Abstract

Keywords
Introduction
Statins are among the most widely prescribed medications in the world. Statins are generally safe and well-tolerated. Seven different statins are available in the United States, and this class of medications has been extensively studied. Statins are a frequent topic of conversation in the media. Patients often have questions and concerns about statins, but in patients who have (or are at risk for) cardiovascular disease, the benefits greatly outweigh any risks.
What are statins and how do they work?
Statins, or HMG-CoA reductase inhibitors, are medications that block a step in the production of cholesterol in the liver, which results in the liver putting more receptors out on its surface to remove low-density lipoprotein cholesterol (LDL-C) or ‘bad’ cholesterol. The lower the LDL-C, the lower the risk of cardiovascular problems. Another type of cholesterol is the high-density lipoprotein cholesterol (HDL-C), or ‘good’ cholesterol, which may increase a small amount with statin therapy. Cholesterol is packaged with triglycerides in lipoproteins and is linked to the development and progression of atherosclerosis, which is the build-up of plaque in the arteries. Blocked arteries can occur throughout the body, especially in the heart (coronary artery disease), the neck (carotid artery disease), and the legs (peripheral artery disease, PAD).
Along with lowering LDL-C, statins may have other beneficial effects, including reducing inflammation, which is thought to be related to the development and progression of atherosclerosis. Statins have a favorable effect on plaque in the arteries. Figure 1 shows an artery with plaque, which the statins help to delay, halt, or potentially even reverse.
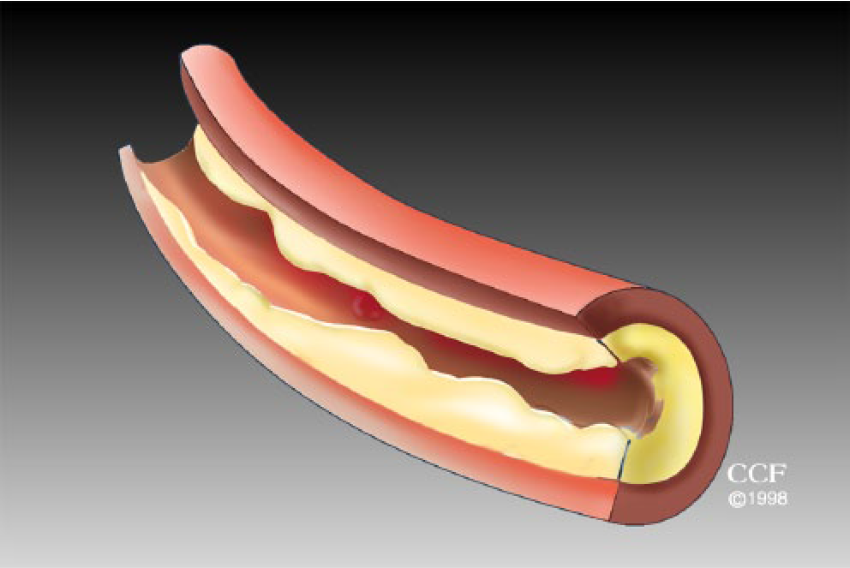
An artery narrowed by atherosclerotic plaque. (Reprinted with permission, Cleveland Clinic Center for Medical Art & Photography © 1998–2017. All rights reserved.)
Why are statins important in vascular disease?
Multiple research studies – in particular, randomized controlled trials, the highest level of evidence – have proven the importance of statins in reducing cardiovascular risk, which means decreasing the chance of heart attack, stroke, and vascular-related death. One of the most important of these clinical trials was the Heart Protection Study, which compared simvastatin 40 mg to placebo. The entire study enrolled >20,000 patients with cardiovascular disease or diabetes, and among these about 7000 had PAD. The risk of a first major vascular event was significantly decreased in the simvastatin group. 1 In total, more than 150,000 patients with and without PAD have participated in clinical trials of statin therapy, and the reduction in vascular events is clearly proportional to the intensity of LDL-C lowering. Similar benefits of statins have been demonstrated among patients with coronary artery disease and prior stroke.
What are the side effects of statins?
Side effects of statins may include muscle, glucose (sugar), or liver enzyme problems. Most patients do not have any side effects. It is critical to know that just because someone has side effects from one statin does not mean that the same side effect will occur with another statin. In addition, the side effects are generally reversible, meaning that they disappear when the medication is stopped.
Many patients are concerned because they have a friend who had muscle problems or other side effects with a statin, and they are thus hesitant to take the statin. In this situation, important considerations include that (1) the risks and benefits of statins are different for each person; (2) the side effects for one person will not necessarily happen to another person; (3) if side effects occur, the medication can be changed (to a different dose or a different statin); and (4) symptoms such as joint or leg pains are common in general and may not be related to the statin (especially if they were there before the statin was started). In research studies in which patients have been blinded (unaware of their treatment group) and crossed back and forth between statin treatment and placebo, similar muscle symptoms have been seen in both groups.
What are the options if side effects occur with the statin?
If side effects occur, then the health care provider who prescribed the statin should be contacted to discuss the next steps, which may include reducing the dose, switching, or stopping the statin. The provider may want to check a blood test called a creatine kinase (CK) level to assess for any muscle breakdown. Blood tests can also detect liver enzyme problems, which are rare.
The overwhelming majority of patients can tolerate a statin, and it is simply a question of finding the best statin medication and dose for each patient. Starting with a low dose and gradually increasing it can be one approach if there are concerns about side effects. If a side effect occurs, then the dose can easily be decreased or the medication can be stopped temporarily (only under the direction of a health care provider) to see if the side effect resolves. Many patients will have resolution of side effects.
Interestingly, studies have found that most patients are then able to re-start the same medication again (either at the same dose or at a lower dose) without any recurrence of the original side effect. This frequent observation suggests that side effects originally thought to be caused by the statin were not actually from the statin but in fact from some other issue at the time (e.g. a viral illness). If the side effect does come back, then the statin may be the cause, and a different statin or dosing regimen can be tried. In some situations, the dosing (such as with rosuvastatin) can be reduced to three times per week if needed. Even low doses of statins like rosuvastatin can produce significant LDL-C lowering (e.g. 30% reduction).
Why are statins controversial?
Statins are recommended for all patients who already have a diagnosis of cardiovascular disease. Statins are also often recommended for patients who do not yet have a diagnosis of cardiovascular disease but who are at risk for cardiovascular disease based on their risk factors (such as smoking, diabetes, high blood pressure, or a family history of early heart disease).
One of the biggest controversies about statins is whether patients should take them for what is called ‘primary prevention’, which means to prevent cardiovascular disease before it occurs. It is important to note, however, that in patients with a diagnosis of cardiovascular disease, the primary prevention debate does not apply because the cardiovascular disease is already there, and the medical evidence is overwhelmingly strong. For example, some may debate about whether a 40-year-old man with slightly high cholesterol but no other medical problems would benefit from starting a statin, which he would then take for the rest of his life. This situation is vastly different from a patient with an established diagnosis of cardiovascular disease, such as PAD, coronary artery disease, abdominal aortic aneurysm, carotid artery disease, or stroke. For the patient with cardiovascular disease (‘secondary prevention’), statins are beneficial regardless of age and regardless of the cholesterol level.
Are all statins the same?
In the current cholesterol guidelines, the two most commonly recommended statins for patients with cardiovascular disease or at high risk are atorvastatin and rosuvastatin. 2 These two are the strongest statins, and both are available as a generic. Statins differ in terms of their potency or strength per milligram. Table 1 shows the relative ‘strength’ of the seven statins available in the United States. Based on the statin and dose, statins are classified as high, moderate, or low intensity.
Approximate dose equivalence of various statins.
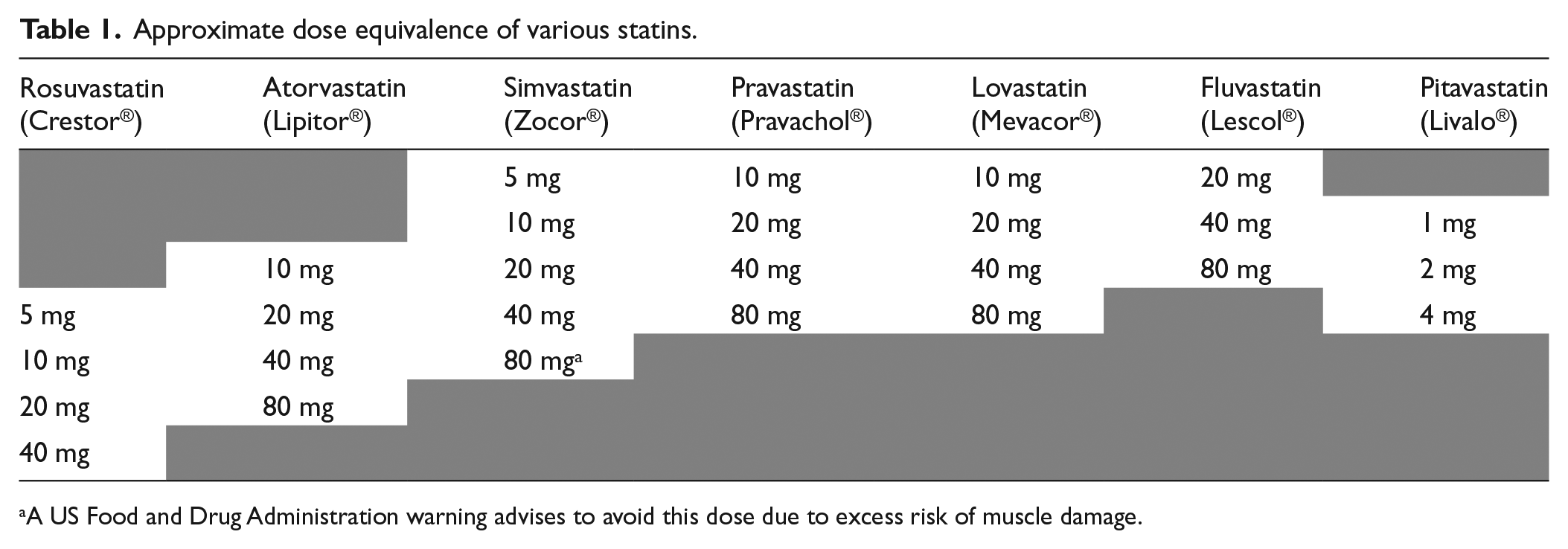
A US Food and Drug Administration warning advises to avoid this dose due to excess risk of muscle damage.
High-intensity statins include atorvastatin (40 or 80 mg/day) and rosuvastatin (20 or 40 mg/day) and should lower the LDL-C by 50% or more. Lower doses of atorvastatin and rosuvastatin are considered moderate intensity, along with a variety of equivalent strength formulations of other statins. Moderate intensity means lowering the LDL-C by about 30–50%. In general, guidelines recommend aiming for moderate or high-intensity therapy, but the overall goal is for each person to find his or her maximum tolerated statin dose to provide the most amount of LDL-C lowering and risk reduction.
Statins vary in their ‘half-life’, which is the time that they remain in the bloodstream after they are taken. Some statins (e.g. simvastatin and lovastatin) do not last long in the bloodstream and thus need to be taken at night, when the liver makes most of the cholesterol. Other statins stay in the bloodstream longer and can be taken in the morning with other medications (e.g. atorvastatin, pravastatin, and rosuvastatin).
Are there other prescription medications for cholesterol?
For patients with cardiovascular disease, statins are by far the most important. Most patients do not need any cholesterol-lowering medications other than the statin. In some cases, other medications may be prescribed either instead of or in addition to a statin.
Ezetimibe (Zetia®) is not a statin. It works completely differently by blocking cholesterol absorption in the intestines. It is taken once per day and lowers LDL-C by ~20% on average. In a large clinical trial of patients with unstable coronary artery disease called the IMPROVE-IT trial, ezetimibe was added to simvastatin and showed a reduction in vascular events compared to simvastatin alone. 3 Therefore, in certain cases, such as in high-risk secondary prevention patients, it may be added to a statin to push the LDL-C down even lower.
More recently, a new class of injectable medications called PCSK9 inhibitors has become available. These medications are human monoclonal antibodies, meaning they are biological agents that bind directly to a target and in this case block its action. They are injected under the skin every 2 weeks or once a month and lower LDL-C by 60% on average. Two PCSK9 inhibitors have been approved by the US Food and Drug Administration (FDA), evolocumab (Repatha®) and alirocumab (Praluent®), to be used in addition to maximally tolerated statin therapy. Research is ongoing regarding these medications and their role in secondary prevention of cardiovascular disease. A challenge with this new therapy is access because the cost is much higher than statins.
Current expert recommendations say to use ezetimibe and PCSK9 inhibition as the next step after statin therapy in patients needing additional LDL-C lowering on top of maximally tolerated statin and lifestyle therapy. Another older but still available class of cholesterol-lowering medications is bile acid sequestrants (such as colesevelam). Other non-statins may be used in certain cases, such as patients with triglyceride disorders. These other medications include fibrates (e.g. fenofibrate), omega-3 fatty acid therapy (fish oil), and niacin.
What about supplements like red yeast rice? How about fish oil?
Supplements are not regulated by the US FDA, may be of variable quality, and have not been proven to improve life expectancy. Over-the-counter red yeast rice or fish oil may improve the cholesterol profile to some degree, but they are not proven to have any direct effect on the plaque itself or on cardiovascular events. Moreover, studies show that over-the-counter products can be contaminated with other unwanted ingredients. Prescription fish oils are used in patients with severely elevated triglycerides.
How often are blood tests needed while taking a statin?
After starting a statin, patients often will have blood tests in 6 to 12 weeks to see if the dose needs to be adjusted in order to reach the target. According to current cholesterol guidelines, for patients under 75 years of age with cardiovascular disease, a high-intensity statin is recommended and the target is usually at least a 50% reduction in the LDL-C. 2 For example, if the LDL-C is 110 mg/dL initially, then the LDL-C goal would be around 55 mg/dL after starting the statin. For patients over 75 years of age, the goal may be more moderate, such that the LDL-C is lowered by approximately 30–50%.
Each time the dose is adjusted, the blood tests may be repeated 6 to 12 weeks later. After the LDL-C goal has been achieved, then blood tests are typically checked once per year. Other testing, such as liver, glucose, or muscle tests, are only necessary in select patients who have a history of such problems or are at high risk for them.
Do statins cause diabetes or memory loss?
Some studies have shown a slight increase in the blood sugar level among patients on statins. However, this finding is typically in patients who were already headed toward the ‘borderline diabetes’ range. In patients who need statins, the benefits of lowering the cardiovascular risk greatly outweigh any small risk related to the blood sugar. If anything, it provides further support to the importance of regular exercise and maintaining a normal body weight for these patients who are near the ‘borderline diabetes’ range. Blood tests such as hemoglobin A1C can be helpful to monitor patients who are at risk for diabetes.
Statins do not cause memory loss. In fact, several studies suggest that statins help to prevent dementia when taken consistently for many years.
What about drinking alcohol or grapefruit juice while taking a statin?
Statins and alcohol are both metabolized by the liver. In general, the current recommendation is not to exceed one drink per day for women or two drinks per day for men to prevent damage to the liver and other detrimental effects of alcohol. This recommendation is independent of the statin question, but holds true while taking the statin as well.
The body has an enzyme named CYP3A, mainly in the liver and intestine, which breaks down medications like statins to remove them from the body. Anything blocking this enzyme can lead to higher than expected statin levels and therefore could make side effects more likely. Notably, grapefruit juice contains compounds called furanocoumarins that block CYP3A. Thus, caution is advised when drinking grapefruit juice for statin users. Simvastatin, atorvastatin, and lovastatin appear to be most affected by grapefruit juice.
Conclusion
Statins are critical medications for patients with cardiovascular disease. Side effects are not as common as many people believe. Working with a health care provider to find the optimal statin regimen is key to maintaining cardiovascular health and preventing cardiovascular events among patients at risk.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
