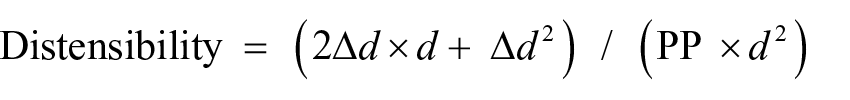Abstract
Arterial distensibility, an independent predictor of cardiovascular events, is transiently increased with acute hyperglycemia (AHG) in healthy individuals. Whether this response interacts with physical inactivity remains unknown. We examined the effects of short-term bed rest (BR) on the response of carotid artery distensibility (CD) to AHG, and the influence of underlying changes in insulin resistance and blood volume. CD was assessed with ultrasonography before as well as 30 and 120 minutes following ingestion of 75 g of glucose prior to and after 3 days of BR in 15 healthy male volunteers. Plasma insulin/glucose concentrations and blood volumes were concomitantly determined. On day 4 of BR, blood volume was re-established to pre-BR levels by albumin infusion and CD and insulin/glucose concentrations were determined as in the previous experimental days. Basal CD was not affected by BR. AHG increased CD before and after BR but reached a higher peak increment after BR (12% vs 60% at 30 min OGTT, p=0.028). BR also increased the plasma insulin concentration during AHG (p=0.007). In regression analyses, plasma insulin and glucose concentrations were positively correlated to CD, particularly after BR (r=0.31, p<0.05). Restoration of the BR-induced loss (5%) in blood volume did not affect the response of CD to AHG. In conclusion, short-term physical inactivity strongly accentuates the initial increase in CD in response to AHG in healthy individuals. This effect is associated with concomitant increases in circulating insulin concentration attributable to early insulin resistance.
Keywords
Introduction
Physical inactivity is a salient independent risk factor for diabetes, cardiovascular disease (CVD) and all-cause mortality.1–3 In western societies, the effect of sedentarism is often accompanied by unhealthy dietary habits generally characterized by excessive consumption of added sugars leading to acute episodes of hyperglycemia (AHG), which increase the risk of CVD. 4 Long-term physical inactivity and recurrent AHG are associated with adverse phenotypic alterations of vascular structure and dilator function.5,6 In this regard, sedentary individuals with poor dietary habits have stiffer arteries, a hallmark of arterial ageing.7–9 Arterial stiffness (AS), and its opposite arterial distensibility, characterize the capacity of an artery to expand and recoil in response to changes in blood pressure and strongly predicts CVD and mortality when assessed in central elastic (e.g. carotid) arteries. 10 However, relatively little is known regarding the early combined impact of physical inactivity and AHG on AS. This may be of particular relevance for a substantial percentage of industrialized populations who alternate short-term periods of relatively physical (in)activity on a weekly basis. 11
The detrimental effects of short-term physical inactivity on the vasculature have been demonstrated using a variety of experimental models (e.g. lower limb suspension, cast immobilization, bed rest (BR)). 12 Increased AS has been noted after only 5 days of BR in healthy young individuals.13,14 These observations parallel those related to glucose tolerance in that insulin resistance and the rise in circulating insulin levels induced by AHG were augmented following 3–5 days of BR.13,15 Given the (in)direct pleiotropic effects of insulin on functional and structural arterial properties,5,16 increased insulin resistance induced by short-term physical inactivity could influence the AS response to AHG. AS is commonly decreased with AHG or post-prandial conditions in healthy insulin-sensitive individuals.17–23 This response has been linked to the vasodilator effect of physiological doses of insulin in large elastic arteries, which is not observed under long-term insulin-resistant conditions.22,24 Indeed, AS is unchanged or even increased by AHG in individuals with type 2 diabetes.18,21 Possible mechanisms underlying the altered effect of AHG on AS in insulin-resistant individuals include: (i) a switch of the vascular effect of insulin from prevailing nitric oxide (NO)-dependent vasodilation to endothelin-1 (ET-1)-dependent vasoconstriction; 25 (ii) increased sympathetic activity responsiveness; 26 and/or (iii) augmented post-AHG inflammatory markers. 21 Whether these phenotypic modifications are present after short-term physical inactivity, thus affecting the AS response to AHG, is uncertain. Intriguingly, there is emerging evidence that prior to established ET-1 predominance and NO depletion, early stages of insulin resistance are characterized by enhanced NO activity and reduced AS. 27
Another variable influenced by physical inactivity and potentially affecting AS is blood volume. BR is typically associated with a rapid (24–48 hours) decrease in plasma volume (~10–20%) due to augmented diuresis in healthy individuals. 28 This may lead to reduced left ventricular preload, stroke volume and a compensatory increase in heart rate to preserve cardiac output. 29 In turn, chronic increases in heart rate are associated with augmented AS.30,31 In addition, acute decreases in blood volume may alter plasma hormone and nutrient intravascular concentrations, possibly influencing AS. However, no previous investigation has experimentally determined the impact of changes in blood volume on AS.
We assessed the effects of short-term (3 days) BR on the response of carotid artery distensibility (CD; inverse of carotid AS) to AHG and to what extent these effects are mediated by changes in insulin resistance and blood volume. We hypothesized that the increase in CD in response to AHG would be (i) accentuated by BR and associated with increases in insulin concentration and (ii) partially restored with the reversal of blood volume to pre-BR values by albumin infusion. Healthy, young adults were studied to limit the confounding influence of cardiovascular risk factors upon the early impact of physical inactivity and AHG on CD.
Methods and procedures
Ethical approval
The study was approved by the Ethical Commission of Zurich (KEK-ZH-Nr. 2015-0453) and conducted in accordance with the Declaration of Helsinki. Prior to the start of the experiments, informed oral and written consents were obtained from all participants. A parallel investigation from this cohort dealing with the effects of plasma volume alterations on glucose tolerance has recently been published. 32
Participants
Fifteen healthy young recreationally active male individuals volunteered to participate in the study. As inclusion criteria, all participants were normotensive (systolic blood pressure (SBP) < 130 mmHg and diastolic blood pressure (DBP) < 85 mmHg), medication free and without history of cardiovascular /metabolic/renal diseases, blood donation or sojourn to > 2000 m altitude within the last month. All individuals completed a screening visit to determine eligibility. Up to a month later, they started the study protocol. Participants were asked to maintain their usual physical activity levels before the study admission and avoid strenuous exercise, alcohol and caffeine from 24 hours prior to testing.
Experimental design
All individuals arrived on the first study day (day 0) in the morning after fasting overnight and had an assessment of CD, glucose tolerance and blood volumes, as described below. On completion of baseline experiments, participants began the BR period and remained in hospital beds for 4 full days (96 hours). Individuals were full-time monitored and permitted to perform quiet activities such as reading, watching television, working on the computer and talking on the phone. Individuals were allotted up to 5 hours per day in a sitting position (2.5 hours at 45° and 90°, respectively) leaning against the backrest of their beds. This included the time necessary to personal hygiene, which was performed with the aid of wheelchairs driven by nursing staff. A stopwatch was used to monitor all time spent out of bed. Ad libitum water and regular healthy food from the canteen at the University of Zurich (Irchel Campus) were served during the whole BR period. A minimum of 2000 ml water or soft drinks per day was assured to avoid the influence of dehydration. On day 3 of BR, CD, glucose tolerance and blood volumes were measured, after fasting overnight, at the same time of day as the baseline evaluation. These measurements were repeated after 24 hours (day 4) immediately following blood volume normalization with albumin infusion but otherwise conditions were identical to previous evaluations. Albumin infusion was performed as previously described. 33 In brief, participants were positioned on a bed with an 18-gauge catheter placed in an antecubital vein. A volume corresponding to the blood volume lost during BR was infused in the form of 20% human albumin (Albumin CSL 20%; CSL Behring, Switzerland). An overview of the timeline of all assessments is shown in Supplementary Table 1.
Experimental measures
Oral glucose tolerance test (OGTT)
The study participants underwent an oral glucose tolerance test (OGTT), where 1 g glucose · kg−1 body mass was diluted in 6 ml water · g−1 glucose. Blood samples for determination of plasma glucose and insulin concentrations were collected in EDTA tubes (Vacutainer; Becton Dickinson AG, Switzerland) immediately prior to and after 30 and 120 minutes of glucose drink intake. Plasma concentrations of glucose were measured using Synchron reagents and UniCel DxC 800 analyzer (Beckman and Coulter, Switzerland), while the plasma insulin concentration was determined with an ELISA kit (ALPCO, Salem, NH, USA).
Carotid artery distensibility (CD)
A single operator obtained properties of the left common carotid artery, with the use of an ultrasound scanner equipped with a 7 MHz linear array probe (M7; Mindray, China), before as well as 30 and 120 minutes after OGTT according to established guidelines. 34 All the measurements were made off-line and in duplicate, including an operator blind to the study conditions, as previously described. 35 Blood pressures were measured on the right arm with an automated system (Dinamap; GE Medical Systems, WI, USA). CD was calculated according to the following formula:
where d is arterial diameter, ∆d is distension and PP is brachial pulse pressure (calculated as systolic minus diastolic blood pressure).
Blood volumes
Plasma volume, blood volume and red blood cell volume were determined using a fully automated carbon monoxide (CO) re-breathing system (OpCO; Detalo, Switzerland) with a typical error of 1.0%. Total plasma volume, blood volume and red blood cell volume were derived from measures of hemoglobin mass and hematocrit. 36
Statistical analysis
Statistical analysis was performed using SPSS 22.0 (IBM Corp., Armonk, NY, USA). To examine the effects of BR and AHG on arterial structure and function, hemodynamics as well as insulin and glucose concentrations, a two-way ANOVA with repeated measures was used. The main effects for BR (pre-BR (day 0), post-BR (day 3)) and time during OGTT (baseline, 30 min, 120 min) as within-subjects factors were determined, along with interaction between these factors. A main effect for an independent variable (e.g. BR) is the effect of BR averaged across the levels (baseline, 30 min, 120 min) of the other independent variable (time during OGTT). The main effects can be essentially understood as the overall effect of a variable. When an interaction is present, the effect of one independent variable depends on the levels of the other independent variable. In addition, the effects of albumin infusion on the aforementioned variables were separately assessed with two-way ANOVA with repeated measures, with the main effects determined for albumin infusion (pre-albumin infusion (day 3), post-albumin infusion (day 4)) and time during OGTT (baseline, 30 min, 120 min) as within-subject factors, along with interaction between these factors. When F was significant in the ANOVA, planned pair-wise specific comparisons were carried out using Student’s paired t test. Furthermore, the relationship of insulin and glucose concentration with CD was assessed by means of Pearson’s correlation coefficients (r). A two-tailed p-value less than 0.05 was considered significant. All data are reported as mean (± SD) unless otherwise stated.
Results
Main baseline characteristics of the study subjects were: age = 24 ± 3 years, body weight = 77 ± 9 kg, height = 181 ± 4 cm, SBP = 126 ± 11 mmHg, DBP = 68 ± 6 mmHg. With respect to the diet provided during the BR period, all food was weighed, registered and caloric intake calculated to 2603 ± 446 kcal per day (14 ± 2% protein, 49 ± 3% carbohydrates, 33 ± 3% fat, 4% fibers).
Blood volumes and insulin/glucose concentrations (Figure 1 and Supplementary Figure 1)
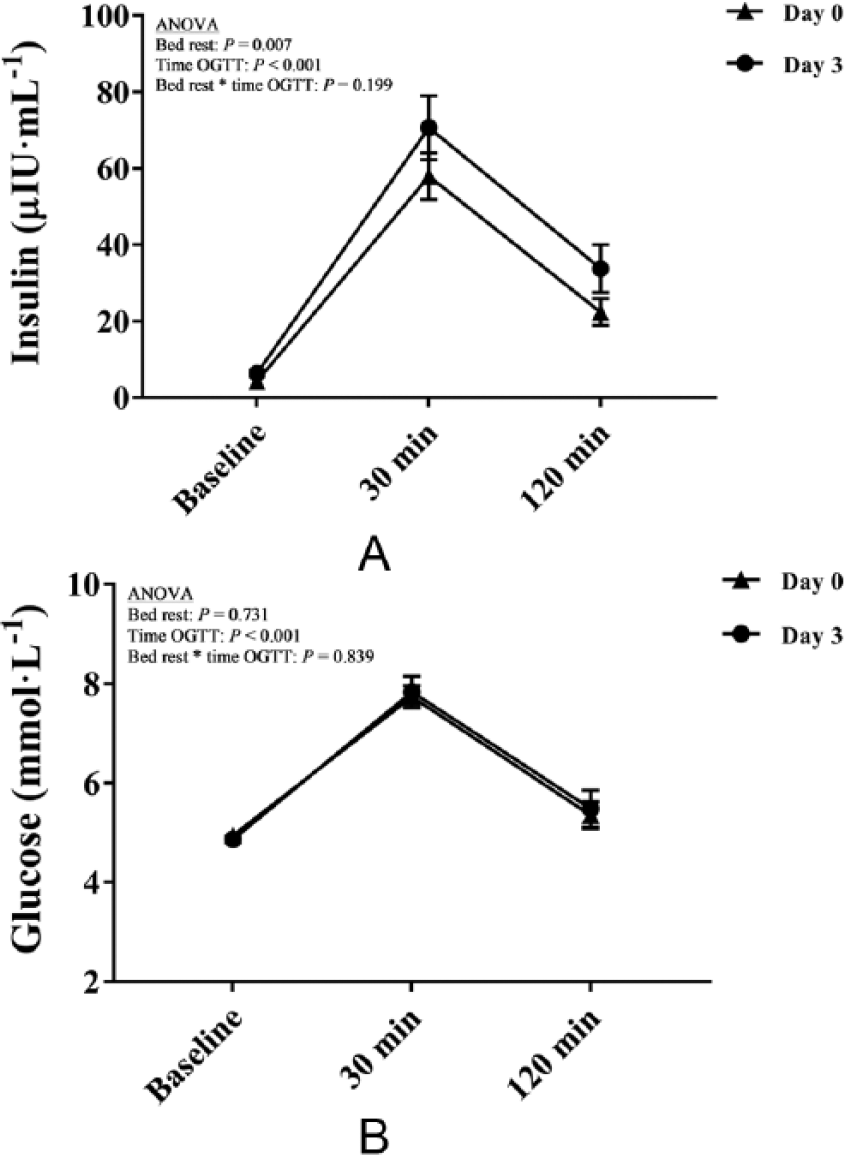
Effects of bed rest and acute hyperglycemia on insulin (A) and glucose (B) concentrations. Data are expressed as mean ± SEM. (OGTT, oral glucose tolerance test.)
The total blood volume decreased (5%) after BR (F = 43.1, p < 0.001) and was reversed to baseline values after albumin infusion (413 ± 99 ml). The contraction blood volume with BR was explained by that of plasma volume, which decreased (10%) with BR (F = 73.2, p < 0.001). Red blood cell volume was not affected by BR. Fasting plasma insulin concentration was increased after BR (F = 10.2, p = 0.007). Fasting plasma glucose concentration was not affected by BR. The overall effects of AHG were found in insulin (F = 58.9, p < 0.001) and glucose (F = 71.3, p < 0.001) concentrations, both being substantially augmented during OGTT. Albumin infusion did not alter insulin and glucose concentrations during OGTT.
Impact of BR and AHG on hemodynamics, carotid artery size and CD (Table 1, Figure 2)
Impact of bed rest, acute hyperglycemia and albumin infusion on blood pressure and carotid artery diameters.
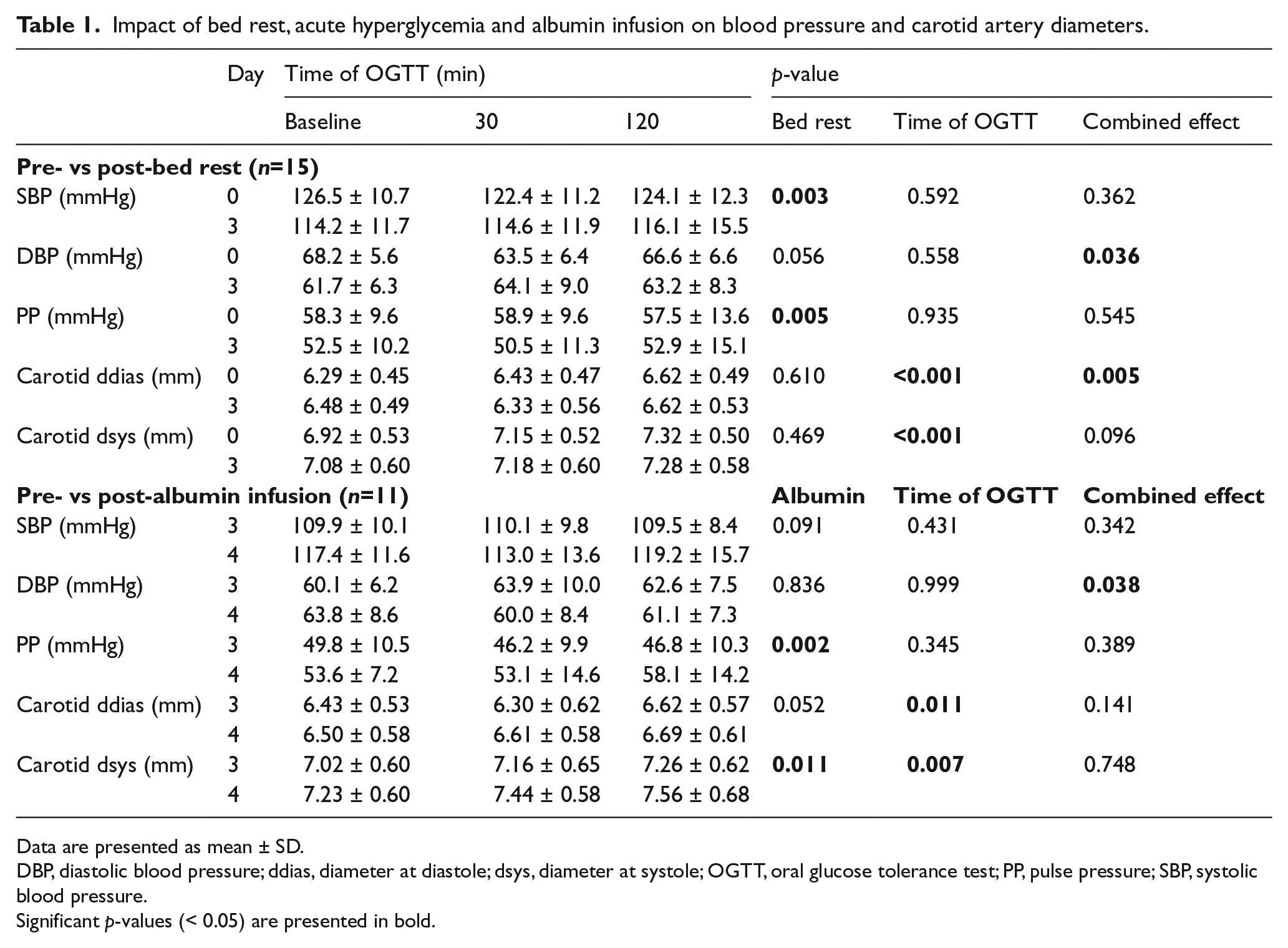
Data are presented as mean ± SD.
DBP, diastolic blood pressure; ddias, diameter at diastole; dsys, diameter at systole; OGTT, oral glucose tolerance test; PP, pulse pressure; SBP, systolic blood pressure.
Significant p-values (< 0.05) are presented in bold.
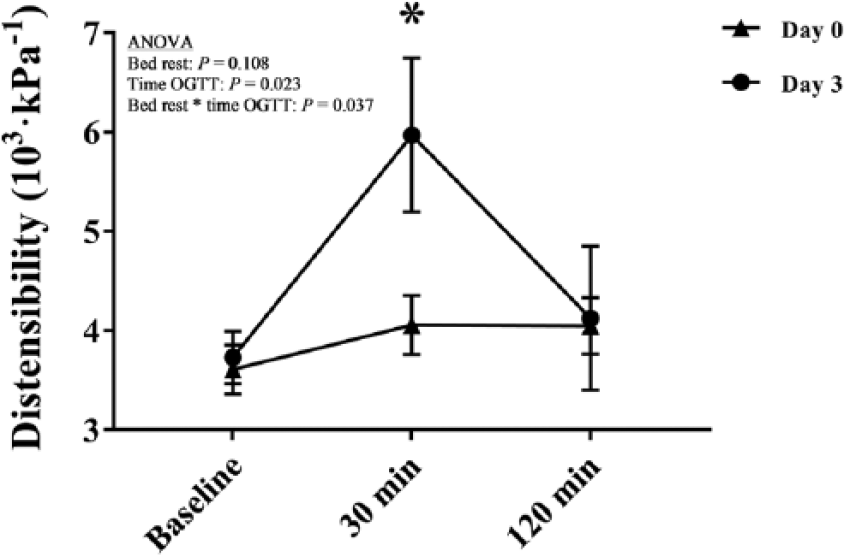
Effects of bed rest and acute hyperglycemia on carotid artery distensibility. Data are expressed as mean ± SEM. (OGTT, oral glucose tolerance test. *p = 0.028, day 0 at 30 min vs day 3 at 30 min.)
The overall effects of BR were observed on SBP (F = 12.8, p = 0.003) and PP (F = 11.0, p = 0.005), both being reduced (10%) after BR. Heart rate (59 ± 9 bpm) was not affected by BR and AHG. Analysis of carotid diastolic diameter revealed an overall effect of AHG (F = 12.4, p < 0.001) and a combined effect of AHG and BR (F = 6.5, p = 0.005), such that the carotid diastolic diameter was increased (2% from baseline) at the 30-minute OGTT on BR day 0 (p = 0.034) but not day 3 (p = 0.090). Carotid systolic diameter increased (up to 5% from baseline) during OGTT (ANOVA overall effect of AHG, F = 16.3, p < 0.001). With respect to carotid distensibility, an overall effect of AHG (F = 4.4, p = 0.023) and a combined effect of AHG and BR (F = 3.7, p = 0.037) were observed. Further analysis revealed that the increment in CD from baseline to the 30-minute OGTT was substantially higher on day 3 (60%) compared with day 0 (12%) of BR (p = 0.028). CD was similar at baseline (p = 0.686) and 120 minutes (p = 0.907) during OGTT on days 0 and 3 of BR.
Impact of albumin infusion and AHG on hemodynamics, carotid artery size and CD (Table 1, Figure 3)
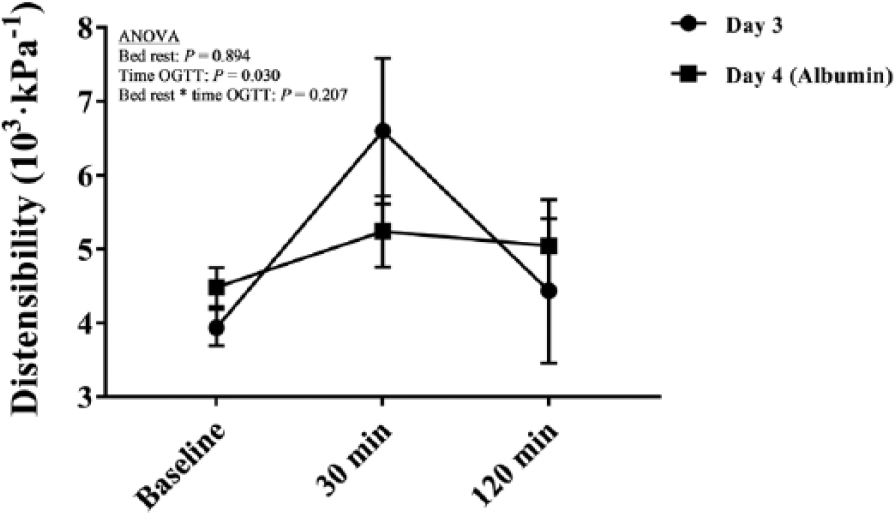
Effects of albumin infusion and acute hyperglycemia on carotid artery distensibility. Data are expressed as mean ± SEM. Analyses were performed on n =11. (OGTT, oral glucose tolerance test.)
Complete data after albumin infusion were not available in four individuals and hence the analyses were performed on n =11. While no overall effect was detected on SBP and DBP, PP (7%) and heart rate (30%) increased with albumin infusion (ANOVA overall effect of albumin infusion on PP, F = 17.3, p = 0.002; ANOVA overall effect of albumin infusion on heart rate, F = 29.9, p < 0.001). An overall effect of AHG was observed on the carotid diastolic diameter (F = 5.7, p = 0.011) and systolic diameter (F = 6.5, p = 0.007), both increasing (up to 5% from baseline) during OGTT. Moreover, the carotid systolic diameter increased (3%) with albumin infusion (ANOVA overall effect of albumin infusion, F = 9.8, p = 0.011). Furthermore, CD was not affected by albumin infusion but increased (up to 17% from baseline) during OGTT (ANOVA overall effect of AHG, F = 4.2, p = 0.030).
Relationship of insulin and glucose concentrations with CD
Using data from the entire study protocol (prior to and during OGTT for days 0, 3 and 4), plasma insulin (r = 0.21, p = 0.037) and glucose (r = 0.28, p = 0.005) levels were positively associated with CD. These relationships were particularly observed on day 3 (r = 0.31, p = 0.041 for insulin-CD and r = 0.31, p = 0.037 for glucose-CD association) but not day 0 (r = 0.08, p = 0.601 and r = 0.12, p = 0.413) or day 4 (r = 0.11, p = 0.557 and r = 0.32, p = 0.069).
Discussion
The major findings of the present study are: (i) 3 days of BR do not affect basal CD but strongly accentuate (fivefold) the initial increase in CD in response to AHG; (ii) this effect is associated with augmented insulin and glucose concentrations after BR; and (iii) restoration of blood volume after BR does not alter CD. These findings denote the combined effects of metabolic and vascular alterations elicited by short-term physical inactivity in healthy individuals.
Sound evidence demonstrates the association between chronic sedentary behavior and altered vascular health as reflected by the stiffening of central elastic arteries. 37 Notwithstanding that sedentary behavior is compounded by additional factors other than physical inactivity, such as unbalanced dietary and sleep patterns, BR is considered a classic experimental model to study the effects of sedentarism in humans.12–15 Early adverse effects have been reported following short-term (5 days) BR, 14 where Nosova et al. found increases in diastolic blood pressure and central augmentation index, an indirect measure of AS, in healthy young adults. However, these variables were unaltered following 5 weeks of BR in a recent study by Palombo et al. 29 Likewise, in the present study, neither blood pressure nor AS were increased by BR. Although the evidence remains scarce, the above divergences could be related, at least in part, to the severity of BR interventions. Notably, the protocol of Nosova et al. 14 did not allow the subjects to adopt any upright position during the whole BR intervention, including eating and excretion periods, plausibly increasing the effects of deconditioning. In that study, 14 subjects presented reduced levels of plasma adiponectin after BR, which may reflect increased psychological and oxidative stress, potentially contributing to hemodynamic alterations. 38 In addition, the early impact of physical inactivity on arterial distensibility may be influenced by the rate of progression of insulin resistance, which may be modulated by the characteristics of the BR intervention. In this respect, early insulin resistance is associated with a transitory increase in NO synthesis and preserved arterial distensibility until NO depletion occurs. 27 This effect has been attributed to high insulin levels in the presence of preserved insulin-mediated distensile properties,22,24 as supported by the current study and discussed in detail below.
In contrast to the deleterious effects of long-term recurrent AHG on arterial distensibility, 7 previous studies have consistently observed increases in arterial distensibility with AHG in insulin-sensitive individuals.17–23 This seemingly counterintuitive reduction in AS has been proposed to be mediated by the vasodilator function of insulin, which facilitates the delivery of insulin and nutrients to target tissues.16,22,24 In this regard, insulin acts on the vasculature via insulin receptor/phosphatidylinositol 3-kinase (PI3K) pathways, 39 which are present in both endothelium and vascular smooth muscle, leading to NO synthase activation, increased NO production and subsequent vasodilation. 40 Conversely, when these pathways are impaired in long-term insulin-resistant conditions, the vasoconstrictor effect of insulin, via Ras/mitogen-activated protein kinase and ET-1 production, predominates. 25 The question arises as to whether the vascular action of insulin is altered at the initial stages of insulin resistance, as induced by short-term physical inactivity. This study demonstrates that increases in CD are augmented following 3 days of BR in association with elevated insulin levels due to early insulin resistance. While greater CD in response to AHG may be seen as a positive adaptation, it is reasonable to expect that longer periods of physical inactivity may prompt opposite alterations due to progressive insulin resistance and/or NO depletion. 25 Further research is warranted to establish the time course of insulin resistance, NO bioavailability and basal arterial distensibility with physical inactivity.
An unexplored factor potentially contributing to changes in arterial distensibility is blood volume. The commonly observed 10–20% loss of plasma volume following short-term BR reduces mean systemic filling pressure, venous return and thereby (upright) stroke volume unless baroreflex feedback mechanisms restore circulatory homeostasis. 28 These comprise increased sympathetic stimulation of blood vessels, particularly the venous low-pressure system – which holds ~80% of the total blood volume – enhancing ventricular contractility and heart rate, all potentially affecting arterial distensibility.30,31 In the present study, plasma volume decreased (10%) after BR but neither heart rate nor basal CD were altered. It could be hypothesized that the supine position (intrinsic to BR) may attenuate the activation of feedback mechanisms with reduced blood volume. Nonetheless, basal CD was also not affected by acute blood volume normalization via albumin infusion, despite increased heart rate. This conflicts with the independent association between heart rate and CD established in large cross-sectional studies.30,31 Chronic effects of increased heart rate may involve arterial wall remodeling and structural stiffening due to gradual fragmentation of elastin fibers as well as accumulation of collagen fibers in the carotid media layer.30,31 In our study, the impact of plasma volume expansion could have been cushioned by intact carotid elastic properties, as supported by the increase in carotid systolic diameter. Therefore, CD seems to be preserved in response to moderate increases in blood volume of an acute nature.
Limitations
Findings were obtained from a sample of healthy young males. Further research will elucidate whether current conclusions can be extrapolated to populations including females and/or older individuals. A control intervention for time was not included, although previous observations indicate minimal fluctuation of arterial function during 5 days in healthy males. 13 This contrasts with marked changes in the CD response to AHG with BR in the present study. In addition, the effects of BR on CD could only be detectable with an acute stressor. Herein we tested AHG but other stressors might prompt similar results. Finally, physical inactivity may be associated with multiple metabolic and vascular phenotypic modifications; 12 thus, a strictly independent relationship between insulin levels and CD remains to be established.
Conclusions
The current study demonstrates that short-term physical inactivity, as elicited by 3 days of BR, has a major impact on the increase in CD induced by AHG in healthy individuals. This effect is related to an augmented insulin response to AHG, reflecting increased insulin resistance after BR. In turn, changes in CD are not influenced by BR-induced reductions in blood volume. These data therefore provide evidence of the early interplay of vascular health and insulin resistance with physical inactivity, independent of underlying alterations in blood volume.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by a grant from the Zürich Center for Integrative Human Physiology to C.L.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.