Abstract
Endothelial dysfunction plays a major role in cardiovascular diseases and pulse amplitude tonometry (PAT) offers a non-invasive way to assess endothelial dysfunction. However, data about the reliability of PAT in cardiovascular patient populations are scarce. Thus, we evaluated the test-retest reliability of PAT using the natural logarithmic transformed reactive hyperaemia index (LnRHI). Our cohort consisted of 91 patients (mean age: 65±9.7 years, 32% female), who were divided into four groups: those with heart failure with preserved ejection fraction (HFpEF) (n=25), heart failure with reduced ejection fraction (HFrEF) (n=22), diabetic nephropathy (n=21), and arterial hypertension (n=23). All subjects underwent two separate PAT measurements at a median interval of 7 days (range 4–14 days). LnRHI derived by PAT showed good reliability in subjects with diabetic nephropathy (intra-class correlation (ICC) = 0.863) and satisfactory reliability in patients with both HFpEF (ICC = 0.557) and HFrEF (ICC = 0.576). However, in subjects with arterial hypertension, reliability was poor (ICC = 0.125). We demonstrated that PAT is a reliable technique to assess endothelial dysfunction in adults with diabetic nephropathy, HFpEF or HFrEF. However, in subjects with arterial hypertension, we did not find sufficient reliability, which can possibly be attributed to variations in heart rate and the respective time of the assessments.
Keywords
Introduction
Endothelial dysfunction (ED) plays a major role in the pathogenesis and progression of cardiovascular diseases (CVD)1–3 and has important prognostic implications for subsequent cardiovascular events.4–6 ED and arterial hypertension showed reciprocal enhancement in their pathophysiologies and reinforce each other. 7 At the same time, ED is a potential prognostic marker as well as an interventional target in the pathogenesis and progression of arterial hypertension, heart failure5,8 and diabetes – as well as ED-induced CVD and renal diseases. 9 In summary, ED as a common pathogenesis factor is important in the progression and the prognosis of these diseases.
Several methods have been established for evaluating ED. Additional to invasive procedures such as the intravascular infusion of vasoactive agents to evaluate the endothelial function, flow-mediated dilation (FMD) is the most common non-invasive technique. Results in FMD may lack reproducibility due to the dependence on both an adequate ultrasound system and the operator’s experience,10,11 especially in large multicentre clinical trials. 12 Only a few devices have been developed to target and reduce this error, among them EndoPATTM (Itamar Medical Ltd, Caesarea, Israel), to measure the endothelial function, using pulse amplitude tonometry (PAT). EndoPAT provides the augmentation index (AI), the baseline pulse amplitude and the reactive hyperaemia index (RHI) which correlates with ED and cardiovascular risk factors.13,14 Studies have found the AI to be a predictor of adverse cardiovascular events in different patient populations, with higher AI indicating increased end-organ damage. 15 Local peripheral arterial tone is reflected by the baseline pulse amplitude and is directly related to most cardiovascular risk factors. 14 The RHI reflects changes in vascular tone that primarily depend on nitric oxide-induced effects. 16 The natural logarithmic transformed reactive hyperaemia index (LnRHI) is calculated using natural log transformation, a monotonic transformation. LnRHI provides a more symmetric distribution that is closer to normal distribution than RHI. An LnRHI of ⩽ 0.51 has been defined as outside the normal range. 17
Digital PAT has been measured in various disease states and results have been published extensively in the past few years. Prior studies detected a relation between a low RHI and future cardiovascular adverse events, 18 cardiovascular risk factors 14 in high-risk patients, 1 patients with early coronary atherosclerosis13,19 and those with heart failure with preserved ejection fraction (HFpEF).5,20 However, before PAT can be tested in clinical trials and clinical risk assessment, the validation of day-to-day variation, especially in patients with increased cardiovascular risk, is essential. Several studies have reported that PAT was reliable in healthy subjects21–24 and in those with metabolic syndrome 25 and coronary heart disease19,26 but knowledge about reproducibility in patients with CVD, especially with heart failure, is limited and data about influences interfering with PAT are rare. Thus, data about reliable assessment of endothelial function is crucial; it allows primary diagnosis, evaluation of severity, and long-term follow-up of patients with CVD. In summary, PAT has been used for many years and the current literature indicates good reliability and feasibility of PAT in general. Nevertheless, data concerning the reliability of PAT in patients with CVD, especially those with heart failure, are lacking.
Thus, the purpose of this study was to analyse for the first time the day-to-day test-retest reliability of PAT as a measurement for ED in patients with HFpEF, heart failure with reduced ejection fraction (HFrEF), diabetic nephropathy and in subjects with arterial hypertension without structural cardiovascular damage, a patient group which has never been looked at in detail before.
Methods
Ethics
This investigator-initiated, prospective study was performed at the Charité – University Medicine Berlin. The protocol and amendments were approved by the Medical Ethics Committee of Charité – University Medicine Berlin. All patients provided written informed consent before enrolment. All procedures related to the trial conformed to the principles outlined in the Declaration of Helsinki and ICH-GCP. This trial is registered at clinicaltrials.gov under the clinical trials number: NCT02299960.
Study subjects
All subjects (n = 91) in this prospective, observational, single-centre study were recruited from the database of the outpatient department at the University Hospital Charité between August 2014 and September 2015. Patients had to be clinically stable and aged between 35 and 80 years. Exclusion criteria were symptomatic coronary heart disease, coronary intervention in the past 4 weeks, myocardial infarction or stroke during the past 3 months, relevant valvular disease, initiation of cardiac resynchronization therapy during the past 3 months or implantable-cardioverter-defibrillator implantation during the past 4 weeks, uncontrolled hypertension (systolic blood pressure > 180 mmHg or diastolic blood pressure > 95 mmHg), anaemia with haemoglobin < 10 mg/dL, or untreated thyroid disease. General inclusion and exclusion criteria are displayed in the online supplement (Appendix 1).
Four groups of patients were included. Firstly, 25 participants with HFpEF were included based on the modified criteria published by Paulus et al. 27 Further criteria were symptomatic heart failure New York Heart Association (NYHA) class I–III 3 months prior to screening and sinus rhythm.
Secondly, 22 participants with HFrEF based on an ischemic or non-ischemic aetiology were included. Inclusion criteria were ejection fraction ⩽ 45%, NYHA class I–III, and on optimized guideline-directed treatment before screening.
The third group were 21 participants with diabetic nephropathy and type 2 diabetes mellitus under treatment other than dietary with the following additional inclusion criteria: glomerular filtration rate (GFR) < 90 mL/min/1.73 m2 (CKD-EPI), 28 macro-albuminuria (urine albumin to creatinine ratio (UACR) > 300 mg/g creatinine) or micro-albuminuria (UACR 30–300 mg/g creatinine).
The last group consisted of 23 participants diagnosed with arterial hypertension ⩾ 6 months prior to screening and without evidence of structural heart disease (clinically and on echocardiography). Inclusion criteria were no changes in medication for ⩾ 4 weeks prior to screening and systolic blood pressure < 140 mmHg during the screening visit (under anti-hypertensive medication). Exclusion criteria were secondary hypertension, intraventricular septal thickness (IVS) ⩾ 13 mm or left atrial volume index (LAVI) ⩾ 25 mL/kg/m2, detection of structural heart disease or systolic blood pressure > 140 mmHg during the screening visit. Three patients were pre-menopausal. Data about the menstrual cycle were not recorded.
Measurement procedure
All subjects underwent two separate PAT measurements in a period of 4–14 days with a median interval of 7.0 (25th/75th percentile: 7.0/9.0) days between the measurements. The aim was to complete the tests in the morning: 69.8% of measurements were performed between 08:30 a.m. and 12:59 p.m. In 81 patients, the tests at visit 1 and visit 2 were performed at the same time of day, with a variance of less than 3 hours. A daytime variance of more than 3 hours between visit 1 and visit 2 was present in 12 cases, five being patients with HFpEF, one patient with HFrEF, three with arterial hypertension and three patients with diabetic nephropathy. All subjects were instructed to remain fasting and not to smoke or consume caffeine for at least 8 hours before measurement. Medication had to be taken as normal except nitrates, which had to be stopped for the visit day. Blood pressure was measured with a digital haemodynamometer (OMRON M6 comfort, Omron, Japan) on the dominant arm and the values were entered into the EndoPAT software (Version 3.5.4) in addition to weight, height and age. All subjects rested for at least 5 minutes (lying down) in a well-tempered and soft-lit room, according to the manufacturer’s instructions. Both arms were resting on the arm support provided by the manufacturer to avoid muscular activity of the upper extremities. The plethysmographic-based finger probes were placed on both index fingers and the cuff on the non-dominant arm. The signal measured from the finger probes, which records the finger arterial pulsatile volume changes and thus the peripheral vasodilator response as a measure of ED, 29 was monitored until it was stable, with a maximum of 5 minutes. After a 6-minute baseline, the cuff was inflated to between minimally 200 mmHg and maximally 300 mmHg (but at least 100 mmHg over the blood pressure measured before, depending on the signal) to occlude the non-dominant arm for 5 minutes. After release of the cuff, the signal was recorded for another 6 minutes (post-occlusion time). As the results of each test, the software calculated the value for endothelial dysfunction (RHI), arterial stiffness (AI), AI normalized to a heart rate of 75 bpm (AI@75) and mean baseline pulse amplitude for each arm by using the pulse wave amplitude. RHI was calculated as the difference in the PAT signal ratio of the occluded arm before and after occlusion, relative to the same ratio in the control arm and corrected for baseline vascular tone. 29 Because the RHI has a skewed distribution, the RHI was transformed to its natural logarithm (LnRHI) to better fit to normal distribution. We also utilized fRHI, which does not include baseline correction and showed stronger cardiovascular risk correlation. 14 All data provided by the software were anonymized and transmitted to the manufacturer for analysis of quality and categorized into three categories. Subjects with ‘good’ or ‘moderate’ data quality were included; subjects with ‘low’ quality data were excluded from the study.
Data analysis
The statistical analysis was performed by an independent external statistician (A.B.). All analyses were conducted with R V 3.3.1. 30 Continuous variables with normal distribution are expressed as mean ± standard deviation (SD). Continuous variables with skewed distribution are summarized as median and 25th/75th percentile. Test-retest reliability was analysed with intra-class correlation (ICC) type II (single random raters), calculated as (MSB – MSE) / (MSB + (nr − 1) * MSE + nr * (MSJ – MSE) / nc) Sb2 – Sw2 / Sb2 + Sw2 (where ‘Sb2’ = between-subject variance; ‘Sw2’ = within-subject variance; MSB = between mean square variance; MSE = variance and mean squared error; MSJ = mean square between judges).
Data were plotted as scatterplots for visit 1 versus visit 2 with expected fit, and estimated linear and non-linear fit, using the non-parametric regression method ‘loess’ (locally weighted scatterplot smoothing). 31
Furthermore, Bland–Altman plots were generated depicting the relation between differences in visit 1 and visit 2 and mean (visit 1 / visit 2). Confidence intervals (CI) for ICC (ICC_CI) were calculated using the method of Shrout and Fleiss. 32
The mean variability is reported as mean ± SD of absolute differences in visit 1 and visit 2. For better comparison of variabilities, coefficients of variation (CV) were computed based on absolute differences. CV was calculated as SD / mean × 100 and was reported and expressed in percentages. Variables were tested for normality and transformed when appropriate. We examined correlations among AI, AI@75, mean baseline pulse amplitude, fRHI, RHI and LnRHI. The mean baseline pulse amplitude (MeanBPA) was calculated as (MeanBPAoccluded arm + MeanBPAcontrol arm) / 2. A p < 0.05 was considered statistically significant. Sample size calculations were computed using R. The necessary minimal sample size is based on the expected ICC of rho1 = 0.8 versus rho0 = 0.5, as the minimal ICC of clinical relevance.
Results
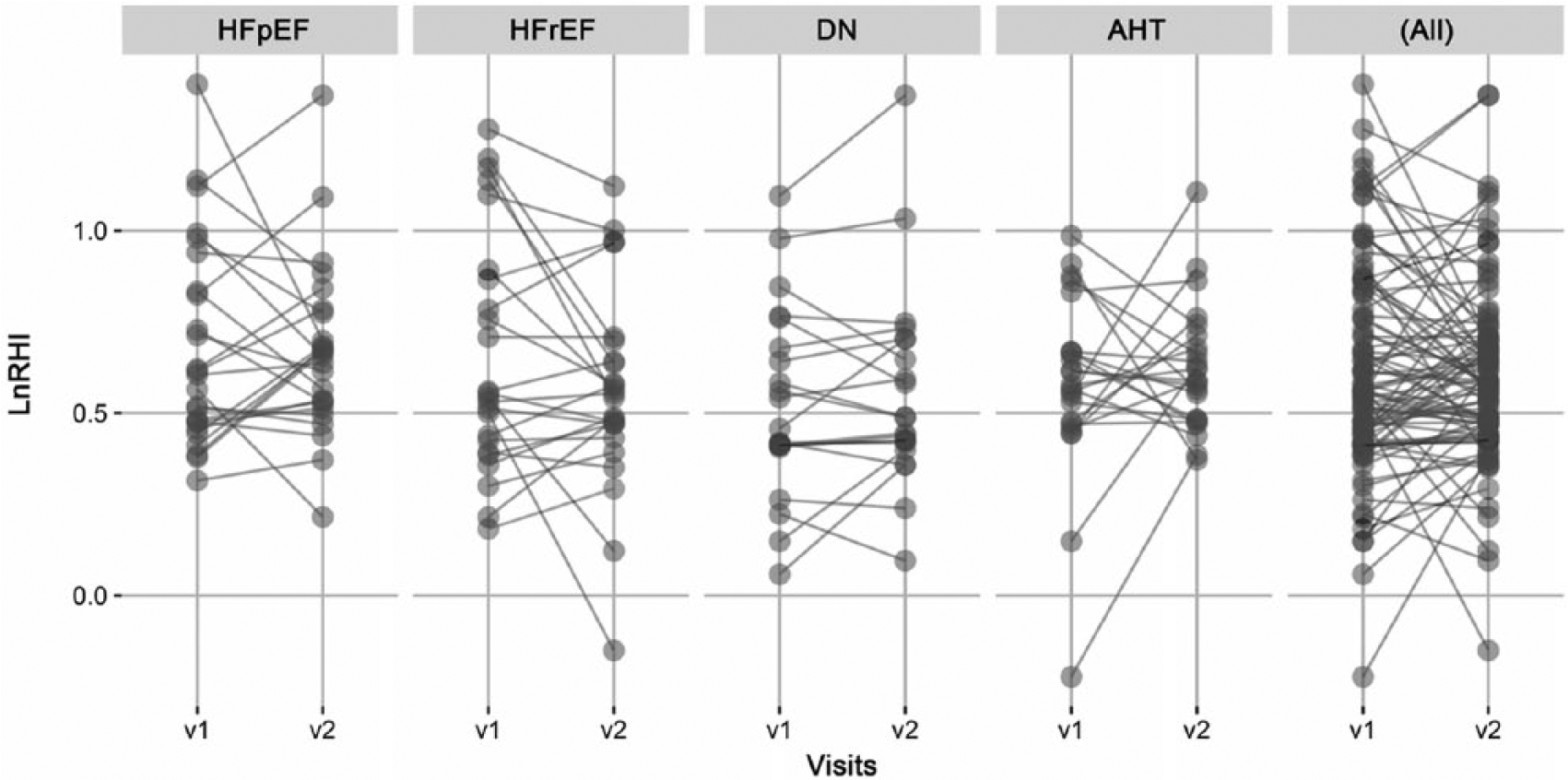
We identified 171 potentially eligible participants for inclusion in this study. Seventy were excluded because they did not meet the inclusion criteria. Of the remaining 101 subjects, 10 were excluded because of poor PAT signal quality (n = 3), failed test because of computer error or incomplete PAT data acquisition (n = 4), missing data file (n = 2) and exceedance of the maximum time interval of 14 days between the visits (n = 1) (Figure 1). Thus, the 91 remaining subjects were included in the study and two PAT measurements were performed and analysed as described in the Methods section. Baseline characteristics are shown in Table 1. Mean variability, CV, ICC and ICC_CI of investigated parameters of visit 1 and visit 2 are displayed in Table 2. A Bland–Altman plot of LnRHI is shown in Figure 2, and raw data for LnRHI in visit 1 and visit 2 are shown in Figure 3. Figure 4 represents the heart rate in visit 1 and visit 2.
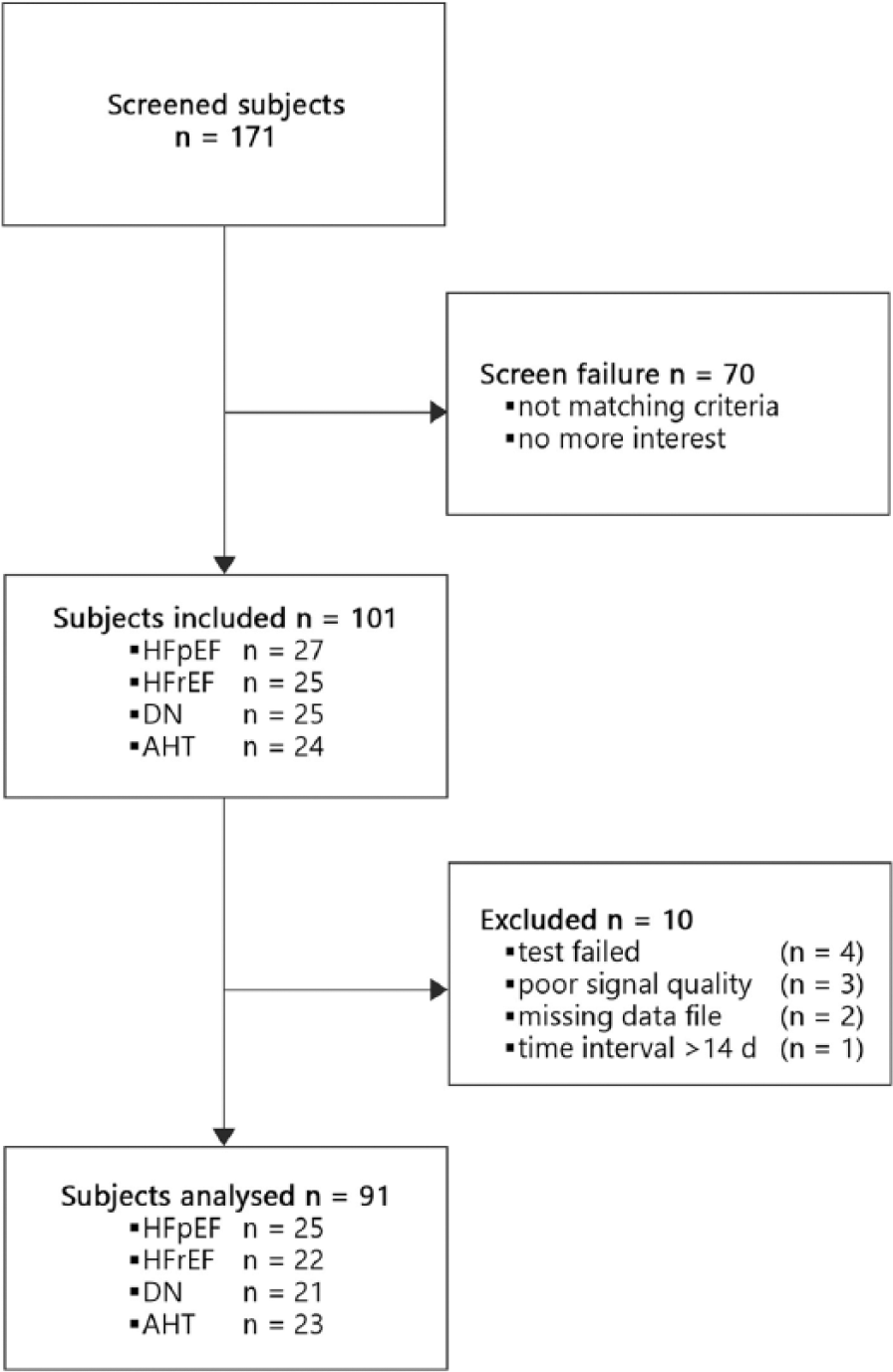
Flow chart showing the recruited collective of patients. (HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; DN, diabetic nephropathy; AHT, arterial hypertension; d, days.)
Baseline characteristics, medication, and laboratory parameters of all subjects at the screening visit, grouped by main diseases.
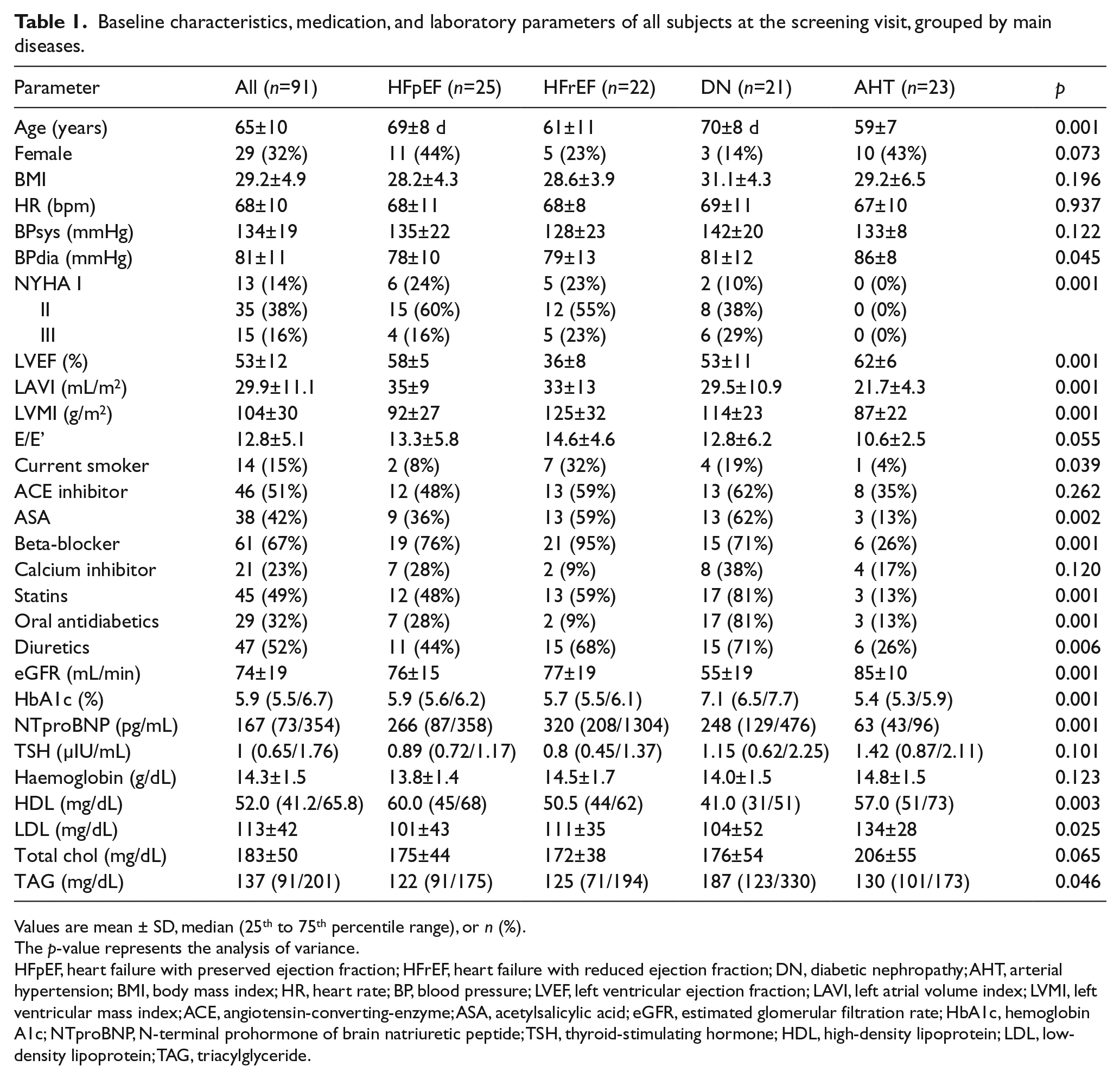
Values are mean ± SD, median (25th to 75th percentile range), or n (%).
The p-value represents the analysis of variance.
HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; DN, diabetic nephropathy; AHT, arterial hypertension; BMI, body mass index; HR, heart rate; BP, blood pressure; LVEF, left ventricular ejection fraction; LAVI, left atrial volume index; LVMI, left ventricular mass index; ACE, angiotensin-converting-enzyme; ASA, acetylsalicylic acid; eGFR, estimated glomerular filtration rate; HbA1c, hemoglobin A1c; NTproBNP, N-terminal prohormone of brain natriuretic peptide; TSH, thyroid-stimulating hormone; HDL, high-density lipoprotein; LDL, low-density lipoprotein; TAG, triacylglyceride.
EndoPAT™ scores measured in fasting subjects across visit days.
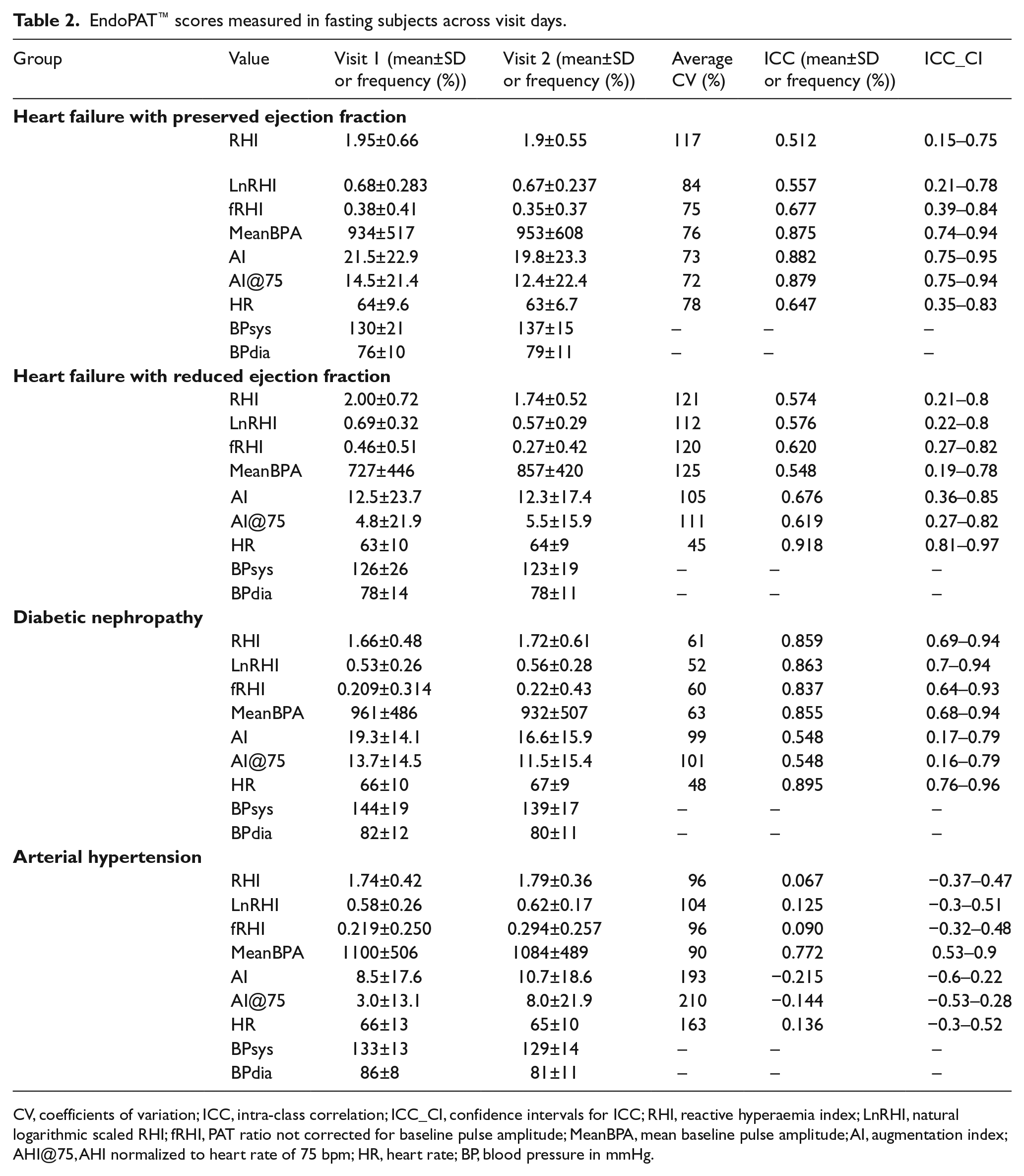
CV, coefficients of variation; ICC, intra-class correlation; ICC_CI, confidence intervals for ICC; RHI, reactive hyperaemia index; LnRHI, natural logarithmic scaled RHI; fRHI, PAT ratio not corrected for baseline pulse amplitude; MeanBPA, mean baseline pulse amplitude; AI, augmentation index; AHI@75, AHI normalized to heart rate of 75 bpm; HR, heart rate; BP, blood pressure in mmHg.
Bland–Altman plot depicting the relation between natural logarithmic transformed reactive hyperaemia index (LnRHI) differences in visit 1 and visit 2 and mean (visit 1 / visit 2) – and showing the regression line between delta and mean to explore potential dependencies. No formal test for significance is included.
Test-retest raw data for natural logarithmic transformed reactive hyperaemia index (LnRHI). (v1, visit 1; v2, visit 2; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; DN, diabetic nephropathy; AHT, arterial hypertension.)
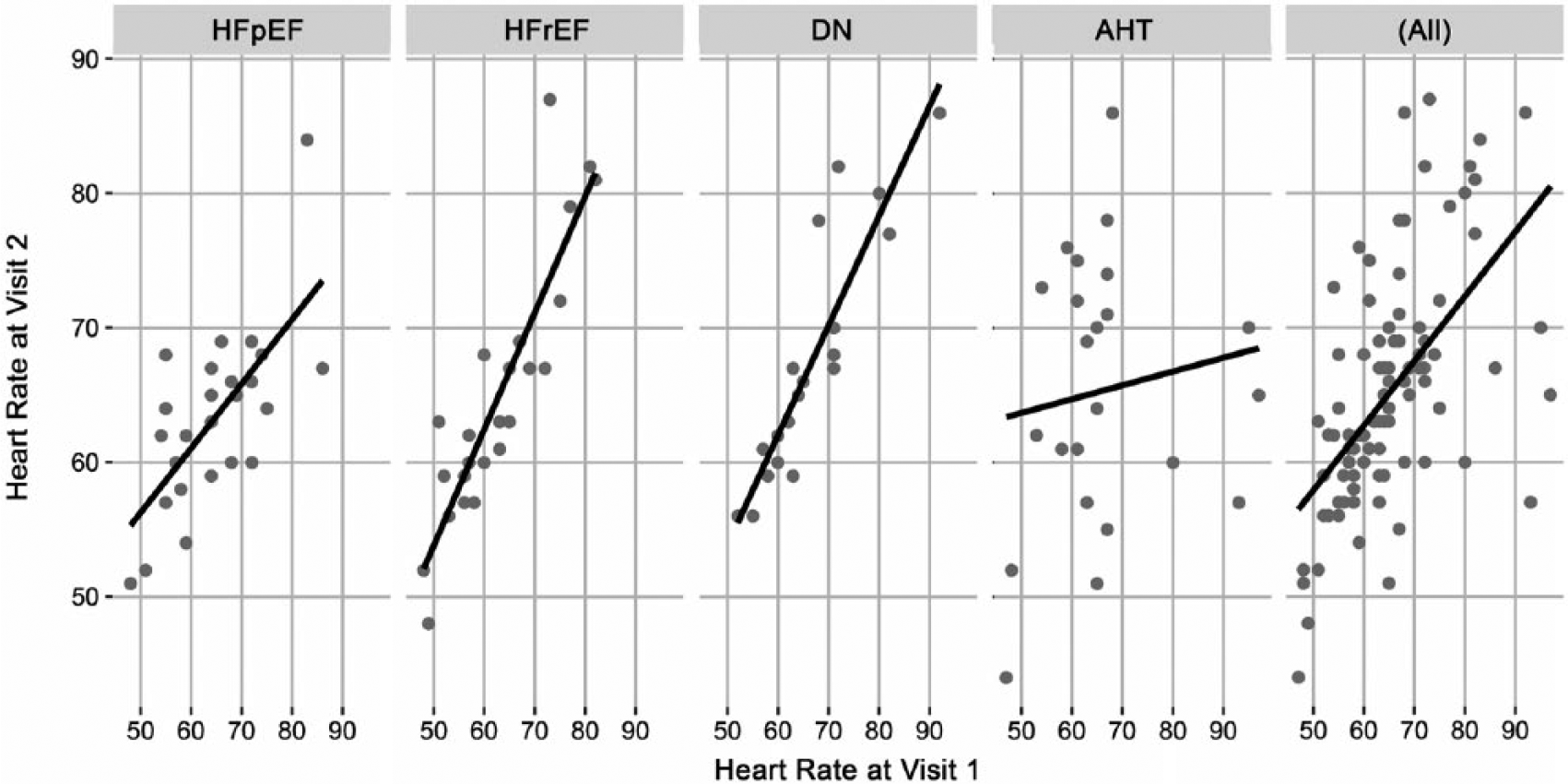
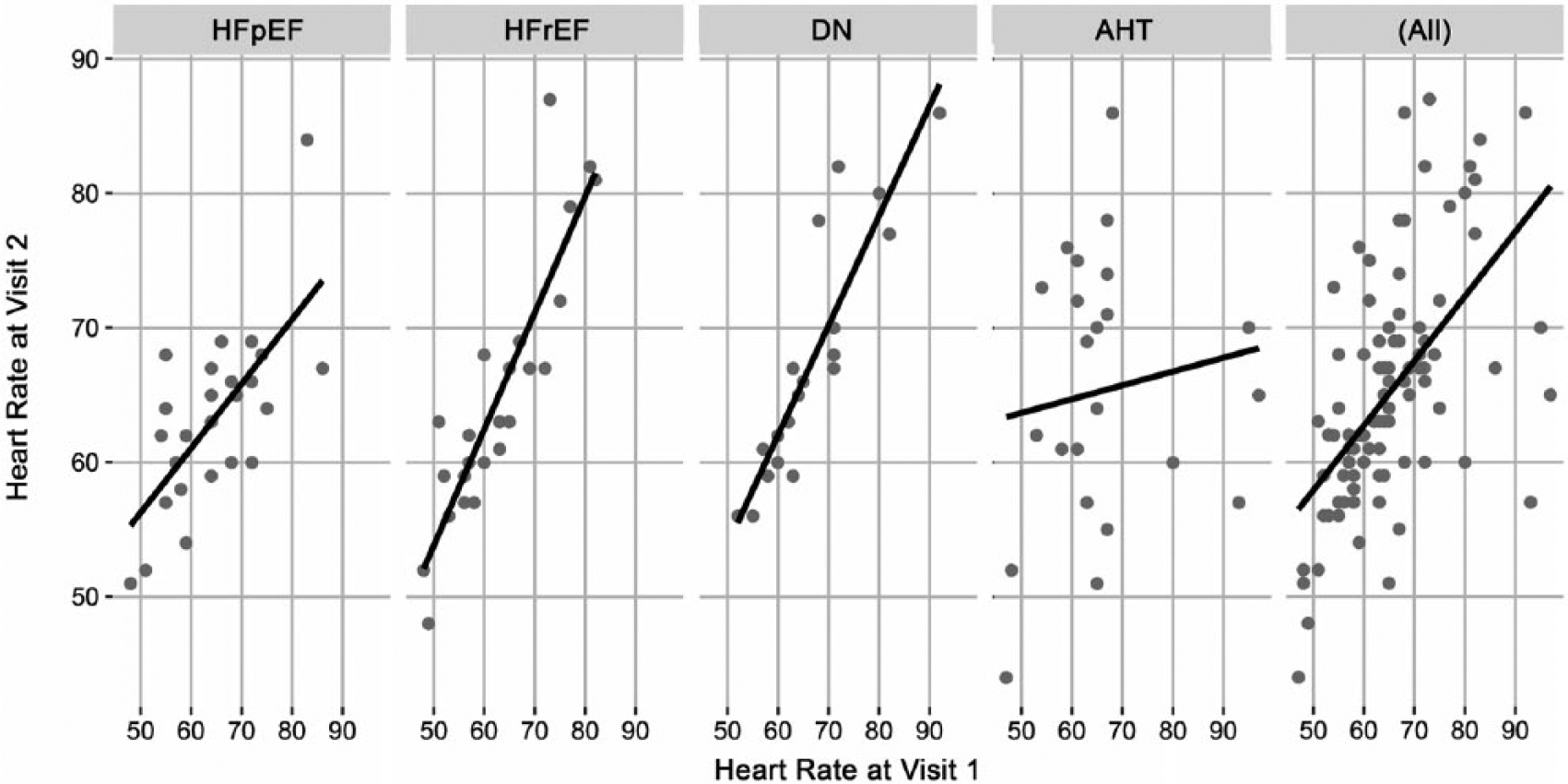
Heart rate (bpm) in visit 1 to visit 2. (HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; DN, diabetic nephropathy; AHT, arterial hypertension.)
Mean LnRHI in all PAT measurements was 0.62±0.29 in visit 1 and 0.61±0.25 in visit 2. Median time between the two measurements was 7 days. Correlations between paired measurements (visit 1 vs visit 2) were moderate to high in strength (ICC ranging from 0.557 to 0.863, AI@75 ranging from 0.548 to 0.879) except in subjects with arterial hypertension in whom we measured poor agreement (LnRHI-ICC = 0.125, AI@75-ICC = 0.144).
In subjects with arterial hypertension, the CV for LnRHI was 104%, LnRHI-ICC = 0.125, RHI-ICC = 0.067, fRHI-ICC = 0.09, AI-ICC = −0.215 and AI@75-ICC = −0.144. In patients with HFpEF, the CV for LnRHI was 84%, LnRHI-ICC = 0.557, RHI-ICC = 0.512, fRHI-ICC = 0.677. For AI and AI@75, the ICC was 0.882 and 0.879, respectively. In patients with HFrEF, the CV for LnRHI was 112%, and the ICC = 0.576, RHI-ICC = 0.574 and fRHI-ICC = 0.620. For AI and AI@75, the ICC was 0.676 and 0.619, respectively. In subjects with diabetic nephropathy, the CV for LnRHI was 52% and the ICC = 0.863, RHI-ICC = 0.859 and fRHI-ICC = 0.837. For AI and AI@75, the ICC was 0.548 and 0.548, respectively.
Discussion
ED plays an important part in CVD and is associated with considerable morbidity and mortality.1,2,5,6,8,33 Consequently, ED is a potential prognostic marker and target for (pharmaceutical) intervention and is expected to gain even more interest in the coming years.
To our knowledge, this is the first study to evaluate the test-retest reliability of PAT in patients with HFpEF, HFrEF, diabetic nephropathy and arterial hypertension since we are in need of new surrogate parameters for the prevention and progression of these diseases. We have demonstrated good reliability of PAT measures of endothelial function in subjects with diabetic nephropathy. In subjects with HFpEF and HFrEF, we assume moderate reliability, considering the wide range of the ICC_CI. These results are similar to those of previously reported studies of healthy participants, patients with metabolic syndrome and CVD19,21–26 where reproducibility of PAT was moderate to high. However, in subjects with arterial hypertension, we measured poor reliability agreement except for baseline pulse amplitude; here we were able to show good reliability in all cohorts. As a quality control for each PAT measurement, we used a stable baseline signal and complete cuff occlusion, both derived by the device, and data were evaluated for quality by the manufacturer.
Reviewing and analysing all collected data, the poor agreement was probably related to the higher variance in heart rate between visit 1 and 2, which was the most obviously varying parameter compared with the other groups (Figure 4). Owing to medication, we could not determine any relation affecting the heart rate variability across these groups. Studies have shown that heart failure and diabetes are associated with decreased heart rate variability.34,35 These results sustain higher heart rate variance in patients with arterial hypertension. Despite considering as many factors as possible affecting the heart rate, we could not finally clarify the reason for the unusually higher heart rate variance in patients with arterial hypertension.
In addition, RHI is influenced by sympathetic nervous system activity, 36 humoral factors and temperature, etc. These factors may have a higher variability in some individuals than in others. 19 Furthermore, the arterial hypertension group showed the greatest daytime variability between the two visits. Here, 13 participants underwent their PAT measurement after 1 p.m. Then again, Liu et al. have shown that the intra-day variability of PAT is low, but the time of day can influence PAT results. 22 Thus, the daytime variance in our subjects with arterial hypertension might also have had an influence on the poor agreement. In combination with the higher heart rate variance, it may be one of the reasons for the poor agreement of the PAT reliability in this group.
In order to control variables known to influence endothelial function, in this study we limited the post-prandial effects and recent exercise, room temperature variations, disallowed the use of nitrates on the visit day and standardized the time of day for each sample as far as possible. Furthermore, alcohol, caffeine and nicotine were prohibited for 8 hours prior to testing and the daily medication was stable for more than 7 days before screening. In accordance with the manufacturer’s manual, the measurement procedure was also standardized in terms of posture, probe placement and resting time before the test to minimize sympathetic effects. However, we cannot exclude that some of these variables may still have been influenced by subjects’ habits and behaviour.
In comparison to PAT, FMD is currently considered the gold standard for non-invasive endothelial function assessment, evaluating cardiovascular risk, prognostic information and need of intervention. 37 Nevertheless, the associated costs and higher investigator dependence and variability are limiting factors for using this technique for clinical decisions or large trials.11,12 As well as FMD, PAT is an independent predictor of cardiovascular events and mortality. 38 In contrast to FMD, however, which measures large artery reactivity, PAT represents microvascular function. 11 This makes LnRHI complementary to FMD, and not a marker to replace it. 39 As micro-vessels are the primary location of ED, 40 PAT could be a more precise technique in earlier disease stages. However, digital PAT is, like FMD, highly responsive to the sympathetic tone. In addition, in patients with impaired vasodilator response of small vessels, such as in diabetes mellitus or rheumatic disease, PAT results could be different. 11 Thus, with an operator-independent procedure, easier use, lower costs, better feasibility and tested applicability in healthy volunteers,21–24 patients with heart failure5,8,20 and CVD, 1 assessing endothelial function with digital PAT is a promising alternative to FMD. As we confirmed in patients with HFpEF, HFrEF and diabetic nephropathy, other studies have shown low day-to-day variability.21–25
In summary, PAT is a promising technique to evaluate the ED, and LnRHI is a potential surrogate parameter for risk stratification, clinical intervention and prognosis, particularly in patients with heart failure and diabetic nephropathy.
Limitations
The present study was limited by the sample size of 21–25 subjects in each group, which is relatively small compared to other studies investigating PAT or FMD reliability. We cannot make any statements about differences in reliability across the groups because sample size was calculated for a comparison against threshold of ICC – considering this sample size, a comparison of groups is underpowered. Owing to the wide range of the ICC_CI in patients with HFpEF, HFrEF and arterial hypertension, we cannot be certain about our findings regarding reliability.
Further, we only performed two measurements within an interval of 4–14 days. Therefore, we cannot draw any conclusions about PAT reliability over a longer period of time. PAT measurements were not strictly standardized regarding time of day and menstrual cycle was not recorded in premenopausal patients; however, both are known for influencing endothelial function.41,42 We did not ascertain the glucose or insulin levels of the samples, but a previous study has shown that the variability in glucose and insulin levels was associated with greater variability in endothelial function using FMD. 43 A similar effect using PAT cannot be excluded.
Conclusions
This study demonstrated for the first time good reliability of PAT in patients with diabetic nephropathy and moderate reliability in patients with HFpEF and HFrEF. However, in subjects with arterial hypertension, PAT did not show sufficient reliability, which can possibly be attributed to variations in heart rate and the respective time of the assessments.
Footnotes
Acknowledgements
We thank Anne Gale for editorial assistance.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Hans-Dirk Düngen received salary as an advisor and travel grants from BAYER Pharma AG; Wilfried Dinh and Thomas Krahn are full-time employees of BAYER Pharma AG. All other authors have no conflicts of interest to disclose.
Funding/Role of Sponsor
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This trial was conducted as an investigator initiated study and supported by an unrestricted grant by BAYER Pharma AG. The sponsor according to the ICH-GCP was the Charité – Universitätsmedizin Berlin, Germany. The sponsor did not influence trial conduct or interpretation of data.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
