Abstract
The 2013 American College of Cardiology/American Heart Association cholesterol guideline recommends moderate to high-intensity statin therapy in patients with peripheral artery disease (PAD) and ischemic cerebrovascular disease (ICVD). We examined frequency and facility-level variation in any statin prescription and in guideline-concordant statin prescriptions in patients with PAD and ICVD receiving primary care in 130 facilities across the Veterans Affairs (VA) health care system between October 2013 and September 2014. Guideline-concordant statin intensity was defined as the prescription of high-intensity statins in patients with PAD or ICVD ≤75 years and at least moderate-intensity statins in those >75 years. We calculated median rate ratios (MRR) after adjusting for patient demographic factors to assess the magnitude of facility-level variation in statin prescribing patterns independent of patient characteristics. Among 194,151 PAD patients, 153,438 patients (79.0%) were prescribed any statin and 79,435 (40.9%) were prescribed a guideline-concordant intensity of statin. PAD patients without ischemic heart disease were prescribed any statin and a guideline-concordant intensity of statin therapy less frequently (69.1% and 28.9%, respectively). Among 339,771 ICVD patients, 265,491 (78.1%) were prescribed any statin and 136,430 (40.2%) were prescribed a guideline-concordant intensity of statin. ICVD patients without ischemic heart disease were prescribed any statin and a guideline-concordant intensity of statin less frequently (70.9% and 30.5%, respectively). MRRs for both PAD and ICVD patients demonstrated a 20% and 28% variation among two facilities in treating two identical patients with statin therapy and guideline-concordant intensity of statin therapy, respectively. The prescription of statins, especially guideline-recommended intensity of statin therapy, is suboptimal in PAD and ICVD patients, with significant facility-level variation not explained by patient-level factors.
Keywords
Introduction
The benefits of statin use in patients with atherosclerotic cardiovascular disease (ASCVD) for secondary prevention of cardiovascular events are well established.1–4 Peripheral artery disease (PAD) is associated with an increase in all-cause mortality, but National Health and Nutrition Examination Survey (NHANES) data suggests an approximately 65% reduction in all-cause mortality in PAD patients prescribed statins. 2 The Heart Protection Study and the Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) trial demonstrate reduced cardiovascular event rates in patients on at least moderate-intensity statin who have PAD and ischemic cerebrovascular disease (ICVD), respectively. The Heart Protection Study demonstrated a reduction in future vascular events and the need for revascularization procedures in patients randomized to moderate-intensity statin therapy compared to placebo. Both of these effects were true regardless of baseline low-density lipoprotein cholesterol. 3 In the SPARCL trial, a total of 4731 patients with known ICVD without ischemic heart disease (IHD) were randomized to receive either daily high-intensity atorvastatin or placebo. The atorvastatin arm benefited from a reduction in the risk of fatal or non-fatal stroke (hazard ratio (HR), 0.85; 95% confidence interval (CI), 0.71 to 0.99; p=0.03) and a 6.6% absolute risk reduction in any cardiovascular event (HR, 0.74; 95% CI, 0.66–0.83; p<0.001) over a follow-up period of 5 years. 4 Because of the strong evidence for statin use in patients with known ASCVD, the guideline published by the American College of Cardiology (ACC) and American Heart Association (AHA) in 2013 recommended use of high-intensity statin therapy in adults ≤75 years of age and moderate-intensity statin in those >75 years with known ASCVD. 5
The 2013 ACC/AHA guideline describes ASCVD as IHD, PAD and ICVD. 5 Patients with PAD and ICVD have similar rates of cardiovascular disease events compared to patients with IHD. 6 Previous studies have shown facility-level variation in prescription of evidence-based statin therapy among patients with IHD;7,8 similar gaps in evidence-based statin prescription exist for diabetic patients. 9 The frequency of evidence-based statin prescription in patients with PAD and ICVD in the United States and whether there is variation in this evidence-based statin prescribing across facilities has not been well investigated. We performed this study to assess the frequency and facility-level variation in baseline statin prescription in patients with PAD and ICVD receiving primary care in the entire Veterans Affairs (VA) Health Care System at the time of initial implementation of the 2013 ACC/AHA cholesterol guideline.
Methods
Patient population
Our patient cohort was identified using a national VA administrative and clinical database comprising data from 1,266,541 veterans with IHD, PAD, and ICVD with primary care visits at 130 VA parent facilities and their community-based outpatient clinics between October 1, 2013 and September 30, 2014. If a patient had multiple primary care visits during the study interval, the most recent primary care visit during the study interval was used as the index visit.
Details of the cohort have been described previously.10–13 We identified patients with PAD (Supplemental Tables 1 and 2) using the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) diagnoses codes for intermittent claudication, atherosclerosis or thrombosis of the renal arteries, abdominal or thoracic aorta, iliac artery, arteries of the upper and lower extremities (with or without rest pain, ulceration or gangrene), or presence of peripheral bypass grafts. Patients with procedure codes for endarterectomy, atherectomy or angioplasty of peripheral arteries with or without stent, bypass grafting of the peripheral arteries or lower limb, and amputations were also included in the PAD group. Based on prior studies,11,14 we also used exclusion criteria to improve specificity for the diagnosis of PAD (Supplemental Table 2). We identified patients with ICVD (Supplemental Table 1) using diagnoses codes for atherothrombosis of the cerebral, vertebral or basilar arteries with or without stroke, cerebral embolism and transient ischemic stroke. We also used procedure codes for carotid artery angioplasty with or without stents and carotid endarterectomy to identify patients with ICVD. 15 Based on a prior chart review of 100 random patients, the positive predictive value for the diagnosis of ASCVD based on our methodology was 88%. 10 Patients with IHD (Supplemental Table 1) were identified by using diagnoses codes for myocardial infarction or unstable angina, and procedure codes for percutaneous coronary intervention or coronary artery bypass grafting, as shown in prior studies.8,10,12,13,16
We excluded patients from our analysis if they were under hospice care, had a diagnosis of metastatic cancer in the past 5 years, or both. Of the remaining patients, further exclusions were made for patients with diagnoses of IHD without concomitant PAD or ICVD (Figure 1).
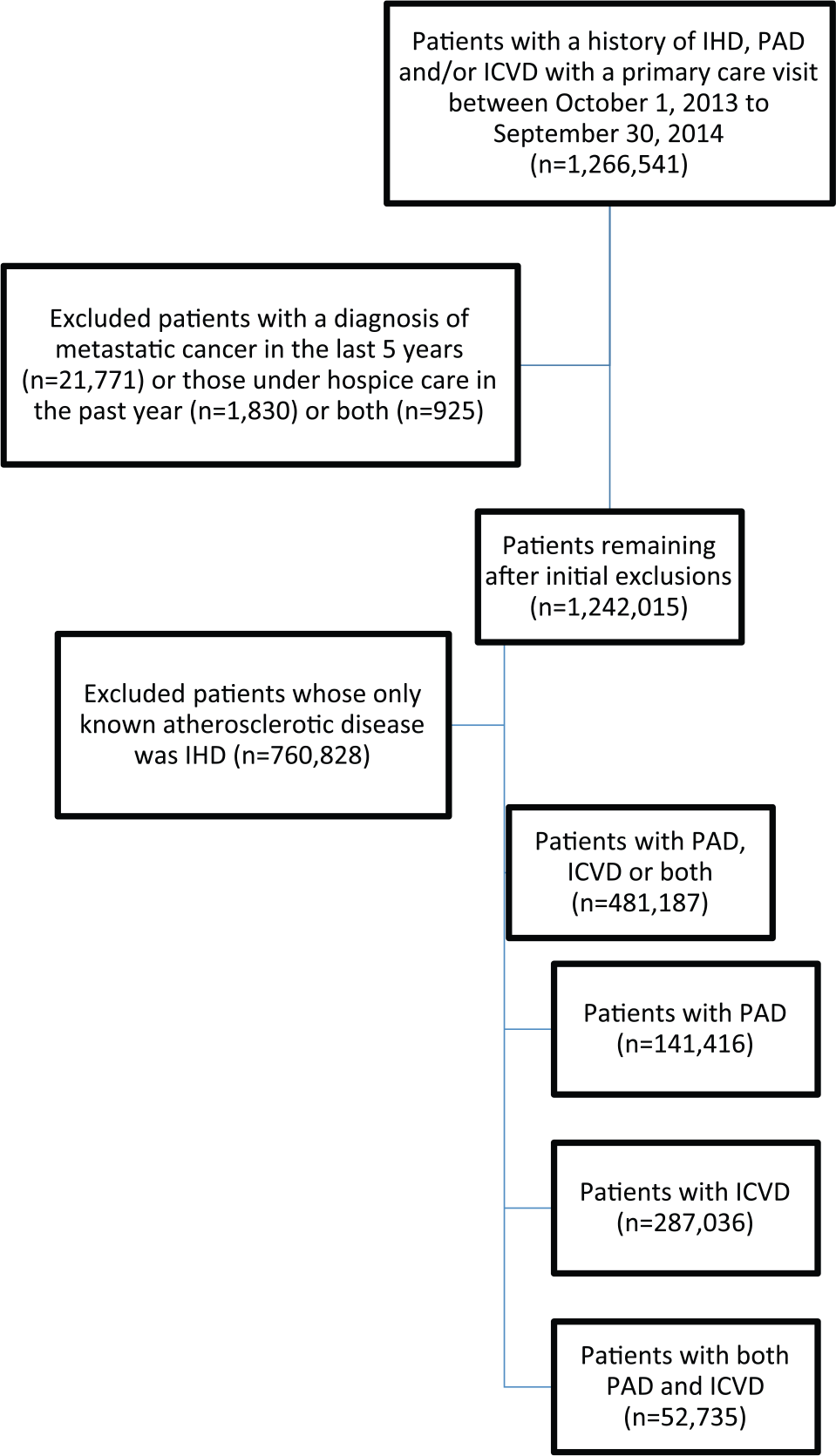
Flow chart of the study cohort identification and exclusions.
Covariate assessment
We first evaluated patient characteristics, including their age, race, sex, history of hypertension or diabetes, and the number of primary care visits in the 12 months prior to the index primary care visit. To assess the impact of a patient’s overall illness burden on evidence-based statin prescribing, we calculated a Diagnostic Cost Group (DCG) Relative Risk Score (RRS) for each PAD or ICVD patient in our cohort. DCG RRS is a ratio of predicted to mean cost and has been used to assess the impact of a patient’s overall illness burden in prior studies.8–10,16,17 DCG RRS are calculated for each patient from diagnoses derived from more than 15,000 ICD-9-CM codes classified into clinically similar groups called condition categories. These categories are ranked based on severity to form hierarchical condition categories (HCCs). These HCCs have been used to develop a custom regression model of VA costs, which produced the DCG RRS for each patient, with a score of 1 being the mean of the population. As an example, a patient with a DCG RRS of 1.5 is expected to incur 50% more cost than an average patient. We also assessed facility and provider-level variables, such as whether patients received their care in a teaching facility or a non-teaching facility, and whether their primary care provider (PCP) was a physician or an advanced practice provider, such as a nurse practitioner or a physician assistant.
Statin use assessment
Statin prescription and statin intensity were assessed up to 100 days prior to a patient’s index primary care visit using outpatient medication lists, which are on record for each patient in the VA electronic medical record. Guideline-concordant intensity of statin therapy was defined as high-intensity statin for patients ≤75 and at least moderate-intensity statin for patients >75 years of age, as per the 2013 ACC/AHA cholesterol guideline. 5 High-intensity statin therapy was defined as 40–80 mg of atorvastatin daily or 20–40 mg of rosuvastatin daily. Moderate-intensity statin therapy included 10–20 mg of atorvastatin, 5–10 mg of rosuvastatin, 20–40 mg of simvastatin, 40–80 mg of pravastatin, 40 mg of lovastatin, 2–4 mg of pitavastatin, 80 mg of fluvastatin XL or 40 mg of fluvastatin daily.
Statistical analysis
We first examined the proportion of PAD or ICVD patients receiving a statin and guideline-concordant intensity of statin prescription. We also calculated facility-level rates of statin prescription in PAD or ICVD patients. PAD patients were grouped into three categories: PAD with or without IHD or ICVD (any PAD), PAD without IHD (can have concomitant ICVD) and PAD alone. Likewise, patients with ICVD were grouped into ICVD with or without IHD or PAD (any ICVD), ICVD without IHD (can have concomitant PAD) or ICVD alone. In each group, we identified the proportion of patients on a statin and guideline-concordant intensity of statin therapy. We then determined the percentage of patients in each category, at each facility, who were on a statin and guideline-concordant intensity of statin therapy. We assessed median facility-level rates (and interquartile intervals (IQI)) for prescription of any statin therapy and guideline-concordant intensity of statin therapy.
Our definition of PAD includes several entities which some clinicians may consider non-typical presentations of PAD. These include the conditions corresponding to ICD-9-CM codes for arterial embolism and thrombosis (444) and atheroembolism (445) (Supplemental Table 1). Similarly, for our ICVD cohort, some clinicians may feel that retinal artery or vascular occlusion (362.30–362.34) or transient paralysis of a limb (781.4) do not fall strictly in the spectrum of stroke or transient ischemic attack (Supplemental Table 1). Because the natural history of these entities may differ from classic presentations of lower extremity PAD and ICVD, we performed sensitivity analyses after excluding these ICD-9-CM codes from the rest of the PAD and ICVD groups (Supplemental Tables 3 and 4).
To investigate whether there was variation in the rates of statin prescription across VA facilities independent of patient-level factors, we constructed multivariable hierarchical regression models to determine median rate ratio (MRR). These two-level hierarchical models adjusted for clustering of patients within facilities and modeled each individual facility as a random effect and patient characteristics as filter effects within each facility. 18 MRR allows for control of confounding between facilities so that patients with similar characteristics are compared to each other. The MRR serves as an index of variability in the rate of any statin prescription and guideline-concordant intensity of statin prescription while preventing confounding between two given facilities. It is a metric that quantifies the degree of variation in clinical practice between two given VA primary care sites in a way that controls for the typical illness burden of patients at a facility, patient age, sex, race, and other demographic factors. As an example, an MRR of 1.5 reflects a 50% likelihood that two facilities would prescribe evidence-based statins in a different way to similar patients. The use of MRR to assess variation in care has been previously described.9,19,20 First, an unadjusted model of MRR was constructed. Subsequently, we created models adjusting for the patient’s age, sex, race, presence of hypertension or diabetes, DCG RRS (marker of a patient’s illness burden), receipt of primary care at a teaching versus non-teaching VA facility, and number of primary care visits 1 year prior to the index visit to assess facility-level variation in statin prescription independent of patient and some provider-level factors. Based on the methodology above, we created unadjusted and adjusted MRRs for each category of PAD and ICVD patients.
Our analyses were conducted using SAS software, version 9.1.3 (SAS Institute Inc., Cary, NC, USA) and Stata statistical software, release 11 (StataCorp LP, College Station, TX, USA). The protocol was approved by the Institutional Review Boards at Baylor College of Medicine and the Michael E DeBakey VA Medical Center. No informed consent was required.
Results
A total of 481,187 patients in our cohort met the inclusion criteria which had PAD, ICVD, or both. Table 1 describes the baseline characteristics of these patients. The mean age was 71.8 ± 10.7 years; hypertension was present in 410,878 (85.4%) and diabetes was present in 221,958 (46.1%) of these patients. Overall, 361,303 (75.1%) were white and 68,661 (14.3%) were African American. A total of 205,234 (42.7%) of these patients were receiving care at a teaching facility and 78% were receiving care from a physician provider. This group of 481,187 patients had an overall high illness burden (mean DCG RRS of 2.4).
Baseline patient characteristics of the study population (n=481,187 with PAD, ICVD, or both).
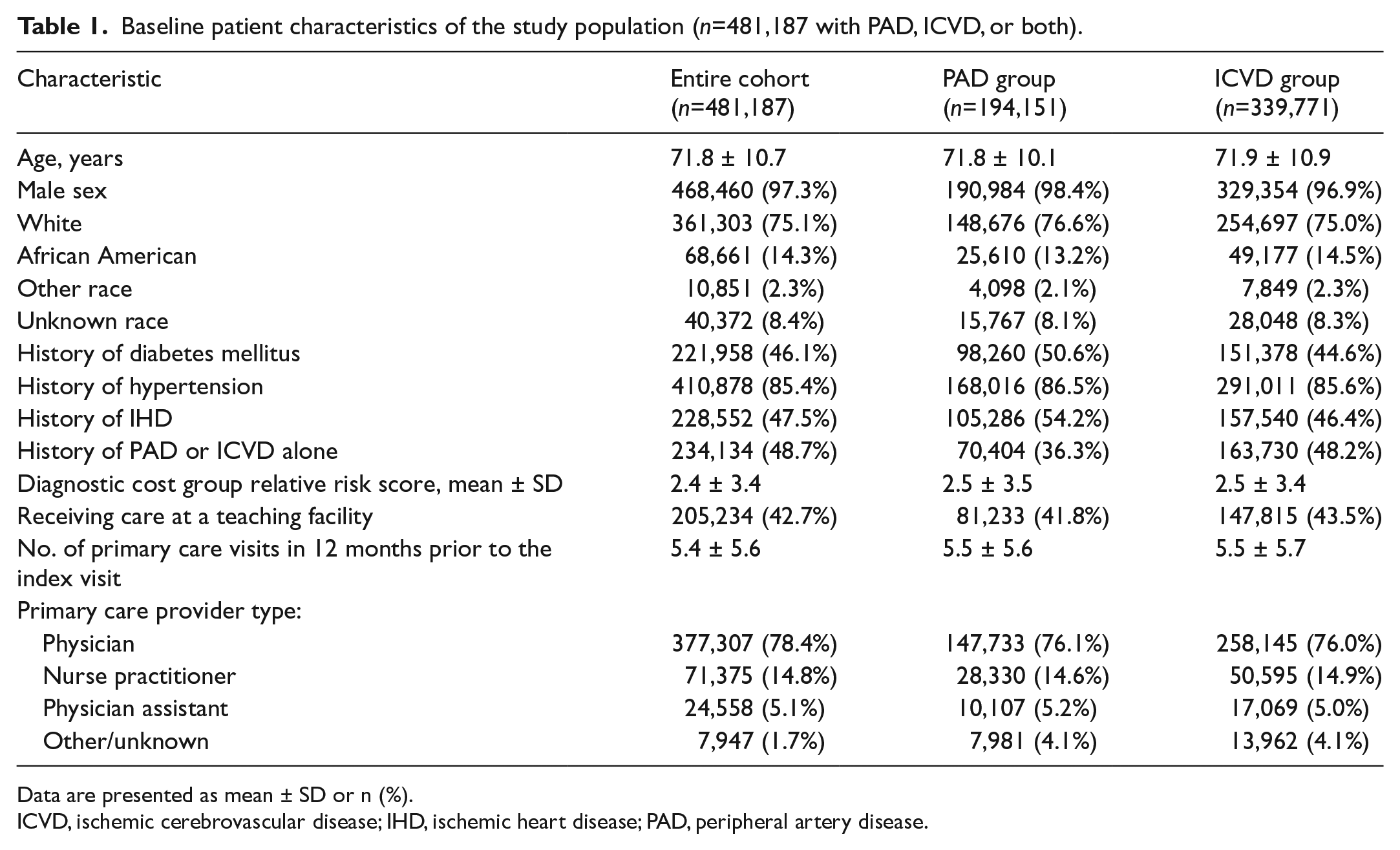
Data are presented as mean ± SD or n (%).
ICVD, ischemic cerebrovascular disease; IHD, ischemic heart disease; PAD, peripheral artery disease.
Statin prescription
There were 194,151 patients with PAD (with or without IHD or ICVD), 88,905 with PAD without IHD, and 70,404 with PAD without IHD or ICVD. The proportion of these groups on statin therapy is described in Table 2 for the entire PAD group and for each subgroup of PAD patients. For the entire PAD group, 153,438 patients (79.0%) were prescribed a statin, and those with PAD without IHD (with or without ICVD) had a 69.1% prescription rate (n=61,385). The lowest proportion of patients prescribed a statin was noted for the PAD group without any concomitant IHD or ICVD; patients with PAD alone were prescribed statins in 66.3% of cases (n=46,694).
Prescription rates of statin therapy and their facility-level variation in patients with PAD.
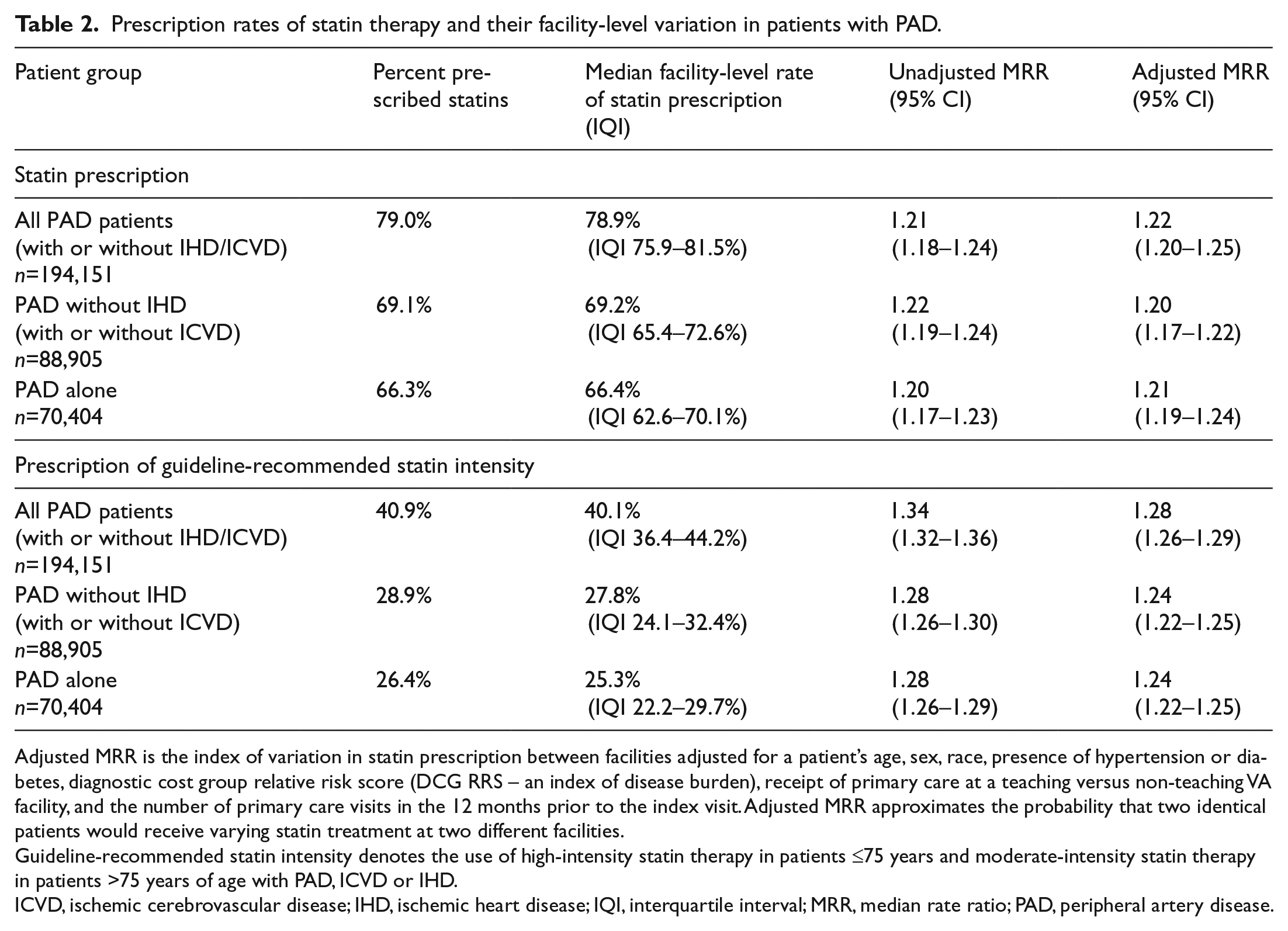
Adjusted MRR is the index of variation in statin prescription between facilities adjusted for a patient’s age, sex, race, presence of hypertension or diabetes, diagnostic cost group relative risk score (DCG RRS – an index of disease burden), receipt of primary care at a teaching versus non-teaching VA facility, and the number of primary care visits in the 12 months prior to the index visit. Adjusted MRR approximates the probability that two identical patients would receive varying statin treatment at two different facilities.
Guideline-recommended statin intensity denotes the use of high-intensity statin therapy in patients ≤75 years and moderate-intensity statin therapy in patients >75 years of age with PAD, ICVD or IHD.
ICVD, ischemic cerebrovascular disease; IHD, ischemic heart disease; IQI, interquartile interval; MRR, median rate ratio; PAD, peripheral artery disease.
Our analysis included 339,771 patients with ICVD. Of those, 182,231 (53.6%) had ICVD without IHD and 163,730 (48.2%) had ICVD without IHD or PAD. The proportion of patients on statin therapy is described in Table 3 for the entire ICVD group and for each subgroup of ICVD patients. For the entire ICVD group, 265,491 (78.1%) were prescribed a statin. The rates of statin prescription for patients with ICVD without IHD (with or without PAD) and ICVD alone (without IHD or PAD) was 70.9% and 69.9%, respectively (n=129,140 and n=114,449, respectively). As noted, the lowest proportion of patients receiving a statin was noted in ICVD patients without IHD or PAD.
Prescription rates of statin therapy and their facility-level variation in patients with ICVD.
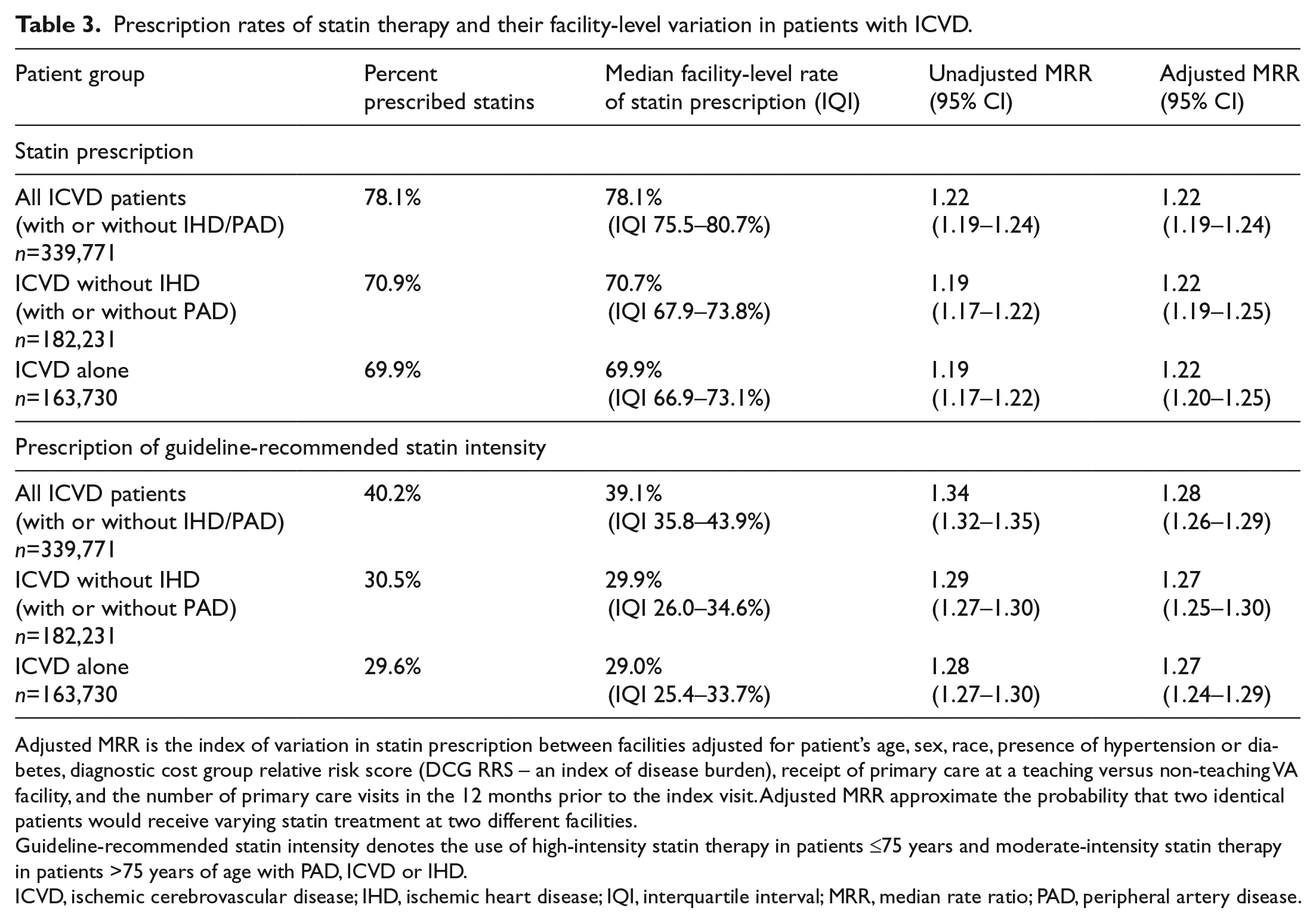
Adjusted MRR is the index of variation in statin prescription between facilities adjusted for patient’s age, sex, race, presence of hypertension or diabetes, diagnostic cost group relative risk score (DCG RRS – an index of disease burden), receipt of primary care at a teaching versus non-teaching VA facility, and the number of primary care visits in the 12 months prior to the index visit. Adjusted MRR approximate the probability that two identical patients would receive varying statin treatment at two different facilities.
Guideline-recommended statin intensity denotes the use of high-intensity statin therapy in patients ≤75 years and moderate-intensity statin therapy in patients >75 years of age with PAD, ICVD or IHD.
ICVD, ischemic cerebrovascular disease; IHD, ischemic heart disease; IQI, interquartile interval; MRR, median rate ratio; PAD, peripheral artery disease.
Statin intensity
For PAD patients, 79,435 (40.9%) were prescribed guideline-concordant intensity of statin therapy at the time of announcement and initial implementation of the 2013 ACC/AHA guideline. The rate of guideline-concordant intensity of statin prescriptions for patients with PAD without IHD (with or without ICVD) was 28.9% (n=25,646) and 26.4% (n=18,598) for patients with PAD alone.
For ICVD patients, 136,430 (40.2%) were prescribed guideline-concordant intensity of statin therapy. Guideline-concordant statin therapy was being prescribed to 30.5% (n=55,528) of patients with ICVD without IHD (with or without PAD) and to 29.6% (n=48,480) of patients with ICVD alone. PAD and ICVD patients who had no other concomitant ASCVD had the lowest proportion of patients on the guideline-concordant statin intensity.
Facility-level rates of statin prescription and variation
Median facility-level rates of any statin prescription for all PAD patients (with or without IHD and/or ICVD), PAD patients without IHD (with or without ICVD) and patients with PAD alone were 78.9% (IQI 75.9–81.5%), 69.2% (IQI 65.4–72.6%) and 66.4% (IQI 62.6–70.1%), respectively. The median facility-level rates for guideline-concordant intensity of statin prescription were 40.1% (IQI 36.4–44.2%), 27.8% (IQI 24.1–32.4%) and 25.3% (IQI 22.2–29.7%), respectively. The unadjusted MRR for the entire PAD group reflected 20–22% facility-level in the rate of any statin prescription. The unadjusted MRR for guideline-concordant intensity of statin prescription in PAD patients demonstrated 28–34% variation between facilities. Neither the MRRs for any statin or the guideline-concordant intensity of statin therapy in the PAD group were much attenuated after adjusting for covariates. There was 20–28% variation in any statin or in guideline-concordant intensity of statin prescription among two similar PAD patients receiving care at two random facilities (Table 2).
Median facility-level rates of any statin prescription for all ICVD patients (with or without IHD and/or PAD), ICVD patients without IHD (with or without PAD) and patients with ICVD alone were 78.1% (IQI 75.5–80.7%), 70.7% (IQI 67.9–73.8%) and 69.9%% (IQI 66.9–73.1%), respectively. Median facility-level rates for guideline-concordant intensity of statin prescription were 39.1% (IQI 35.8–43.9%), 29.9% (IQI 26.0–34.6%) and 29.0% (IQI 25.4–33.7%), respectively. The unadjusted MRR for any statin and guideline-concordant intensity of statin prescription reflected an overall variation of 19–22% and 28–34%, respectively. MRRs adjusted for covariates were not attenuated for any statin prescription compared to unadjusted MRR and were only slightly attenuated for guideline-concordant intensity of statin prescription (27–28% variation). Overall, there was 22–28% variation in statin prescription or guideline-concordant intensity of statin prescription among two similar ICVD patients receiving care at two random facilities (Table 3).
The demographics, comorbidities and location at which the patients in our cohort sought primary care altered their likelihood of receiving appropriate statin therapy. For patients in both the PAD and ICVD group, they were more likely to receive any statin and guideline-concordant statin therapy if they had hypertension, diabetes, ischemic heart disease or an increasing number of primary care visits during the study period. Patients in both groups were less likely to be prescribed any statin as their age increased or if they received primary care at a teaching facility. In both the PAD and ICVD groups, women and patients with higher DCG RRS demonstrated a reduced likelihood of being prescribed any statin and a guideline-concordant statin (Table 4).
Odds ratios expressing likelihood of any statin and guideline-concordant statin prescription based on baseline characteristics of patients.
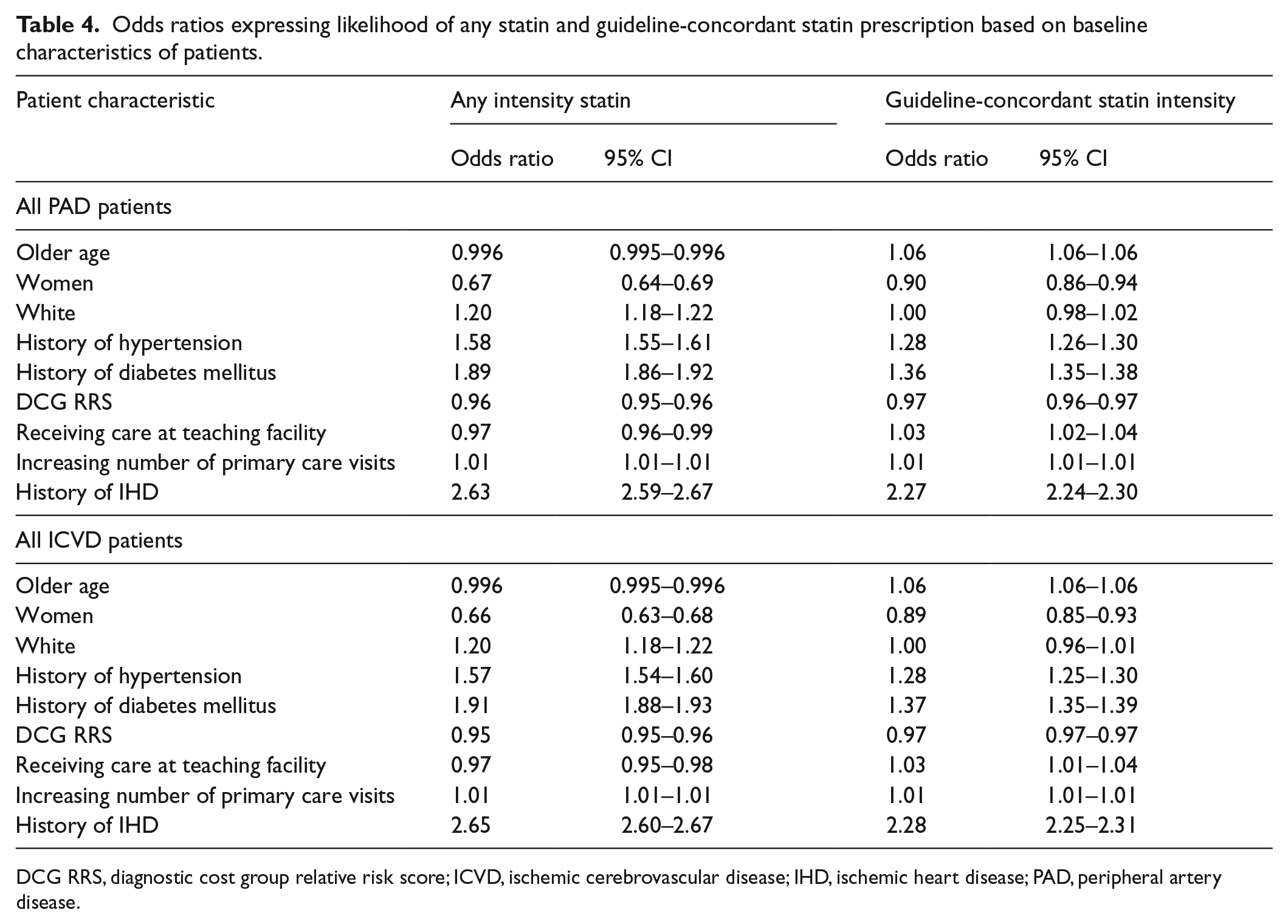
DCG RRS, diagnostic cost group relative risk score; ICVD, ischemic cerebrovascular disease; IHD, ischemic heart disease; PAD, peripheral artery disease.
By excluding patients with ICD-9-CM codes for arterial embolism and thrombosis of peripheral arteries and atheroembolism, the number of patients in the PAD group was reduced by 3,359, leaving 190,792 PAD patients. In the ICVD group, exclusion of codes 362.30–362.34 and 781.4 resulted in the reduction of the group size by 15,039 patients, leaving 324,732 patients with ICVD. Our sensitivity analyses in which we analyzed rates of any statin and guideline-concordant statin after exclusion of these codes were nearly identical to the aforementioned results in which these codes were included (Supplemental Tables 3 and 4).
Discussion
In our analyses of 130 VA facilities across the country, approximately one-fifth of patients with PAD and ICVD were not prescribed a statin, and roughly 60% were not on guideline-concordant intensity of statin therapy. For both PAD and ICVD patients, both statin use and guideline-concordant intensity of statin use was lowest among patients without concomitant IHD. Patients with PAD or ICVD alone had the lowest likelihood of being prescribed a statin, or especially, guideline-concordant intensity of statin therapy. In fact, patients with PAD alone and ICVD alone were prescribed guideline-concordant intensity of statin therapy in only 26.4% and 29.6% of cases, respectively. We found significant facility-level variation in statin prescribing patterns, which were minimally explained by patient-level factors. Female patients with PAD and ICVD were less likely than males to receive any and guideline-concordant statin therapy, as were patients with higher DCG RRS scores. We have previously described a similar disparity in female patients with ASCVD receiving optimal medical therapy. 8 The finding that patients with greater illness burden, represented by higher DCG RRS scores, receive less appropriate statin therapy and that older age is associated with less overall statin prescription is congruent with the previously described treatment-risk paradox. 21
The low rates of guideline-concordant intensity of statin therapy reveal that a majority of patients with ICVD and PAD would require alteration of their statin prescription (whether to increase intensity or begin prescribing at all) to attain compliance with the guideline. This analysis demonstrates the significant challenge and opportunity for changing practice. Because the data for this study were obtained during the fiscal year of the announcement of the 2013 ACC/AHA guideline, an important future direction for this work includes repeating the same analysis with a more contemporary cohort to assess whether guideline compliance improved over time.
Patients with ASCVD, and especially those with PAD, are undertreated with statins. In the Reduction of Atherothrombosis for Continued Health (REACH) registry, which comprised data from 67,888 patients across 44 countries with ASCVD, roughly 36% of patients with PAD and 44% of the patient with ICVD were not receiving statins. 22 One study of patients from 350 primary care practices included in the PAD Awareness, Risk and Treatment: New Resources for Survival (PARTNERS) program reported that patients in their dataset were only prescribed anti-hyperlipidemic medications in 56% of cases and a subset of these patients were on anti-lipid drugs other than statins. 23 A recent analysis of United States ambulatory care data between 2006 and 2013 from the National Ambulatory Medical Care Survey (NAMCS) and National Hospital Ambulatory Medical Care Survey (NHAMCS) described similar statin prescribing patterns. Over the 8 years studied, statin use in PAD patients rose from 30.5% in 2006–2007 to 38.8% in outpatient visits occurring in 2012–2013. 24 Comparatively, 79.0% of PAD patients and 78.1% of ICVD patients in our analyses were prescribed a statin, which is markedly better than the contemporary overall US rate described by the NAMCS and NHAMCS analyses. However, the use of guideline-concordant intensity of statin therapy remains suboptimal in this high-risk secondary prevention group.
The presence of IHD in our cohort had an effect on the prescribed statin therapy. The likelihood that a patient with PAD or ICVD was prescribed any statin or guideline-concordant intensity of statin therapy was lower if they did not have concomitant IHD. Furthermore, it decreased even further if they had PAD or ICVD in the absence of other known atherosclerotic disease. This suggests that clinicians more aggressively treat to reduce the risk of cardiovascular events and adhere more closely to statin prescribing guidelines in PAD or ICVD patients when they have concomitant IHD. Primary care providers may perceive that PAD or ICVD patients who do not have known concomitant IHD have a lower risk of serious cardiovascular events and therefore do not need aggressive medical therapy. This is supported by prior work showing a significantly lower prescription of evidence-based therapies in patients with isolated PAD or ICVD compared with those with IHD. 10 International data from the REACH registry reported that 76.2% of IHD patients in their cohort received statin therapy. In comparison, statin therapy was prescribed in 56.4% and 64.2% of patients with ICVD and PAD, respectively. 22 This phenomenon has even been demonstrated in patients undergoing percutaneous vascular intervention (PVI) for PAD during a hospital admission. Krishnamurthy and colleagues reported rates of prescription of cardioprotective medications, including statins, before and after PVI in a cohort of 4459 patients with critical limb ischemia. Patients with PAD alone were prescribed a statin less frequently than patients who also had concomitant ICVD or IHD both pre-procedure (51.8% vs 76.1%, respectively; p<0.0001) and post-procedure (61.5% vs 80.0%, respectively; p<0.0001). 25 Note that this occurred despite both groups of patients having PAD of a severity warranting invasive management.
Ideally, statin prescribing patterns across VA facilities should be uniform and guideline directed, but our calculated MRRs reveal heterogeneity in practice patterns. Unadjusted MRRs showed significant variation among facilities in prescribing patterns for any statins and guideline-concordant statin intensity in PAD and ICVD patients. MRRs did not decrease significantly after adjusting for patient level factors, suggesting that differences in patient populations across facilities does not account for much of the variation seen in our study. These results therefore suggest that this variation in evidence-based statin prescribing patterns at these facilities is likely related to practice patterns of individual providers at these facilities, rather than patient factors. System-level interventions could be used to address the deficiencies in statin prescription. A recent multi-center study of 10,169 patients who underwent inpatient PVI found that prescription of cardioprotective medications at the time of discharge significantly increased the frequency of continued outpatient use of these medications at 6 months post-discharge. In this study, 21.8% of those not prescribed a statin at discharge were on statin therapy at the follow-up versus 84.6% of those who were prescribed statin therapy at discharge (p<0.001). 26 In the absence of contraindications, inpatients with ASCVD should receive evidence-based medical therapies, including statins, before leaving the hospital.
Study limitations
Our results should be considered in the context of the limitations of our study. Because patients who seek care at the VA are a predominately male population, our findings may not be generalizable to health care systems with larger proportions of female patients. Although this is possible, our analyses included 12,727 women with PAD or ICVD. One could argue that some VA patients could be receiving statin medications from health care providers outside the VA system. Although this is possible, our analyses accounted for non-VA medications entered by primary care providers in the electronic medical records. Our analyses were based on statin-prescribing patterns for PAD and ICVD patients, but did not assess patient compliance with taking the prescribed medications. Data comprising patients’ smoking status were not used for the adjustment of MRR. In our definition of PAD, we included ICD-9-CM codes associated with atherosclerosis and thrombosis of arteries in the thoracic and abdominal cavities including the renal arteries, thoracic aorta and abdominal aorta, which are not universally treated as a single group in other studies. The natural histories, clinical implications and potential treatment strategies for these pathologies may differ and therefore providers caring for patients with these conditions may not have approached these patients with the same medical strategy as they would for a patient with classic lower extremity PAD. This may make our reported statin prescription rates for PAD patients less comparable to other studies whose focus is medical therapy in lower extremity PAD. The timing of our data collection was during the announcement and initial year of implementation of the 2013 ACC/AHA guideline for management of serum cholesterol; therefore, our results show early implementation of the 2013 ACC/AHA cholesterol guideline. The ICD diagnosis and procedure codes used to identify patients with PAD are meant to be comprehensive and include several codes which are not exclusively used for PAD patients; an example are codes for arterial thrombosis and embolism of the aorta and extremities. We believe, however, that using these codes for inclusion along with the exclusion codes mentioned in Supplementary Table 2 allows us to collect data for a nearly complete list of veteran patients with PAD with acceptable specificity. We’ve previously found that these codes in combination identify PAD patients with 88% positive predictive value. 10 Finally, some of this treatment could be related to partial or complete statin intolerance which is not well captured in structured datasets like ours. The strengths of our study include a large number of patients with PAD and ICVD seeking care in the entire VA health care system with good ascertainment of patient characteristics using well-validated VA national data sources.
Conclusion
The use of statin therapy is suboptimal in patients with PAD and ICVD, especially among those without concomitant IHD. In our cohort, a majority of patients with PAD and ICVD without other known atherosclerotic disease required a change in statin prescription and statin intensity to achieve guideline compliance. There is also a significant facility-level variation in statin prescribing which is minimally explained by patient-level factors. System-level interventions are needed to improve the quality of secondary prevention among patients with PAD or ICVD and to reduce variation in care. Because the data for this study overlapped with the announcement of the 2013 ACC/AHA guideline, an important future direction for this work includes repeating the same analysis with a more contemporary cohort to assess whether guideline compliance has improved over time.
Supplemental Material
VMJ758914_Supplementary_tables – Supplemental material for Statin prescription rates and their facility-level variation in patients with peripheral artery disease and ischemic cerebrovascular disease: Insights from the Department of Veterans Affairs
Supplemental material, VMJ758914_Supplementary_tables for Statin prescription rates and their facility-level variation in patients with peripheral artery disease and ischemic cerebrovascular disease: Insights from the Department of Veterans Affairs by Cameron L McBride, Julia M Akeroyd, David J Ramsey, Vijay Nambi, Khurram Nasir, Erin D Michos, Ruth L Bush, Hani Jneid,Pamela B Morris, Vera A Bittner, Christie M Ballantyne, Laura A Petersen and Salim S Virani in Vascular Medicine
Footnotes
Acknowledgements
The views expressed in this article are those of the authors and do not necessarily represent the views of the Department of Veterans Affairs.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Nambi reported receiving a research grant from Merck (paid to Baylor College of Medicine), reported serving as a consultant to and on an advisory board for Sanofi-Regeneron, reported receiving an honorarium for event adjudication for a clinical trial by Siemens Healthcare Diagnostics and reported holding provisional patent 61721475 entitled ‘Biomarkers to Improve Prediction of Heart Failure Risk’ filed by Baylor College of Medicine and Roche. Dr Morris reported serving as a consultant for Amgen, AstraZeneca, Sanofi-Regeneron and having received research funding from Amgen. Dr Nasir reported serving as a consultant for Sanofi-Regeneron and serving on the advisory board for Quest Diagnostics. Dr Michos reported receiving an honorarium for event adjudication for a clinical trial by Siemens Healthcare Diagnostics. Dr Bittner reported serving on steering committees (her institution [University of Alabama at Birmingham] was compensated) for the ODYSSEY trial (Sanofi-Regeneron) and for development of the CETP inhibitor (Eli Lilly), reported being national coordinator of the STRENGTH trial (AstraZeneca), reported serving on advisory boards for Eli Lilly and Amgen, reported being co-investigator on a University of Alabama School of Public Health–Amgen contract related to Medicare analyses, and reported holding the position of past local site principal investigator for a Pfizer Inc. Studies of PCSK9 Inhibition and the Reduction of Vascular Events (SPIRE) trial. Dr Ballantyne reported receiving grant and research support (all paid to Baylor College of Medicine) from Abbott Diagnostics, Amarin, Amgen, Eli Lilly, Esperion, Novartis, Pfizer Inc., Otsuka, Regeneron, Roche Diagnostics, Sanofi-Synthelabo, Takeda, National Institutes of Health, American Heart Association and American Diabetes Association, and reported serving as a consultant to Abbott Diagnostics, Amarin, Amgen, AstraZeneca, Eli Lilly, Esperion, Genzyme, Isis, Matinas BioPharma Inc., Merck, Novartis, Pfizer Inc., Regeneron, Roche, and Sanofi-Synthelabo. Dr Virani reported serving on the steering committee (with no financial remuneration) for the Patient and Provider Assessment of Lipid Management (PALM) Registry at the Duke Clinical Research Institute.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the American Heart Association Beginning Grant-in-Aid (14BGIA20460366) and the American Diabetes Association Clinical Science and Epidemiology award (1-14-CE-44). This work was also supported by the Houston VA HSR&D Center for Innovations grant (HFP 90-020) and the VA HSR&D Investigator Initiated Grant (IR 16-072).
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
