Abstract
This study was conducted to identify specific abnormalities using the results from air plethysmography in legs with lymphedema. A routine air plethysmography exercise protocol was performed in 31 patients with unilateral leg lymphedema, and the results were compared with those of 53 patients with unilateral great saphenous vein reflux and 15 normal subjects. The venous filling index in legs with lymphedema (2.1 ± 1.2 mL/sec) was smaller than in legs with great saphenous vein reflux (6.4 ± 4.1 mL/sec, p < 0.05), but was not different from that in normal legs (1.9 ± 1.2 mL/sec). The ejection fraction was similar in all groups. The residual volume fraction in legs with lymphedema (35 ± 32%) was larger than that in normal subjects (13 ± 23%, p < 0.05), but was not significantly different from that in the contralateral leg of the lymphedema patients (32 ± 27%). In conclusion, we found no specific air plethysmography findings in uncomplicated lymphedema.
Introduction
There have been few reports on venous hemodynamics in legs with lymphedema. Kim et al. 1 studied venous hemodynamics in 41 patients with unilateral leg lymphedema using air plethysmography. They found abnormal air plethysmography parameters, namely increased venous volume, venous filling index, and ambulatory venous pressure, represented by the residual volume fraction, as well as decreased ejection fraction in legs with lymphedema, despite the absence of specific disorders in the venous system. However, the parameters derived from air plethysmography should not be abnormal in the absence of an anatomical venous disorder. The current study compared air plethysmography findings in legs with lymphedema with those in legs with great saphenous vein reflux (GSVR) and in normal legs in order to identify any abnormal air plethysmography parameters despite the absence of a confirmed venous disorder.
Patients and methods
This retrospective study was approved by the Institutional Review Board of Yamaguchi University Hospital (Center for Clinical Research, Ube, Yamaguchi, Japan), and the need for individual patient consent was waived. Between April 2012 and March 2016, 103 patients with leg lymphedema visited our clinic. The study enrolled 31 (30%) patients with unilateral leg lymphedema (two primary and 29 secondary lymphedema) classified as International Society of Lymphedema stage II 2 who could complete the air plethysmography exercise protocol. In these patients, the median age of onset of lymphedema was 64 (range 17–84) years, median duration of lymphedema was 3 (range 0.1–42) years, and median lymphedema volume calculated from the truncated cone method was 1827 (range 59–6329) mL. Seven of these patients (23%) experienced cellulitis at a frequency of 0.5 (range 0.1–2.7) per year. No evidence of insufficiency and/or occlusion in the deep veins, saphenous veins, and perforators was confirmed using a duplex venous scan in the affected legs (lymphedema) or contralateral asymptomatic legs. Axial great saphenous vein reflux (GSVR) was found in 12 legs of 10 patients (10%) with lymphedema, and the affected legs (lymphedema + GSVR) were included in the comparison. The study also included 53 patients with unilateral varicose veins solely due to axial GSVR and a clinical score of C2 in the Clinical, Etiological, Anatomical, Pathophysiological (CEAP) classification3,4 (affected leg: GSVR). Fifteen normal right legs of healthy volunteers were included as controls. Characteristics of the subjects and legs are summarized in Table 1. Air plethysmography exercise testing was performed as described by Christopoulos et al. 5 Duplex scanning and air plethysmography were performed by a qualified cardiovascular technician (KN).
Subject characteristics.
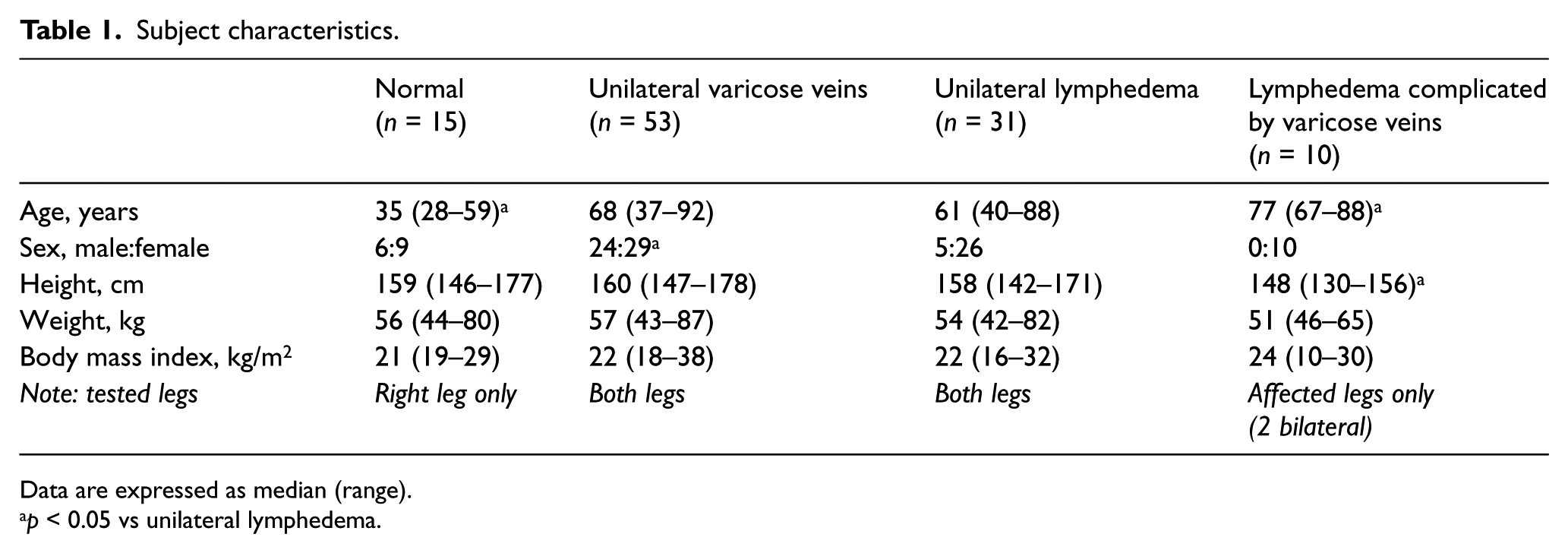
Data are expressed as median (range).
p < 0.05 vs unilateral lymphedema.
Statistical analysis
Results are reported as median (range) or count (percentage), unless otherwise indicated. The Kruskal–Wallis test was used for differences in subject characteristics and for each air plethysmography parameter among the groups. The Mann–Whitney U-test was used for multiple comparisons. Statistical analyses were performed using JMP 11.0 (SAS Institute, Cary, NC, USA). A p-value < 0.05 was considered significant.
Results
Venous volume in different legs is demonstrated in Figure 1. The venous volume in legs with GSVR was larger than that in contralateral asymptomatic legs with GSVR. This difference in venous volume was not observed between legs with lymphedema and contralateral legs with lymphedema. The venous volume in legs with lymphedema was smaller than that in legs with GSVR and that in legs with lymphedema + GSVR, but was not different from that in normal legs. Interestingly, venous volume in contralateral legs with lymphedema was significantly smaller than that in contralateral legs with GSVR (p < 0.01), although patient habitus was similar.
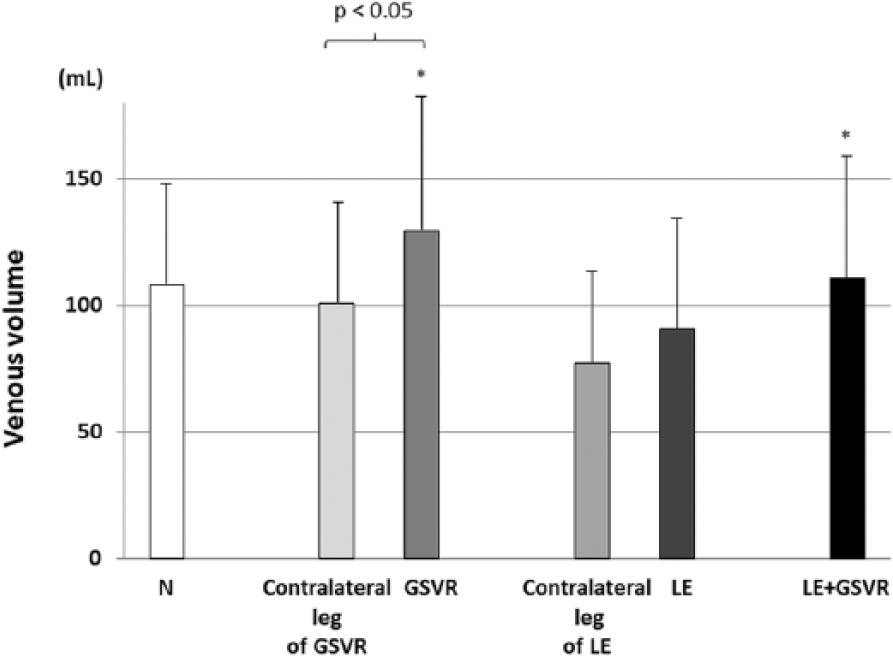
Venous volume.
The venous filling index in different legs is demonstrated in Figure 2. The venous filling index in legs with GSVR was larger than that in contralateral legs with GSVR. This difference in the venous filling index was not observed between legs with lymphedema and contralateral legs with lymphedema. The venous filling index in legs with lymphedema was smaller than that in legs with GSVR or lymphedema + GSVR, but was not different from that in normal legs.
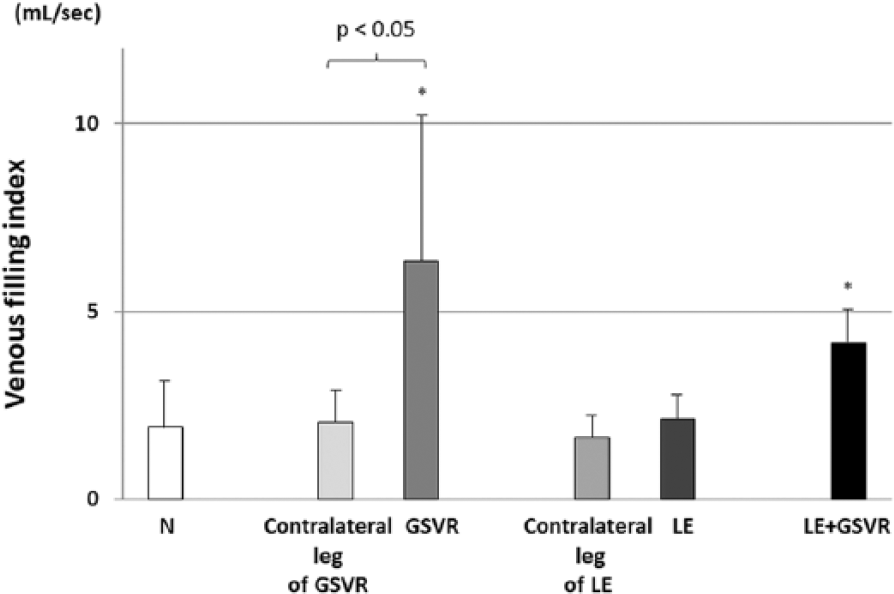
Venous filling index.
The ejection fraction in different legs is demonstrated in Figure 3. The ejection fraction in legs with GSVR was smaller than that in contralateral legs with GSVR. This difference in ejection fraction was not observed between legs with lymphedema and contralateral legs with lymphedema. There was no significant difference in ejection fraction among legs with lymphedema, GSVR, and lymphedema+GSVR, and normal legs.
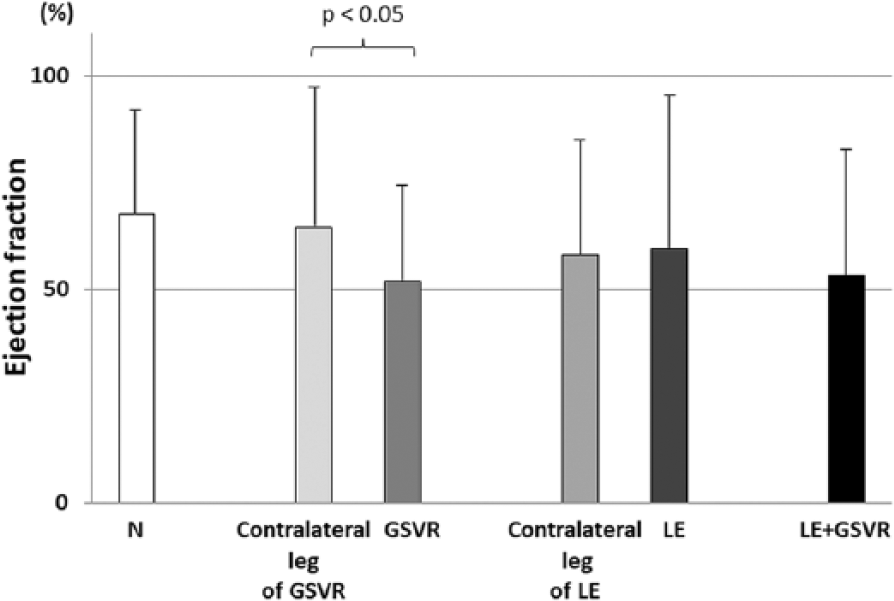
Ejection fraction.
The residual volume fraction in different legs is demonstrated in Figure 4. The residual volume fraction in legs with GSVR was larger than that in contralateral legs with GSVR. This difference in residual volume fraction was not observed between legs with lymphedema and contralateral legs with lymphedma. The residual volume fraction in lymphedema was not significantly different from that in legs with GSVR or lymphedema + GSVR, but was larger than that in normal legs.
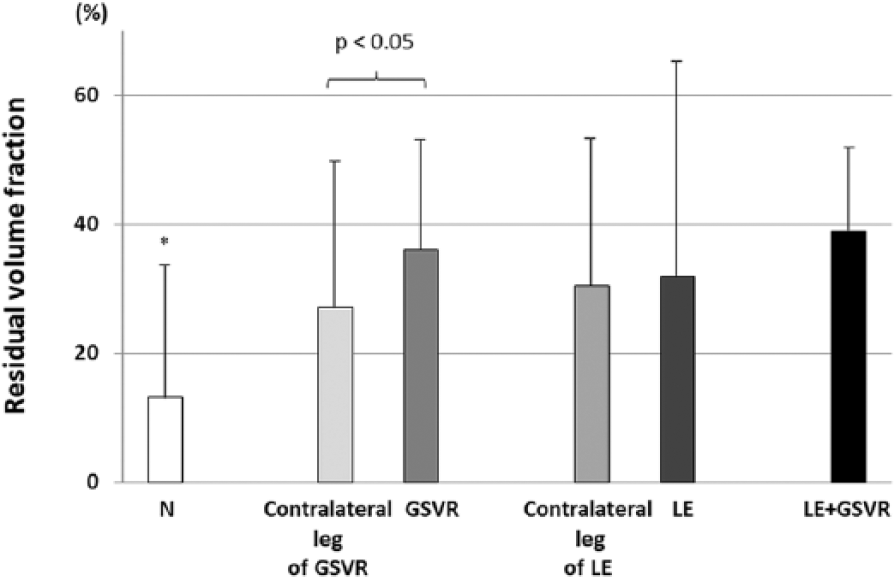
Residual volume fraction.
Discussion
In the current study, we demonstrated that air plethysmography parameters (i.e. venous volume, venous filling index, ejection fraction, and residual volume fraction) were affected only when a confirmed venous disorder was present and were not affected solely by the presence of lymphedema.
As venous volume can vary among individuals, direct comparison of venous volume among different groups may not be appropriate. Venous volume in legs with GSVR was larger than that in the contralateral legs with GSVR, as previously reported. 5 However, in a comparison between venous volume in lymphedema and that in the contralateral legs with lymphedema, the presence of lymphatic insufficiency itself did not seem to increase venous volume. In the current study, venous volume in the contralateral leg with lymphedema was smaller than that in the contralateral leg with GSVR, although patient habitus was similar. This might simply be due to individual variation, but it is possible that the venous system in the contralateral leg with GSVR might have undergone some stress before presentation.
The venous filling index, which represents venous blood reflux from the thigh to the calf, was elevated in legs with GSVR and lymphedema + GSVR, but not in legs with lymphedema. The venous filling index can also be elevated by increased arterial inflow. 6 Arterial inflow in limbs with lymphedema was reportedly increased7–9 or unchanged.10,11 Even if the increase is due to chronic low grade inflammation or excess fat accumulation, 12 the impact on the venous filling index should be limited compared to that derived from axial GSVR, as Shiraishi reported, 13 and as verified in the current study.
The ejection fraction represents calf muscle function and ankle joint motion. The ejection fraction can be reduced when proximal venous obstruction is present or venous blood is pooled in varices. Venous outflow obstruction of various degrees was reportedly present in legs with secondary lymphedema but not in legs with primary lymphedema. 14 However, duplex scanning did not identify venous outflow obstruction or dilated veins in the current study. Therefore, as Struckmann et al. 15 reported, the ejection fraction in limbs with lymphedema seemed to be similar to that in normal legs.
Christopoulos et al. first reported that residual volume fraction values were almost identical to those for ambulatory venous pressure as expressed in mmHg, 5 although this is not necessarily the case. 16 In the current study, residual volume fraction in legs with lymphedema was larger than that in normal legs, but was not different from that in the contralateral leg with lymphedema. This should not be interpreted as indicating that the increased residual volume fraction was caused by lymph stasis and/or resultant skin manifestations in lymphedema. One possible explanation is the age-related reduction in calf muscle function due to decreased mass and strength 17 and/or decreased calf venous compliance, 18 because patients with lymphedema were older than the healthy volunteers; however, this will require further investigation.
Limitations
Air plethysmography testing is highly patient- and operator-dependent, and exercise protocol is not easy to complete, particularly for elderly people; therefore, only the patient who could complete the protocol was assessed in the current study. Because of these reasons, and because this was a single-center study that included a limited number of subjects, reaching a definite conclusion may be difficult. Although no particular abnormal findings were found in lymphedema using air plethysmography, Saito et al. demonstrated impaired venous function in limbs with lymphedema using near-infrared spectroscopy to monitor changes in deoxygenated hemoglobin levels during exercise. 19 Therefore, it was speculated that air plethysmography might not be sensitive enough to detect specific venous abnormalities in lymphedema.
Conclusion
We could not demonstrate specific air plethysmography abnormalities in legs with lymphedema. This suggests that the presence of stage II lymphedema symptoms (i.e., edema and tissue fibrosis/proliferation) do not alone significantly affect venous function as assessed using air plethysmography.
Footnotes
Acknowledgements
The authors thank Dr Yasushi Shiraishi (Shiraishi Cardiovascular Clinic, Kagawa, Japan) for his suggestions and encouragement during this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
