Abstract
In this single center, retrospective cohort study we wished to compare early and total mortality for all patients treated for abdominal aortic aneurysms (AAA) with open surgery who were taking statins compared to those who were not. A cohort of 640 patients with AAA was treated with open surgery between 1999 and 2012. Patients were consecutively recruited from a source population of 390,000; 21.3% were female, and the median age was 73 years. The median follow-up was 3.93 years, with an interquartile range of 1.79–6.58 years. The total follow-up was 2855 patient-years. An explanatory strategy was used. The propensity score (PS) was implemented to control for selection bias and confounders. The crude effect of statin use showed a 78% reduction of the 30-day mortality. A stratified analysis using the Mantel–Haenszel method on quintiles of the PS gave an adjusted effect of the odds ratio equal to 0.43 (95% CI: 0.18–0.96), indicating a 57% reduction of the 30-day mortality for statin users. The adjusted rate ratio was 0.62 (95% CI: 0.45–0.83), indicating a reduction of long-term mortality of 38% for statin users compared to non-users for a median follow-up of 3.93 years. This retrospective cohort study showed a significant beneficial effect of statin use on early and long-term survival for patients treated with open surgery. To be conclusive, our results need to be replicated by a randomized clinical trial.
Introduction
Statin therapy is associated with improved survival after open surgery and endovascular aortic repair (EVAR) for patients with abdominal aortic aneurysms (AAA). Lipid-modifying medication (LMM) is recommended for all patients who have coronary and non-coronary arteriosclerotic disease. In this single center, retrospective study we wished to compare early and total mortality for all patients treated for AAA who were taking statins compared to those patients who were not. Patients treated with EVAR were excluded from this study in order to decrease the known beneficial effect of EVAR on perioperative survival.
Materials and methods
A cohort of 640 patients with AAA was operated on between 20 July 1999 and 15 July 2012, with a closing date of 15 July 2012. Patients were consecutively recruited from a source population of 390,000; 21.3% (137) were female, and the median age was 73 years. A total of 23.4% (150) of the patients had emergency ruptured AAA (rAAA), verified by computed tomography angiography (CTA).
Early and total mortality were evaluated. The median follow-up was 3.93 years, with an interquartile range of 1.79–6.58 years; total follow-up was 2855 patient-years. An explanatory strategy was used. We used the propensity score (PS) to control for selection bias and confounders.
Epidemiological design and statistical methods
This was an exposed (statin user) versus a non-exposed cohort with the outcomes of early mortality and total mortality with long-term survival. The crude effect of statin use on the outcome of early (30-day) mortality was estimated by the odds ratio (OR) and its 95% confidence interval (CI). For total mortality, we used the incidence rate ratio (IRR) and its 95% CI using the patient-years model. 1 In addition, survival curves using the Kaplan–Meier method and the log-rank test were implemented to compare equality of the curves. 1
The adjusted effect of statin use on outcomes was done using stratification of the PS into quintiles using the Mantel–Haenszel method. 2
This was complemented by use of the logistic model by controlling the PS as a continuous variable for the 30-day mortality end point. For the total mortality end point, we complemented by using the Cox model and estimating the hazard ratio (HR) of statin use and stratifying the Cox model into quintiles of the PS.
The PS is the conditional probability to use statins given the individual covariates, adjusting for observed use of statins with the probability of non-use (propensity) creates a quasi-randomized experiment. Stratification of the PS into quintiles will remove 90% of measured selection bias due to the difference in baseline covariates between a user and non-user of statins.3,4
Our model is in agreement with the limitations by Peduzzi et al., which pinpoints the need to have no less than 10 events per covariate, especially for the outcome of total mortality. 5
As we are in a prognostic strategy in the PS estimation analysis, the predictive accuracy of the model was evaluated by calibration and discrimination. Calibration was evaluated by the Hosmer–Lemeshow (H-L) goodness-of-fit test. Discrimination was evaluated by the analysis of the area under the ROC (receiver operating characteristic) curve. If the area under the curve (AUC) is greater than 0.7, it can be concluded that the model has an acceptable discriminatory capability. The reporting and analysis of our study followed the suggestions for improvement of Weitzen et al. 6 when combining classical methods and the PS to remove 90% of measured selection bias due to baseline differences using quintiles stratification. 3 Unlike randomization, PS techniques cannot control for unknown or unmeasured potential confounding factors.
Results
Generation of the propensity score (PS)
Because of a difference in distribution of covariates between statin users and non-statin users (Table 1), we constructed a PS. We generated the PS for probability of statin use by the logistic model considering 13 variables: diabetes mellitus, American Society of Anesthesiology (ASA) physical status classification system group, beta-blockers, hypertension, previous myocardial infarction (MI), atrial fibrillation, chronic obstructive pulmonary disease (COPD), renal failure, smoker, sex, age, diameter of AAA, and emergency rAAA.
Distribution of clinical profile between users and non-users of statins.
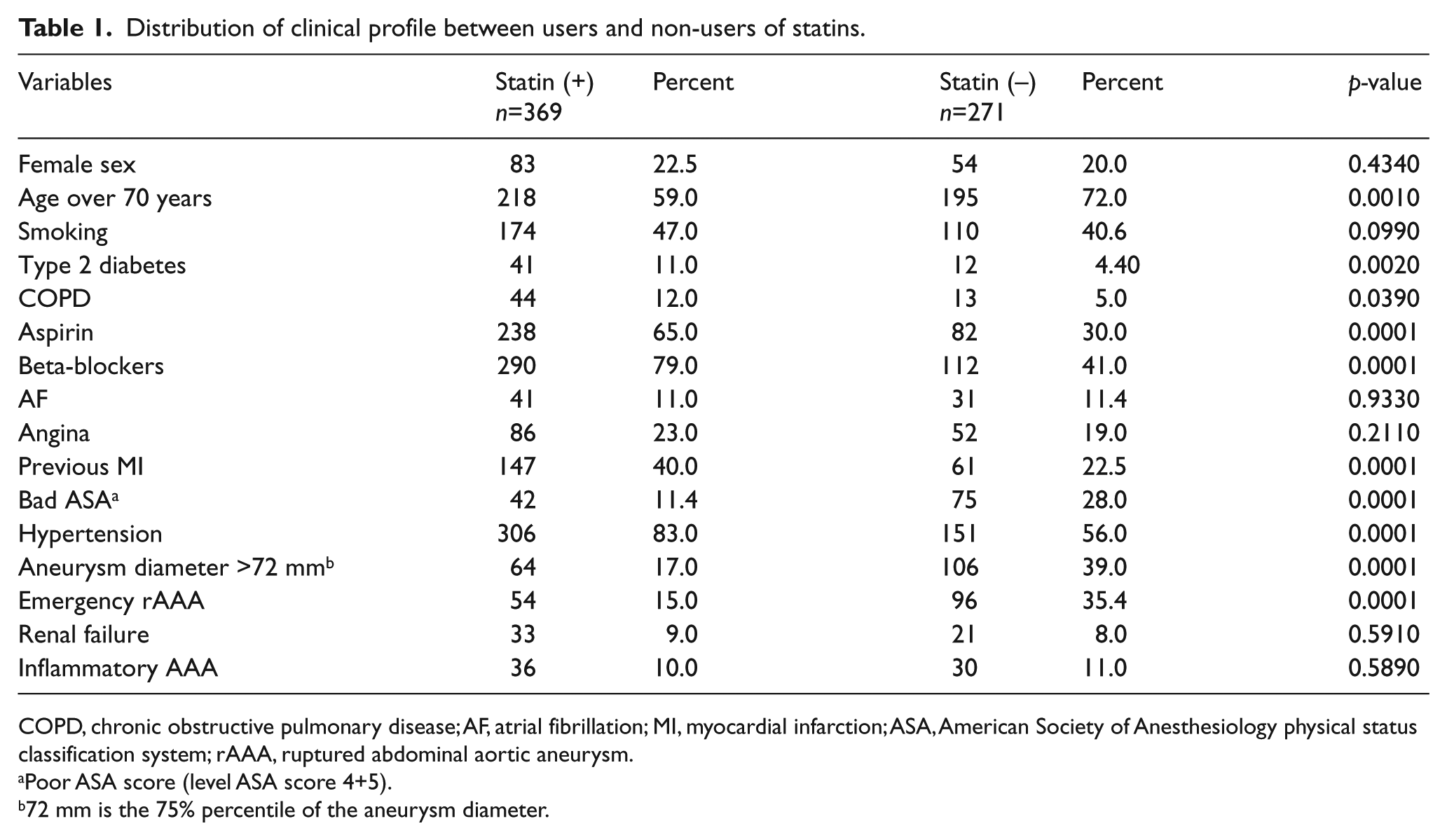
COPD, chronic obstructive pulmonary disease; AF, atrial fibrillation; MI, myocardial infarction; ASA, American Society of Anesthesiology physical status classification system; rAAA, ruptured abdominal aortic aneurysm.
Poor ASA score (level ASA score 4+5).
72 mm is the 75% percentile of the aneurysm diameter.
The generated model was satisfactory as far as discrimination of the area under the ROC curves, with a value of 0.8082, which was markedly over the 0.70 borderline (Figure 1).
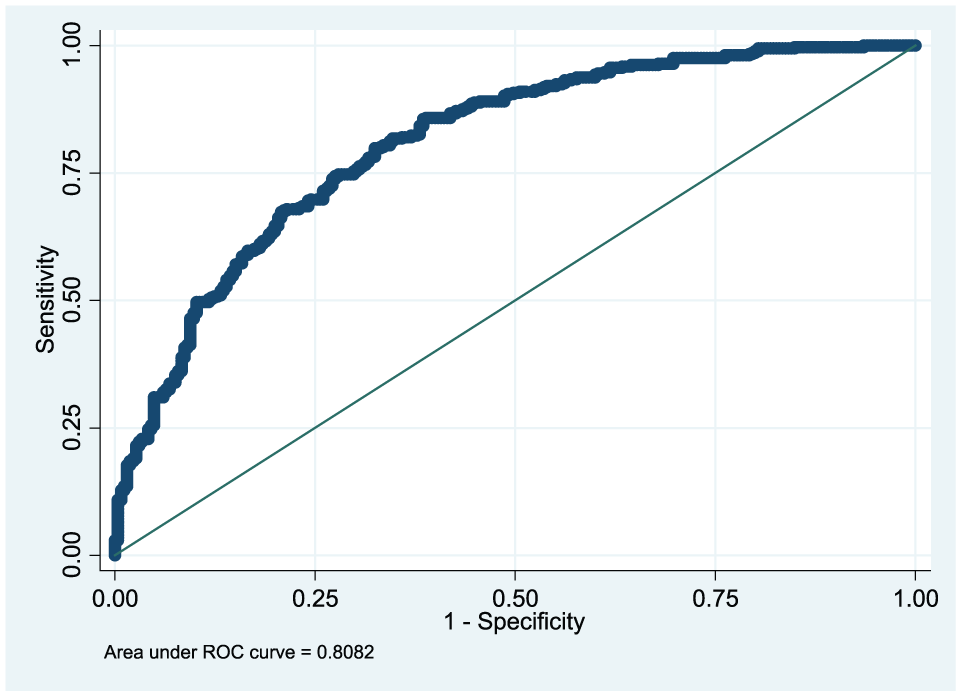
ROC curve of the discrimination of the propensity score.
Also, the calibration of the model was satisfactory with the H-L goodness-of-fit test and had a p=0.9946 indicating a goodness-of-fit for the model data.
Outcome: Early 30-day mortality
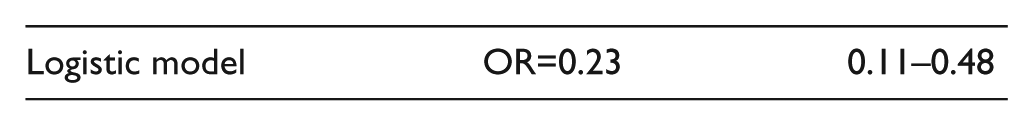
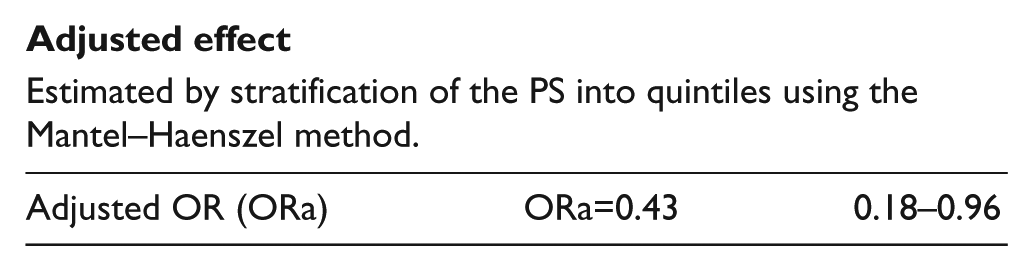
The crude effect of statin use showed a 78% reduction of 30-day mortality (Table 2) for patients using statins compared to non-users. A stratification analysis using the Mantel–Haenszel method on quintiles of the PS gave an adjusted effect on the 13 variables included in the PS (adjusted odds ratio (ORa) = 0.43, 95% CI: 0.18–0.96), indicating a 57% reduction of the 30-day mortality for statin users versus non-statin users. There was homogeneity of the effect in the five strata (the test of homogeneity had a p-value = 0.2659).
End-point: 30-day mortality.
Crude effect
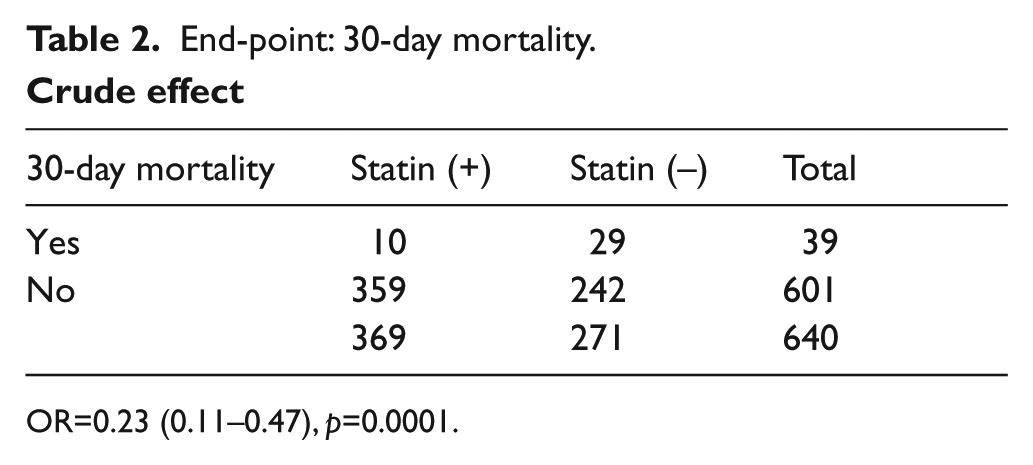
OR=0.23 (0.11–0.47), p=0.0001.
Adjusted effect
Estimated by stratification of the PS into quintiles using the Mantel–Haenszel method.
Test of homogeneity over five strata: p=0.3919.

OR, odds ratio; ORa, adjusted odds ratio; PS, propensity score.
A complementary analysis using the logistic model gave a very close result (OR=0.41, 95% CI: 0.04–0.91) when adjusting for the PS in the model (Table 2).
Outcome: Total mortality
The survival curves were markedly different between statin users and non-statin users (Figure 2). This difference was highly significant (p=0.0001) according to the log-rank test.
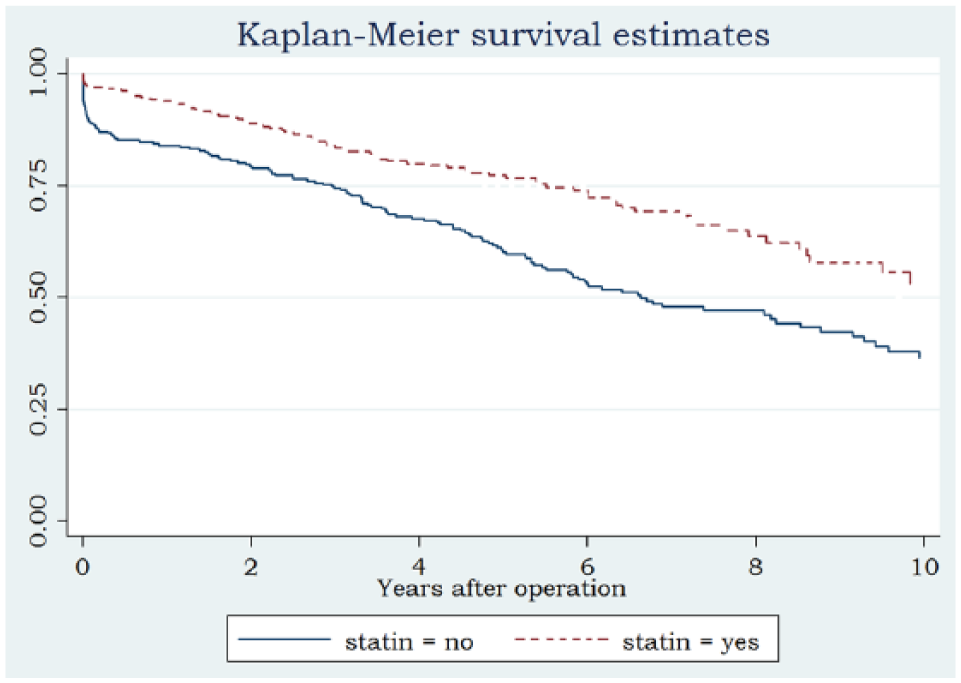
Survival of statin users versus non-users.
The crude effect of statin use on total mortality showed an IRR=0.56 (95% CI: 0.42–0.73; p=0.0001). An adjusted effect using the person-years stratification of the PS into quintiles using the Mantel–Haenszel method is shown in Table 3.
End-point: Total mortality.
Crude effect
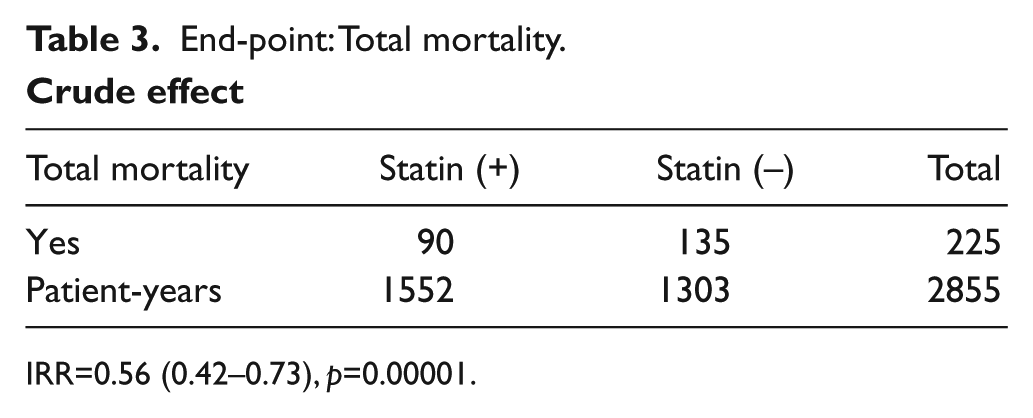
IRR=0.56 (0.42–0.73), p=0.00001.
Adjusted effect
Estimated by stratification of the PS into quintiles using the Mantel–Haenszel method.
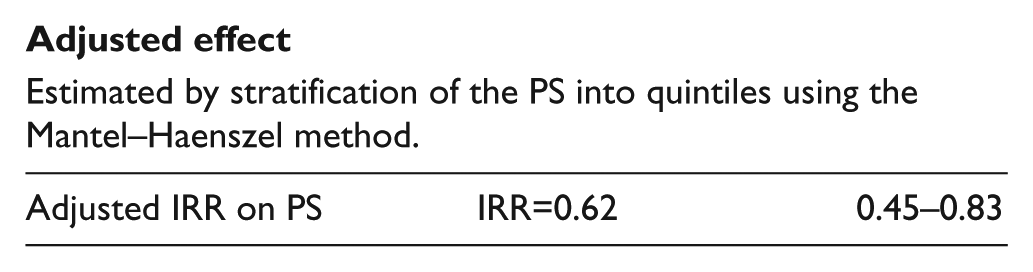
Test of homogeneity over five strata, p=0.3987.
IRR, incidence rate ratio; HR, hazard ratio; PS, propensity score.
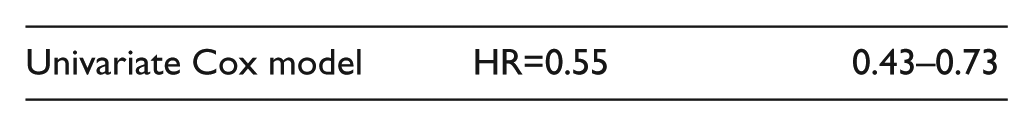
The results showed an adjusted rate ratio using the Mantel–Haenszel of 0.62 (95% CI: 0.45–0.83). This indicates a reduction of the long-term mortality of 38% for statin users compared to non-statin users. This result was adjusted for the 13 variables when generating the PS. The test of homogeneity had a p-value of 0.3987, which indicates homogeneity of the effect on statin use in the five strata of the PS.
A complementary analysis was done using the Cox model to stratify the PS into quintiles, and this gave close results (HR=0.60, 95% CI: 0.44–0.82).
Discussion
AAA patients were not optimally treated with the following prophylactic medications: beta-blockers, platelet inhibitors, angiotensin converting enzyme (ACE) and statins. In particular, where 75% of the patients should have been taking statins, only 41% were. 7 In a Danish study, AAA repair patients had an annual MI incidence of 2.5% (HR=2.1%; 95% CI: 1.9–2.2) compared to the general population. Stroke had an annual incidence of 2.9% (HR=1.8; 95% CI: 1.6–1.9). There was also a 2.4-fold 95% CI increase (2.3–2.4) in the HR of all-cause mortality compared with the general population. 8
What are the plausible biological mechanisms of the prophylactic protective effect of statin medication? Reduced incidence of MI and stroke is, by some authors attributed to the pleiotropic effect on the vascular wall. It has been shown in a pilot study that statin use had a pleiotropic effect on aortic wall atheroma that both stabilized and reduced the size of the plaque. 9 When looking at the effect of statin medication on the expansion rate and the risk of rupture in the aortic wall of AAA patients, irrespective of the decrease in serum cholesterol levels, there was also cellular activity. This activity included effects on endothelial cells, smooth muscle cells, platelets, monocytes/macrophages, and the inflammatory process/cascade. 10 Further pleiotropic effects were documented. Statins decrease the levels of matrix metalloproteinase (MMP-9, MMP-3) in the AAA wall. MMPs are implicated in reducing extracellular matrix proteins (ECM) in the AAA wall. 11 It has been postulated that an imbalance between MMPs and their inhibitors (TIMPS) is a cause of aneurysmal and atherosclerotic disease.12,13 In addition, an association between statin therapy and reduced expansion in patients with small AAA has been shown.14,15 In post deployment of EVAR, it has been documented that statin therapy can be predictive of AAA sac diameter reduction. 16
Going from the cellular level to the clinical setting, there are quite a number of investigative teams that have shown reduced mortality when using statins. Several studies have suggested a beneficial effect of perioperative statin therapy in reducing cardiovascular complications and death directly after vascular surgery.17,18
The Dream Study group has shown that statin therapy upon enrollment in their study was independently associated with better overall survival after AAA open surgery and EVAR (HR=0.5, 95% CI: 0.3–0.8; p=0.004). Statin therapy was especially associated with fewer cardiovascular deaths (HR=0.4, 95% CI: 0.2–0.9; p=0.025). 19 Statin therapy has been shown to give better perioperative outcomes in several studies with patients treated for AAA.20–24 Previously, only a few studies have shown a possible beneficial effect of statin therapy on long-term survival after AAA repair, but these were all retrospective.25–27
Statin use undeniably reduces coronary events and stroke in AAA patients. Our study also demonstrated a protective effect of statin use. This was true both for early (30-day) and long-term survival. We registered statin use at the time of admission: 369 patients were taking statins and 271 were not. Five patients had unknown status. Our elective perioperative work-up includes a cardiologist-reviewed heart duplex study, stress test, and spirometry. Medications are adjusted at this time. In an elective setting, those patients requiring coronary angiography receive either percutaneous coronary intervention (PCI) or a coronary artery bypass graft (CABG), if required.
The PS is quite extensively used in thoracic surgery. Bias and confounding factors are thus reduced. But, it does not control for unmeasured confounders that always exist in an observational design. Here, we chose to use the stratified Cox model and the logistic model to complement our analysis.
We have shown that the adjusted effect for confounders and the selection bias of statin therapy reduces early adjusted 30-day mortality by 57% and total mortality in long-term survival is reduced by 38%.
Conclusion
This retrospective cohort study showed a significant beneficial effect of statin use on early mortality and long-term survival for patients treated for AAA with open surgery. The PS was used to control for major selection bias and confounding factors. To be conclusive, our results need to be replicated by a randomized clinical trial.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
