Abstract
An abnormally high level of homocysteine (Hcy) has been consistently observed in the blood of abdominal aortic aneurysm (AAA) patients. However, the expression of Hcy in human AAA tissues has not been investigated. In this study, the expression of Hcy in aneurysmal tissues from AAA patients (n=30) was compared with non-aneurysmal tissues from organ donors (n=31) by dot blotting and immunohistochemistry. A significantly higher expression of Hcy was observed in AAA than control tissues (p<0.001). Furthermore, the associations of methylenetetrahydrofolate reductase (MTHFR) C677T polymorphism, detected by polymerase chain reaction-restriction fragment length polymorphism, with both AAA and tissue Hcy expression were evaluated. Our results showed MTHFR C677T polymorphism was not significantly associated with AAA or tissue Hcy expression. Lastly, the expression of Hcy in vascular smooth muscle cells (VSMCs), which were isolated from human aortic tissues by explant culture, and their release to cultured media was investigated by dot blotting. The AAA VSMCs expressed and released a significantly higher level of Hcy than the control VSMCs (p<0.001). In summary, our novel findings showed Hcy expression was abnormally elevated in human AAA tissues, which may not be dependent on MTHFR C677T polymorphism.
Keywords
Introduction
Abdominal aortic aneurysm (AAA) is a complex vascular disease, affecting 1.7% to 7.2% of men aged over 65 years across different populations.1–4 Despite a growing amount of literature and the advancements in surgical treatment of AAA in the past decades, the mortality rate of AAA is not declining globally. 5 There is an urgent need to investigate the pathogenesis of AAA and develop an effective therapeutic therapy for preventing the formation and progression of AAA.
Homocysteine (Hcy) is a non-essential amino acid and is an intermediate product in the cellular metabolic pathways.6–8 Hcy can be removed by remethylation to methionine or conversion to cysteine.6–8 Disruption of Hcy metabolism might lead to an abnormal accumulation of unmetabolized Hcy, resulting in a high Hcy level in the blood plasma which is known as hyperhomocysteinemia. 7 The association between hyperhomocysteinemia and several vascular events, such as ischemic heart disease, stroke and venous thrombosis, has been extensively reported.9–13 Consequently, a number of studies have been initiated to investigate the association between hyperhomocysteinemia and AAA, showing a high circulating Hcy in AAA patients.14–19 Furthermore, prior studies using different animal models have reported that Hcy would induce significant disruption of medial elastin in the aortic wall, which is a pathophysiological feature of AAA.20,21 Although these studies suggested that Hcy might play a critical role in the pathogenesis of AAA, the expression of Hcy in human AAA tissues has not been investigated.
In relation to Hcy metabolism, it has been proposed that AAA may be genetically related to a single nucleotide polymorphism in the methylenetetrahydrofolate reductase (MTHFR) gene. MTHFR is a key enzyme to facilitate the removal of Hcy by generating 5-methyltetrahydrofolate for the remethylation of Hcy back to methionine.22–24 The genetic polymorphism in the MTHFR gene (C677T), where the cytosine located at the 677 locus is substituted by thymine, will result in the conversion of an alanine to a valine in the encoded protein.24–26 Such variant protein was reported to be a thermolabile enzyme with a reduced catalytic activity in an in vitro study. 25 Although the association between the C677T polymorphism and AAA has been investigated in prior studies, the findings were controversial.14,18,19,27–31 Therefore, it is still uncertain whether AAA is associated with MTHFR C677T polymorphism through the disruption of Hcy metabolism.
The first aim of this study is to investigate the expression of Hcy in human AAA tissues. Moreover, the association between AAA and MTHFR C677T polymorphism will also be investigated, followed by the detection of any association between Hcy expression in aortic tissues and MTHFR C677T polymorphism. Lastly, the expression of Hcy in vascular smooth muscle cells (VSMCs), which is the dominant cell type in aortic wall and plays a critical role in the pathogenesis of AAA, from AAA and non-aneurysmal tissues will also be investigated.
Methods
Collection of human abdominal aortic tissue specimens
Full-thickness aneurysmal infrarenal abdominal aortic tissues were excised at the point of maximum aortic dilatation from 30 AAA patients who were free from connective tissue disorders (i.e. Marfan syndrome and Ehlers–Danlos syndrome) during open surgical aneurysmal repair. For the control group, full-thickness non-aneurysmal infrarenal abdominal aortic tissues were dissected out from 31 organ donors without known cardiovascular diseases or connective tissue disorders during liver harvesting for transplantation. The aortic tissue specimens collected were used for all analyses except four specimens (three AAA and one control), which were limited by their size and not sufficient for performing explant culture of VSMCs. All subjects were of Chinese ethnicity. Demographics are shown in Table 1. All relevant clinical data were obtained from the clinical medical system. All experiments were performed with approval from the Institutional Review Board of the University of Hong Kong/Hospital Authority Hong Kong West Cluster. Written informed consents were obtained from AAA patients and the next of kin of organ donors.
Demographics of the abdominal aortic aneurysm and control groups.
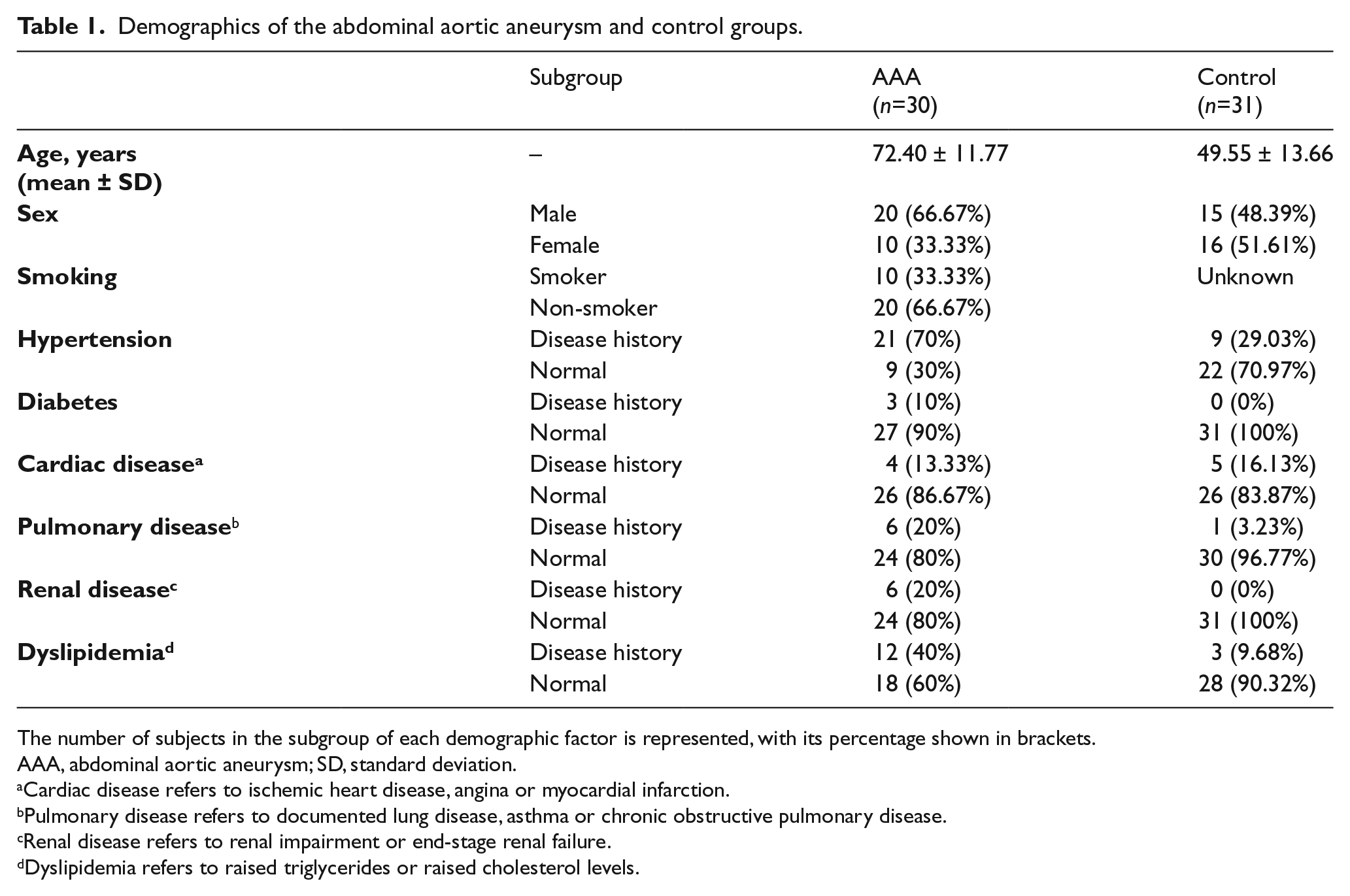
The number of subjects in the subgroup of each demographic factor is represented, with its percentage shown in brackets.
AAA, abdominal aortic aneurysm; SD, standard deviation.
Cardiac disease refers to ischemic heart disease, angina or myocardial infarction.
Pulmonary disease refers to documented lung disease, asthma or chronic obstructive pulmonary disease.
Renal disease refers to renal impairment or end-stage renal failure.
Dyslipidemia refers to raised triglycerides or raised cholesterol levels.
Analysis of Hcy expression in human abdominal aortic tissues by dot blotting
The expression of Hcy in human abdominal aortic tissues was determined by dot blotting. Human aortic tissues were first homogenized and lysed in radioimmunoprecipitation lysis buffer (Cell Signaling Technology, Inc., Danvers, MA, USA) with 1-mM phenylmethanesulfonylfluoride (Sigma-Aldrich Co., St. Louis, MO, USA) according to the protocol provided by the manufacturer. Extracted lysate was blotted on polyvinylidene difluoride membrane (GE Healthcare, Little Chalfont, UK) by vacuum filtration using Bio-Dot® Microfiltration Apparatus (Bio-Rad Laboratories, Inc., Hercules, CA, USA) according to the manufacturer’s protocol. The membrane was blocked and incubated at 4°C overnight with the following primary antibodies: rabbit polyclonal antibody against Hcy (Abcam plc, Cambridge, UK) at a dilution of 1:5000; mouse monoclonal antibody against β-actin (Santa Cruz Biotechnology, Inc., Dallas, TX, USA) at a dilution of 1:1000 as internal control. Incubation with horseradish peroxidase (HRP)-conjugated goat anti-rabbit or mouse IgG (Invitrogen, Carlsbad, CA, USA) at a dilution of 1:10,000 at room temperature for 1 hour was followed. ECL™ Prime Western Blotting Detection System (GE Healthcare) was used for development. The chemiluminescent signal was detected and transformed into digital images using myECL™ Imager (Thermo Scientific, Waltham, MA, USA), followed by analysis using ImageJ software (National Institutes of Health, Bethesda, MD, USA). The Hcy expression in aortic tissues was normalized with the expression of internal control.
Detection of Hcy in human abdominal aortic tissue sections by immunohistochemistry (IHC)
Detection of Hcy in the formalin-fixed and paraffin-embedded aortic tissue sections from humans was done by IHC. The tissue section (6-μm thickness) on Polysine® slides (Thermo Scientific) were dewaxed and hydrated, followed by permeabilization, blocking and overnight incubation at 4°C with the rabbit polyclonal antibody against Hcy (Abcam plc) at a dilution of 1:100. Negative control was performed side by side under the same conditions but with antibody diluent (Dako, Glostrup, Denmark) alone. HRP-labeled polymer-conjugated goat anti-rabbit antibody (Dako) was incubated for 1 hour at room temperature. Liquid 3,3’-Diaminobenzidine Substrate Chromogen System (Dako) was applied for visualization, followed by counterstaining with Mayer’s Hematoxylin Histological Staining Reagent (Dako). The slides were dehydrated and mounted with Permount™ Mounting Medium (Fisher Scientific, Hampton, NH, USA). Images of positive staining signal were captured by inverted microscope (Nikon Eclipses E600; Nikon Corp., Tokyo, Japan).
Detection of MTHFR C677T polymorphism by polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP)
The MTHFR C677T polymorphism was detected by PCR-RFLP. Genomic DNA was first extracted from human abdominal aortic tissues according to the protocol of TaKaRa MiniBEST Universal Genomic DNA Extraction Kit (Takara Bio Inc., Shiga, Japan). PCR was then performed according to the protocol of Taq PCR Master Mix Kit (QIAGEN, Venlo, The Netherlands) using primers amplifying a 198-bp fragment of the MTHFR gene containing the polymorphic nucleotide 677 (sense primer: 5’-TGAAGGAGAAGGTGTCTGCGGGA-3’; antisense primer: 5’-AGGACGGTGCGGTGAGAGTG-3’) (Invitrogen) in the Thermal Cycler 2720 (Applied Biosystems, Foster City, CA, USA) under the following cycling conditions: an initial denaturation for 3 minutes at 94°C; 35 cycles of 30 seconds at 94°C, 30 seconds at 60°C and 1 minute at 72°C; and a final extension for 10 minutes at 72°C.25,32 PCR products were purified according to the protocol of the Monarch PCR & DNA Cleanup Kit (New England Biolabs, Ipswich, MA, USA), followed by overnight digestion by the restriction enzyme HinfI (New England Biolabs) according to the protocol provided by the manufacturer. The C→T substitution at the nucleotide 677 will create a HinfI cut site. Hence, the 198-bp fragment containing the T nucleotide will be digested by HinfI into 175- and 23-bp fragments, while the 198-bp fragment containing the C nucleotide will remain intact. 25 Digested products were then resolved by electrophoresis in 3% agarose gel (Gibco BRL, Grand Island, NY, USA) with 0.5-μg/mL ethidium bromide (Pharmacia Biotech Inc., Sweden) using the Horizon 11-14 Gel Electrophoresis System (Life Technologies, Carlsbad, CA, USA). DNA bands were visualized and captured under the G:BOX Gel Documentation and Analysis System (Syngene, Cambridge, UK).
Analysis of Hcy level in VSMCs explant cultured from human abdominal aortic tissues
VSMCs were first grown out of human abdominal aortic tissues by explant culture prior to analysis of Hcy by dot blotting. An approximately 2 mm × 2 mm-sized segment from human aortic tissues was dissected out and placed onto a complete cell culture medium containing Dulbecco’s modified eagle medium (DMEM), 10% fetal bovine serum, 2-mM
Statistical analysis
SPSS 20.0 (IBM Corp.) was used for statistical analysis. The statistical difference of the relative Hcy level between AAA and control groups was tested by Mann–Whitney U-test. Frequencies of MTHFR alleles and genotypes were compared between AAA and control groups by chi-squared test, and Fisher’s exact test was used if the expected frequency was less than 5. The Hcy expression level among MTHFR genotypes was compared using the Kruskal–Wallis test. Adjustment for the demographic factors (i.e. age, sex, hypertension, diabetes, cardiac disease, pulmonary disease, renal disease, and dyslipidemia) was done using a linear model. Data were expressed as mean ± standard error of the mean. The level of significance was set at 0.05.
Results
Elevated Hcy expression in AAA compared with control aortic tissues
The Hcy expression in human AAA and control aortic tissues was examined by dot blotting (Figure 1). The relative Hcy expression level in AAA aortic tissues (n=30; 3.24±0.36) was significantly higher than that in control aortic tissues (n=31; 1.00±0.08) (p<0.001). Such differential Hcy expression between AAA and control aortic tissues remained statistically significant after adjustment for the demographic factors (i.e. age, sex, hypertension, diabetes, cardiac disease, pulmonary disease, renal disease, and dyslipidemia) (p<0.01).
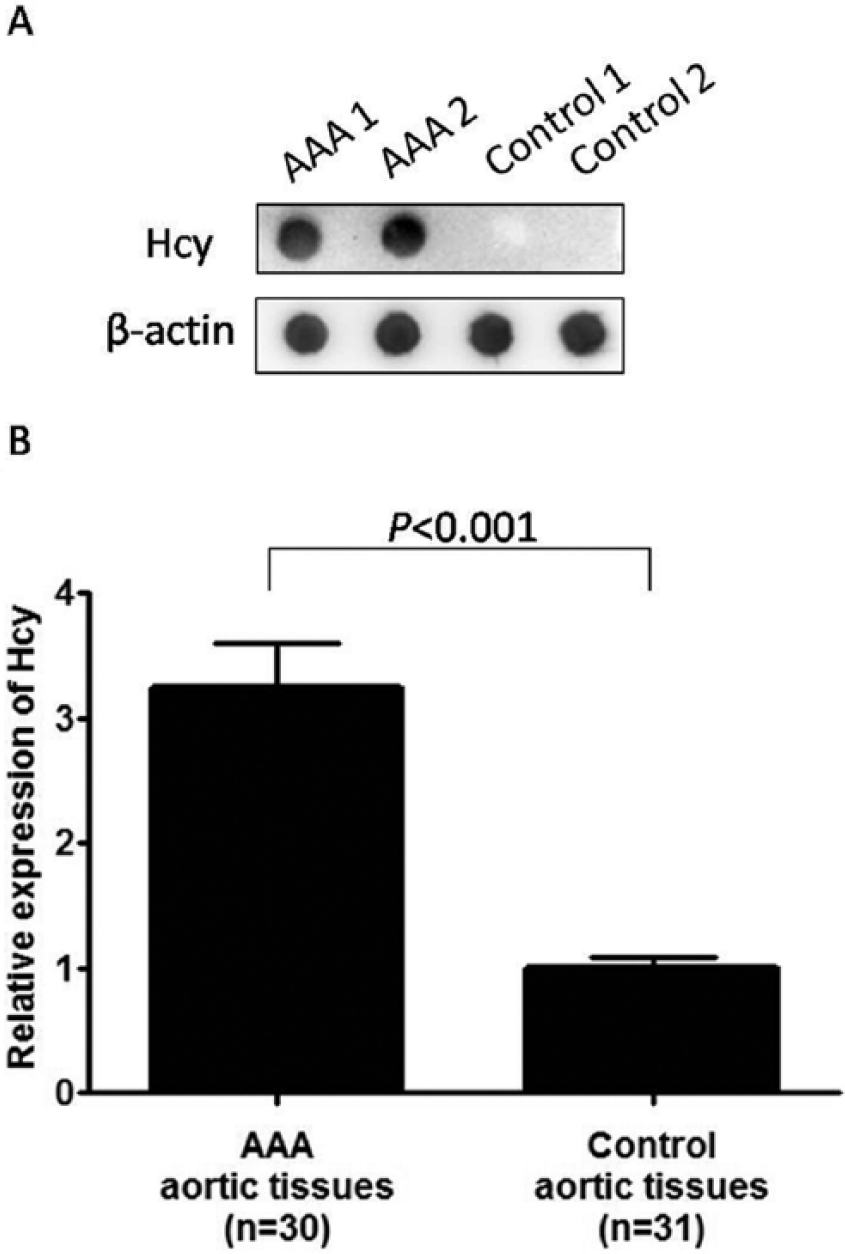
Detection of homocysteine (Hcy) expression level in abdominal aortic tissues from the abdominal aortic aneurysm (AAA) and control groups by dot blotting. (A) Representative dot blot showing two AAA and two control tissue samples, probing for Hcy and β-actin. (B) Statistical analysis showed the relative Hcy expression in AAA tissues (n=30; 3.24±0.36) was significantly higher than that in control aortic tissues (n=31; 1.00±0.08) (p<0.001).
The observed differential expression of Hcy was validated by IHC staining of the AAA and control aortic tissues (Figure 2). Both the medial and adventitial regions in AAA tissue sections showed more staining of Hcy than those in control aortic tissue sections. Furthermore, within the AAA tissue sections, the medial region showed slightly stronger staining than the adventitial region. Therefore, it is suggested that the Hcy expression level was elevated in AAA compared with control aortic tissues from humans.
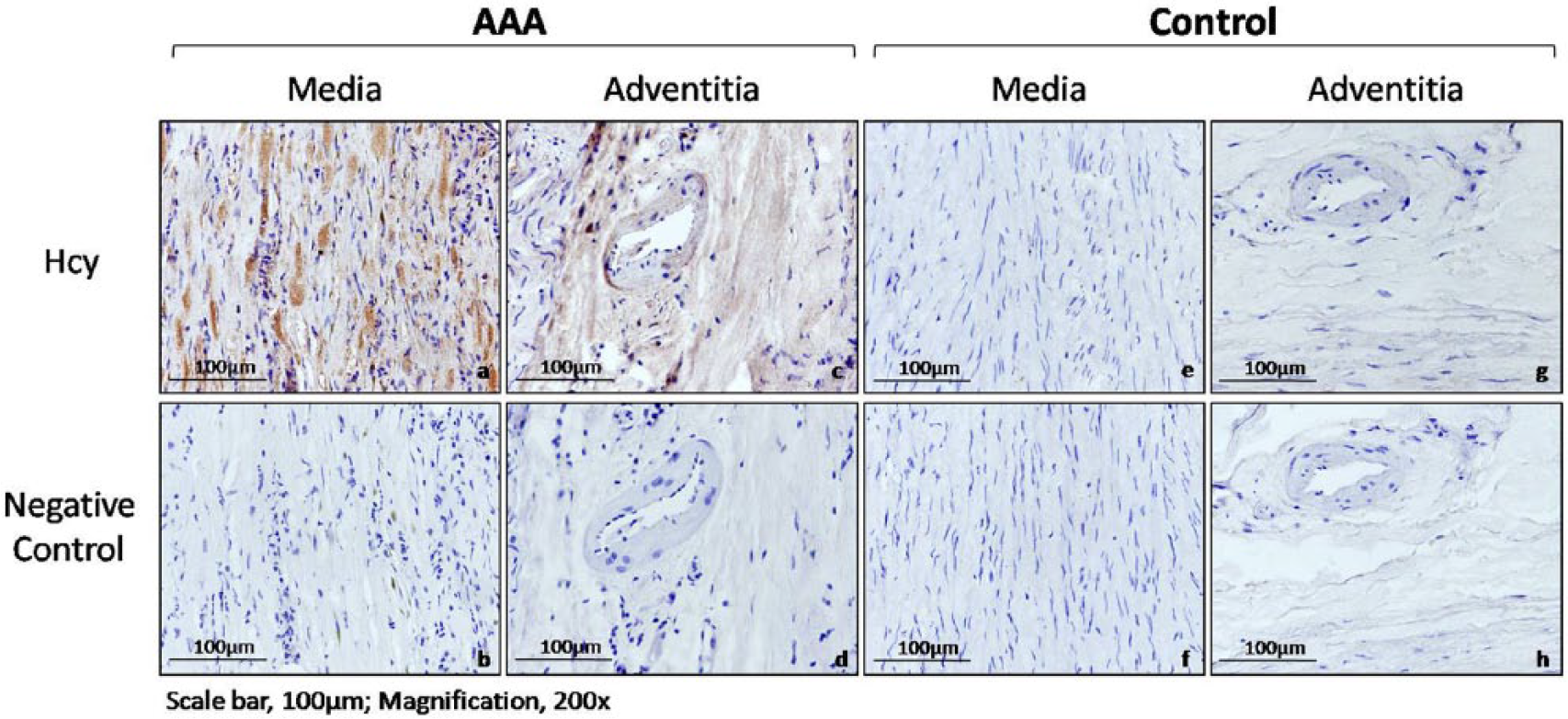
Detection of homocysteine (Hcy) in abdominal aortic tissues from the abdominal aortic aneurysm (AAA) and control groups by immunohistochemistry (IHC). Representative IHC images of different vascular regions, that is, the media (a,b,e,f) and adventitia (c,d,g,h) of AAA and control tissue section samples, probing for Hcy under 200× magnification. AAA tissue sections showed more positive staining (brown) of Hcy than control aortic tissue sections. Note: the figure is in color online.
No association between MTHFR C677T polymorphism and AAA
The MTHFR C677T polymorphism in AAA and control groups was investigated by PCR-RFLP. The allele frequencies of MTHFR C677T polymorphism in AAA and control groups were analyzed (Table 2). No significant difference was found in the T allele frequency between AAA (25%) and control (29.03%) groups (p=0.62). In addition, the genotype frequencies of MTHFR C677T polymorphism in AAA and control groups were analyzed (Table 3). The frequencies of CC, CT and TT genotypes in the AAA group were 60%, 30% and 10%, respectively, whereas the corresponding frequencies in the control group were 51.61%, 38.71% and 9.68%, respectively. No significant difference was observed in the frequencies of CT and TT genotypes between AAA and control groups (CT vs CC: p=0.47; TT vs CC: p=1.00). Therefore, it is suggested that there was no significant association between the MTHFR C677T polymorphism and AAA.
Allele frequencies of MTHFR C677T polymorphism in the AAA and control groups.
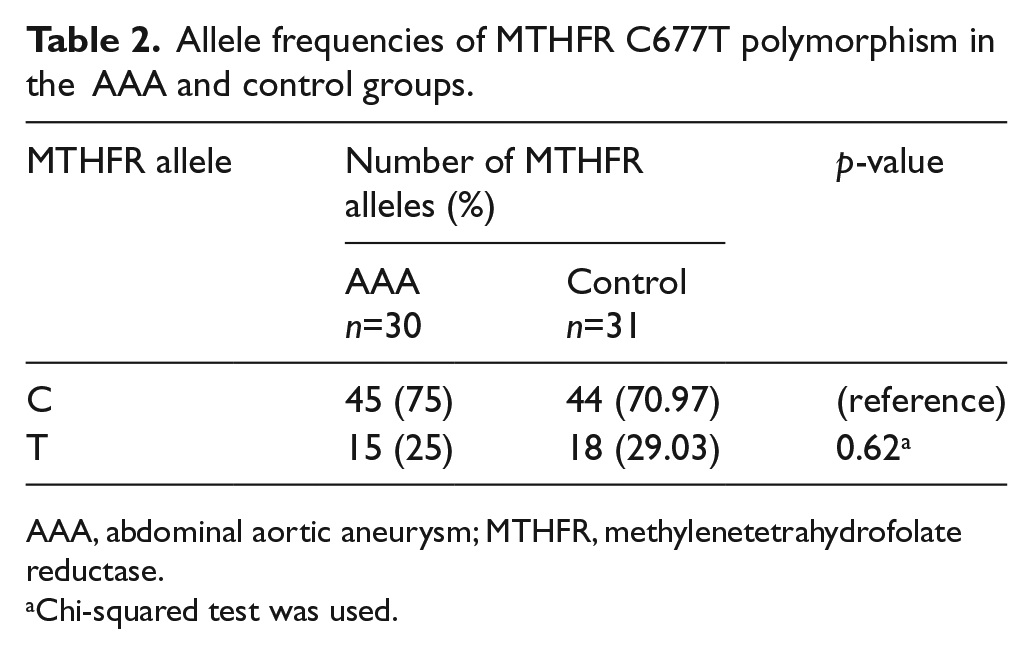
AAA, abdominal aortic aneurysm; MTHFR, methylenetetrahydrofolate reductase.
Chi-squared test was used.
Genotype frequencies of MTHFR C677T polymorphism in the AAA and control groups.
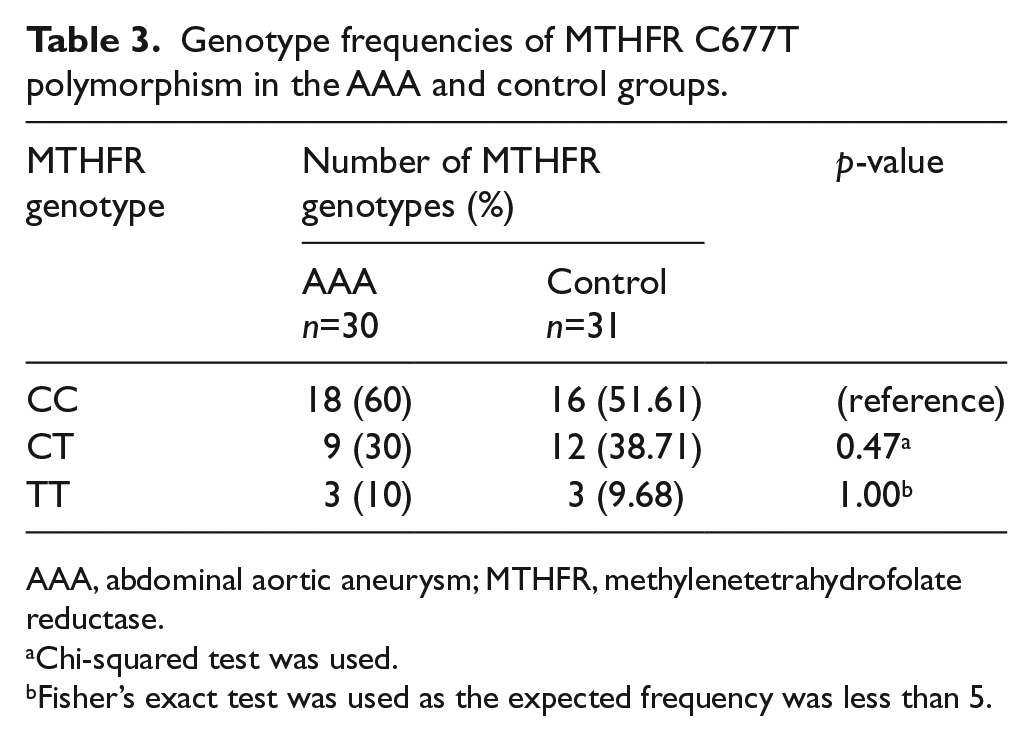
AAA, abdominal aortic aneurysm; MTHFR, methylenetetrahydrofolate reductase.
Chi-squared test was used.
Fisher’s exact test was used as the expected frequency was less than 5.
No difference in Hcy expression in aortic tissues among MTHFR C677T genotypes
The relative Hcy expression in aortic tissues among MTHFR C677T genotypes was assessed. No significant difference in the relative Hcy expression was observed among the CC (n=34; 2.35±0.32), CT (n=21; 1.70±0.33) and TT (n=6; 2.11±1.05) genotypes (p=0.20), suggesting that there was no significant association between tissue Hcy expression and MTHFR C677T polymorphism.
Elevated Hcy level in VSMCs cultured from human AAA tissues
The expression of Hcy in VSMCs isolated from AAA and control aortic tissues from humans was examined by dot blotting (Figure 3A). The relative Hcy expression in AAA VSMCs (n=27; 1.44±0.07) was significantly higher than that in control VSMCs (n=30; 1.00±0.02) (p<0.001). After adjustment for the demographic factors, such differential Hcy expression between AAA and control VSMCs remained statistically significant (p<0.001). In addition, the amount of Hcy released to the cultured media from AAA and control VSMCs was also examined (Figure 3B). The relative Hcy level in the cultured media of AAA VSMCs (n=27; 1.33±0.04) was significantly higher than that of control VSMCs (n=30; 1.00±0.01) (p<0.001). Such differences between AAA and control VSMC-cultured media remained statistically significant after adjustment for the demographic factors (p<0.001). Therefore, the level of Hcy in AAA VSMCs was higher than that of control VSMCs.
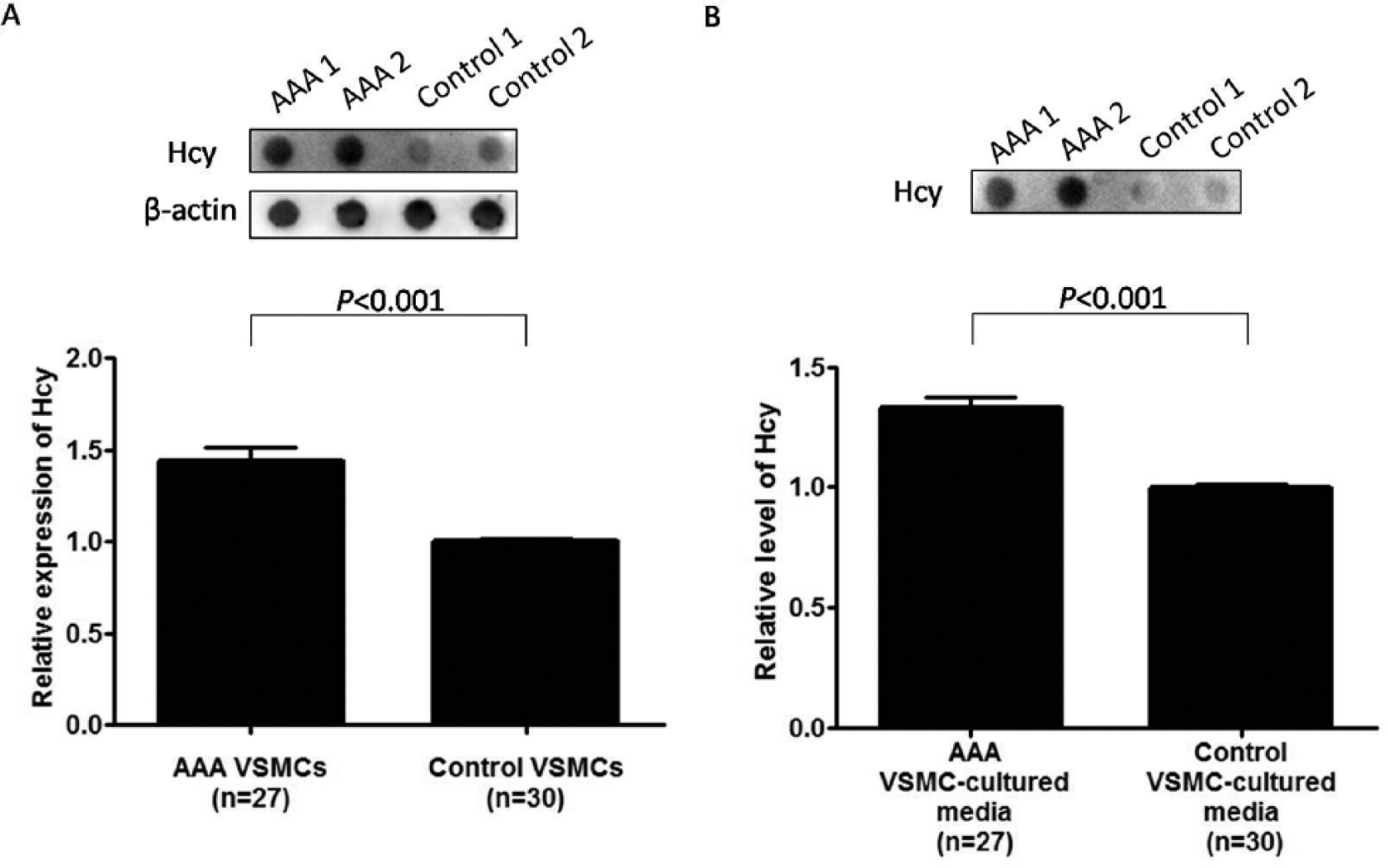
Detection of homocysteine (Hcy) level in (A) vascular smooth muscle cells (VSMCs) explant cultured from human abdominal aortic aneurysm (AAA) and control aortic tissues and (B) their cultured media by dot blotting. (A) Representative dot blot showing two AAA and two control VSMCs, probing for Hcy and β-actin (upper). Statistical analysis showed the relative Hcy expression in AAA VSMCs (n=27; 1.44±0.07) was significantly higher than that in control VSMCs (n=30; 1.00±0.02) (p<0.001) (lower). (B) Representative dot blot showing two AAA and two control VSMC-cultured media, probing for Hcy (upper). Statistical analysis showed the relative Hcy level in the cultured media of AAA VSMCs (n=27; 1.33±0.04) was significantly higher than that of control VSMCs (n=30; 1.00±0.01) (p<0.001) (lower).
Discussion
Firstly, this study aimed to investigate the tissue expression level of Hcy in human AAA tissues. Results from dot blotting showed a significantly higher Hcy expression in AAA compared with control aortic tissues from humans, which was further validated by IHC staining of tissue sections, suggesting an elevated tissue Hcy expression may be associated with AAA. Secondly, this study also aimed to investigate the association between AAA and MTHFR C677T polymorphism. No significant difference was found in the frequency of the variant T allele between AAA and control groups. On the other hand, no significant differences were observed in the frequencies of CT and TT genotypes between AAA and control groups. These findings implicated that there is no association between MTHFR C677T polymorphism and AAA, suggesting MTHFR C677T polymorphism may not be a risk factor of AAA pathogenesis. Thirdly, this study aimed to investigate the association between Hcy expression in aortic tissues and MTHFR C677T polymorphism. No significant difference was observed in the tissue Hcy expression level among the MTHFR C677T genotypes, suggesting there is no association between the tissue Hcy expression level and MTHFR C677T polymorphism. Therefore, the observed elevated Hcy expression in human AAA tissues may not be contributed by MTHFR C677T polymorphism. Lastly, the level of Hcy in VSMCs isolated from human AAA and control tissues was also investigated. The level of Hcy in AAA VSMCs was significantly higher than that in control VSMCs. Furthermore, the amount of Hcy released to cultured media from AAA VSMCs was significantly higher than that from control VSMCs. These findings showed an elevation of Hcy may be associated with AAA at the cellular level.
Although previous studies showed a high circulating Hcy in AAA patients, tissue Hcy expression from AAA patients has never been investigated.14–19 Our current finding of significantly higher Hcy expression in AAA than control tissues from humans may further indicate a disruption of Hcy metabolism is associated with AAA. Moreover, our findings of elevated Hcy level in both AAA VSMCs and the cultured media might possibly suggest a disruption of Hcy metabolism locally in VSMCs, which might contribute to hyperhomocysteinemia as reported in AAA patients.14–19 However, the other possibility that a disrupted Hcy metabolism might have taken place remotely, resulting in a high circulating Hcy, which is then diffused into the cells at the aortic wall, should not be eliminated. Future investigation of the Hcy level in AAA VSMCs and cultured media in a multiple time-point assay is necessary to differentiate these two possibilities.
Degradation of extracellular matrix elastin in the aortic wall is a pathophysiological feature of AAA. Hcy has been proposed to have a role in inducing proteolytic degradation of elastin, resulting in AAA. 6 Previous animal studies reported that Hcy could induce significant disruption of medial elastin in the aortic wall.20,21 Additionally, prior in vitro studies also showed Hcy could induce the production of serine elastase and matrix metalloproteinase (MMP)-2, which are well-known elastin-degrading proteases abundantly present in human AAA tissues, in arterial smooth muscle cells.33–35 It is thus hypothesized that Hcy may promote AAA formation by inducing proteases for proteolytic degradation of elastin in the aortic wall. An in vitro study should be performed to investigate the effect of elastinolysis and expressions of elastase and MMP-2 upon Hcy induction in VSMCs explant cultured from human AAA tissues.
Elevated Hcy was observed in the adventitial region of human AAA tissues from an IHC result. It is thus proposed that Hcy may interact with the adventitial cells, such as fibroblasts, for the pathogenesis of AAA. Furthermore, a prior study reported Hcy could aggravate aneurysm formation in mice, at least in part, by activating NADPH oxidase 4 in adventitial fibroblasts, although the importance of adventitial fibroblast Smad2/3 activation remained elusive. 21 This needs to be further investigated in human AAA samples.
The association between MTHFR C677T polymorphism and AAA has been widely investigated over the years but the findings were controversial. Studies with a relatively small sampling size showed a significant association, while studies with larger sampling sizes showed no significant association.4,18,19,27–31 Although these inconsistent findings may be due to the difference in sampling size, the confounding factor (i.e. ethnicity) should not be neglected as variation in the allele frequency of MTHFR C677T polymorphism could exist in different ethnicity and geographical areas. 36 In addition, the finding of a lack of association between MTHFR C677T polymorphism and AAA in this study is in concordance with the finding from the study conducted by Liu et al. in the Chinese population. 19 This possibly suggested that MTHFR C677T polymorphism may not be a risk factor of AAA in the Chinese population. A further large scale study is necessary to validate this finding. Furthermore, there are three other single nucleotide polymorphisms in the MTHFR gene (i.e. rs1801131, rs2274976 and rs4846049). Unlike C677T polymorphism, the associations between AAA and these single nucleotide polymorphisms are rarely investigated, although a prior study reported no significant associations. 37 In addition, associations between these polymorphisms and tissue Hcy expression were not investigated in that study. Therefore, further investigation is necessary.
MTHFR C677T polymorphism was once proposed to contribute to elevated Hcy expression in human AAA tissues as its variant protein showed a reduced catalytic activity in Hcy metabolism, but no association was found between the tissue Hcy expression level and MTHFR C677T polymorphism in this study. 25 Furthermore, together with the finding of no association between MTHFR C677T polymorphism and AAA shown in this study, it was hypothesized that such genetic polymorphism may not be involved in the pathogenesis of AAA. Moreover, it should be noted that the finding showing a reduced catalytic activity in variant MTHFR protein from the aforementioned in vitro study was performed on lymphocytes. 25 It is still uncertain whether such a reduction in catalytic activity of MTHFR arisen from genetic polymorphism is cell type-specific.
Although the disruption of Hcy metabolism in AAA may not be dependent on MTHFR C677T polymorphism, the tight down-regulation of MTHFR expression mediated by microRNAs (miRNAs), which are short RNAs for silencing specific gene expression, may be a possible cause of such disruption. 38 Furthermore, miRNA microarray analyses had identified several AAA differentially expressed-miRNAs, and, more importantly, those miRNAs were predicted to regulate MTHFR expression.39,40 It is thus hypothesized that the abnormally elevated Hcy in AAA VSMCs may be caused by a dysregulation of MTHFR through miRNAs. Further investigation is warranted.
Vitamin B12 has been proposed to play a role in AAA as it is a co-factor facilitating the remethylation of Hcy to methionine. 41 In fact, a significantly higher circulating Hcy level and a significantly lower vitamin B12 level have been reported in the same cohort of AAA patients in a prior study. 15 In view of this, correlation between circulating vitamin B12 level and Hcy expression in VSMCs should also be examined in a future study. Therefore, the potential therapeutic effect of supplemental vitamin B12 to treat AAA through restoring the disrupted Hcy metabolism remains to be investigated.
Limitations
There are several limitations in this study. One of the limitations is the small sampling size as human AAA tissue specimens are getting scarce due to the prevalence of endovascular aneurysm repair over open surgical repair. Moreover, there is a lack of data on circulating Hcy level and vitamin status since this is a retrospective study. The correlation between Hcy expression in tissue or VSMCs and circulating Hcy level, and their relationships with vitamins, could not be evaluated. Moreover, it is still uncertain if the lack of other demographic factors, such as body mass index, would affect the current observations, although several typical demographic factors were found to have no effect on the significant findings in this study. Therefore, a further study with a larger sampling size and collection of more comprehensive clinical data for analysis is warranted.
Conclusion
In conclusion, this is the first study showing an abnormally elevated Hcy expression in human AAA tissues. Such differential tissue Hcy expression may not be associated with MTHFR C677T polymorphism. Future study on the pathogenic mechanism of the disruption of Hcy metabolism may provide a therapeutic strategy to prevent the formation of AAA.
Footnotes
Acknowledgements
We are grateful for colleague surgeons in the Division of Hepatobiliary & Pancreatic Surgery and Liver Transplantation, Department of Surgery, University of Hong Kong Medical Centre, Pokfulam, Hong Kong, for obtaining human aortic tissues from organ donors.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was supported by the division fund from the Division of Vascular Surgery, Department of Surgery, University of Hong Kong Medical Centre, Pokfulam, Hong Kong.
