Abstract
Introduction:
This study aims to determine the effectiveness, cost savings and feasibility of implementing a systematic process of deprescribing medications for symptomatic management, namely, acid suppressants, laxatives, analgesics and antiemetics for patients of a Singapore rehabilitation hospital.
Methods:
A total of 200 patients were randomized to a deprescribing intervention (n = 100) or control (usual care) group (n = 100). The patient-centred deprescribing process was utilized. Symptomatic medications were deprescribed following initial pharmacist assessment, discussion with doctors and consideration of patients’ preferences regarding discontinuation or dose reduction. Symptom recurrence, adverse drug withdrawal events (ADWEs) and the need for drug re-initiation or initiation of new symptomatic medications after deprescribing were monitored in the first, second and sixth weeks.
Results:
The mean age of patients was 72.8 years and 68.6 years in the intervention and control groups. There were no significant reductions in the monthly cost and total number of medications between both intervention and control groups. Systematic deprescribing of acid suppressants was the highest among the four target drug classes. Recurrence of pain and re-initiation of analgesics occurred in two out of seven cases of discontinuation. However, no ADWEs or constipation were noted in the intervention group. On average, a total of 19 minutes was required by pharmacists and doctors to complete the deprescribing process.
Conclusion:
The systematic deprescribing of symptomatic medications did not reduce costs nor the total number of medications. The risk of symptom recurrence and adverse events was negligible. Knowledge, attitudes and collaboration among healthcare professionals regarding deprescribing are critical.
Trial registration:
Clinicaltrials.gov, number NCT03354845.
Introduction
Deprescribing is an organized method of identifying and withdrawing drugs, which are currently or potentially causing more harm than benefit to patients, based on each individual patient’s condition and treatment goals. 1 Deprescribing is conducted as part of the prescribing process to enhance the quality of life, and involves shared decision-making between patients or caregivers and the multidisciplinary care team.1–4 The five steps of deprescribing comprise: 1) performing a comprehensive medical/medication history; 2) considering the overall risk and appropriateness of drugs; 3) assessing the possibility of and planning drug discontinuation; 4) initiating and documenting drug discontinuation; and 5) monitoring effects and providing patient support. 1
Deprescribing is even more important in elderly patients aged 65 years and above due to the higher prevalence of polypharmacy.1,5–7 Fifty percent or more of inpatients in a Singapore acute restructured hospital were discharged with at least five chronic medications in 2013. 8 Statistics were similar in three Singapore nursing homes where significant inappropriate medication use and drug-related problems were also identified.9,10 Symptomatic control medications, such as acid suppressants, laxatives and painkillers, are most frequently prescribed and continued in inpatient elderly populations.11,12 However, such medications are rarely needed on a long-term basis.13,14
Therefore, symptomatic medications were targeted for deprescribing in this study so that polypharmacy, unnecessary medication use and healthcare costs could be reduced. The discontinuation of medications has been trialled without causing significant adverse effects or symptom recurrence.15–24 Positive outcomes demonstrated include enhanced cognition and reduced falls.18,21 However, it is necessary to investigate the effects and feasibility of deprescribing in the present local setting, where healthcare perceptions, and associations between medication discontinuation and patient clinical outcomes may be different from other studied populations. 22 This study was conducted in a Singapore rehabilitation hospital, where subacute and rehabilitative care is provided to patients discharged from acute general hospitals.
Methods
Study design
This study was conducted as a single blind trial with two parallel groups. A total of 200 patients were randomized to an intervention (deprescribing) or control (usual care) group in a 1:1 ratio. The randomization sequence was obtained by an independent administrator using GraphPad randomization sequence software ©2017. The allocation sequence was administered by an executive from the medical administration office, who was not part of the study team. Whenever a patient has consented to be randomized, the executive will be contacted for the allocation group in accordance with the allocation sequence. Group assignment was made known to the pharmacists and doctors involved in the deprescribing intervention but not to the patients recruited. All intervention and control patients were treated by the same group of doctors who were allowed to alter patients’ medication regimens as wished in both groups. In the control group, doctors maintained the usual practice of medication review, altering and discontinuing medications as necessary, without receiving deprescribing recommendations from pharmacists. Apart from initial interventions regarding medication orders where necessary, pharmacists did not participate in doctors’ routine medication reviews.
Newly admitted patients in six subacute and rehabilitative wards of a Singapore rehabilitation hospital were screened for inclusion and recruited in the study between April 2016 and March 2017 by three hospital pharmacists. The inclusion criteria comprised all patients, regardless of age, who were on at least one of the following target symptomatic control medications for deprescribing: acid suppressants/proton pump inhibitors (PPIs), laxatives, analgesics (paracetamol, nonsteroidal anti-inflammatory drugs (NSAIDs), codeine, tramadol) and antiemetics. Patients with terminal illness or cancer, and patients with documented dementia who had no accompanying caregiver were excluded from the study. Patients who were discharged or transferred out from the rehabilitation hospital less than six weeks after recruitment were followed up for the partial duration of the study until leaving the hospital.
This study was approved by the SingHealth Centralised Institutional Review Board (CIRB) in March 2016 (CIRB reference number: 2016/2149). All recruited patients provided written informed consent. Additionally, consent was provided for medication discontinuation or dose reduction in the study. The study was registered in Clinicaltrials.gov database in November 2017 (Clinicaltrials.gov, number NCT03354845).
Study objectives
The primary study objective was to determine the cost savings of systematic deprescribing. The secondary objective was to determine the reduction in the total number of medications. Possible adverse consequences of deprescribing, which comprise constipation indicated by no bowel movement (BNO) in the past two days (BNO ⩾2/7), other symptom recurrence, adverse drug withdrawal events (ADWEs) and the need for deprescribed medications to be restarted, or initiation of new symptomatic control medications after deprescribing, were monitored. The feasibility of implementation was also determined through the time required to complete the deprescribing process and the limitations and challenges encountered, as documented by the pharmacists.
Intervention
The five-step patient-centred deprescribing process was utilized.1,4 Steps one to three of the process (review of patient’s history and drug appropriateness and planning for deprescribing) were performed by pharmacists and steps four to five (initiation and monitoring effects of deprescribing) by the doctor-in-charge in the rehabilitation hospital.
Pharmacists reviewed patients’ medical history and current medications from case notes and inpatient medication records (IMRs), and assessed for appropriateness of symptomatic medications and potential for discontinuation by acquiring information from patients and caregivers if necessary. Pharmacists would continue to review the symptomatic medications for one week if patients were found to require the medications for their ailments at that juncture. If at least one of the deprescribing criteria in the algorithm was met, the pharmacist would discuss with the doctor-in-charge regarding the suitability of discontinuing or reducing the dose of target symptomatic medications. Examples of deprescribing criteria included lack of clear drug indication, resolution of symptoms and preference of patient or caregiver for medication cessation or dose reduction.
Doctors would review the indication, dosing and duration of medications to be deprescribed, and take note of patients’ and caregivers’ preferences regarding discontinuation or dose reduction. After deprescribing, patients were monitored by doctors for symptom recurrence and ADWEs. The incidence of symptom recurrence and ADWEs and the need for drug re-initiation or initiation of new symptomatic control medications were documented by pharmacists in the first, second and sixth weeks of the deprescribing study intervention (or upon discharge from the hospital if earlier).
Statistical analysis
PS Power and Sample Size Calculations version 3.1.2 was used to determine the sample size required. Assuming that the deprescribing intervention would produce an improvement of 20% in cost savings from the current usual practice (this assumption was made with reference to a study by Garfinkel et al. 17 ), 81 patients were needed in each of the intervention and control groups (α = 0.05, β = 0.2). 17 To offset potential patient drop-out, a target recruitment of 100 patients was established in each arm.
Guided by the intention-to-treat principle, patients who fit the inclusion criteria and were randomized but could not be followed up were included in the data analysis. Missing data of patients, who were discharged less than one week after recruitment, were imputed as zero for reduction in the cost and number of medications, and coded as two, representing no incidence, for BNO ⩾2/7 in the analysis.
The independent-samples t test was used to compare the reduction in the total cost and total number of medications between the intervention and control groups. The chi-square test of independence was used to compare the incidence of BNO ⩾2/7 between both groups. IBM SPSS Statistics 22 was used and a p-value of less than 0.05 was considered statistically significant.
Results
Patient recruitment and baseline data
Ten ineligible patients (5%), six who had been randomized to the intervention group and four to the control group, were erroneously recruited though they met the exclusion criteria and were excluded in the data analysis (figure 1). Ten patients (5%), six from the intervention group and four from the control group, were discharged or transferred from the rehabilitation hospital and dropped out of the study shortly after being recruited (figure 1). These patients were included in the intent-to-treat analysis.
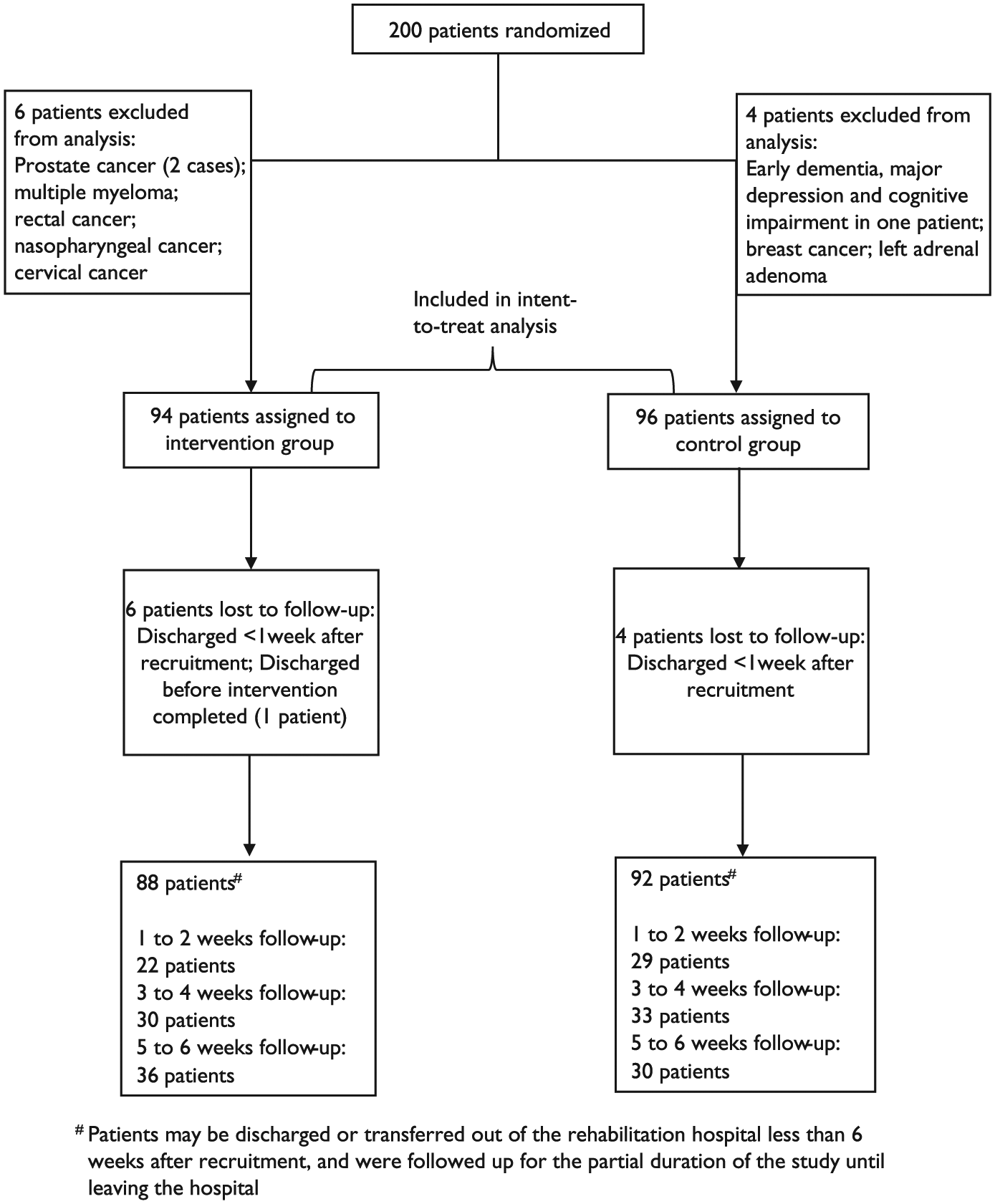
The patient demographics, medical conditions and medications at baseline are presented in Table 1. A greater proportion of patients in the study were female (55%); though there were significantly more male patients in the intervention group than in the control group (51 versus 34, p < 0.01). The mean age of patients in the intervention group was higher compared to the control group (72.8 versus 68.6 years, p < 0.01). The majority of patients in the intervention and control groups had five co-morbidities and were on 14–15 medications at baseline. The incidence of chronic medical conditions such as hypertension, hyperlipidaemia and diabetes was similar in both groups. Likewise, the incidence of osteoarthritis and fracture, which required analgesics, was similar in both the intervention and control groups. However, more patients in the intervention group had gastritis, gastrointestinal (GI) ulcer or upper GI bleed (12 versus 7 in the control group, p = 0.21) and more patients in the control group had BNO ⩾2/7 at baseline (10 versus 5 in the intervention group, p = 0.19), though these differences were not statistically significant. The proportion of patients on target symptomatic control medications was similar in both groups, with the exception of antiemetics.
Baseline demographics, medical conditions and medications.

BNO ⩾2/7: no bowel movement in the past two days; NSAIDs: nonsteroidal anti-inflammatory drugs; SD: standard deviation; GERD: gastroesophageal reflux disease; GI: gastrointestinal.
Cost savings of deprescribing intervention
The cost of one month of medications for each patient was calculated at baseline and at the sixth week or upon discharge using the drug formulary list and selling prices of the rehabilitation hospital. The cost of symptomatic medications prescribed on an as-needed basis was calculated as 15 days instead of a one-month supply, except paracetamol, paracetamol/codeine and paracetamol/orphenadrine, which were calculated as 100 tablets supply. External preparations (including topicals, eye/ear drops, mouthwashes, gargles), nebulizations and short-term medications with a duration of less than a month (including cough syrups, lozenges, intravenous and oral antibiotics) were excluded in the calculation of the overall cost of medications.
The implementation of a systematic process of deprescribing symptomatic control medications did not lead to increased cost savings; although the reduction in the total cost of medications from baseline was higher in the intervention group than in the control group by SGD$0.17, this difference was not significant (Table 2).
Comparison of cost savings, reduction in number of medications and adverse effect of constipation between groups.

At sixth week from date of recruitment or upon discharge (if earlier).
BNO ⩾2/7: no bowel movement in the past two days; CI: confidence interval.
Reduction in the total number of medications
Every medication in each patient’s IMR was counted in the overall number of medications at baseline and at the sixth week or upon discharge. The reduction in the total number of medications was higher in the control group, but the mean difference between the groups was −0.1 and not significant (Table 2).
Change in bowel movement following deprescribing
Constipation indicated by BNO ⩾2/7 was recorded in both intervention and control patients at baseline and at the sixth week or upon discharge so that adverse effects of decreased bowel movement and recurrence of constipation as a result of deprescribing could be determined.
The number of patients with BNO ⩾2/7 was 15 (7.9%) at baseline (Table 1) and 20 (10.5%) at the sixth week or upon discharge (Table 2). The incidence of BNO ⩾2/7 at the sixth week or upon discharge was not significantly different between the intervention and control groups (9 and 11 patients, respectively). As there was no statistically significant difference in BNO ⩾2/7 at baseline between the groups, it can be inferred that the deprescribing of laxatives was not associated with adverse effects of decreased bowel movement and recurrence of constipation.
Feasibility of deprescribing intervention
Among the 27 patients (17 + 10) for whom systematic deprescribing was conducted (Table 3), 14 patients had discontinuation of target symptomatic medications, 12 patients had dose reduction of target symptomatic medications, and one patient had both discontinuation and dose reduction. Doses were reduced to an as-needed basis in four of the 13 patients with dose reduction. Discontinuation of analgesics and dose reduction of acid suppressants were the highest compared to other target drug classes (Table 4). Following dose reduction using the deprescribing protocol, omeprazole and laxatives were discontinued by the doctor in two cases. The deprescribing criteria applied in all cases of discontinuation and dose reduction were a lack of clear drug indication and resolution of symptoms. For example, patients were still on omeprazole though the precipitating NSAID had been discontinued. In other cases, omeprazole was discontinued or its dose reduced in the study as the patient had no gastric complaints, nor history of gastroesophageal reflux disease (GERD) or peptic ulcer. In 11 cases, patients preferred to take a reduced dose of or to cease taking symptomatic medications.
Deprescribing of target symptomatic medications in intervention group categorised based on systematic and informal deprescribing.
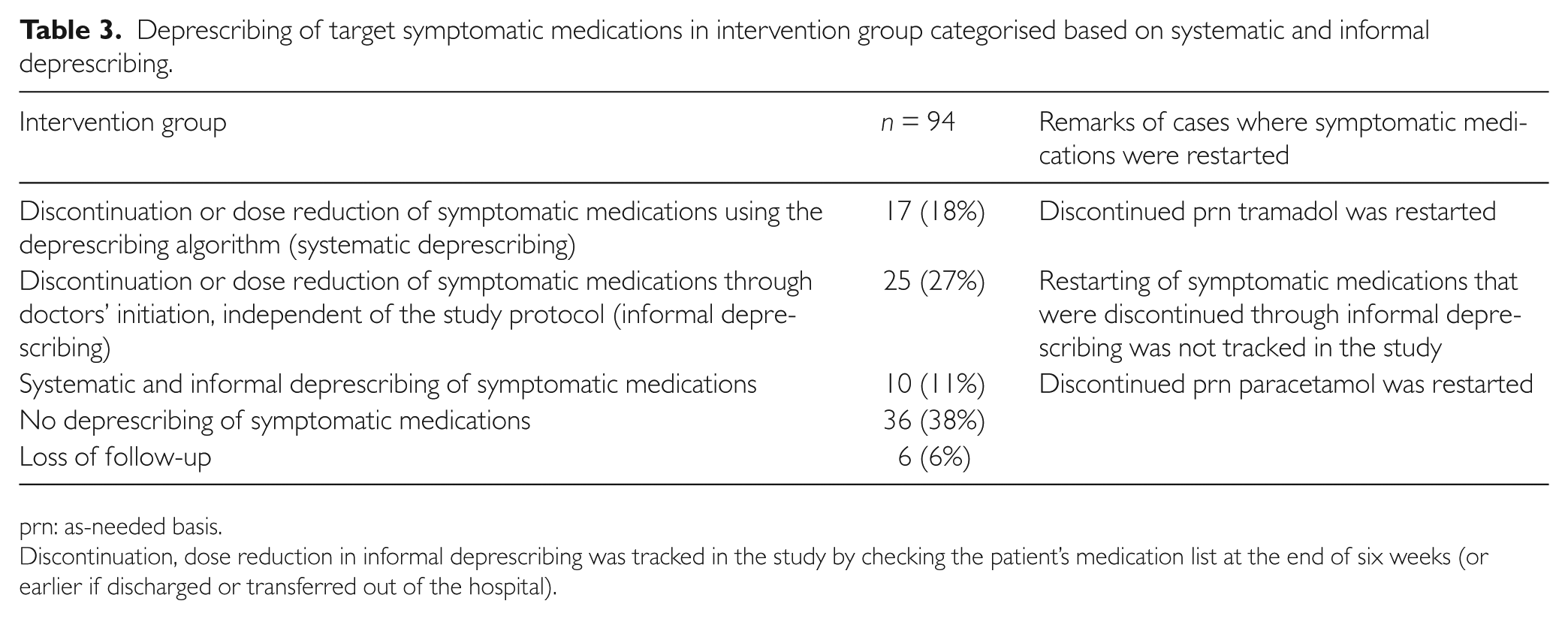
prn: as-needed basis.
Discontinuation, dose reduction in informal deprescribing was tracked in the study by checking the patient’s medication list at the end of six weeks (or earlier if discharged or transferred out of the hospital).
Deprescribing of target symptomatic medications in intervention group categorised by drug class.

Including one patient where omeprazole 40 mg/day was changed to famotidine 20 mg/day.
Systematic deprescribing: using the study protocol; informal deprescribing: through doctors’ initiation, independent of the protocol.
There were two reports of recurrence of pain which required re-initiation of discontinued analgesics (Table 3), out of seven cases overall where analgesics were discontinued using the deprescribing protocol (Table 4). However, no complaints of ADWEs were reported. With the exception of these two cases, re-initiation or dose increase of target symptomatic medications following deprescribing was not necessary. Initiation of new symptomatic control medications was also not required.
On average, pharmacists required 11 minutes to review each patient’s medication history, and assess the appropriateness of drug use and potential for discontinuation. An additional average of eight minutes was required for discussion with the doctor about the suitability to deprescribe, and for assessment and deprescribing by the doctor.
Discontinuation of laxatives and analgesics observed in the control group was higher than in the intervention group when both systematic and informal deprescribing were taken into account (Table 5). Dose reduction across all target drug classes in the control group was lower than in the intervention group with both systematic and informal deprescribing (Table 5). Dose reduction of acid suppressants observed in the control group and achieved through systematic deprescribing were similar (Tables 4 and 5).
Comparison of deprescribing of target symptomatic medications between intervention and control groups.
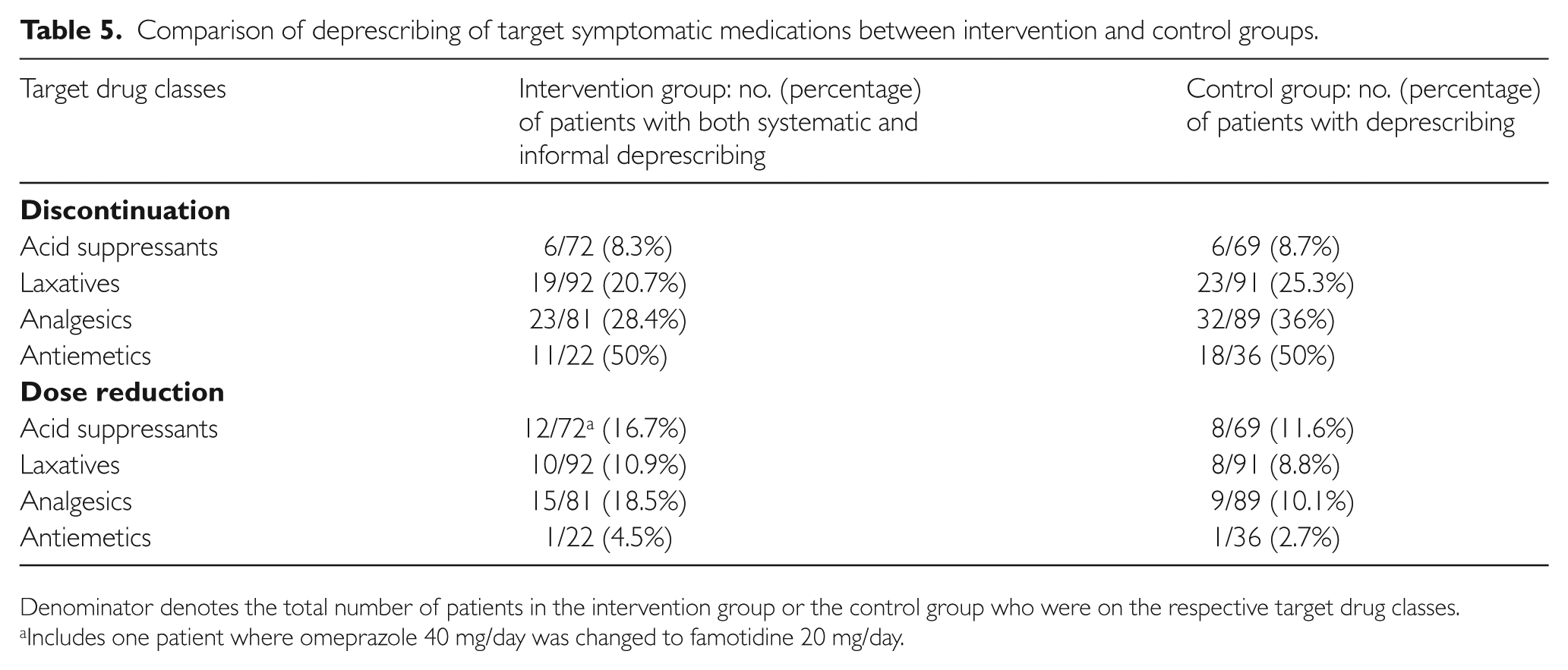
Denominator denotes the total number of patients in the intervention group or the control group who were on the respective target drug classes.
Includes one patient where omeprazole 40 mg/day was changed to famotidine 20 mg/day.
Acid suppressants, laxatives, analgesics and antiemetics were deemed necessary and continued in most patients. Discontinuation of these medications through systematic deprescribing using the study protocol and through informal deprescribing initiated by the doctors ranged from 8.3% to 50% in the intervention group (Table 4). Rather than discontinuing the medications if symptoms had resolved, there was some preference among doctors to maintain as-needed dosing for standby in case patients required relief (Table 6). In addition, the current dose of acid suppressants was maintained for gastroprotection and not reduced to the lowest possible in a few patients who had no history of gastritis, GERD nor peptic ulcer (Table 6). Informal deprescribing by doctors independent of the study accounted for a larger proportion of symptomatic medication discontinuation and dose reduction compared to systematic deprescribing in the intervention group (Table 4). This could be explained by individual doctors’ preferences to review and deprescribe a few weeks later despite having noted deprescribing recommendations from pharmacists.
Non-utilisation of deprescribing criteria and algorithm in the intervention group.

Systematic deprescribing: using the study protocol; informal deprescribing: through doctors’ initiation, independent of the protocol; OM: every morning; BD: twice daily; BO: bowel open; GI: gastrointestinal; prn: as-needed basis.
Discussion
Implementation of systematic deprescribing in the study did not lead to cost savings nor a reduction in patients’ overall number of medications. These findings could be attributed to the low deprescribing rate of symptomatic medications in the study and the possibility that deprescribing symptomatic medications alone was insufficient to produce marked cost savings. A prospective controlled study which discontinued all drugs that met the deprescribing criteria in the geriatric-palliative algorithm demonstrated a significant reduction of $0.46 in a patient’s daily drug cost and successful discontinuation of 2.8 drugs per patient across 10 different drug types, including H2 antagonists, antihypertensives and sedatives. 17
Deprescribing of symptomatic medications in the study was not associated with decreased bowel movement, symptom recurrence and ADWEs. Recurrence of pain and re-initiation of analgesics occurred in two out of seven cases following discontinuation. Thus, systematic deprescribing can be implemented safely and this finding is consistent with other deprescribing studies.15–19
The relatively low rate of deprescribing reflected in this study could be explained by considerations of the necessity of symptomatic medications, especially laxatives and analgesics, in elderly patients and patients undergoing physical rehabilitation in the hospital. Laxatives were required in conditions such as limited mobility and acute urinary retention. However, such requirements could not be assessed adequately using the deprescribing algorithm. The discontinuation rates of laxatives and analgesics in this study (20.7% and 28.4% respectively) were comparable to those reported in a randomized controlled trial in Australian residential aged care facilities (28% and 34% respectively), where patients were of the same age group (> 65 years) as our study. 16
Among the four target symptomatic drug classes, deprescribing of acid suppressants using the five-step algorithm was the most successful. A stepwise structured process of deprescribing or a deprescribing checklist might be more suitable for acid suppressants compared to other drug classes as doctors were agreeable to discontinue the drug in the absence of a clear indication or to reduce to the lowest appropriate dose. The utility of deprescribing algorithms in detecting inappropriate or unnecessary use of acid suppressants can be confirmed in other deprescribing studies.15,19 Discontinuation and dose reduction of acid suppressants using the deprescribing study protocol (16.7% of patients on PPIs) were comparable with a study which also utilized the five-step patient-centred deprescribing process (10.5% of recruited PPI users). 15 A pilot intervention study, however, achieved higher discontinuation of acid suppressants (48%) in general medicine wards through the implementation of an education program and a deprescribing decision support tool. 19 Lower deprescribing rates of acid suppressants in our sample could be attributed to a larger number of study patients with co-morbidities that required PPIs.
The total time required to complete medication review and deprescribing through the five-step patient-centred process was relatively manageable. In general, patients and doctors were receptive and agreeable to deprescribing recommendations. However, a stepwise approach to deprescribing might not be entirely suited in patients with multiple, complicated co-morbidities, as such methods do not aid clinicians to consider various patient factors holistically when deprescribing. Thus, this aspect should also be considered in evaluating the overall feasibility.
A few limitations exist in this study. Firstly, the open trial design adopted, while resembling actual clinical practice where doctors could freely adjust patients’ medication regimens, might have introduced confounding effects in the overall implementation of the deprescribing protocol. Secondly, due to resource limitations, the planned follow-up period for pharmacists to review the appropriateness of symptomatic medications was restricted to one week, which might have been insufficient to ensure that the medications were deprescribed once symptoms had resolved. Thirdly, the utilization of a randomized controlled parallel group study design restricted collaboration and support sought from doctors in actively deprescribing symptomatic medications. However, as the same pool of doctors were involved in the treatment of both the intervention and control groups, review of recruited patients by pharmacists was conducted independently from doctors, apart from discussions about deprescribing recommendations, to prevent bias in doctors’ clinical judgements and decisions to deprescribe.
The knowledge and attitudes of healthcare professionals regarding deprescribing, such as establishing clear treatment goals and justification for the clinical value of drugs, are critical. More importantly, collaboration between pharmacists and doctors is needed to ensure judicious and sustained deprescribing in clinical practice.3,25
Conclusion
The implementation of systematic deprescribing of symptomatic medications through the five-step patient-centred process did not improve cost savings nor reduce the overall number of medications. There was negligible risk of symptom recurrence and adverse events. Deprescribing of acid suppressants was the highest compared to other drug classes. A structured approach could be effective in guiding deprescribing and reducing unnecessary and inappropriate use of acid suppressants.
Supplemental Material
Proceedings_Supplementary_material_for_deprescribing_5-step_deprescribing_process_28.4.18 – Supplemental material for Effectiveness and feasibility of deprescribing of symptomatic medications in a Singapore rehabilitation hospital
Supplemental material, Proceedings_Supplementary_material_for_deprescribing_5-step_deprescribing_process_28.4.18 for Effectiveness and feasibility of deprescribing of symptomatic medications in a Singapore rehabilitation hospital by Charissa Ann Jia Ming Ee, Kheng Hock Lee, Hee Lim Tan and Lian Leng Low in Proceedings of Singapore Healthcare
Footnotes
Acknowledgements
The authors thank medical administrators Belinda Soh and Sadhana Binte Snin for assistance with randomization, and pharmacists Jessica Chong and Lin Xiuting for assistance with recruitment and data collection.
Authors’ contributions
All authors contributed to the study development, drafting or revision of this article.
Availability of data and materials
Access to files containing primary data and data analysis can be obtained by contacting corresponding author:
Declaration of conflicting interests
None declared.
Ethical approval
Ethical approval to report this case was obtained from the SingHealth Centralised Institutional Review Board (CIRB reference number: 2016/2149).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Informed consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
