Abstract
Elderly patients with acute pulmonary embolism (PE) have higher mortality than non-elderly patients, but receive systemic fibrinolysis less frequently. In this sub-analysis of the SEATTLE II trial, we evaluated the efficacy and safety of ultrasound-facilitated, catheter-directed, low-dose fibrinolysis in elderly patients with submassive and massive PE. We compared patients ⩾65 years old with those <65 years old. Eligible patients had proximal PE and a right ventricular-to-left ventricular (RV/LV) diameter ratio ⩾0.9 on chest computed tomography (CT). The primary efficacy outcome was the change in chest CT-measured RV/LV diameter ratio at 48 hours after procedure initiation. The primary safety outcome was major bleeding within 72 hours. Sixty-two patients were ⩾65 years of age and 88 were <65 years of age. The RV/LV diameter ratio decreased in both groups 48 hours post-procedure, with a mean change of −0.47 in those ⩾65 and −0.39 in those <65 years old, with no difference between groups (p = 0.31). Major bleeding occurred in nine (15%) of those ⩾65 and in six (7%) of those <65 years old (p = 0.17). Ultrasound-facilitated, catheter-directed, low-dose fibrinolysis resulted in a similar reduction in RV/LV diameter ratio in elderly patients with massive and submassive PE compared with non-elderly patients.
Introduction
Pulmonary embolism (PE) is the third most common cause of cardiovascular death in the United States, and its incidence is increasing.1–4 PE occurs more frequently with advancing age and results in higher mortality in the elderly population.1,5 Systemic fibrinolysis decreases mortality and prevents cardiovascular collapse, particularly in the highest risk patients.6–8 Despite these findings, its utilization remains low, particularly among elderly patients, owing to concerns of increased bleeding, specifically intracranial hemorrhage (ICH).1,9,10 The overall risk of bleeding increases incrementally by 4% with each year of age. 10 The risk of ICH appears to be significantly greater in the elderly population >75 years old, which has been considered a relative contraindication for the administration of systemic fibrinolysis in recent guidelines.6,11,12
Ultrasound-facilitated, catheter-directed, low-dose fibrinolysis may improve right ventricular function with a reduced bleeding risk. The recommended dose of tissue-plasminogen activator (t-PA) is 24 mg, compared with the United States Food and Drug Administration (FDA)-approved dosing regimen of 100 mg of t-PA for peripheral venous administration. The SEATTLE II trial examined the safety and efficacy for reversal of RV dysfunction of ultrasound-facilitated, catheter-directed, low-dose fibrinolysis in 150 patients with massive or submassive PE. 13 The trial showed decreased right ventricular (RV) size, computed tomography (CT) angiographic thrombus burden, and pulmonary artery systolic pressure 48 hours post-procedure. The rate of major bleeding (10%) was lower than historical rates for full-dose systemic fibrinolysis, and no ICH was observed.13,14 Similarly, the ULTIMA trial in submassive PE patients showed improvement in RV function with ultrasound-facilitated, catheter-directed, low-dose fibrinolysis compared to anticoagulation alone, with no major bleeding events. 15
Given the increased mortality rate of PE and the risks of complications from systemic fibrinolysis in the elderly population, we evaluated the safety and efficacy of ultrasound-facilitated, catheter-directed, low-dose fibrinolysis in this high-risk population by performing a subgroup analysis of the SEATTLE II trial.
Methods
Study design
The methodology of SEATTLE II has been previously described in detail, 13 but, in brief, 159 hospitalized patients with acute massive or submassive PE were screened and 150 were prospectively enrolled between June 2012 and February 2013 at 22 sites across the United States.
Patients were eligible if they had a massive or submassive PE with a filling defect in at least one main or lobar pulmonary artery and an right ventricular-to-left ventricular (RV/LV) diameter ratio of ⩾0.9 on contrast-enhanced chest CT. The EkoSonic Endovascular System (EKOS, Bothell, WA, USA) catheters were placed into the pulmonary arteries via the common femoral or internal jugular vein. Baseline right heart pressures were measured prior to delivery of fibrinolytic therapy or activation of ultrasound. The fixed-dose regimen of t-PA (Genentech, South San Francisco, CA, USA) was 24 mg at 1 mg/hour for both unilateral and bilateral PEs. For patients with predominantly unilateral PE, a single catheter was placed with infusion of t-PA over 24 hours. For patients with bilateral PE, two catheters were placed with infusion of t-PA over 12 hours in each catheter. After completion of the procedure, but prior to catheter removal, right heart pressures were measured. Follow-up contrast-enhanced chest CT and transthoracic echocardiography were performed within 48 ± 6 hours after initiation of the procedure. The changes in the right ventricular (RV)-to-left ventricular (LV) diameter ratio and modified Miller angiographic obstruction index score were assessed by the chest CT performed at baseline and at 48 hours. 16 Echocardiography was performed to estimate pulmonary artery systolic pressure at 48 ± 6 hours. Bleeding complications were assessed for 72 hours after the procedure. Overall, 149 of 150 patients completed the required clinical follow up.
Outcomes
The primary efficacy outcome was the core laboratory-measured change in RV/LV diameter ratio from baseline and at 48 hours post-initiation of the procedure. 17 Secondary efficacy outcomes included the change in pulmonary artery systolic pressure before the procedure, measured at the conclusion of the procedure, and estimated by transthoracic echocardiography at 48 hours. An additional secondary outcome was the change in core laboratory-measured modified Miller score for angiographic obstruction evaluated by contrast-enhanced chest CT and follow-up scan performed at 48 hours.
The primary safety outcome was major bleeding within 72 hours of initiation of the procedure. Bleeding events were classified by the Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries (GUSTO) bleeding criteria. 18 All safety outcomes were adjudicated by a designated independent Study Safety Monitor.
Statistical analysis
Sub-analysis was performed comparing patients <65 and ⩾65 years of age. Continuous variables were generally compared between the two groups using a two-sample t-test. The Fisher’s exact test was used for binary data. All reported p-values were two-sided and a p-value <0.05 was considered statistically significant. All statistical analyses were performed using SAS statistical software version 9.2 (SAS Institute Inc., Cary, NC, USA).
Results
Baseline demographics and clinical characteristics
The elderly patient cohort comprised 62 patients who were ⩾65 years of age with a mean age of 74 years compared with 88 patients <65 years of age who had a mean age of 48 years. Those ⩾65 years old had higher rates of comorbidities, including hypercholesterolemia, hypertension, diabetes, atherosclerotic disease, hepatic or renal insufficiency, and chronic obstructive pulmonary disease (Table 1). Those <65 years old had significantly greater weight and body mass index. There were no differences in rates of prior transient ischemic attack or stroke, cancer, or prior PE or deep vein thrombosis. There were similar rates of risk factors for development of PE, including immobility, family history of venous thromboembolism (VTE), or recent trauma or surgery.
Baseline demographics and clinical characteristics.
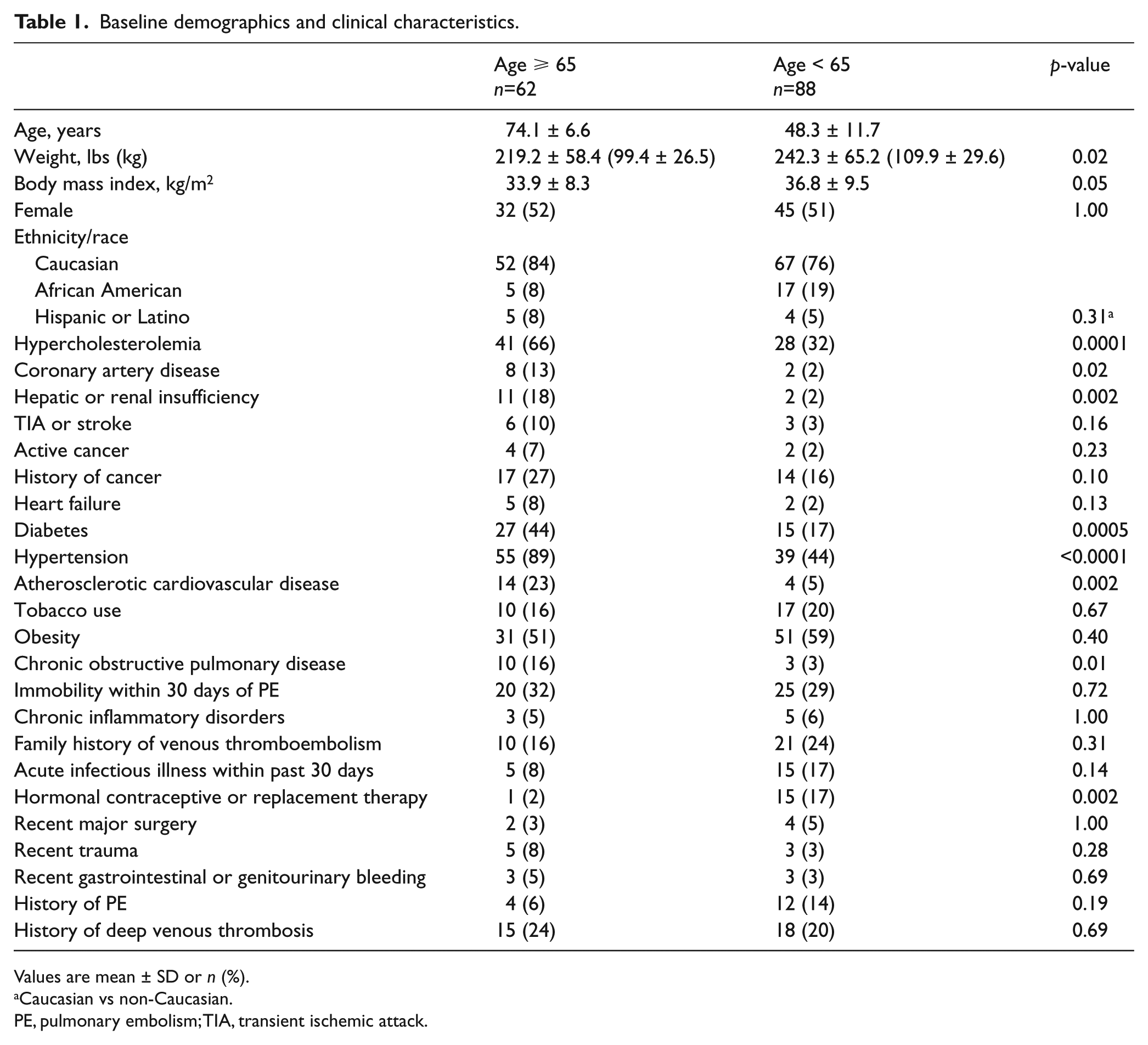
Values are mean ± SD or n (%).
Caucasian vs non-Caucasian.
PE, pulmonary embolism; TIA, transient ischemic attack.
There were no differences in PE symptoms, duration, or severity upon presentation (Table 2). There was a higher rate of antiplatelet use prior to the procedure in the elderly group. Procedural characteristics were similar with respect to the total dose of t-PA received, rate of successful placement of the device, and number of catheters placed per patient (Table 3).
Characteristics of pulmonary embolism and initial anticoagulation.
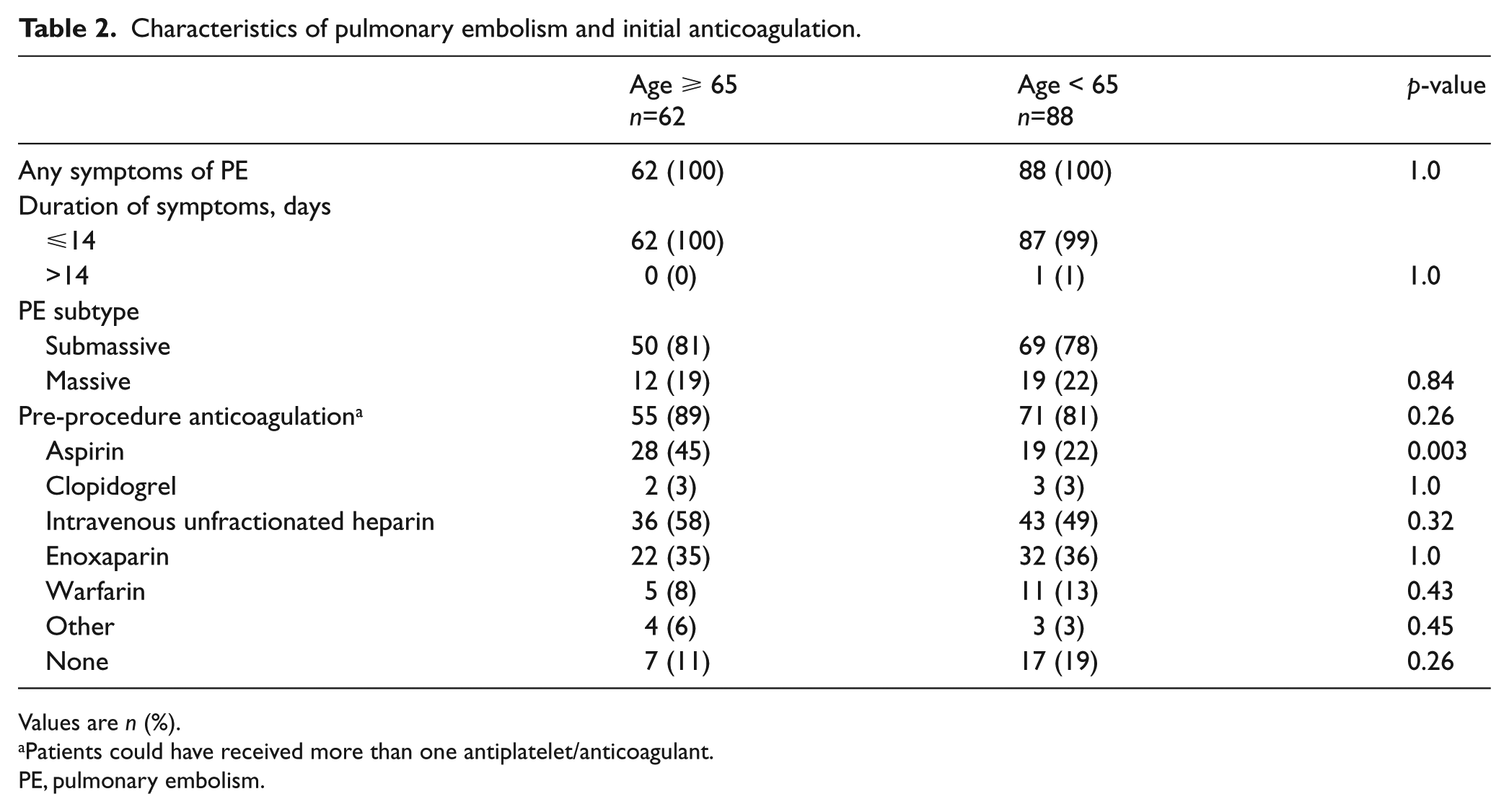
Values are n (%).
Patients could have received more than one antiplatelet/anticoagulant.
PE, pulmonary embolism.
Procedural characteristics.
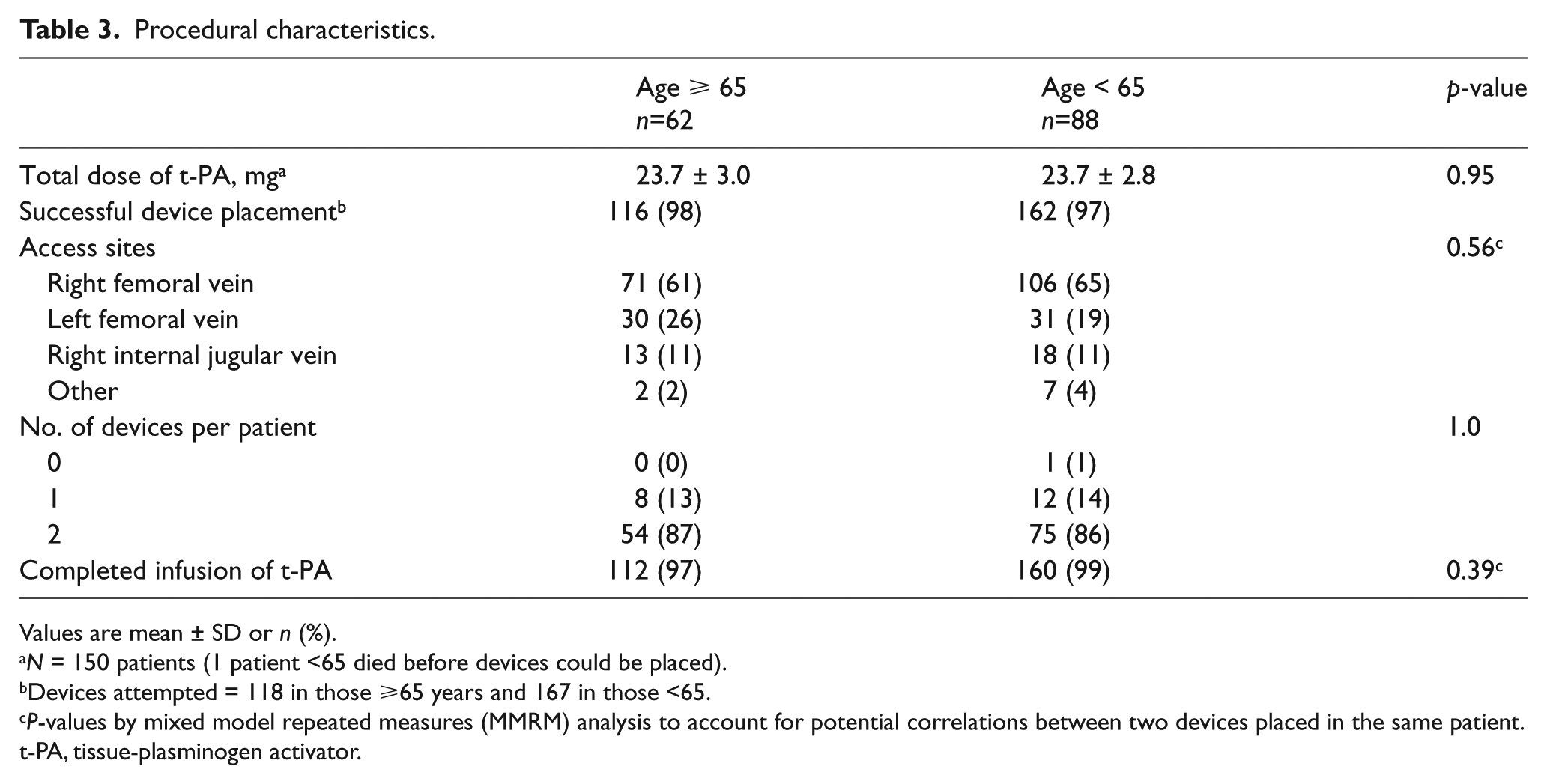
Values are mean ± SD or n (%).
N = 150 patients (1 patient <65 died before devices could be placed).
Devices attempted = 118 in those ⩾65 years and 167 in those <65.
P-values by mixed model repeated measures (MMRM) analysis to account for potential correlations between two devices placed in the same patient.
t-PA, tissue-plasminogen activator.
Efficacy outcomes
A follow-up CT scan was performed within the 48 ± 6-hour window in 42 (68%) of those ⩾65 years old and in 74 (84%) of those <65 years old (p = 0.03) (Table 4). The mean baseline RV/LV diameter ratio on CT was similar between the two groups (1.58 in those ⩾65 vs 1.53 in those <65, p = 0.48) (Figure 1A). The mean RV/LV diameter ratio at 48 hours decreased in both age groups (mean absolute change of −0.47 in those ⩾65 (p < 0.0001) and −0.39 in those <65 (p < 0.0001)). The degree of decrease in RV/LV diameter ratio was similar between the two groups (p=0.31). When also including patients with CT outside of the pre-specified window (89% in those ⩾65 and 97% in those <65, p = 0.09), the findings of similar improvement in mean change in RV/LV diameter ratio between the two groups persisted (−0.5 vs −0.41, p = 0.28). Modified Miller scores were also similar at baseline (23 in those ⩾65 vs 22.2 in those <65, p = 0.40), with a decrease at 48 hours within each group (mean change of −7.4 in those ⩾65 (p < 0.0001) and −6.2 in those <65 (p < 0.0001)), with no difference between groups (p = 0.31) (Figure 1B).
Efficacy outcomes.
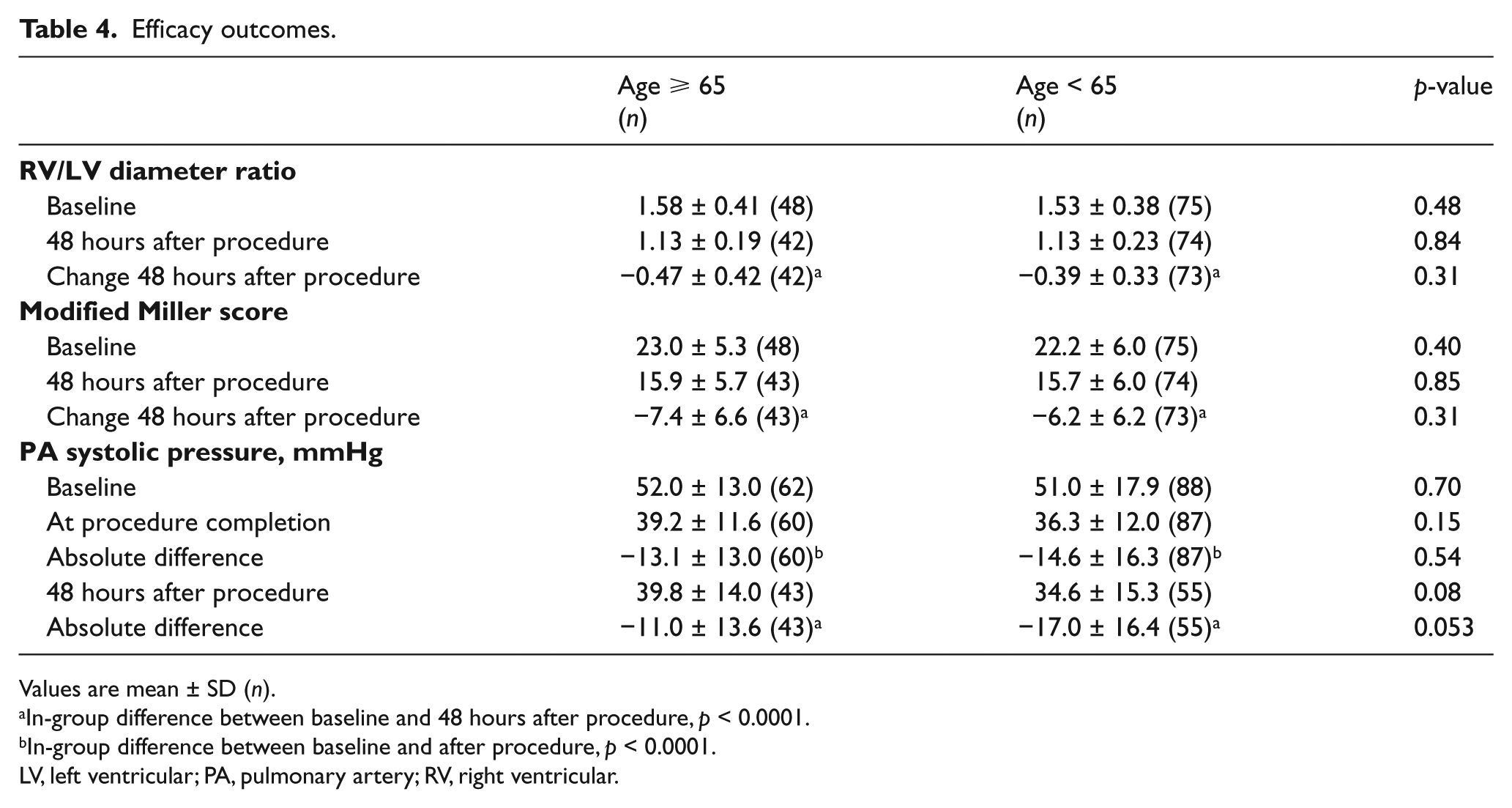
Values are mean ± SD (n).
In-group difference between baseline and 48 hours after procedure, p < 0.0001.
In-group difference between baseline and after procedure, p < 0.0001.
LV, left ventricular; PA, pulmonary artery; RV, right ventricular.
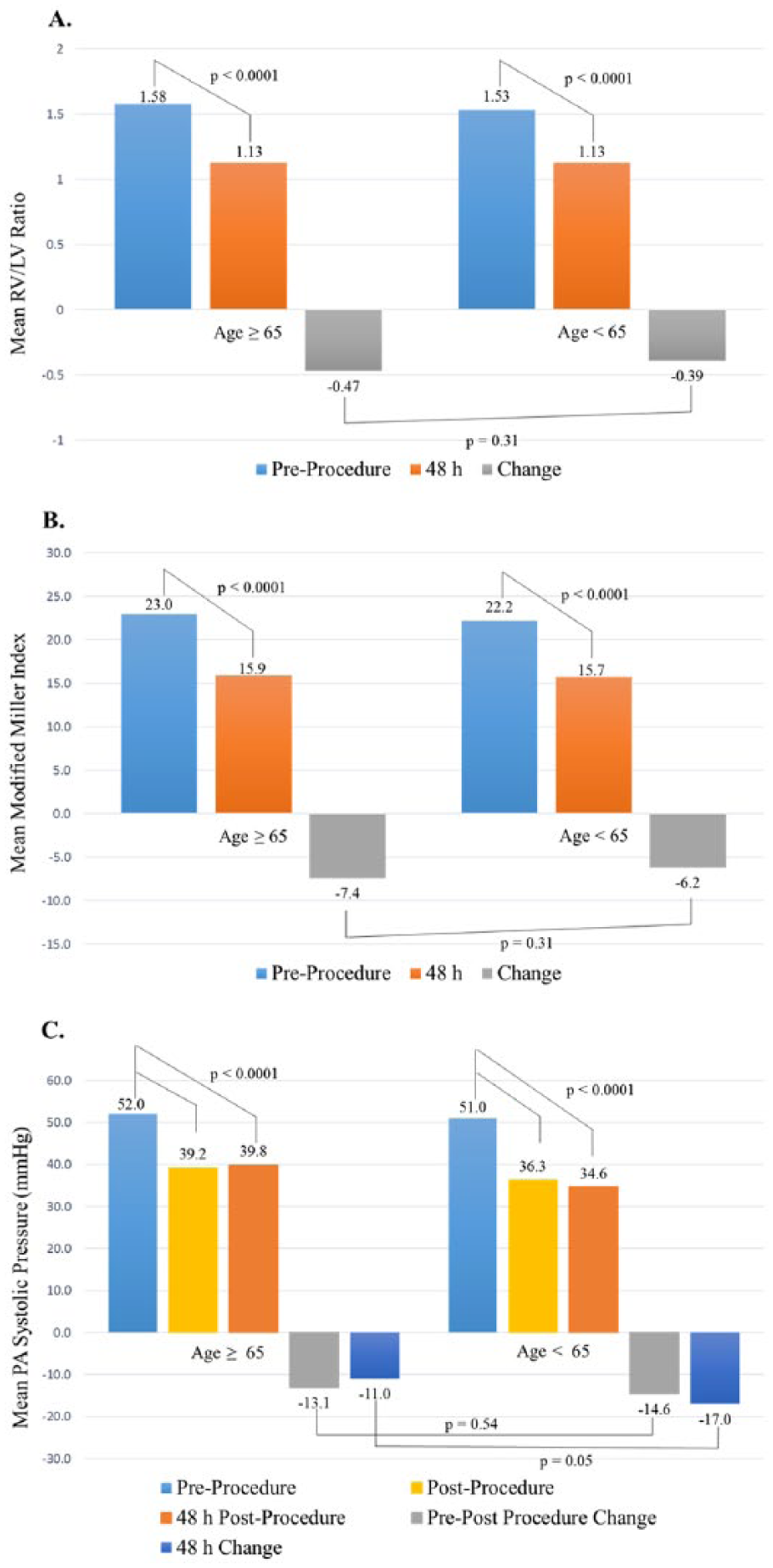
(A) Change in contrast-enhanced CT-determined mean RV/LV diameter ratio from baseline to 48 ± 6 hours after initiation of the ultrasound-facilitated, catheter-directed, low-dose fibrinolytic procedure. (B) Change in contrast-enhanced CT-determined mean modified Miller pulmonary angiographic obstruction index score from baseline to 48 ± 6 hours after initiation of ultrasound-facilitated, catheter-directed, low-dose fibrinolytic procedure. (C) Change in invasively measured mean systolic pulmonary artery pressure from baseline to completion of the ultrasound-facilitated, catheter-directed, low-dose fibrinolytic procedure and to 48 ± 6 hours after the procedure estimated by transthoracic echocardiogram.
Pulmonary artery systolic pressures were similar between the two groups at baseline (52.0 mmHg in those ⩾65 vs 51.0 mmHg in those <65 years old, p = 0.70) (Figure 1C). Both groups had decreases in pulmonary pressure at completion of the procedure compared with baseline measurements, with no difference in mean change (−13.1 mmHg in those ⩾65 vs −14.6 mmHg in those <65, p = 0.54) or absolute value (39.2 mmHg in those ⩾65 vs 36.3 mmHg in those <65, p = 0.15) between groups. Forty-three (69%) patients ⩾65 and 55 (63%) <65 had a transthoracic echocardiogram performed within the 48 ± 6-hour post-procedure window. The estimated average pulmonary artery systolic pressure from echocardiogram at 48 hours post-procedure was not significantly different between the two groups (39.8 mmHg in those ⩾65 vs 34.6 mmHg in those <65, p = 0.08). Both groups had a decrease in estimated pulmonary pressures on echocardiogram compared to baseline (p < 0.0001 in both groups), with a trend toward greater improvement in the younger group (mean change of −11.0 mmHg in those ⩾65 vs −17.0 mmHg in those <65, p = 0.053). A small percentage of patients underwent transthoracic echocardiography outside of the 48 ± 6-hour window in both groups (10% vs 13%). When including all patients who underwent follow-up echocardiography, there was a trend toward greater improvement in pulmonary artery systolic pressure in the younger cohort (−11.4 mmHg vs −17.3 mmHg, p = 0.05).
The elderly and non-elderly groups had a similar proportion of patients with a massive PE (19% in those ⩾65 and 22% in those <65 years old). There were similar improvements in the RV/LV ratio between groups in those with massive PE (−0.42 in those ⩾65 and −0.53 in those <65, p = 0.51) and submassive PE (−0.48 in those ⩾65 and −0.35 in those <65, p = 0.13). There were also similar improvements in the modified Miller Index ratio between groups in those with massive PE (−11.0 in those ⩾65 and −9.3 in those <65, p = 0.52) and submassive PE (−5.2 in those ⩾65 and −6.6 in those <65, p = 0.30), along with a decrease in PA pressure at 48 hours in massive PE (−10.7 mmHg in those ⩾65 and −13.3 mmHg in those <65, p = 0.71) and submassive PE (−11.1 mmHg in those ⩾65 and −18.1 mmHg in those <65, p = 0.05).
Safety outcomes
Length of stay was similar between groups (9.5 days in those ⩾65 vs 8.3 days in those <65 years old, p = 0.17) (Table 5). One patient <65 years old died prior to the procedure being completed. Both in-hospital mortality (1 in those ⩾65 vs 2 in those <65, p = 1.0) and 30-day mortality (2 in those ⩾65 vs 2 in those <65, p = 1.0) were similar between groups. There were no significant differences in adjudicated adverse events related to the device (2 in those ⩾65 vs 1 in those <65, p = 0.57) or t-PA (2 in those ⩾65 vs 0 in those <65, p = 0.17). Major bleeding occurred in nine (15%) of those ⩾65 and in six (7%) <65 (p = 0.17). No patient ⩾65 and one patient <65 experienced GUSTO-defined severe bleeding. No patients in either group suffered intracranial hemorrhage.
Safety outcomes.
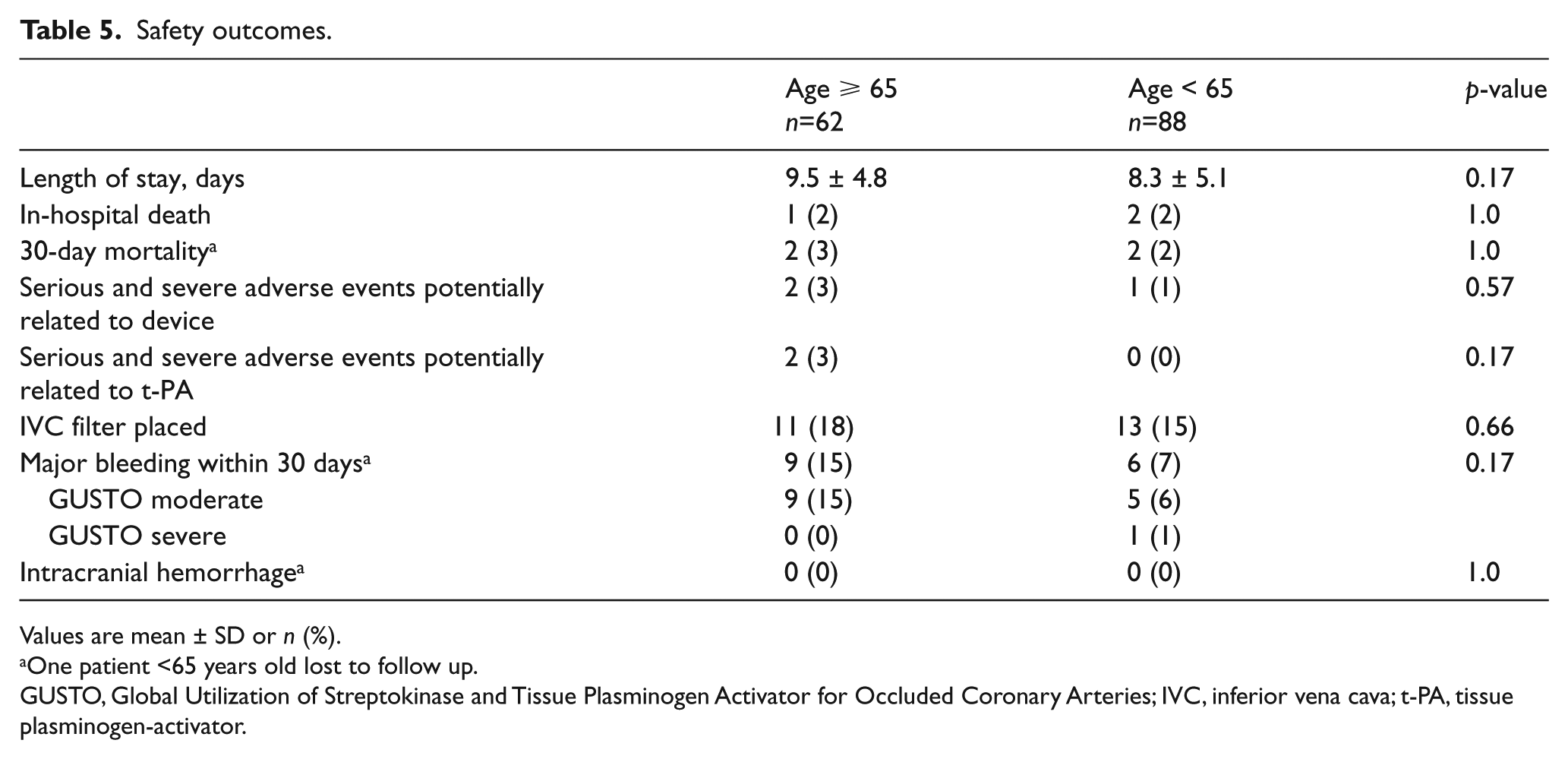
Values are mean ± SD or n (%).
One patient <65 years old lost to follow up.
GUSTO, Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries; IVC, inferior vena cava; t-PA, tissue plasminogen-activator.
Discussion
We did not observe a significant difference in primary safety or efficacy outcomes between elderly and non-elderly patients undergoing ultrasound-facilitated, catheter-directed, low-dose fibrinolysis for acute PE. We did observe similar decreases in RV/LV ratio, pulmonary artery angiographic obstruction, and pulmonary artery pressures over 48 hours in those ⩾65 years old compared with those <65 years old. The rate of major bleeding was similar in both groups but trended toward more frequent bleeding in the elderly.
Utilization of thrombolysis to treat submassive and massive PE has been limited due to bleeding concerns. 19 The risk of bleeding with fibrinolysis, particularly of intracranial hemorrhage, increases when treating elderly individuals.9,10 Factors that contribute to increased bleeding in elderly patients who receive thrombolysis include higher rates of hypertension, 20 hepatic or renal insufficiency,21,22 diabetes,22,23 antiplatelet utilization, 24 and lower weight.25,26 We observed a higher prescription of antiplatelet therapy before the procedure in elderly patients. In the randomized-controlled trial PEITHO, which compared full-dose systemic fibrinolysis plus anticoagulation versus anticoagulation alone in high-risk submassive PE patients, 2% who received systemic fibrinolysis sustained an intracranial hemorrhage. 6 All were ⩾65 years old. Increased major extracranial bleeding was also seen in the elderly subgroup analysis of PEITHO, with a rate of 11.1% in those >75 years old compared with 4.1% in those ⩽75 years old.
In PEITHO, there was a decreased rate of death or hemodynamic decompensation with systemic thrombolysis in those ⩽75 years old, which was not observed in those >75 years old. Our analysis showed similar efficacy with comparable decreases in the RV/LV diameter ratio from baseline to 48 hours across different age groups.
Ultrasound-facilitated, catheter-directed fibrinolysis, with its low dose of t-PA of 24 mg compared with 100 mg as the full systemic dose, may be an attractive option to rapidly reduce RV pressure overload in the elderly population. An ongoing clinical trial is testing lower t-PA dosing regimens and shorter durations of therapy, which may further improve safety and efficiency without compromising efficacy (see NCT02396758).
Limitations and strengths
Although the elderly patient subgroup was compared with a non-elderly group, there was no comparator group that received an alternative treatment. Therefore, we cannot comment on the efficacy or safety outcomes of this treatment approach compared to alternatives such as anticoagulation alone, systemic fibrinolysis, or catheter-directed fibrinolysis without ultrasound-assistance. No data were collected regarding follow up beyond 30 days, and clinical efficacy outcomes were limited to mortality. The rate of comorbidities was higher in the elderly group, which might confound outcomes. Fewer elderly patients had a follow-up CT scan within the 48-hour window. Finally, sample size may have limited the power of our analysis to detect differences in outcomes between elderly and non-elderly patients, including the comparison of outcomes in massive and submassive PE.
We report the largest prospective case series of elderly patients to undergo ultrasound-facilitated, catheter-directed, low-dose fibrinolysis for both submassive and massive PE. We assessed three parameters for short-term improvement in patients with PE, including decreased RV/LV ratio, pulmonary artery systolic pressures, and anatomic extent of thrombus. We also utilized a robust and inclusive assessment of bleeding.
Conclusions
Ultrasound-facilitated, catheter-directed, low-dose fibrinolysis improved RV function, reduced pulmonary artery angiographic obstruction, and decreased pulmonary artery systolic pressure in those ⩾65 years old without intracranial hemorrhage. Ultrasound-facilitated, catheter-directed, low-dose fibrinolysis appears to be a reasonable treatment option for elderly patients with massive or submassive PE.
Footnotes
Declaration of conflicting interests
Dr Piazza receives research grant support from EKOS, a BTG International Group company, Bristol-Myers Squibb, Daiichi-Sankyo, and Janssen. Dr Liu receives consulting fees from EKOS, a BTG International Group company. Dr Goldhaber receives research grant support from EKOS, a BTG International Group company, Bristol-Myers Squibb, Daiichi-Sankyo, and Janssen.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was funded by a research grant from EKOS, a BTG International group company. The sponsor had no role in data interpretation or writing the manuscript. BJC, SZG, and GP had full access to the data and had final responsibility for the decision to submit for publication. The sponsor of the trial was in possession of the database.
