Abstract
Both open surgery and endovascular peripheral interventions have been shown to effectively improve outcomes in patients with peripheral artery disease, but minimal data exist comparing outcomes performed at and below the knee. The purpose of this study was to compare outcomes following infrageniculate lower extremity open bypass (LEB) versus peripheral vascular intervention (PVI) in patients with critical limb ischemia. Using data from the 2008–2014 Vascular Quality Initiative, 1-year primary patency, major amputation, and mortality were compared among all patients undergoing LEB versus PVI at or below the knee for rest pain or tissue loss. Overall, 2566 patients were included (LEB=500, PVI=2066). One-year primary patency was significantly worse following LEB (73% vs 81%; p<0.001). One-year major amputation (14% vs 12%; p=0.18) and mortality (4% vs 6%; p=0.15) were similar regardless of revascularization approach. Multivariable analysis adjusting for baseline differences between groups confirmed inferior primary patency following LEB versus PVI (HR 0.74; 95% CI, 0.60–0.90; p=0.004), but no significant differences in 1-year major amputation (HR 1.06; 95% CI, 0.80–1.40; p=0.67) or mortality (HR 0.71; 95% CI, 0.44–1.14; p=0.16). Based on these data, we conclude that endovascular revascularization is a viable treatment approach for critical limb ischemia resulting from infrageniculate arterial occlusive disease.
Keywords
Introduction
There is a multitude of data comparing outcomes following infrainguinal lower extremity revascularization for critical limb ischemia using an open bypass versus endovascular approach.1–3 However, the majority of studies on this topic involve the treatment of proximal infrainguinal disease. For example, in the Bypass Versus Angioplasty in Severe Ischemia of the Leg (BASIL) trial, which is the only randomized controlled trial to compare outcomes after open lower extremity bypass (LEB) versus endovascular peripheral vascular interventions (PVI) with balloon angioplasty, more that 80% of procedures involved revascularization of the femoral artery. 1
Importantly, diabetes mellitus (DM) is associated with more infrageniculate arterial occlusive disease compared to that observed in non-diabetic patients with peripheral arterial disease (PAD). 4 Although successful infrainguinal revascularization has been previously shown to reduce rates of amputation with good patency using both LEB and PVI approaches in DM patients,4–7 the infrageniculate disease distribution of this population may alter the outcomes of revascularization performed at or below the knee. 4
Recently, we demonstrated favorable outcomes following infrageniculate LEB and PVI in both diabetic and non-diabetic cohorts. 8 However, there is a paucity of data comparing outcomes following LEB versus PVI performed at or below the knee. As a result, there is still considerable debate over which approach is most appropriate for the treatment of critical limb ischemia resulting from infrageniculate disease. The aim of our study was to compare 1-year outcomes following infrageniculate LEB versus PVI in patients with critical limb ischemia.
Methods
Study cohort
Data from the 2008–2014 Vascular Quality Initiative (VQI) were analyzed. The VQI is the official quality improvement registry of the Society for Vascular Surgery (SVS). It is governed by the SVS Patient Safety Organization, which performs internal audits at participating centers on a regular basis and oversees quality measure analyses.9,10 Participating centers are subject to data entry audits on a regular basis, and must comply with long-term follow-up data recording in order to be included in key outcome analyses. The VQI definition for long-term follow-up requires postoperative follow-up between 9 and 15 months.
Baseline demographics are listed in Table 1. All patients undergoing infrageniculate revascularization for critical limb ischemia were included. Critical limb ischemia was defined as the presence of rest pain or tissue loss. Infrageniculate revascularization was defined as any procedure performed at or below the popliteal artery as recorded in the VQI database. For the LEB group, this included all patients undergoing open bypass with the graft origin at or distal to the popliteal artery. For the PVI group, this included all patients undergoing revascularization at or below the popliteal artery (Supplemental Table 1). A total of 59,511 patients in the VQI database were excluded from analysis. Patients were excluded if they underwent a concomitant common femoral endarterectomy (n = 3524); LEB inflow or PVI cephalad to the popliteal artery (n = 50,246); LEB with concurrent ipsilateral PVI (n = 3147); revascularization for an indication other than rest pain or tissue loss (i.e. claudication or acute limb ischemia) (n = 10,885); or were missing primary outcome data (n = 5851) (note: numbers total more than 59,511 because some patients had multiple exclusion criteria). Patients from centers with <50% long-term follow-up were also excluded per SVS VQI reporting standards.
The VQI Patient Safety Organization and the Johns Hopkins Hospital Institutional Review Board approved this work prior to initiation. The need for patient consents was waived because the VQI data are available as part of a quality improvement initiative through the Society for Vascular Surgery. 9
Sample size calculation
Data from the BASIL trial demonstrated 3-year amputation-free survival rates of 79% in the LEB group and 66% in the PVI group. 2 Based on an 80% power, alpha of 0.05, and a presumed sample ratio for PVI versus LEB of 20:1, the minimum sample size required to detect a statistically significant difference in outcomes was calculated to be 2121 (LEB = 101, PVI = 2020).
Outcomes
The primary outcomes of the study were 1-year primary patency, major amputation, and mortality, all of which were defined according to VQI definitions. 10 Specifically, primary patency was defined as a revascularization with uninterrupted patency with either no procedure performed on it or a procedure to deal with disease progression in the adjacent native vessel, in accordance with SVS Reporting Standards. 11 Primary patency and major amputation were assessed according to center-specific data input. Primary patency was diagnosed primarily by duplex (58%) or a palpable distal pulse (14%) in the LEB group, and by duplex (41%) or a palpable distal pulse (36%) in the PVI group (Supplemental Table 2). Mortality was assessed via linkage of the VQI database with the Social Security Death Index. The 1-year time interval for the study was chosen because this is the length of time for which long-term follow-up data are available in the VQI.
Statistical analyses
Outcomes were compared for patients undergoing LEB versus PVI. Owing to a priori concerns that outcomes may differ based on disease distribution and presentation,4,12 subanalyses were then performed comparing outcomes following LEB versus PVI among patients with DM, and among patients stratified by surgical indication (i.e. rest pain or tissue loss). Descriptive statistics were summarized as median (interquartile range) or proportion (count), as appropriate. Univariable analyses including Pearson’s chi-squared or Fisher’s exact test (categorical variables), Student’s t-test (continuous variables), and Kruskal–Wallis test (non-normally distributed continuous variables) were used to compare preoperative, intraoperative and postoperative patient characteristics between the LEB versus PVI groups. Kaplan–Meier curves with log rank test analyses were used to compare outcomes between groups. Cox proportional hazards models were then developed to assess the overall odds of each outcome occurring among patients undergoing LEB compared to PVI. Covariates in the multivariable model included age, gender, race, smoking, American Society of Anesthesiologists (ASA) class, hospital transfer status, diabetes, hypertension, congestive heart failure, chronic obstructive pulmonary disease, coronary artery disease, history of coronary revascularization, and dialysis. All multivariable models were examined for potential interactions and collinearities.
All analyses were performed using Stata version 13.0 (StataCorp, College Station, TX, USA). A significance level of p<0.05 was used for all comparisons.
Results
Study cohort
Overall, 2566 patients were identified for analysis: 500 patients (19.5%) undergoing LEB and 2066 patients (81%) undergoing PVI. Indication for surgery was rest pain in 580 patients (23%) and tissue loss in 1986 patients (77%), and did not significantly differ for the LEB versus PVI groups (p = 0.28; Table 1).
Baseline demographics.
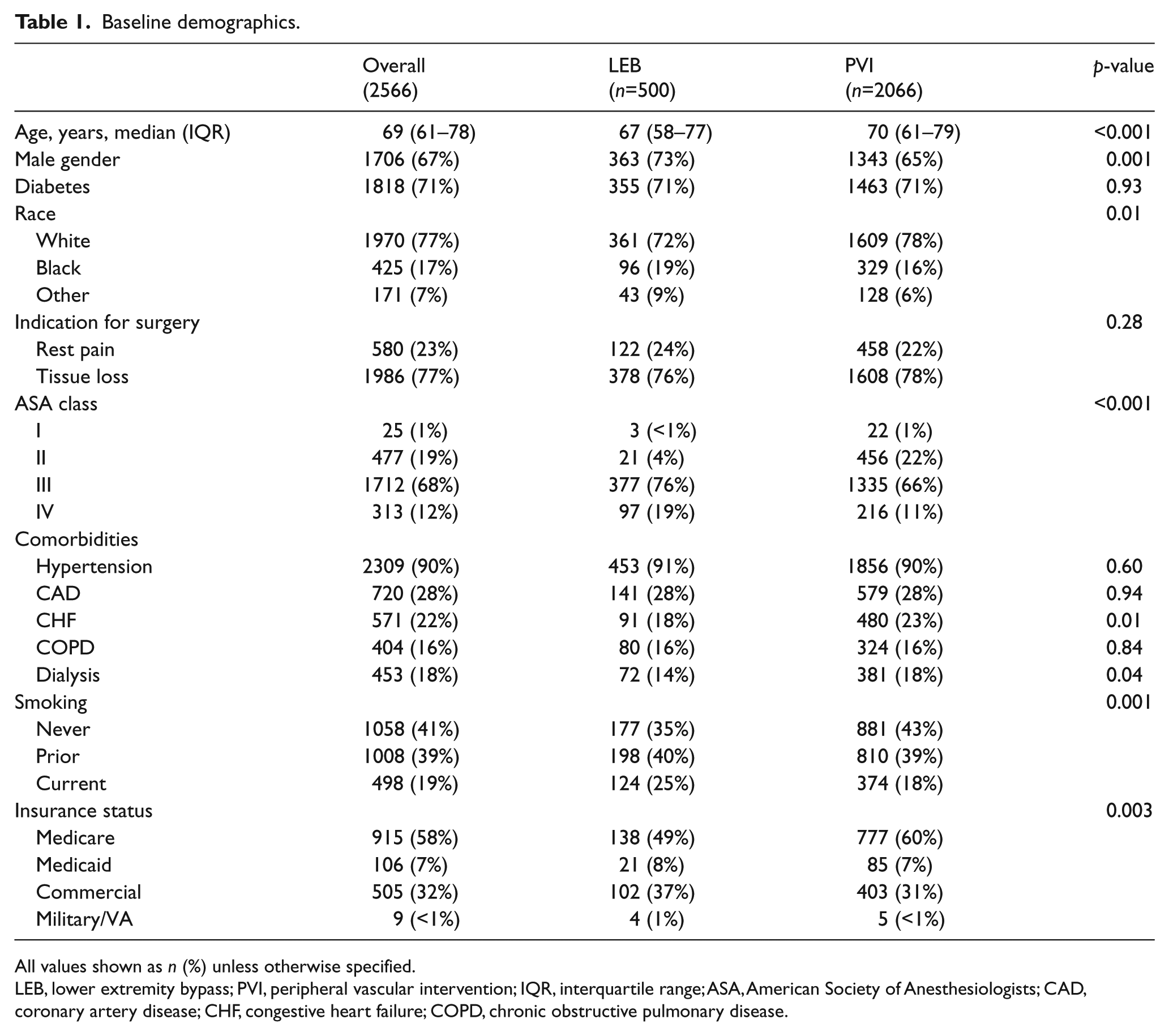
All values shown as n (%) unless otherwise specified.
LEB, lower extremity bypass; PVI, peripheral vascular intervention; IQR, interquartile range; ASA, American Society of Anesthesiologists; CAD, coronary artery disease; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease.
Patients undergoing LEB were younger (67 vs 70 years) and more frequently male (73% vs 65%) and of black race (19% vs 16%) compared to patients undergoing PVI (p ⩽ 0.01; Table 1). Patients undergoing LEB had higher ASA scores and were more frequently current smokers (25% vs 18%), but had a lower prevalence of CHF (18% vs 23%) and end stage renal disease (14% vs 18%) compared to PVI patients (p ⩽ 0.04; Table 1). The prevalence of DM was similar between groups (71% vs 71%; p = 0.93).
The majority of procedures for the LEB bypass group involved grafts to the dorsalis pedis (32%) and the posterior tibial (29%) arteries, followed by the anterior tibial (12%) and below-knee popliteal (10%) arteries. Autogenous vein was used for the bypass conduit in nearly all patients (99%, n = 495). The majority of procedures for the PVI group involved revascularization of the popliteal artery (44%), followed by the anterior tibial (30%) and posterior tibial (12%) arteries. The most common intervention was balloon angioplasty (85%), followed by stent placement (20%) and atherectomy (15%) (Supplemental Table 1). The median number of arteries treated per patient in the PVI group was 1 (IQR 1–2). A total of 30.4% of patients underwent PVI to two vessels and 13.7% underwent intervention to ⩾3 vessels. A complete summary of treated vessels is provided in Table 2.
Distribution of procedures in critical limb ischemia patients undergoing lower extremity bypass and peripheral vascular intervention.
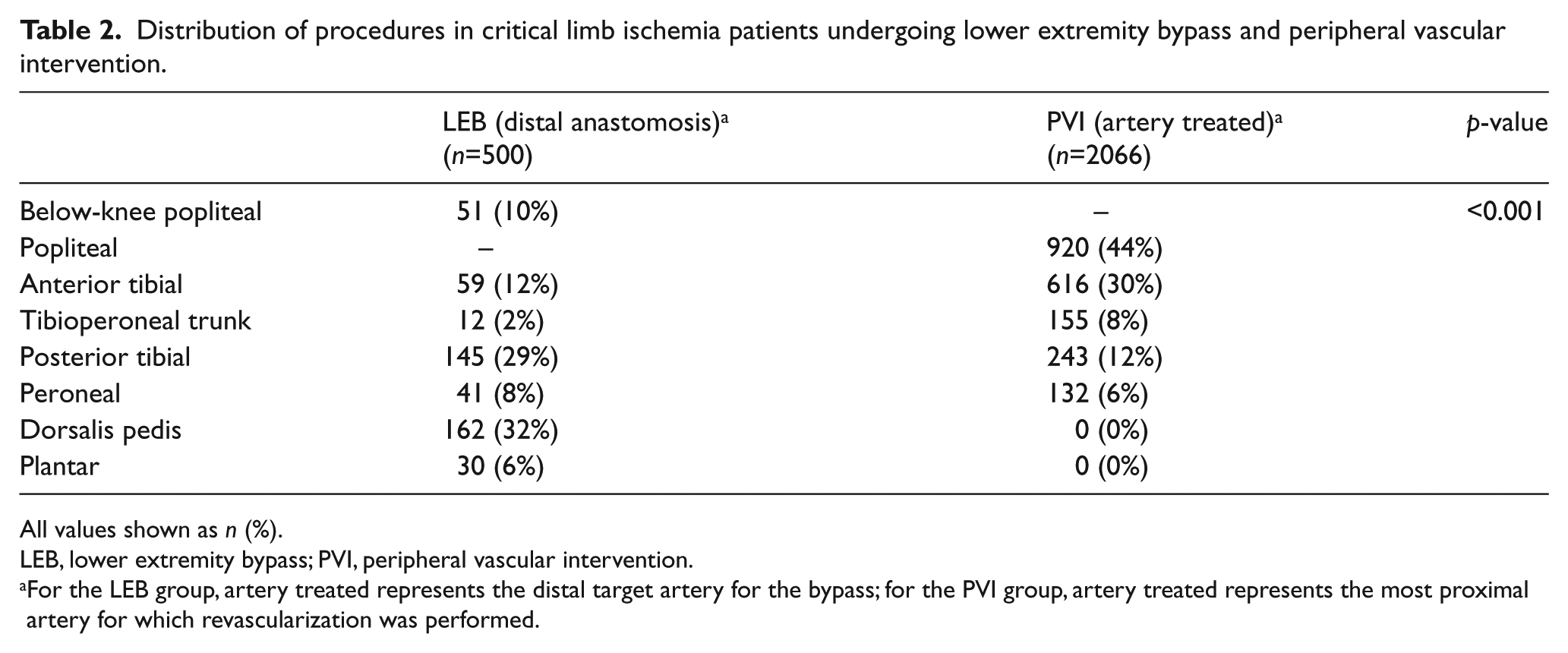
All values shown as n (%).
LEB, lower extremity bypass; PVI, peripheral vascular intervention.
For the LEB group, artery treated represents the distal target artery for the bypass; for the PVI group, artery treated represents the most proximal artery for which revascularization was performed.
Outcomes for patients undergoing LEB vs PVI
One-year primary patency was significantly worse following LEB versus PVI (73% vs 81%; p < 0.001; Figure 1). In a subgroup analysis confined to only those patients assessed by duplex ultrasound, 1-year primary patency was consistently worse in the LEB versus the PVI group (61% vs 73%; p < 0.001). Among all patients, primary-assisted patency (83% vs 90%; p < 0.001) and secondary patency (86% vs 92%; p < 0.001) were also worse for LEB. One-year major amputation (14% vs 12%; p = 0.18) and mortality (4% vs 6%; p = 0.15) outcomes were similar between groups (Table 3).
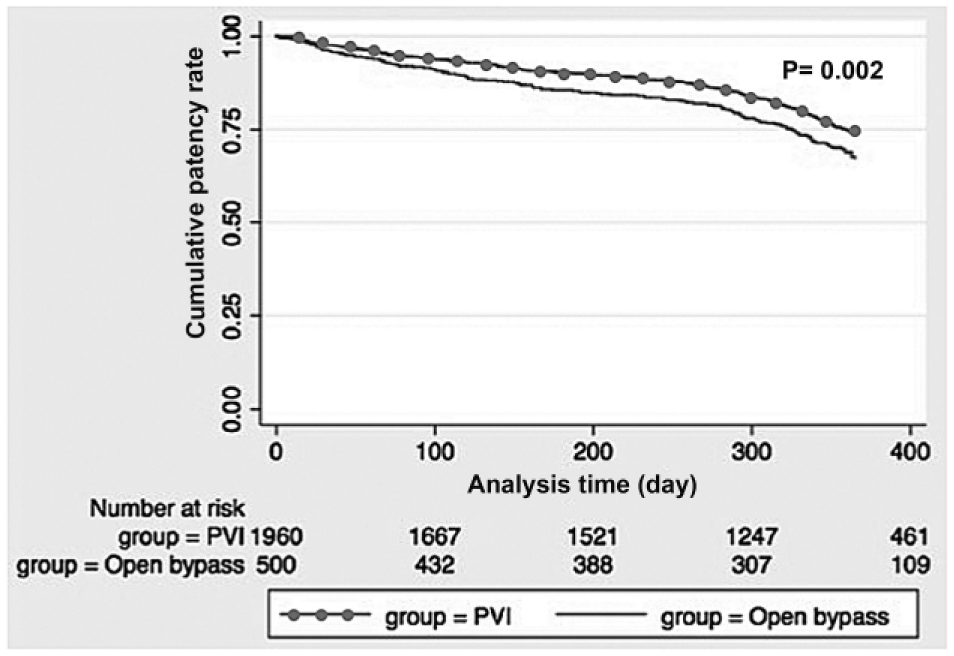
Kaplan–Meier curve showing 1-year primary patency for critical limb ischemia patients undergoing infrageniculate lower extremity bypass (LEB) versus peripheral vascular intervention (PVI). One-year primary patency was better for patients undergoing PVI vs LEB (p = 0.002, log-rank test).
One-year outcomes for critical limb ischemia patients undergoing infrageniculate lower extremity bypass versus peripheral vascular intervention.
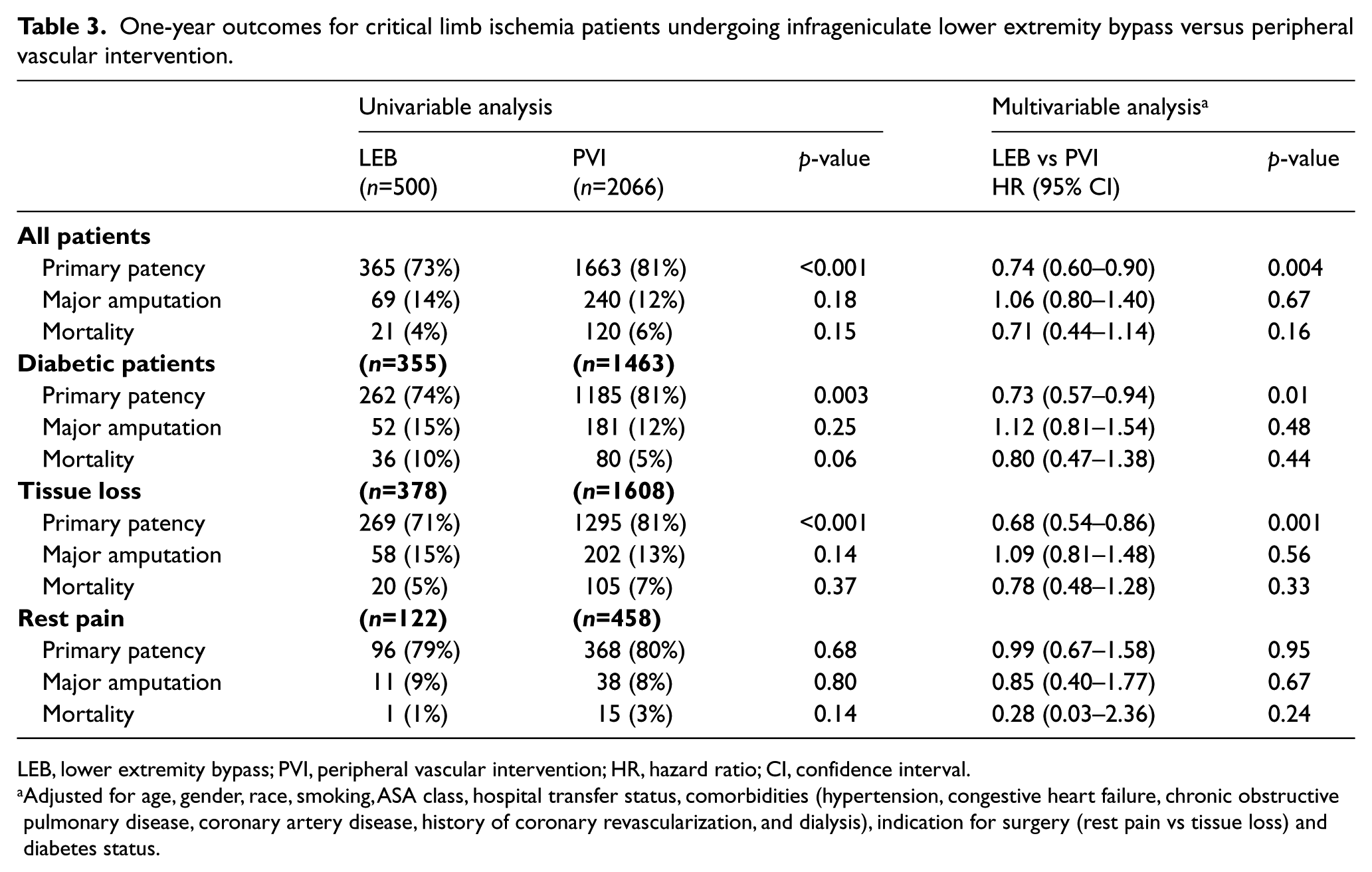
LEB, lower extremity bypass; PVI, peripheral vascular intervention; HR, hazard ratio; CI, confidence interval.
Adjusted for age, gender, race, smoking, ASA class, hospital transfer status, comorbidities (hypertension, congestive heart failure, chronic obstructive pulmonary disease, coronary artery disease, history of coronary revascularization, and dialysis), indication for surgery (rest pain vs tissue loss) and diabetes status.
On multivariable analysis adjusting for baseline differences between groups, primary patency remained significantly worse following LEB versus PVI (HR 0.74; 95% CI, 0.60–0.90; p = 0.004). One-year major amputation and mortality were similar regardless of revascularization approach (both, p ⩾ 0.16; Table 3).
Outcomes for patients with diabetes
Examining outcomes within patients with diabetes specifically, 1-year primary patency was also significantly worse following LEB versus PVI (74% vs 81%; p = 0.003). There was a trend toward higher mortality after LEB, but this was not statistically significant (10% vs 5%; p = 0.06). Major amputation (15% vs 12%; p = 0.25) was similar regardless of revascularization approach.
Multivariable analysis confirmed these findings. One-year primary patency was significantly worse for LEB versus PVI in diabetic patients (HR 0.73; 95% CI, 0.57–0.94; p = 0.01), while risk of major amputation and mortality were similar (both, p ⩾ 0.44; Table 3).
Outcomes stratified by surgical indication
On multivariable analysis, tissue loss was associated with worse outcomes than rest pain overall, with significantly higher rates of major amputation (HR 1.45; 95% CI, 1.04–2.02; p = 0.03) and mortality (HR 1.78; 95% CI, 1.04–3.06; p = 0.04), and a trend toward worse primary patency (HR 0.82; 95% CI, 0.65–1.02; p = 0.08). When stratified by treatment approach, tissue loss was associated with worse primary patency than rest pain following LEB (HR 0.54; 95% CI, 0.32–0.89; p = 0.02). There were no significant differences in risk of major amputation (HR 1.67; 95% CI, 0.81–3.43; p = 0.17) or mortality (HR 4.98; 95% CI, 0.59–41.9; p = 0.14) for patients with tissue loss versus rest pain undergoing LEB. There were also no significant differences between tissue loss versus rest pain for primary patency (HR 0.91; 95% CI, 0.70–1.17; p = 0.45), major amputation (HR 1.37; 95% CI, 0.94–2.00; p = 0.11), or mortality (HR 1.58; 95% CI, 0.90–2.77; p = 0.11) within the PVI cohort.
On subanalysis limited to only those patients presenting with tissue loss, 1-year primary patency was worse following LEB versus PVI (HR 0.68; 95% CI, 0.54–0.86; p = 0.001). There were no differences in 1-year major amputation or mortality between groups (both, p ⩾ 0.33; Table 3).
On subanalysis limited to only those patients presenting with rest pain (Table 3), there were no significant differences comparing 1-year primary patency, major amputation, or mortality for the LEB versus PVI groups (all, p ⩾ 0.24; Table 3).
Discussion
There is considerable debate regarding the efficacy of lower extremity revascularization using endovascular techniques versus open bypass. Both approaches have been used to treat infrainguinal critical limb ischemia with good limb salvage rates.1,3 However, there is a paucity of data on revascularization outcomes for procedures performed at and below the knee. In the current study, we compare infrageniculate lower extremity bypass versus PVI for patients presenting with critical limb ischemia. We demonstrate that, overall, endovascular procedures result in better 1-year primary patency than open surgery, with similar risks of major amputation and mortality. The same is true within the diabetic population, and among patients presenting with tissue loss: 1-year primary patency was better following PVI than open bypass, while major amputation and mortality were similar. Taken together, these data suggest that endovascular revascularization is a viable treatment approach for critical limb ischemia resulting from infrageniculate arterial occlusive disease.
Our finding that primary patency was better following infrageniculate PVI compared to open bypass is novel, and somewhat in discordance with current revascularization guidelines. 13 One explanation for these differences could be that the efficacy of endovascular versus open revascularization attempts may be different for proximal versus distal disease. Previous studies reporting outcomes after infrainguinal revascularization are based on patients with primarily femoral artery disease.1–3 In contrast, our study focused on infrageniculate interventions. Distal arteries are of a smaller diameter and thus known to have a higher risk of failure after attempted revascularization compared to larger, more proximal vessels.14,15 Consistent with this notion, Siracuse et al. used VQI data to report morbidity and mortality outcomes following lower extremity revascularization in 7897 patients with critical limb ischemia arising from infrainguinal PAD. 16 The authors found similar rates of death and major amputation for PVI versus open bypass at 1 year, but lower rates of major adverse limb events with PVI. However, a subanalysis of patients undergoing treatment limited to the superficial femoral artery (SFA) demonstrated no benefit with PVI, suggesting that outcomes were different for patients depending on their anatomic treatment location.
Based on the fact that diabetic patients are more likely to have an infrageniculate pattern of PAD than non-diabetic patients,4,17,18 we performed a subanalysis of open bypass versus PVI among diabetic patients. A few prior studies have reported variable outcomes following revascularization in diabetic cohorts, but nearly all are based on heterogeneous populations undergoing either endovascular or open revascularization at a range of anatomic levels for a wide variety of indications.15,19–23 These data are often used to extrapolate projected outcomes following infrageniculate PVI or open bypass interventions, but to date there is no level I evidence regarding major outcomes for either of these procedures within the diabetic population.24,25 In our study, endovascular interventions had better 1-year primary patency than open bypasses both overall and within the diabetic population, with similar rates of major amputation and death. To date, this is the first report that we know of to compare outcomes following LEB versus PVI among a group of diabetic patients undergoing infrageniculate revascularization for critical limb ischemia.
Of note, we also found that patients with critical limb ischemia and tissue loss are at higher risk for poor outcomes compared to those with rest pain. Furthermore, endovascular revascularization at and below the knee may result in better primary patency outcomes and similar rates of major amputation and mortality compared to an open surgery approach in the tissue loss group. Data using the Society for Vascular Surgery Wound, Ischemia, and foot Infection (WIfI) classification system have recently demonstrated that patients with stage 3 WIfI wounds benefit significantly from early revascularization. 26 In a recent study by Causey et al., infrainguinal surgical revascularization was associated with better limb salvage than an endovascular approach among patients with severely threatened limbs (i.e. WIfI stage 4). 27 However, there are currently no data to guide the use of an open versus endovascular revascularization approach in patients presenting PAD at and below the knee. Our findings are important because they suggest that an endovascular-first approach may be appropriate for infrageniculate revascularization of critical limb ischemia with associated tissue loss. Additional studies are needed examining the efficacy of open bypass versus PVI in this group based on WIfI staging.
Limitations
The data presented here have some inherent limitations, including differences in the anatomic levels treated with open bypass and PVI, and limited follow-up. We do not have data documenting patients’ Rutherford classification because it is not part of the VQI database. Per our study’s inclusion criteria, all patients must have had Rutherford stage 4–6 disease, but it is impossible to determine the clinical degree of PAD for any individual patient. It is also difficult to compare the overall anatomy between the open and endovascular groups in the VQI because, while PVIs are performed to a specific vessel and may involve more than one vessel, open bypasses are performed to a documented target vessel. The VQI does not provide data on Trans-Atlantic Inter-Society Consensus Document on Management of Peripheral Arterial Disease (TASC) classifications, or data regarding direct or indirect angiosome revascularization. As a result, it is impossible to determine the extent of disease being treated. Patients who underwent LEB may have had more extensive PAD compared to patients who underwent PVI, or may have previously failed endovascular attempts at revascularization. Because all patients in the PVI group in our study underwent technically successful procedures, our data are subject to a possible treatment bias and must be interpreted accordingly. However, the same limitations could also apply to the PVI group: patients undergoing endovascular revascularization were older and had more heart disease than those undergoing LEB, and may have had more diffuse and distal disease with poor outflow and pedal arch patency.
Finally, the VQI database is an administrated database and, as such, it has the potential for reporting errors or inaccuracies that are not found in randomized controlled trials. It is also recognized for having poor long-term follow-up and, as noted in the study by Siracuse et al., conclusions using these data must be interpreted accordingly. 16 Outcomes within the VQI are also only reported out to 1 year postoperatively, so long-term outcomes following infrageniculate open bypass versus PVI for critical limb ischemia are unknown. In the BASIL trial, 6-month outcomes were similar for surgery versus endovascular revascularization, initially prompting trial investigators to advocate for a balloon angioplasty-first strategy. 1 It was only on a later subgroup analysis of patients surviving more than 2 years after intervention that the benefit with open bypass was observed. 28 The Kaplan–Meier curve in our study does not show any trends to suggest potential future differences in the outcomes we report, but our conclusions are limited to mid-term outcomes only, and should not be extended beyond that until longer-term data are available. The Best Endovascular Versus Best Surgical Therapy in Patients With CLI (BEST-CLI) trial, which is a prospective, randomized, multicenter trial designed to compare best endovascular therapy with best open surgical therapy in 2100 patients with critical limb ischemia followed to a minimum of 2.25 years, should help to clarify longer-term outcomes. 29
Conclusions
Infrageniculate PAD resulting in critical limb ischemia can be treated successfully with both open surgical bypass or endovascular interventions. Endovascular revascularization may provide better mid-term primary patency rates than an open approach, especially in diabetic patients and in patients presenting with tissue loss, with similar risks of major amputation and death. Thus, endovascular techniques are a viable treatment approach for limb salvage in patients with infrageniculate critical limb ischemia, and can be considered as a first-line approach in patients presenting with diabetes and/or tissue loss.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
