Abstract
Studies have reported an association between obstructive sleep apnea (OSA) and cardiovascular disease (CVD) morbidity and mortality. Proposed mechanisms include endothelial dysfunction and atherosclerosis. We aimed to investigate the associations of OSA with endothelial dysfunction and subclinical atherosclerotic coronary artery disease (CAD), and assess the impact of race on these associations. We used data from the Heart Strategies Concentrating on Risk Evaluation (Heart SCORE) study, a community-based prospective cohort with approximately equal representation of black and white participants. OSA severity was measured in 765 individuals using the apnea-hypopnea index (AHI). Endothelial dysfunction was measured using the Endo-PAT device, expressed as Framingham reactive hyperemia index (F_RHI). Coronary artery calcium (CAC), a marker of subclinical CAD, was quantified by electron beam computed tomography. There were 498 (65%) female participants, 282 (37%) black individuals, and 204 (26%) participants with moderate/severe OSA (AHI ≥15). In univariate models, moderate/severe OSA was associated with lower F_RHI and higher CAC, as well as several traditional CVD risk factors including older age, male sex, hypertension, diabetes, higher body mass index, and lower high-density lipoprotein cholesterol levels. In a multivariable model, individuals with moderate/severe OSA had 10% lower F_RHI and 35% higher CAC, which did not reach statistical significance (p=0.08 for both comparisons). There was no significant interaction of race on the association of OSA with F_RHI or CAC (p-value >0.1 for all comparisons). In a community-based cohort comprised of black and white participants, moderate/severe OSA was modestly associated with endothelial dysfunction and subclinical atherosclerotic CAD. These associations did not vary by race.
Keywords
Background
Obstructive sleep apnea (OSA), a disorder characterized by the repetitive interruption of ventilation during sleep due to complete or partial airway collapse, affects at least 15 million people in the United States 1 and is associated with an increased prevalence of cardiovascular disease (CVD),2,3 including hypertension,4–7 coronary artery disease (CAD),8,9 cardiac arrhythmia,10–13 heart failure, 14 and stroke.15–17 Although the mechanism of these associations is not completely understood, it may be related to downstream effects of OSA-induced chronic intermittent hypoxia, sleep fragmentation, and, to a lesser extent, changes in intra-thoracic pressure. 18 Epidemiologic data indicate that endothelial dysfunction, a well-established early manifestation of atherosclerosis, 19 may modulate that association between OSA and the genesis or worsening of CVD.20–25 It has been proposed that intermittent hypoxia and rapid reoxygenation result in sympathetic excitation, inflammation, and oxidative stress/reduced nitric oxide bioavailability, all of which ultimately contribute to endothelial dysfunction and downstream subclinical and clinical atherosclerosis.26–28 In addition to endothelial dysfunction, the presence and severity of OSA has been independently associated with the degree of coronary artery calcium (CAC) burden, another marker of subclinical atherosclerosis. 29 The extent of CAC has been shown to correlate with atherosclerotic plaque and future cardiac events.30,31 Therefore, understanding the association between OSA and markers of subclinical atherosclerosis may help guide therapeutic interventions to prevent or reduce CVD morbidity and mortality in individuals with OSA.
Although several studies have found OSA to be more prevalent and more severe in black compared to white individuals,32–34 large community-based studies linking OSA severity to markers of subclinical atherosclerosis in a racially diverse population are limited.35,36 In the Multi-Ethnic Study of Atherosclerosis (MESA) prospective cohort (28% black), OSA was associated with CAC score progression over a period of 8 years after adjustment for demographics, behaviors, and body mass index. 35 Subsequent stratified analysis suggested possible effect modification by race. However, among black participants, there was no difference in CAC progression between those with OSA and those without OSA. 35 In another analysis of data from MESA, participants who reported physician-diagnosed OSA did not demonstrate a clinically important reduction in brachial artery flow-mediated dilation (FMD), a measure of macrovascular endothelial function. Of note, the diagnosis of OSA in the MESA study was based on self-reported information, which may be greatly influenced by recall bias. Our group has previously shown that black race is independently associated with microvascular endothelial dysfunction. 37 To our knowledge, there are no studies assessing the independent association of OSA with microvascular endothelial dysfunction and advanced subclinical atherosclerosis together in a racially diverse population. Whether the association of OSA with endothelial dysfunction or CAC varies by race remains unknown. Accordingly, the present study sought to investigate the independent associations of OSA with microvascular endothelial function and CAC, and examine the potential interaction effect of race on these associations in a community-based, prospective cohort of racially diverse participants in the Heart Strategies Concentrating On Risk Evaluation (Heart SCORE) study.
Methods
Study population
Heart SCORE is an ongoing community-based prospective cohort study of the greater Pittsburgh metropolitan area. It is comprised of 1933 adults (43% black, 54% female) and is aimed at identifying population differences, including racial disparities, in CVD outcomes. 38 Eligibility criteria included adults aged 45–75 years who could undergo annual follow-up visits and were without a comorbid condition that would limit life expectancy to <5 years. The full methodology has been previously described in other Heart SCORE studies.38,39 The present study was confined to 765 individuals (37% black, 65% female) who underwent overnight evaluation for OSA using an ApneaLink Plus™ (ResMed Corp., San Diego, CA, USA) portable home monitor. Individuals not previously diagnosed with OSA were included and those on home continuous positive airway pressure or home oxygen therapy were excluded. The University of Pittsburgh’s Institutional Review Board approved the protocol and all participants provided written informed consent.
Data collection
Detailed demographic data, medical histories and information on lifestyle characteristics were collected at a baseline visit. Race was self-reported. Physical examination included vital signs and anthropometric measurements. Body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared. Diabetes mellitus was defined as a fasting glucose of ≥126 mg/dL or a history of previously diagnosed diabetes treated with diet, oral agents, and/or insulin. Blood pressure was measured using a standard protocol, as previously described. 40 Hypertension was defined as a systolic blood pressure of 140 mmHg or a diastolic pressure of 90 mmHg, history of physician-diagnosed hypertension, or current use of anti-hypertensive medication. Data on smoking were self-reported. At the baseline visit, blood samples were drawn for assessment of lipid panel, fasting glucose concentrations and inflammatory biomarkers: high-sensitivity C-reactive protein (hsCRP) and interleukin-6 (IL6).
Obstructive sleep apnea assessment
A total of 765 individuals underwent overnight evaluation for OSA using an ApneaLink Plus portable home monitor. The ApneaLink Plus is a well-validated tool for identifying OSA in the unattended testing environment at home.41,42 Nasal airflow was measured using a nasal cannula connected to a nasal pressure transducer for the identification of snoring and other respiratory events. Respiratory efforts were measured by inductance plethysmography using a single abdominal band. Oxygen saturation and heart rate were measured with digital pulse oximetry. Studies were considered acceptable if they had a minimum of a 2-hour signal duration as monitored by nasal pressure. The scoring algorithm of the ApneaLink Plus software, version 7.0, which has been shown to be an accurate method for scoring, was used without editing. 43
Hypopneas are defined as a 30–80% reduction in nasal pressure flow for at least 10 seconds with a concomitant oxygen desaturation of ≥4%. Apneas were defined as a flow reduction ≥80% in the nasal pressure signal for at least 10 seconds. Oxygen desaturation was scored if there was a ≥4% drop. An apnea-hypopnea index (AHI), defined as the number of apneas and hypopneas per hour of analyzed recording time, was used to risk stratify the severity of OSA. OSA severity was categorized into no/mild OSA (AHI < 15 events/h), moderate OSA (AHI 15–29 events/h), and severe OSA (AHI ≥ 30 events/h).
Assessment of endothelial function
Endothelial function was measured using an Endo-PAT2000 device (Itamar Medical, Caesarea, Israel) as previously reported using the protocol described by the Framingham Heart Study. 44 Subjects fasted and refrained from smoking for at least 8 hours prior to measurement of endothelial function. Home medications were not withheld. Digital pulse amplitude was measured using the peripheral arterial tonometry (PAT) device placed on the tip of both the left and right index fingers. One finger served as the study and the other as the control finger. The baseline PAT signal was measured for 5 minutes on both fingers. Arterial flow was then interrupted on the study arm by occlusive pressure with a blood pressure cuff inflated to 60 mmHg above systolic blood pressure or at least 200 mmHg (whichever was higher). After 5 minutes of occlusion, the cuff was rapidly deflated and endothelium-mediated vasodilation was assessed as a PAT signal for an additional 5 minutes. Data were recorded electronically and analyzed using a computerized, automated algorithm (Itamar Medical). The PAT ratio, also known as the reactive hyperemia index (RHI), was calculated as follows: ratio of average pulse amplitude in the post-hyperemic phase of the occluded finger divided by the average baseline amplitude prior to inflation (normalized to the corresponding ratio of the non-occluded control finger). The Framingham reactive hyperemia index (F_RHI) was calculated as the natural log-transformation of the RHI. A lower F_RHI indicates a blunted hyperemic response, which would suggest poorer endothelial function. 44
Assessment of coronary artery calcium (CAC)
CAC was measured on cardiac-gated chest computed tomographic images using an electron-beam computed tomography scanner (Imatron C150 scanner; GE Imatron Inc., South San Francisco, CA, USA). To evaluate the coronary arteries, 30–40 contiguous 3-mm thick transverse images were obtained from the level of the aortic root to the apex of the heart during maximal breath holding. Images were acquired by using electrocardiogram triggering (80% of the RR interval) of 100-ms exposure during the same phase of the cardiac cycle. CAC scores were calculated by the Agatston method using a densitometric program. 45 Scans were interpreted by an experienced reader blinded to subject identities. An Agatston CAC score above zero is considered positive. A significant CAC score was defined as an Agatston score of >100, which corresponds to significant CAC burden, as previously described.45,46
Statistical methods
We assessed the correlates of OSA by summarizing several socio-demographic, biophysical and biochemical variables within two AHI groups representing no/mild OSA and moderate/severe OSA (AHI <15 events/h and AHI ≥15 events/h, respectively). Test parameters were z-values from regression of AHI on each covariate adjusted for age and sex. To assess cross-sectional association of OSA with F_RHI and CAC, we plotted median (interquartile range; IQR) values of the dependent variables across three categories of OSA (i.e. no/mild, moderate, and severe OSA). The p-values for these plots were determined using the non-parametric Kruskal–Wallis test. We used linear regression models to determine the magnitude of association of F_RHI and CAC with moderate/severe OSA. We assessed interaction of race on the association of OSA with F_RHI or CAC by fitting an interaction term in linear regression models. In order to investigate the shape of association between OSA, and F_RHI and CAC, we categorized OSA into quintiles and plotted the mean values of F_RHI and CAC across the quintiles. We then fitted the quintiles of OSA in a linear regression model as continuous and categorized variables. We performed a likelihood ratio test comparing the continuous and categorized models to determine if the shape of the association differed significantly from linearity. Because this test was not statistically significant, indicating that the shape of the association did not significantly deviate from linearity, we report the p-value from the continuous model as the p-value for linear trend. Adjustment was made for traditional CVD risk factors. Adjustment variables included age, sex, smoking status, blood pressure, diabetes, body mass index, total cholesterol and high-density lipoprotein cholesterol (HDL-c). All analyses were performed using Stata software, version 11 (StataCorp LP, College Stations, TX, USA). All p-values <0.05 were considered statistically significant.
Results
A total of 765 individuals (65% female, 37% black) had OSA severity assessed, with 204 (27%) having moderate/severe OSA (AHI ≥15 events/h). The prevalence of moderate/severe OSA among black participants was 22% versus 29% among white participants. Baseline characteristics of study participants by AHI severity (no/mild OSA vs moderate/severe OSA) are shown in Table 1. The mean age of the study population was 59 ± 7 years, while the mean F_RHI and CAC score were 0.76 ± 0.46 and 123 ± 210, respectively.
Baseline characteristics by apnea hypopnea index in the study population.
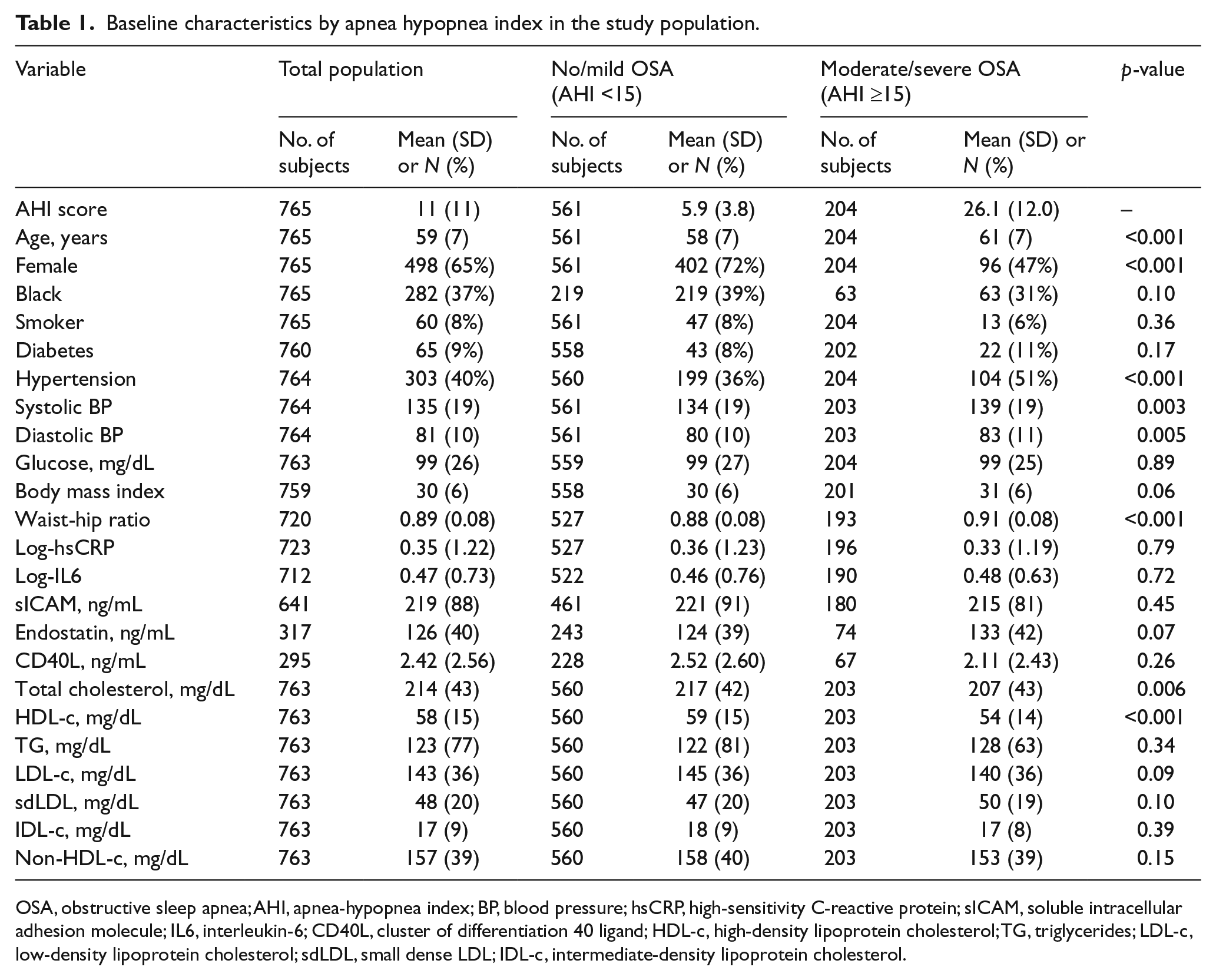
OSA, obstructive sleep apnea; AHI, apnea-hypopnea index; BP, blood pressure; hsCRP, high-sensitivity C-reactive protein; sICAM, soluble intracellular adhesion molecule; IL6, interleukin-6; CD40L, cluster of differentiation 40 ligand; HDL-c, high-density lipoprotein cholesterol; TG, triglycerides; LDL-c, low-density lipoprotein cholesterol; sdLDL, small dense LDL; IDL-c, intermediate-density lipoprotein cholesterol.
In a univariate model, OSA severity was directly associated with age (mean age 61 years in moderate/severe OSA vs 58 years in mild/no OSA), male sex (53% male in moderate/severe OSA vs 28% male in mild/no OSA), waist-hip ratio, hypertension, and diabetes, and inversely associated with HDL-c. OSA severity was not associated with race, current smoking status, or elevated inflammatory markers (hsCRP, IL6). Figure 1 depicts a box-plot of F_RHI and CAC score by OSA severity (no/mild OSA, moderate OSA, and severe OSA). As shown in Figure 1, individuals with more severe OSA had a significantly lower F_RHI and a higher CAC score (p-values 0.0080 and 0.0012, respectively). The F_RHI values (mean (IQR)) for the subgroups with no/mild, moderate, and severe OSA were 0.71 (0.43–1.16), 0.62 (0.4–0.8) and 0.57 (0.30–0.88), respectively. The corresponding values for CAC were 17 (0–107), 44 (8–143) and 101 (11–321), respectively (Figure 1).
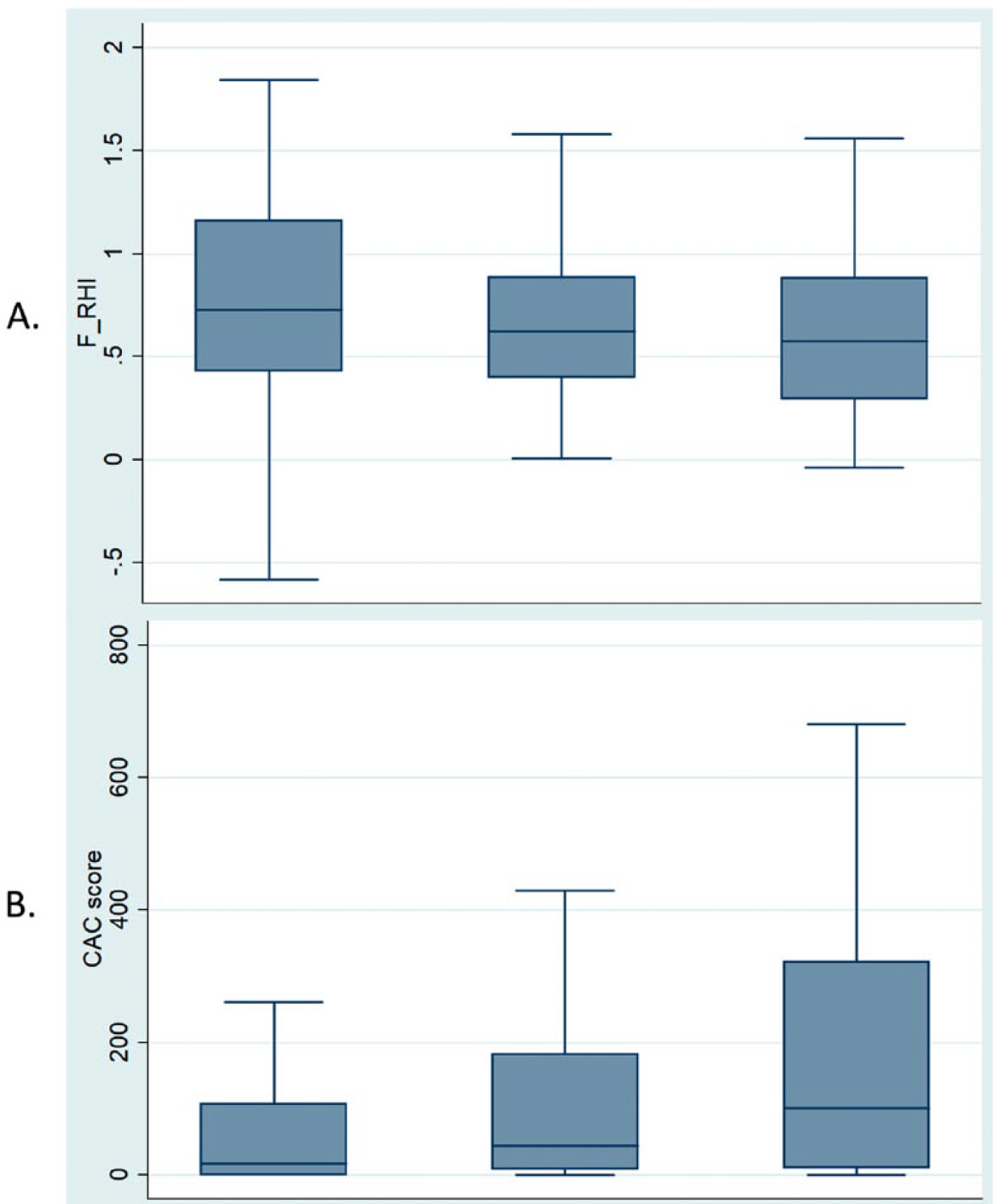
Median values of (A) Framingham reactive hyperemia index (F_RHI) and (B) coronary artery calcium (CAC) score by obstructive sleep apnea (OSA) status.
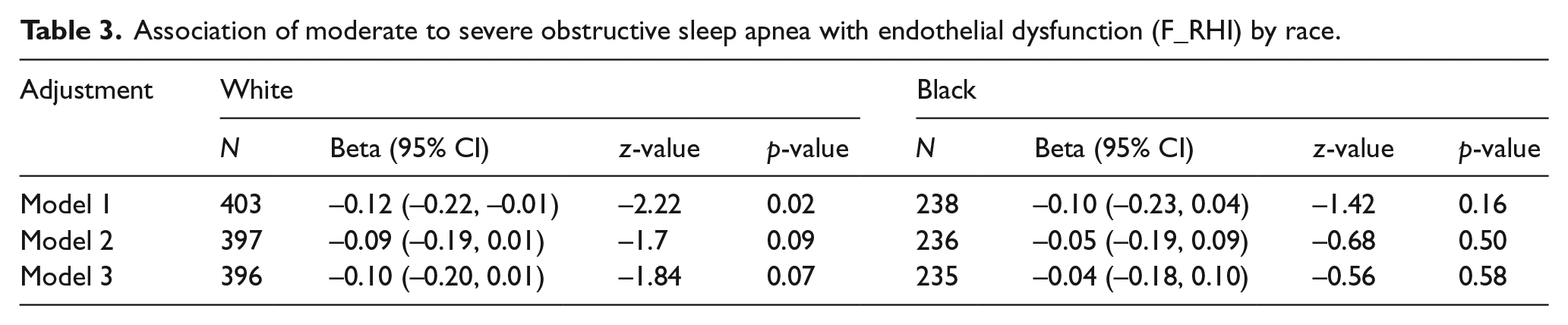
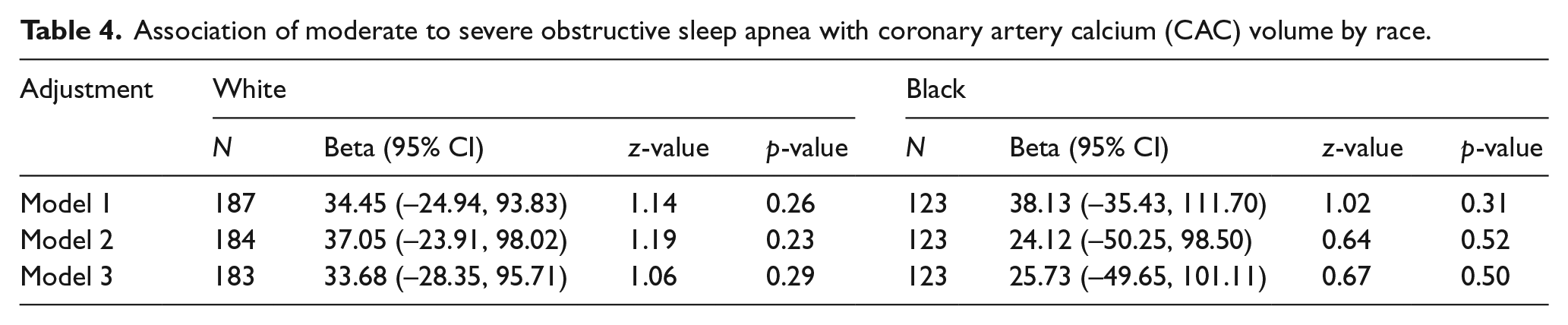
In order to better assess the shape of association, we divided participants into quintiles by OSA severity. Figure 2 depicts F_RHI and CAC values by quintiles of AHI. Beginning with the fourth quintile of AHI (median AHI: 14, IQR: 12–16; mean AHI: 14, SD: 2), there is a notable decrease in F_RHI values (mean (95% confidence interval (CI)): first quintile 0.80 (0.72, 0.87); second quintile 0.81 (0.72, 0.90); third quintile 0.81 (0.73, 0.88); fourth quintile 0.67 (0.59, 0.75); and fifth quintile 0.69 (0.61, 0.77)); however, the shape of the association did not significantly deviate from linearity (p-value for linear trend across the quintiles = 0.005). Similarly, the CAC score (first quintile 90.63 (39.46, 141.79); second quintile 75.59 (10.05, 141.14); third quintile 114.73 (65.09, 164.37); fourth quintile 143.76 (93.37, 194.14); and fifth quintile 160.65 (116.25, 205.05)) increased significantly, beginning with the third quintile of AHI (median AHI: 8, IQR: 7–9; mean AHI: 8, SD: 1); however, the shape of the association did not significantly deviate from linearity (p-value for linear trend = 0.008). In a multivariate model, the associations of OSA with CAC and F_RHI were attenuated and did not reach statistical significance on progressive adjustment for several traditional CVD risk factors (p=0.08 for both comparisons, Table 2). In the model adjusting for traditional CVD risk factors, a decrease in F_RHI of 0.07 (p=0.08) was observed in the moderate/severe OSA group compared with the no or mild OSA group (this corresponds to an approximately 10% lower F_RHI). Similarly, in adjusted models, the CAC score was higher by 42 units (p=0.08) in the moderate/severe OSA group (this corresponds to an approximately 35% higher CAC score). Sensitivity analyses using log-transformed CAC values yielded comparable results (p=0.06). As shown in Tables 3 and 4, there was no significant interaction of race on the association of OSA with F_RHI or CAC (p-value >0.5 for all comparisons).
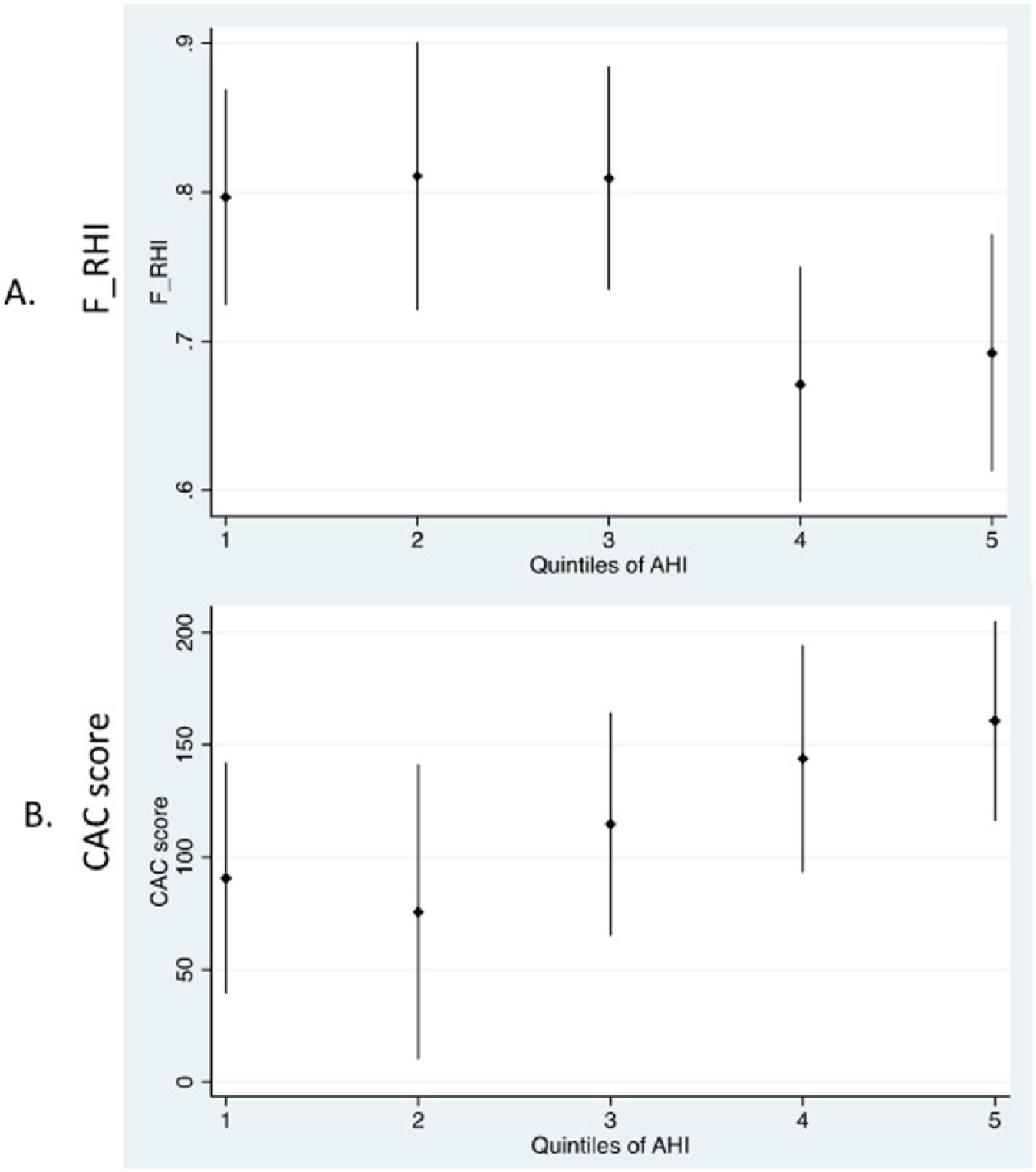
Graphical plot of (A) Framingham reactive hyperemia index (F_RHI) and (B) coronary artery calcium (CAC) score by fifths of apnea hypopnea index (AHI).
Association of moderate to severe obstructive sleep apnea with vascular dysfunction and subclinical atherosclerosis.
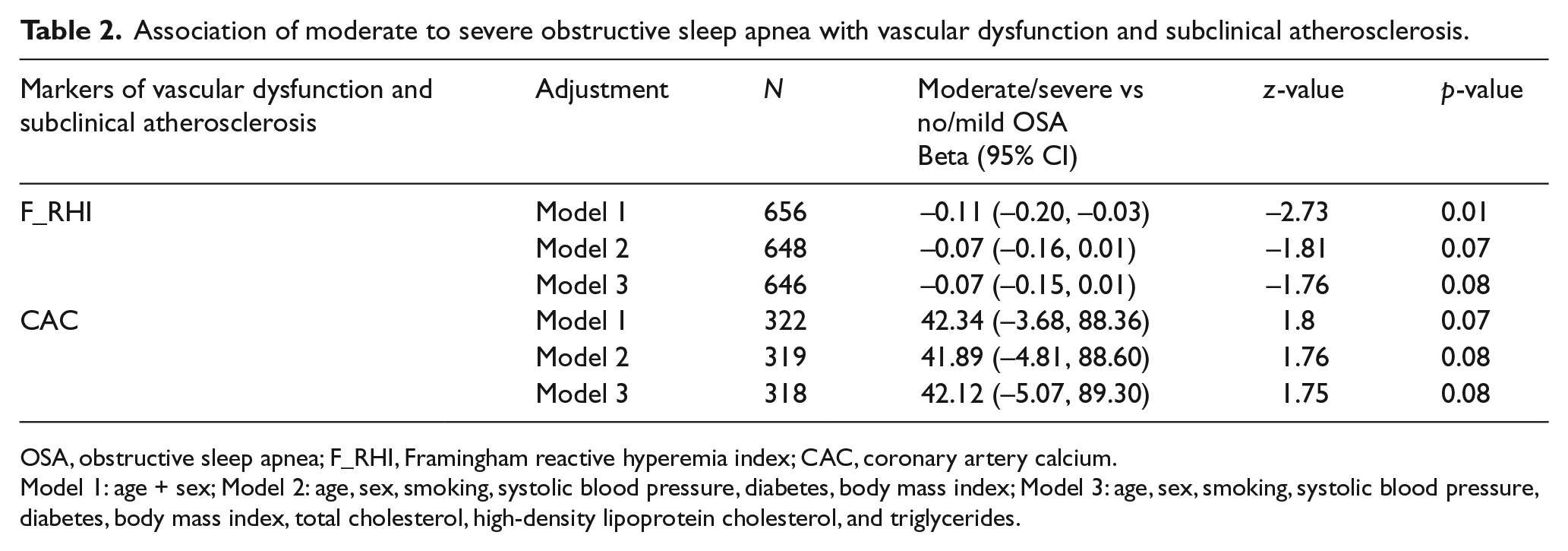
OSA, obstructive sleep apnea; F_RHI, Framingham reactive hyperemia index; CAC, coronary artery calcium.
Model 1: age + sex; Model 2: age, sex, smoking, systolic blood pressure, diabetes, body mass index; Model 3: age, sex, smoking, systolic blood pressure, diabetes, body mass index, total cholesterol, high-density lipoprotein cholesterol, and triglycerides.
Association of moderate to severe obstructive sleep apnea with endothelial dysfunction (F_RHI) by race.
Association of moderate to severe obstructive sleep apnea with coronary artery calcium (CAC) volume by race.
Discussion
This diverse community-based study of middle-aged US adults showed that moderate/severe OSA is associated with a lower F_RHI (endothelial dysfunction) and a higher CAC score. However, adjusting for traditional Framingham risk factors for CVD weakened these associations. Additionally, the association of moderate/severe OSA with F_RHI and CAC score did not vary by race. Although the etiologies of associations between subclinical atherosclerosis and OSA are not entirely clear, potential pathophysiological mechanisms include OSA-induced intermittent hypoxemia and generation of reactive oxygen species. 47 These physiologic changes cause oxidative stress47,48 and stimulate pro-inflammatory processes49,50 which result in endothelial dysfunction.19–25 Over time, endothelial dysfunction results in atherosclerotic plaque which tends to be calcified. Additionally, OSA may contribute to atherosclerosis indirectly by causing dyslipidemia 51 and insulin resistance. 52
Our results suggesting that endothelial dysfunction is associated with severity of OSA (albeit statistically not significant in the analysis) is consistent with prior reports. Data from the Sleep Heart Health Study (SHHS) indicate that OSA patients who are free from any overt CVD exhibit macrovascular endothelial dysfunction, determined by brachial artery FMD, which correlates with the severity of OSA. 22 Other studies have found similar associations of increased OSA severity with endothelial dysfunction as measured by RHI. One observational study of 133 Japanese individuals found that severe OSA was an independent predictor of RHI. 53 Another study involving 267 individuals with OSA found a positive association of increasing F_RHI (indicating healthier endothelial function) with increasing AHI (worsening sleep apnea) when the AHI value was less than 18.4. 54 Interestingly, in that study, when the AHI value was greater than 18.4, there was a negative association of decreasing F_RHI with increasing AHI, although this was not statistically significant. The reason for this disparate result is not entirely clear, but it may be that more severe OSA results in endothelial dysfunction, but not mild OSA. This notion is corroborated by data from the SHHS, which demonstrated that coronary artery disease, 9 heart failure, 9 and mortality 55 increase most at moderate to severely elevated AHI levels. We appear to corroborate these findings by demonstrating similar results in a racially diverse population of middle-aged adults in the United States. While moderate/severe OSA was strongly associated with microvascular endothelial dysfunction determined by F_RHI in our study population, adjustment for multiple traditional risk factors weakened the association. Notably, we did not find a significant interaction effect of race on the association of moderate/severe OSA with F_RHI, even though the burden of microvascular endothelial dysfunction is known to be higher in black compared to white participants in our Heart SCORE cohort. 37
CAC has been shown to be highly specific for coronary atherosclerosis, 30 and associated with overall atherosclerotic plaque burden, coronary events, 31 and CVD in middle and older age populations. These observations resulted in the clinical utilization of CAC scores as a tool for risk stratification. Results from community-based studies examining the association between sleep apnea and CAC show that more severe OSA is independently associated with CAC.29,56 Recent analysis of data from MESA showed that OSA was associated with CAC score progression after adjustment for demographics, behaviors, and body mass index. However, the association was not significant after accounting for cardiovascular risk factors that may mediate the association between OSA and CAC. 35 Consistent with this observation, we found moderate/severe OSA to be strongly associated with CAC in unadjusted analyses. However, adjustment for traditional risk factors weakened the association of OSA with CAC. Of note in the present study, we did not find any significant interaction effect of race on association of moderate/severe OSA with higher CAC. These findings suggest that the apparent deleterious effects of OSA on vascular function and structure may not vary meaningfully by race/ethnicity.
Limitations
Some limitations of our study merit consideration. First, our analysis was cross-sectional in design and precludes conclusions regarding the causality of the markers of subclinical atherosclerosis assessed. While multivariable models were employed in the analyses, residual confounding cannot be excluded. Second, we conducted sleep assessments using a portable ApneaLink Plus monitoring system. Polysomnography may have provided more detailed sleep characteristics for analysis. However, prior studies have shown that the ApneaLink Plus device is highly sensitive and specific for the diagnosis of OSA.41–43 Our scoring of respiratory events using a portable ApneaLink Plus monitor without electroencephalogram (EEG) was based on flow reductions coupled with oxygen desaturation, and, thus, did not include non-hypoxic events causing arousals. Considering that the mechanism by which OSA impacts endothelial function may be partly through recurrent arousal and sleep fragmentation, we may have underestimated the strength of the association. Finally, we measured digital pulse amplitude at the finger to determine microvascular endothelial function and not ultrasound-derived brachial artery FMD which measures macrovascular endothelial function. Prior studies have validated the Endo-PAT-derived RHI as a marker of peripheral and coronary endothelial function.44,57 Our study, however, has an important virtue. To our knowledge, it is the first study to assess the association of OSA with early microvascular endothelial dysfunction and advanced markers of subclinical atherosclerosis together in a racially diverse population.
Conclusions
In a community-based cohort, moderate/severe OSA was modestly associated with microvascular endothelial dysfunction and coronary artery calcification. These associations did not vary meaningfully by race. Further large multiracial studies are needed to further elucidate the impact of OSA on subclinical atherosclerosis.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was funded by the Pennsylvania Department of Health (ME-02-384) and National Institutes of Health (R01HL089292, R21DK113486).
