini is supported by the Department of Veterans Affairs. The remaining authors have no financial disclosures.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
