Abstract
Amphetamine and its related derivatives and analogues (ADRA) are highly addictive central nervous system stimulants that are used commonly in the treatment of attention-deficit/hyperactivity disorder and narcolepsy. These medications are associated with many side effects but reports of peripheral arterial manifestations associated with ADRA usage are scarce. We retrospectively reviewed the records of 16 patients (median age 37 years (IQR 31–47), 13 females) referred to a single tertiary referral service while receiving ADRA. Follow-up was available for a median of 3 years (IQR 3–4.5). The most common presentation (62.5%) was mild vasospastic symptoms involving the upper, lower or both extremities. Six patients developed severe manifestations including tissue loss and the need for lower extremity amputation. Most patients (75%) refused to stop the medication during follow-up. Underlying rheumatologic disorders were found in 25% of the patients, and the presence of rheumatologic disease seemed to be associated with more severe vascular manifestations. In conclusion, it is important to search for ADRA usage as part of the differential diagnosis of digital ischemia.
Introduction
Amphetamine and its related derivatives and analogues (ADRA) are highly addictive as central nervous system (CNS) stimulants and have been listed as a substance of abuse. 1 Amphetamines have been used for the treatment of several disorders, including attention-deficit/hyperactivity disorder and narcolepsy. 2 However, unlabeled uses such as promoting wakefulness and physical activity and decreasing appetite as a weight loss supplement have become a growing problem worldwide. 3 The main pharmacological effect of amphetamine is the increase of serum and intracranial concentrations of norepinephrine, dopamine, and serotonin through multiple mechanisms. 4 Most reported ADRA-related side effects include CNS and cardiac phenomena.5–7 Most recently, ADRA usage has been reported in association with acute coronary syndrome. 8 However, reports of peripheral arterial manifestations associated with ADRA in adults are scarce.
This case series aims to describe the clinical characteristics and outcomes of patients with peripheral arterial manifestations and a history of ADRA use, and to identify the potential risk factors associated with the development of severe peripheral arterial adverse outcomes in these patients.
Methods
This is a retrospective case series study. Clinical records of patients aged > 18 years who presented to the Massachusetts General Hospital vascular medicine service for evaluation of vascular symptoms while on ADRA were reviewed. Patients were identified by chart review. Patients whose symptoms were considered to be related to ADRA use based on review of medical records were included in the analysis. The following data were included in the analysis: patient demographics; medical history; details of ADRA usage, including type of stimulant, daily dosage, and duration of usage; details of the arterial manifestation; and the clinical status of these patients. Clinical presentations were categorized into mild symptoms, which included self-limiting acrocyanosis and Raynaud’s phenomenon (as defined by the European Society of Vascular Medicine consensus 9 ), and severe manifestations, which were defined as tissue loss, need for amputation, and need for advanced therapy (defined as hospital admission and/or invasive intervention – either endovascular or surgical). Considering the small number of subjects, Spearman’s correlation was used to identify individual variables significantly associated with severe manifestations (defined as a two-tailed p-value < 0.05). The protocol of this study was approved by the Partners Healthcare Institutional Review Board.
Results
From May 2000 to December 2017, 16 patients (13 females, median age 37 (IQR 31–47)) with a history of ADRA usage were referred to our service for evaluation (Table 1). Median follow-up was 3 years (IQR 3–4.5). Adderall (amphetamine and dextroamphetamine; DSM Pharmaceuticals Inc., Greenville, NC, USA) was the ADRA used in all the patients and the most common daily dosage was 30 mg. Fifty per cent of patients had one or more cardiovascular risk factors (diabetes, hypertension, tobacco use, and/or chronic renal disease), 75% had a history of a psychiatric diagnosis, and 25% had a history of rheumatologic disease. The most common rheumatologic diagnosis was systemic sclerosis (three out of four). There was concomitant use of vasoactive medication by 37.5% of patients, and 25% were on a beta-blocker. Most (62.5%) vascular manifestations were mild (Figure 1). Most of the patients had either axial imaging, physiological testing, such as upper extremity pulse volume recording (PVR) (Figure 2), or both, as part of the investigation. Physiological tests were utilized in 44% of the patients. Digital subtraction angiography was performed in patients with severe manifestations (Figure 3). Most patients (75%) continued ADRA usage through their last follow-up.
Summary of all patients.
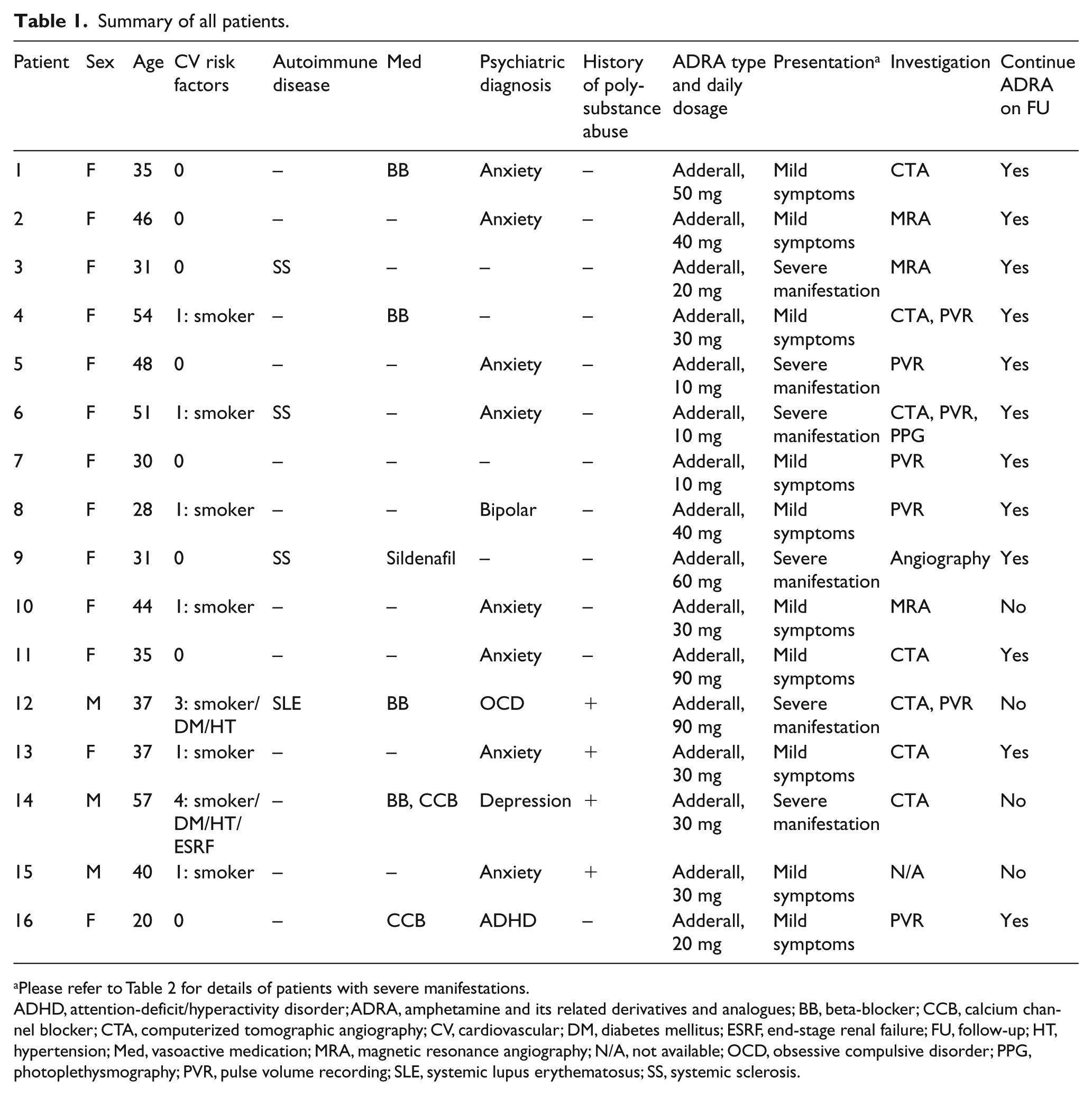
Please refer to Table 2 for details of patients with severe manifestations.
ADHD, attention-deficit/hyperactivity disorder; ADRA, amphetamine and its related derivatives and analogues; BB, beta-blocker; CCB, calcium channel blocker; CTA, computerized tomographic angiography; CV, cardiovascular; DM, diabetes mellitus; ESRF, end-stage renal failure; FU, follow-up; HT, hypertension; Med, vasoactive medication; MRA, magnetic resonance angiography; N/A, not available; OCD, obsessive compulsive disorder; PPG, photoplethysmography; PVR, pulse volume recording; SLE, systemic lupus erythematosus; SS, systemic sclerosis.
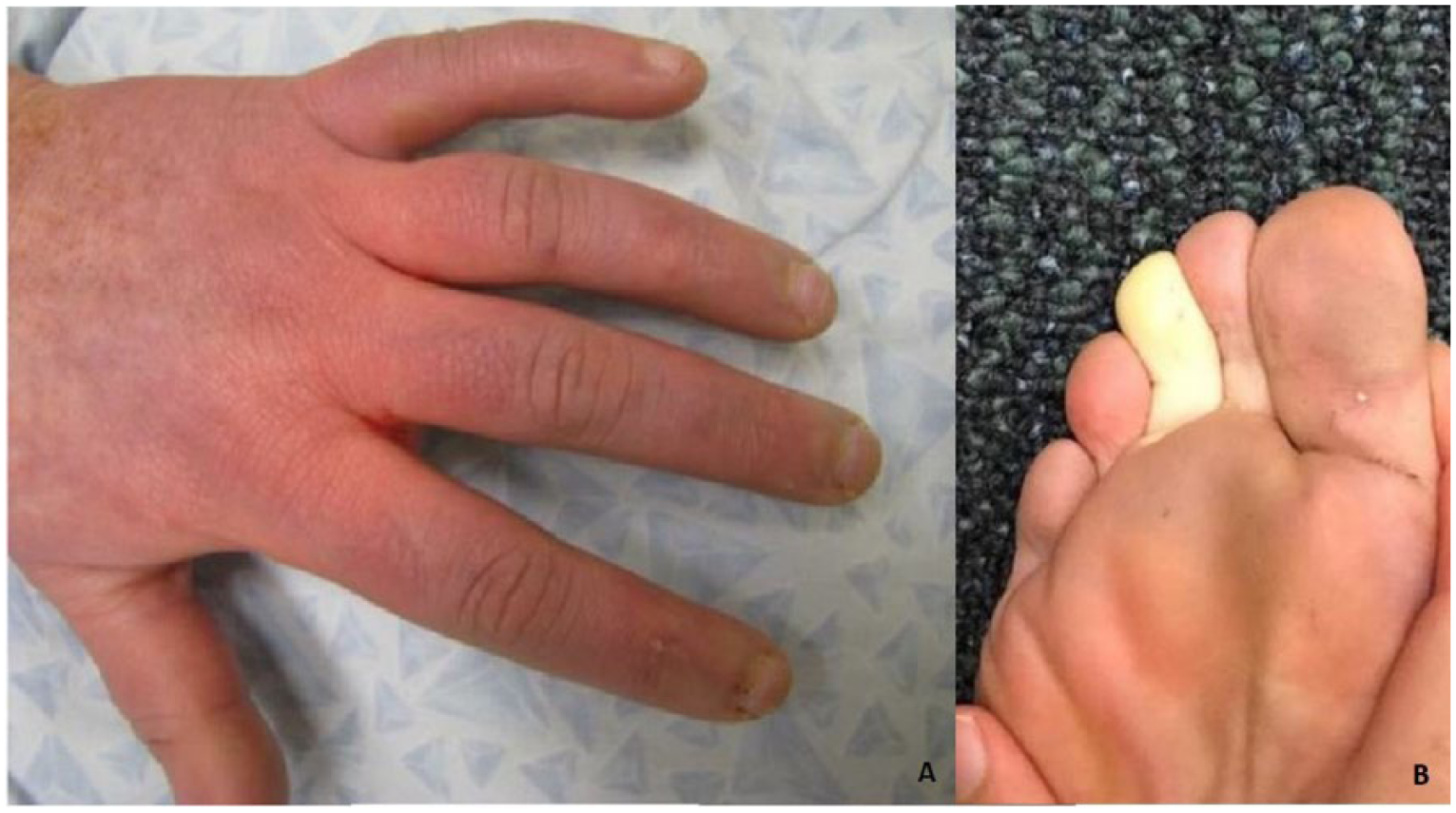
Clinical photograph of acrocyanosis in patient 16 (A) and Raynaud’s phenomenon in patient 8 (B).
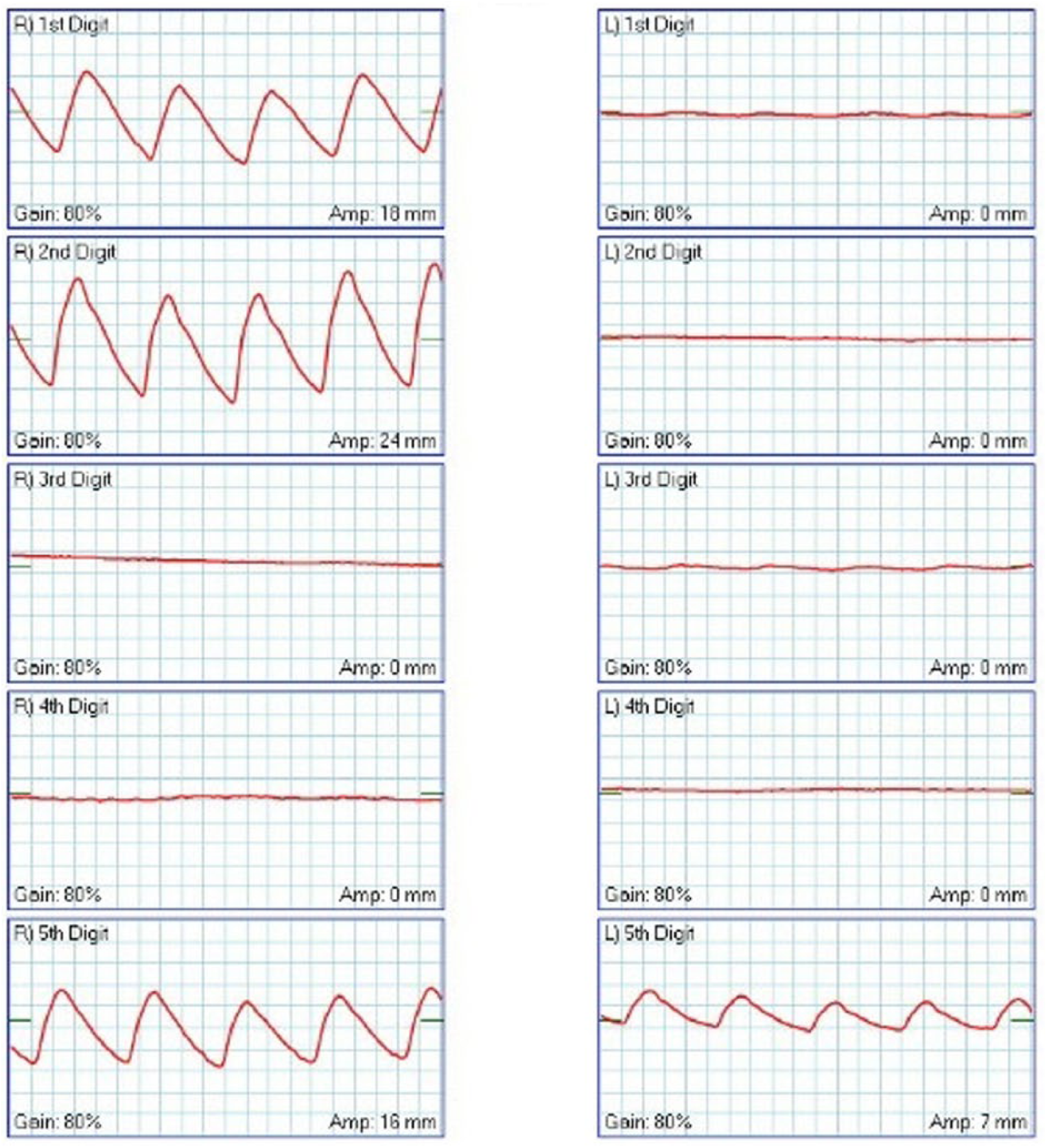
Upper extremity pulse volume recording of patient 6 showing severe impairment of the arterial circulation in bilateral digits.
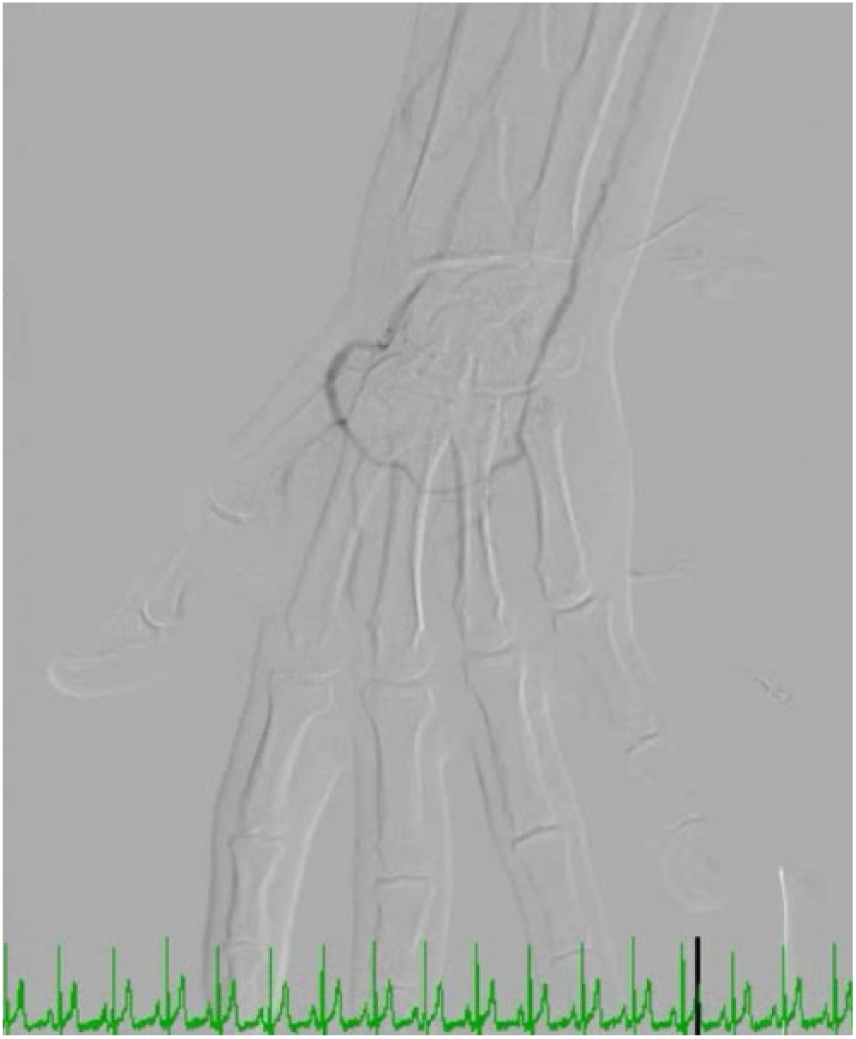
Digital angiography of patient 9 showing abrupt cut-off of the first and second digital arteries.
Six of the patients developed severe vascular manifestations (Table 2). Two patients had severe Raynaud’s phenomenon with digital gangrene and tissue loss. Two patients had critical lower limb ischemia requiring revascularization and one patient required below-knee amputation because of an uncontrolled infection; both patients had multiple cardiovascular co-morbidities. In the correlation analysis, only the presence of rheumatologic disease seemed to be associated with more severe vascular manifestations. Neither cardiovascular risk factors nor age were associated with severe adverse events.
Summary of patients with severe manifestations.
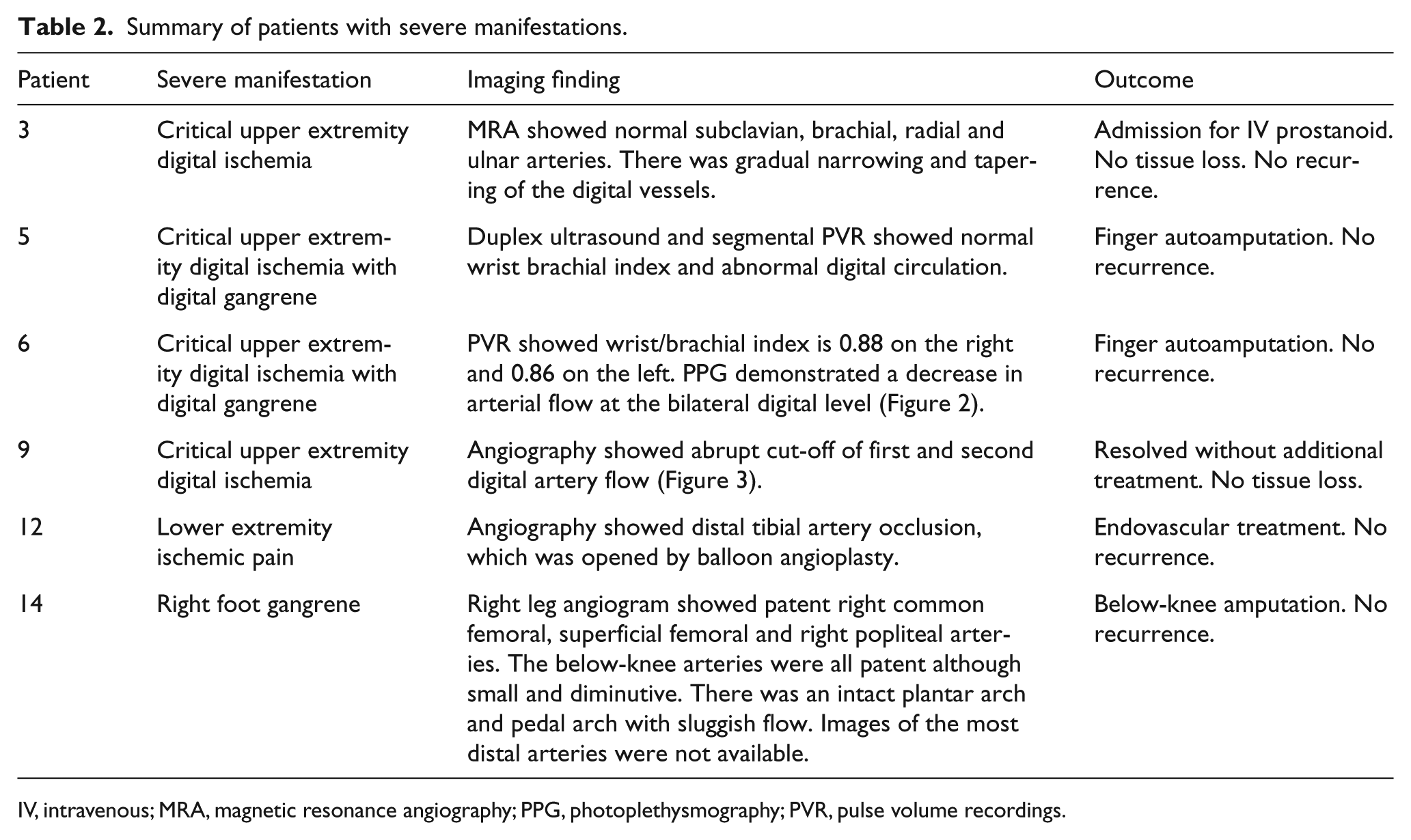
IV, intravenous; MRA, magnetic resonance angiography; PPG, photoplethysmography; PVR, pulse volume recordings.
Discussion
Amphetamine has been used for the treatment of several disorders including attention-deficit/hyperactivity disorder and narcolepsy. However, its use for other indications such as to promote wakefulness and physical activity and as an appetite suppressant, has been a growing problem worldwide. 3 For example, the prevalence of ADRA use among secondary school students in the United States was estimated to be 1.4% in 2016 1 and there has been an increase in amphetamine prescriptions in the United States. 2
Unfortunately, ADRA use is not devoid of side effects. Common side effects of ADRA include insomnia, anorexia, irritability, weight loss, abdominal pain, and headaches. 5 Serious side effects such as myocardial infarction, arrhythmia, and intracranial hemorrhage have been reported.5–7 Peripheral vascular adverse events of ADRA such as Raynaud’s syndrome (RS) 10 have only been reported in the pediatric population11–13 and in a single case report in an adult patient 14 (Table 3). We believe that the current report is the largest and most comprehensive series of peripheral vascular manifestation of ADRA in adults.
Summary of literature.
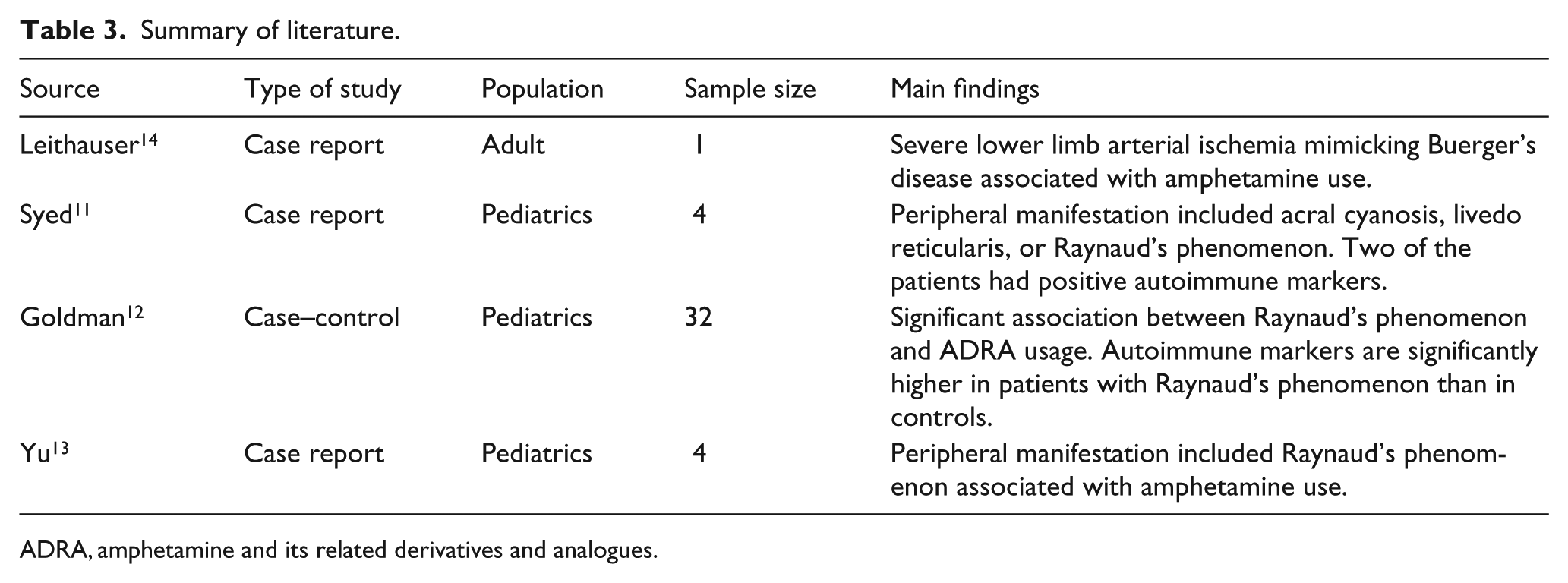
ADRA, amphetamine and its related derivatives and analogues.
In our study, most patients were young females, a finding that is consistent with recent trends in amphetamine prescription in the United States. 2 The majority of our patients had a psychiatric diagnosis, but only one had the diagnosis of attention-deficit/hyperactivity disorder. The indications of ADRA in some of our patients were not well documented. The most common clinical presentation in our series was mild vasospastic symptoms. This is similar to the finding in the pediatric population. 12 Theoretically, the mechanism for this side effect may be the sympathomimetic property of ADRA which may result in vasoconstriction similar to beta-blockers. 15 In our series, however, the concomitant use of beta-blockers was not associated with severe adverse events.
In our study, we showed that a concomitant rheumatologic disease was strongly associated with the development of adverse events. A similar association was demonstrated in the pediatric population, in which patients with Raynaud’s phenomenon had higher levels of inflammatory markers. 12
Interestingly, patients in our series continued to use ADRA despite being given a provisional diagnosis of possible drug-induced vasculopathy. Thus, we cannot comment about the reversibility of symptoms in our population. The addictive property of amphetamine was highlighted in a US Food and Drug Administration (FDA) report and a recent World Drug Report.1,3 Data from the pediatric population showed that discontinuation of ADRA was associated with resolution of symptoms.11,12
Study limitations
Our report has several limitations. Firstly, it was retrospective and thus has the limitations typical of such studies. Specifically, we could not confirm the etiology of symptoms and had to rely on contemporaneous notes taken by treating clinicians that suggested ADRA as the cause of patients’ symptoms. We could not confirm the exact temporal correlation between the onset of symptoms and ADRA use in any of the cases because this has not been documented. Moreover, while this series represents a sample of patients on ADRA with arterial manifestations, it was not designed to assess the prevalence of arterial manifestations among all ADRA users. Thus, we could only comment about the association of adverse events among patients who already presented with symptoms and not in the general ADRA population. Furthermore, as a tertiary referral center, some preliminary investigations, such as axial imaging, had already been arranged prior to our consultation and this resulted in the heterogeneous pattern of investigations among our patients. Lastly, the number of patients included in this study is small and we could not identify a matching control group for comparison. As a result, we could not ascertain the causality of some of the clinical parameters with the development of adverse events. Nonetheless, our series represents the largest such description in adults and should serve to raise awareness for the potential of the association between ADRA and arterial pathology.
Conclusion
Peripheral vasospasm and digital gangrene can be seen in patients receiving ADRA. Among patients who present with arterial manifestations, the presence of an underlying rheumatologic disease seemed to be associated with more severe vascular manifestations, such as ischemia requiring amputation. It is important to search for ADRA usage in the history of young patients presenting with vasospastic disorders and to advise them to withhold ADRA, especially if rheumatologic disease co-exists.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
