Abstract
Introduction:
High-intensity interval exercise (HIIE) is more effective than moderate-intensity interval exercise (MICE) for improving macrovascular function (e.g., flow-mediated dilation), but less is known regarding the effect of HIIE on microvascular function. We used peripheral artery tonometry to measure the reactive hyperemia index (RHI) and examine the acute effects of HIIE and MICE on microvascular function.
Methods:
Ten healthy participants (50% men, age: 26 ± 5 years, mass: 75.6 ± 15.1 kg, height: 170 ± 10 cm, body mass index: 26.0 ± 3.1 kg∙m–2) performed single bouts of HIIE and MICE cycling on separate occasions. The MICE protocol was 20 min at 60% of maximum power output. The HIIE protocol was a 12-min warm up at 50% of maximum power output immediately followed by an 8-min Tabata protocol where participants alternated between cycling at ⩾ 100% max power (20 sec) and rest (10 sec). The RHI was measured before, immediately after, and 1 h after exercise and compared by two-way repeated measures analysis of variance (condition [MICE, HIIE] and time [pre-, post-, and 1-h postexercise]).
Results:
Compared to baseline, RHI increased 1 h after MICE only (p = 0.02). Heart rate was higher during MICE at 5 and 10 min (p = 0.02) and higher during HIIE at min 20 (p < 0.01).
Conclusion:
Within a sample of healthy adults, the RHI was improved 1 h after a single session of MICE but not HIIE. Future research is needed to determine the significance of the differential effects of exercise regimens on the macro- and microvasculature.
Keywords
Introduction
A growing body of literature supports high-intensity interval exercise (HIIE) as a powerful tool for improving and maintaining health and fitness.1,2 Compared to moderate-intensity continuous exercise (MICE), similar or greater improvements in cardiovascular disease (CVD) risk factors and aerobic fitness may be attained through HIIE, despite typically shorter training sessions.2,3 Vascular health in particular may benefit more from HIIE than MICE. 4
Shear stress, which is an endothelium-dependent vasodilation stimulus that has been speculated to be higher during HIIE than MICE, 5 may be a mechanism for the reported differential vascular adaptations. Shear stress is friction on the endothelium resulting from blood flow. In functional endothelium, shear stress causes activation of endothelial nitric oxide synthase (eNOS) and nitric oxide production, which precedes vasodilation. 6 In dysfunctional endothelium, a lack of vasodilatory capacity is secondary to eNOS dysfunction. 7 Chronic exercise training which produces acutely increased shear stress can upregulate eNOS. This augments nitric oxide bioavailability, therefore improving chronic vascular function. 8 Indeed, shear stress increases with exercise intensity, 9 so it is likely to be higher during HIIE than MICE.
Brachial artery flow-mediated dilation (FMD) has frequently been used to quantify endothelial function of the macrovasculature and the acute and long-term effects of exercise on vascular health. 10 Though it is clear that HIIE is beneficial for macrovascular function (e.g., FMD), 10 less is known regarding its effect on microvascular function. The reactive hyperemia index (RHI) is a measure used to evaluate microvascular function noninvasively via peripheral artery tonometry. Similar to FMD, a low RHI is predictive of future cardiovascular events. 11 However, because RHI is a relatively newer metric of CVD risk, data describing the effects of various exercise modalities on RHI are limited. Furthermore, to our knowledge, there has yet to be a comparison of the acute RHI responses to HIIE and MICE. Therefore, the aim of this study was to compare the effects of an acute bout of HIIE and MICE on microvascular function. Specifically, we used the RHI to assess microvascular reactivity before, immediately after, and 1 h after cycling exercise.
Methods
This was a randomized crossover controlled study investigating the effects of MICE and HIIE on microvascular function. Volunteers were included if they were free of chronic diseases and aged 18–49 years. Individuals were excluded for contraindications to high-intensity exercise, diagnosis of heart disease, clotting disorders, diabetes mellitus, hypertension, asthma, use of prescription medication known to alter cardiovascular responses to exercise, and tobacco use. Participants were instructed to not exercise or consume caffeine or alcohol 12 h before each laboratory visit, which were separated by at least 48 h. Participants did not consume food for 12 h before MICE and HIIE trials. Use of over-the-counter medications, such as allergy medications, were stopped 24 h before a study visit. The protocol was approved by the University at Buffalo Institutional Review Board and all participants provided informed consent after study risks and benefits were explained.
Following initial screening, height, mass, baseline heart rate (HR; Polar Electro Inc., Bethpage, NY, USA), and blood pressure (BP; measured by manual auscultation) were measured. Participants were asked to describe their average physical activity routine over the previous 3 months, and this was used to calculate their aerobic training minutes per week (see Table 1). To assess fitness status and prescribe cycling intensity for the MICE and HIIE trials, participants completed a graded exercise test on a cycle ergometer (Ergoselect; COSMED, Rome, Italy) with expired gasses analyzed by a metabolic cart (TrueOne 2400; ParvoMedics, Murray, UT, USA). Ventilatory data were averaged in 15-s intervals. Participants began cycling with a resistance of 80 W and maintained a cadence of 60–70 revolutions per min (rpm). Every 2 min, resistance was increased by 35 W or 30 W for men and women, respectively. The test continued until the participant failed to maintain 60–70 rpm. At the end of each 2-min stage, the OMNI (scale of 0–10) rate of perceived exertion (RPE) 12 was collected. Upon completion, VO2 max and maximum power output, defined as the highest power output maintained for ⩾ 30 s, were recorded. VO2 max was considered valid if at least three of the following criteria were achieved: VO2 plateau (< 150 mL*min–1), respiratory exchange ratio (RER) > 1.1, HR within 95% of age-predicted maximum, and RPE ⩾ 9. Following the graded exercise test, participants were familiarized with the vascular reactivity test, as detailed below.
Participant characteristics.
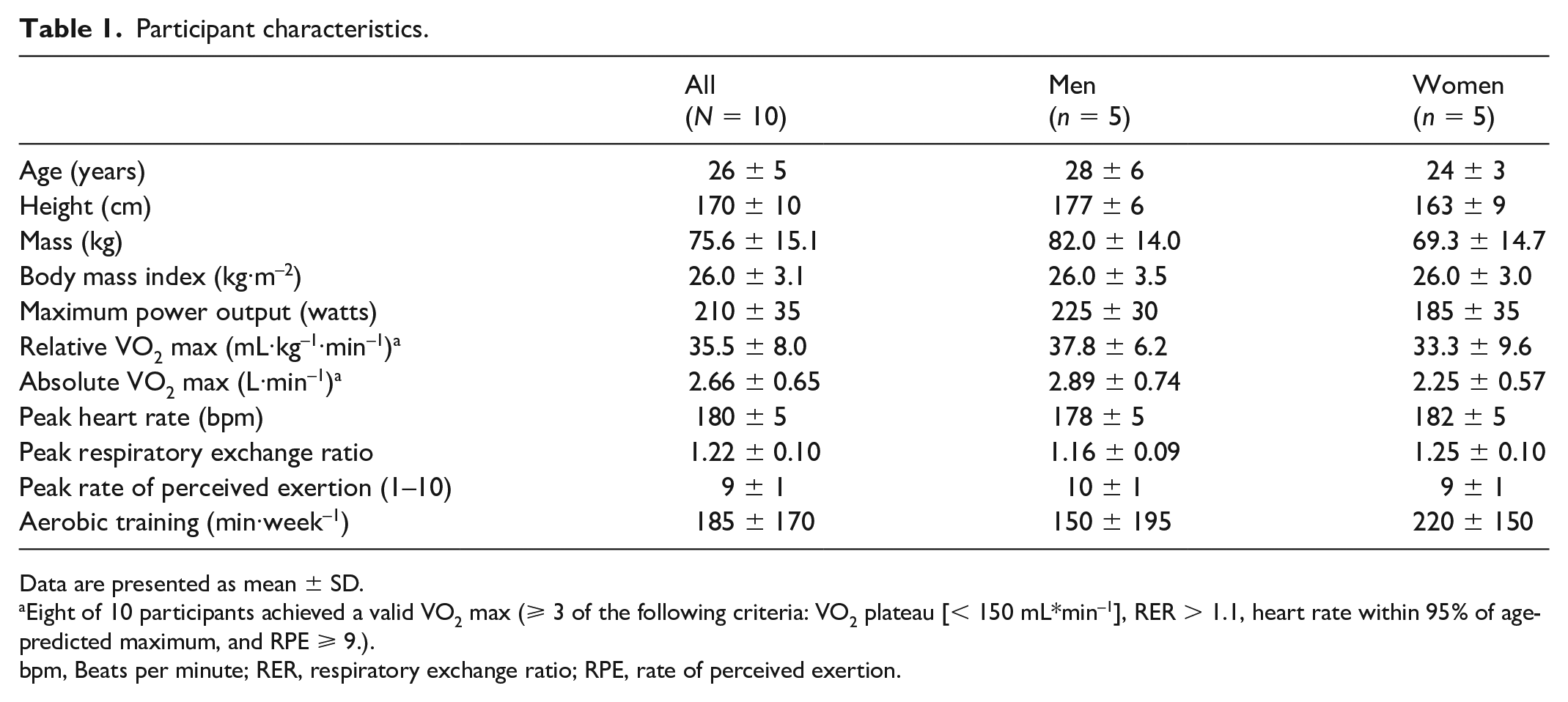
Data are presented as mean ± SD.
Eight of 10 participants achieved a valid VO2 max (⩾ 3 of the following criteria: VO2 plateau [< 150 mL*min–1], RER > 1.1, heart rate within 95% of age-predicted maximum, and RPE ⩾ 9.).
bpm, Beats per minute; RER, respiratory exchange ratio; RPE, rate of perceived exertion.
In two separate laboratory visits beginning between the hours of 0700 and 0900, participants completed the MICE and HIIE exercise protocols with microvascular function assessed before, after, and 1 h after exercise. Total exercise time was matched in both groups such that MICE exercised continuously at a single intensity (60% of participants’ maximum power output) whereas the HIIE visit provided a warm up followed by the high-intensity intervals (12-min warm up of cycling at 50% of maximum power output immediately followed by an 8-min Tabata of alternating efforts of 20 s > 100% max power and 10 s of rest).
13
Intensity for the Tabata protocol started at 100% of participants’ maximum power output and increased by 10–40 W every 2 min until a HR within 10 beats per min (bpm) of the peak HR observed during the graded exercise test was achieved. For both trials, participants were instructed to maintain a cadence of 60–70 rpm. Blood pressure, HR, and OMNI RPE
12
were collected before and immediately after exercise. The RPE and HR were also collected every 5 min during exercise. Mean arterial pressure was calculated as
Microvascular function was assessed by peripheral artery tonometry (PAT) (EndoPAT 2000; ZOLL Itamar Ltd, Atlanta, GA, USA). For each PAT test, participants were seated for 15 min and BP was measured. Beat-to-beat pulse amplitude was recorded as 30-s averages in the left and right index fingers during a 5-min baseline, 5 min of right arm blood flow occlusion, and 5-min recovery. Right arm blood flow occlusion was accomplished by rapid cuff inflation (E20 Rapid Cuff Inflator; DE Hokanson, Inc., Bellevue, WA, USA) with the cuff placed on the upper arm, inflated to the higher pressure of 200 mmHg or 60 mmHg above systolic BP. The RHI was generated by an automated algorithm within the PAT software, which calculates the ratio of mean pulse amplitude before and after the 5-min occlusion. This ratio is calculated for the right arm, then divided by the same ratio in the left arm which serves as a control for endothelium-independent systemic blood flow perturbations.
Statistical analyses
Data are reported as mean ± SD. Before analyses, all data were checked for the assumptions of normality and equal variance using the Shapiro–Wilk and Levene’s test, respectively. Separate two-way repeated measures analysis of variance (ANOVAs) (condition [MICE, HIIE] and time [pre-, post-, and 1-h postexercise]) analyzed differences in RHI, BP, and rate pressure product. Separate two-way repeated measures ANOVAs (condition [MICE, HIIE] and time [min 0, 5, 10, 15, and 20]) were used to analyze differences in HR and perceived exertion during MICE and HIIE. If the ANOVA detected a significant effect, Tukey’s multiple comparisons test was used for post hoc comparisons and Cohen’s d was calculated to measure effect size. A paired t-test assessed differences in mean HR and RPE during MICE and HIIE. All statistical analysis was performed in GraphPad Prism (version 10.0.2; Boston, MA, USA) with statistical significance set at p < 0.05.
Results
Participant characteristics, including VO2 max data from the graded exercise test, are presented in Table 1. Cycling intensity (W) demonstrated a time by condition effect (p < 0.01) such that W were higher during MICE min 0–10 (p < 0.01, d = 0.86), but higher during HIIE in min 15–20 (p < 0.01, d ⩾ 2.41), after the Tabata protocol began (Figure 1). Following these differences in intensity, there was a time by condition effect (p < 0.01) such that HR was higher during MICE at min 5 (MICE: 134 ± 25 bpm, HIIE: 125 ± 17 bpm; p = 0.02, d = 0.42) and min 10 (MICE: 142 ± 27 bpm, HIIE: 128 ± 20 bpm; p < 0.01, d = 0.59), and higher during HIIE at min 20 (MICE: 149 ± 27 bpm, HIIE: 169 ± 14 bpm; p < 0.01, d = 0.93) (Figure 2). Similarly, RPE demonstrated a time by condition effect (p < 0.01) such that RPE was higher during MICE (5 ± 1) compared to HIIE (3 ± 1) at min 10 (p = 0.02, d = 2.0) and higher during HIIE at min 15 (6 ± 1) and min 20 (8 ± 1) compared to MICE (min 15: 5 ± 1, min 20: 5 ± 2; p ⩽ 0.05, d ⩾ 1.0). However, mean RPE was not different between conditions (MICE: 5 ± 1, HIIE: 5 ± 1; p = 0.19, d = 0.0), nor was mean HR (MICE: 143 ± 26 bpm, HIIE: 144 ± 17 bpm; p = 0.73, d = 0.05).
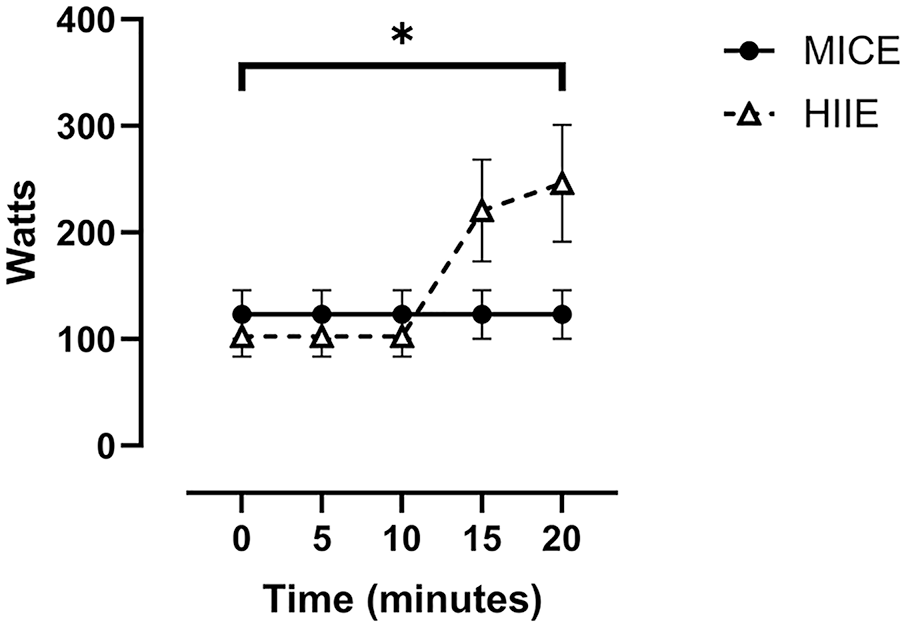
Cycling intensity during MICE and HIIE.
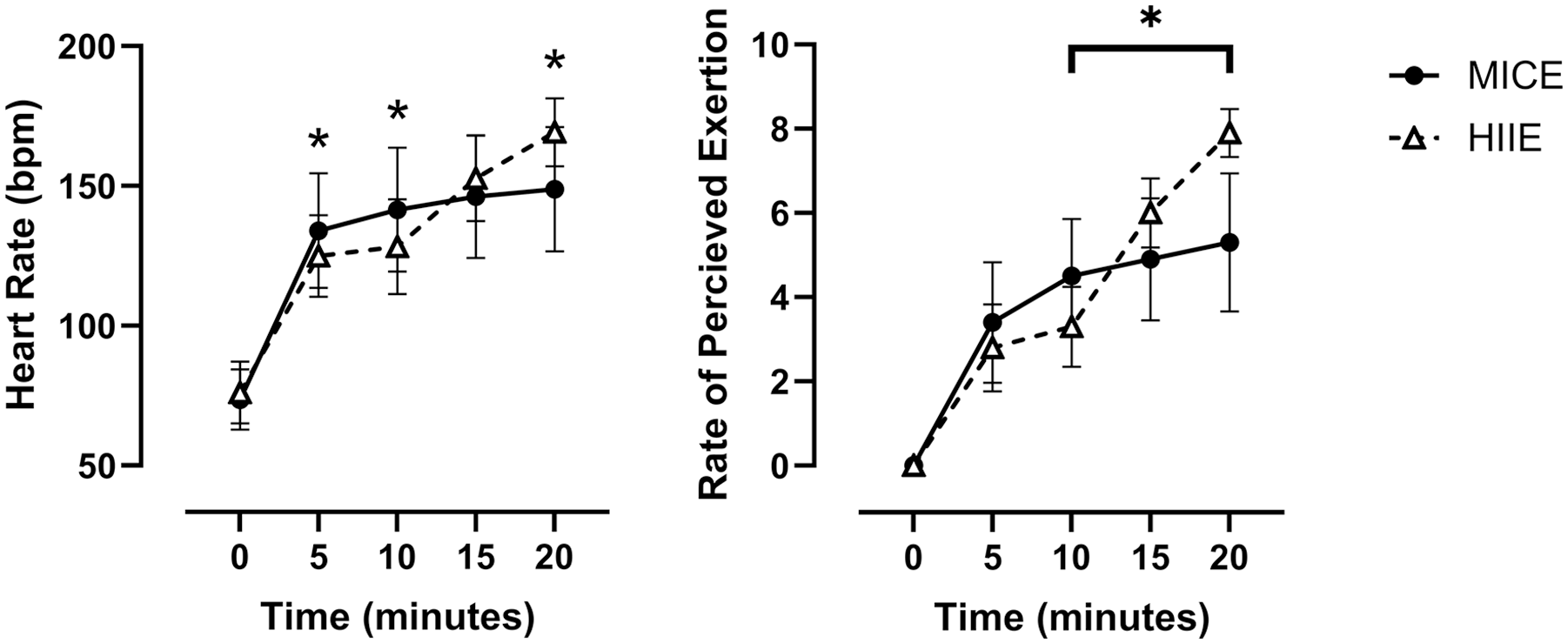
Heart rate responses and rate of perceived exertion (scale of 0–10) during MICE and HIIE.
There was a main effect of time on SBP and mean arterial pressure regardless of condition where SBP and mean arterial pressure were elevated immediately postexercise (p ⩽ 0.01, d ⩾ 1.2) (Table 2). There was also a main effect of time for DBP (p = 0.008), but post hoc analyses did not identify differences by time. Rate pressure product demonstrated main effects of both time (p < 0.01) and condition (p = 0.04) such that it was increased after exercise compared to before exercise in both conditions (p < 0.01, d = 5.1) and was greater after HIIE than after MICE (p = 0.04, d = 0.59) (Table 2). There was a main effect of time (p < 0.01) such that, compared to baseline, RHI increased 1 h after the MICE condition only (p = 0.02, d = 1.21) (Figure 3).
Blood pressure and rate pressure product responses to MICE and HIIE.
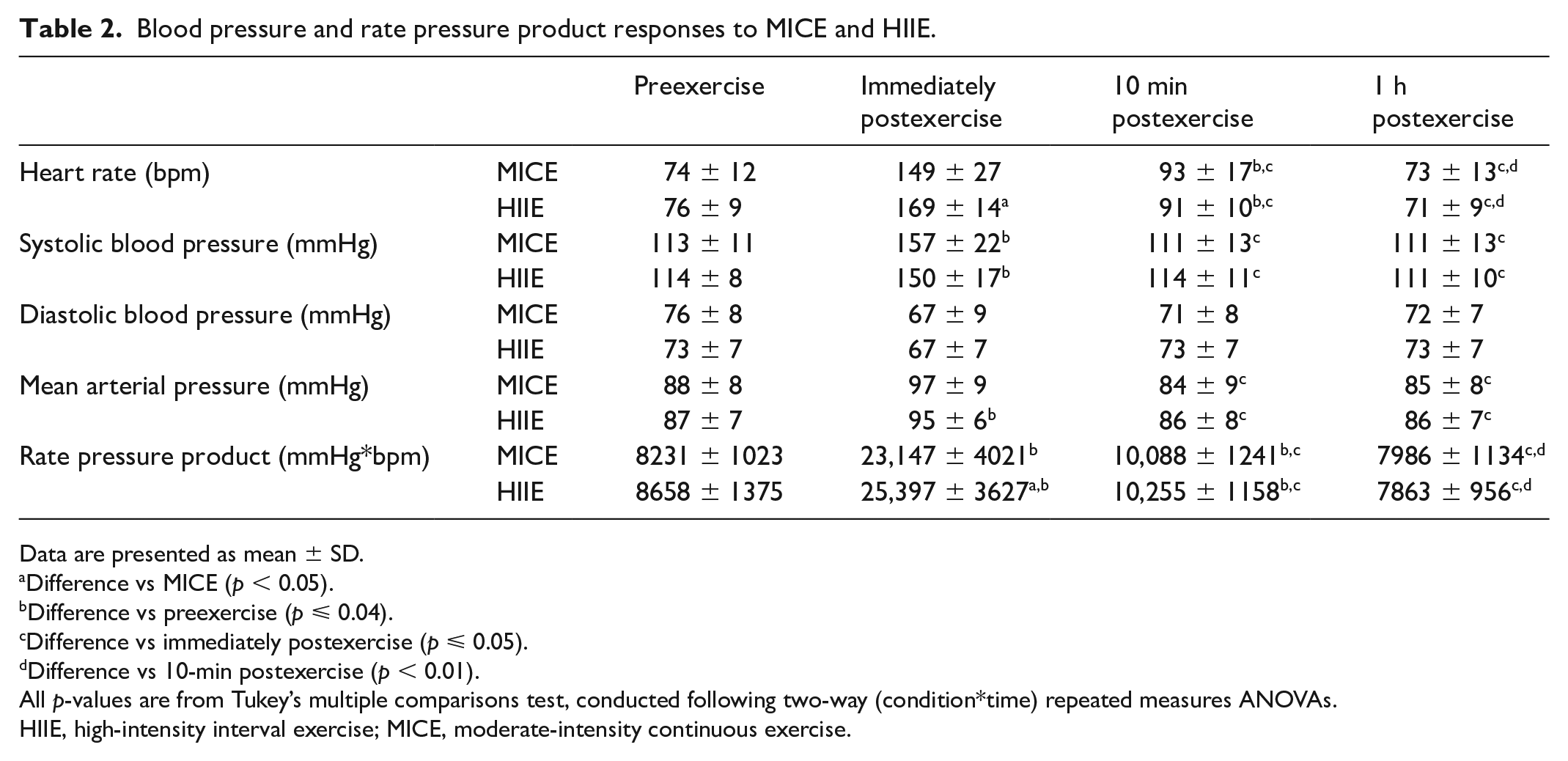
Data are presented as mean ± SD.
Difference vs MICE (p < 0.05).
Difference vs preexercise (p ⩽ 0.04).
Difference vs immediately postexercise (p ⩽ 0.05).
Difference vs 10-min postexercise (p < 0.01).
All p-values are from Tukey’s multiple comparisons test, conducted following two-way (condition*time) repeated measures ANOVAs.
HIIE, high-intensity interval exercise; MICE, moderate-intensity continuous exercise.

Microvascular function before, immediately after, and 1 h after MICE and HIIE.
Discussion
To our knowledge, our study is one of the first to compare the effects of HIIE and MICE on microvascular function. We found that despite similar mean HR and BP responses (Figure 2), RHI was increased 1 h after exercise in the MICE condition but not after HIIE (Figure 3). Before exercise, RHI was similar to baseline RHI reported in previous studies of healthy adults.14–16 However, RHI responses to exercise in the present study are contrary to some previous studies of microvasculature function following MICE and HIIE. One study in which young, healthy, male participants cycled for 30 min at 50% of their HR reserve reported increased RHI immediately after exercise, but not after a control condition of time-matched rest. 16 Another study examined macrovascular and microvascular responses to MICE (~26 min cycling at 90% gas exchange threshold) and HIIE (8 × 1-min cycling at 90% peak power, 75 s active recovery) within a sample of adolescents. 17 That study reported improved microvascular function after both exercise protocols, and greater improvements after HIIE, compared to MICE.17,18 Though it is difficult to directly compare those studies to this present study due to methodologic and differences in population, in conjunction they support the idea that different populations may have disparate vascular responses to a given exercise protocol.
Also contrary to our findings, a bout of HIIE increased RHI in a sample of obese participants. 19 Because the participants in that study had a baseline RHI indicative of endothelial dysfunction, 20 they may have been more likely to improve RHI than the healthy participants in the present study. Similarly, a study in which participants trained at anaerobic threshold three times per week for 6 months increased RHI in participants with baseline endothelial dysfunction, defined as RHI < 1.67, whereas participants with baseline RHI > 1.67 did not improve. 21 Furthermore, recent meta-analyses have reported greater improvement (d = 0.54) in FMD following HIIE (3.8%) compared to MICE (2.2%) training interventions in populations with cardiovascular and metabolic conditions, but not healthy people.4,22 Therefore, fitness and/or baseline RHI may impact the efficacy of HIIE for improving RHI. However, because those data come from exercise training studies, more research is needed to determine if baseline RHI affects RHI responses to acute exercise.
Previously, many studies have used FMD to quantify the effect of HIIE and MICE on vascular function, both acutely and after chronic training. 10 Though there is strong evidence that HIIE training can produce superior vascular adaptation, as assessed by FMD, 22 studies reporting acute responses are less conclusive. In populations with endothelial dysfunction, 30 min of HIIE and MICE performed on a cycle ergometer or treadmill have acutely improved FMD similarly.23,24 Studies investigating healthy populations have reported a variety of effects on FMD. Similar improvements in FMD have been reported following HIIE and MICE, 25 no change regardless of exercise intensity, 26 or augmented responses after HIIE, compared to MICE.27,28 One study also reported FMD was improved immediately after HIIE, but not MICE, and improved 1 h after MICE, but not HIIE. 29 In contrast to the present report, none of these studies using FMD to assess vascular function have reported more favorable responses after MICE, compared to HIIE.
These differential effects of exercise modalities on macro- and microvasculature could be due to nuanced vasodilatory mechanisms within the vascular beds versus the conduit vessels. As described in the introduction, shear stress is an endothelium-dependent vasodilation stimulus which may be greater during HIIE. 9 This may be more important for large blood vessels, such as the brachial artery, than for the much smaller arterioles assessed by the PAT. 30 Furthermore, the potential for HIIE to improve microvascular function could be limited by the resultant reactive oxygen species and retrograde shear stress.9,31,32 Reactive oxygen species have previously been shown to inactivate nitric oxide, thereby limiting the benefit of high-intensity exercise on endothelial function. 31 High-intensity continuous exercise increases oxidative stress and therefore reactive oxygen species. 18 However, HIIE with longer rest intervals than those of the present study may result in similar amounts of reactive oxygen species compared to MICE. 33 It is possible that the shorter rest intervals of the HIIE protocol in this study may have created a metabolic milieu which is intermediate to high-intensity continuous exercise and HIIE with longer rest intervals. 31 Furthermore, though increasing exercise intensity beneficially increases shear stress, it also results in increased retrograde shear stress which may act to diminish endothelial function. 32 These detrimental effects do not seem to outweigh the benefits of high-intensity exercise for macrovascular function, but they may counteract the beneficial effects in the microvasculature.
Limitations
This study has limitations which must be considered. Because we did not analyze expired gases during the MICE and HIIE protocols, we were unable to measure energy expenditure to have a valid measurement of workload. Future research should also investigate relationships between RHI and markers of metabolic and oxidative stress following HIIE and MICE, and extend RHI measurement times beyond 1 h. We also did not include a measure of lower body vascular function responses, which are of importance as these vascular beds have greater increases in blood flow and shear stress during traditional exercise. Furthermore, participants included in this study were healthy and recreationally active and the results of the present report may not apply to other populations. As previously described, fitness and health status are key determinants of endothelial function and the response to exercise.
We did not control for menstrual cycle and we did not have the statistical power to assess sex-dependent differences; however, these are both unlikely to affect RHI responses. Though the menstrual cycle may affect macrovascular endothelial function, a recent meta-analysis did not show an effect on the microvasculature. 34 Data regarding the effect of menstrual cycle on the measurement of microvascular function used in this study (reactive hyperemia index measured by peripheral artery tonometry) is scarce, but one study reported no differences across the menstrual cycle and no difference between men and women in response to orthostasis. 35
Conclusion
In conclusion, within a sample of healthy adults, microvascular function assessed using the RHI was improved 1 h after a single session of MICE but not HIIE, in contrast to results from previous studies comparing MICE and HIIE effects on macrovascular function. To determine the significance of these differences, future research should consider comparing the acute versus longer-term effects of MICE and HIIE on microvascular and macrovascular function within both healthy people and populations with cardiovascular and metabolic diseases.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
