Abstract
It is unclear whether thrombophilia causes resistance to anticoagulant therapy. Post hoc analyses of data from RE-COVER®, RE-COVER™ II, and RE-MEDY™ were performed to compare dabigatran etexilate with warfarin for the treatment and prevention of venous thromboembolism (VTE) in patients with thrombophilia or antiphospholipid antibody syndrome (APS). There were no significant differences in symptomatic VTE/VTE-related deaths between dabigatran etexilate and warfarin in patients with or without thrombophilia. All bleeding event categories were less frequent with dabigatran etexilate than with warfarin, regardless of whether patients had thrombophilia, no thrombophilia, or were not tested. However, these differences did not reach significance in every group. In patients with APS, there was no significant difference in VTE/VTE-related deaths between the two treatment arms. Rates of bleeding events tended to be lower with dabigatran etexilate than with warfarin, reaching statistical significance for any bleeding event. In conclusion, the efficacy and safety of dabigatran etexilate were not significantly affected by the presence of thrombophilia or APS.
ClinicalTrials.gov
Introduction
Thrombophilias describe inherited and acquired hypercoagulable states that increase the risk of venous and, in some cases, arterial thrombosis. 1 Thrombophilias may increase the likelihood of venous thromboembolism (VTE) recurrence.2–4 A major controversy and unresolved management issue is whether identified hypercoagulable states cause resistance to anticoagulant therapy, with a higher than expected rate of treatment failure. Large pivotal trials have compared direct oral anticoagulants (DOACs) – specifically dabigatran, rivaroxaban, apixaban, and edoxaban – versus warfarin and other vitamin K antagonists (VKAs) for acute and extended treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE).5–10 None of these trials required thrombophilia testing, and known thrombophilia did not exclude patients from participation. Small case series have reported DOAC treatment failures in VTE patients with antiphospholipid antibody syndrome (APS).11,12 Therefore, controversy persists regarding the efficacy and safety of DOACs compared with VKAs to manage VTE in patients with thrombophilia.
Two randomized trials of dabigatran versus warfarin for the treatment of acute VTE (RE-COVER® and RE-COVER™ II), as well as a third trial of dabigatran versus warfarin for the extended treatment of VTE (RE-MEDY™), provide a rich database. The overall results showed that in RE-COVER and RE-COVER II, dabigatran etexilate was associated with similar rates of recurrent VTE/VTE-related death, with fewer bleeding events compared with warfarin.9,10 When administered for the extended treatment of VTE in RE-MEDY, dabigatran etexilate was non-inferior to warfarin for the prevention of recurrent VTE, with a lower risk of bleeding. 13 These three phase III trials demonstrated that dabigatran etexilate is as effective as warfarin for the treatment of acute VTE (up to 6 months) and for the extended treatment of VTE (up to 6–36 months following an initial 3–12 months of anticoagulation).
We performed post hoc subgroup analyses of pooled data from the RE-COVER and RE-COVER II trials, and of data from the RE-MEDY trial, to investigate the efficacy and safety of dabigatran etexilate versus warfarin in patients with VTE with and without thrombophilia (congenital or acquired) at baseline. We also performed a post hoc subgroup analysis of pooled data from these trials to evaluate the safety and efficacy of dabigatran etexilate versus warfarin in patients with VTE who had APS at baseline.
Methods
Study population and trial design
The study designs, populations, and outcomes of the RE-COVER, RE-COVER II, and RE-MEDY trials have been published previously.9,10,13 The institutional review board at each participating clinical center approved the study, and all subjects provided informed consent. In RE-COVER and RE-COVER II, patients aged ⩾ 18 years with objectively confirmed, symptomatic, proximal DVT or PE were eligible for inclusion. They were randomized to warfarin or warfarin–placebo plus parenteral anticoagulation for ⩾ 5 days, until the International Normalized Ratio (INR) was ⩾ 2 at two consecutive measurements in the warfarin group. After discontinuing parenteral therapy, patients continued warfarin (therapeutic INR range 2.0–3.0) or received dabigatran etexilate 150 mg twice daily for 6 months (double-dummy, oral-only treatment period). In RE-MEDY, patients aged ⩾ 18 years with objectively confirmed, symptomatic, proximal DVT or PE that had been treated with an approved anticoagulant for 3–12 months, or with dabigatran in RE-COVER or RE-COVER II, were eligible for inclusion. These patients were randomly allocated to receive dabigatran etexilate 150 mg twice daily or warfarin (INR range 2.0–3.0) for 6–36 months. Testing for thrombophilia and APS was not mandatory in the RE-COVER, RE-COVER II, or RE-MEDY trials. However, when tests were performed, results were recorded. Thrombophilia was considered present if diagnosed locally prior to or at the time of the screening visit for the trials and as reported by the investigators. APS was defined as at least one positive test for lupus anticoagulant and/or for anticardiolipin antibodies combined with symptomatic, objectively verified VTE. To avoid double counting of patients, criteria were established to account for rollover patients who were treated in RE-COVER or RE-COVER II and then enrolled in RE-MEDY.
Study outcomes
Clinical outcomes were centrally adjudicated. In the pooled analysis of the RE-COVER trials, the primary efficacy outcome was recurrent, symptomatic, objectively confirmed VTE/VTE-related death from randomization to the end of the prespecified treatment period (6 months). Safety outcomes were major bleeding events (MBEs) according to the International Society on Thrombosis and Haemostasis (ISTH) criteria, the composite of MBEs and clinically relevant bleeding events (CRBEs), and any bleeds, counted during the double-dummy, oral-only treatment period. In RE-MEDY, the primary efficacy outcome was recurrent, symptomatic, objectively confirmed VTE/VTE-related death from randomization to the end of the planned treatment period (6–36 months). Safety outcomes were MBEs according to the ISTH criteria, the composite of MBEs and CRBEs, and any bleeds, from randomization to the end of the planned treatment period.
Statistical analyses
The hazard ratios (HRs) and 95% confidence intervals (CIs) for within-subgroup treatment comparisons were based on the Cox regression analysis model with factor treatment, stratified by study (for pooled analyses). Interaction p-values for treatment/subgroup interaction were from the Cox model, stratified by study (for pooled analyses), with treatment and subgroup as the main factors. Statistical analyses were performed with SAS® version 9.2 (Cary, NC, USA).
Results
Patients
The RE-COVER and RE-COVER II dataset for the overall pooled efficacy analysis consisted of 2553 and 2554 patients randomized to dabigatran versus warfarin, respectively, as long as they received at least one dose of the study drug. 9 The safety analysis for bleeding events included 2456 and 2462 patients randomized to dabigatran versus warfarin, respectively, during the double-dummy, oral-only treatment period. Among randomized patients, 34% were tested for thrombophilia. Overall, 209/2553 (8.2%) patients randomized to dabigatran etexilate and 199/2554 (7.8%) to warfarin had thrombophilia identified at baseline, representing about one-quarter of the tested patients. Factor V Leiden was the most common thrombophilia (Table 1).
Thrombophilia status and type in patients receiving dabigatran etexilate or warfarin in the RE-COVER/RE-COVER II and RE-MEDY trials.
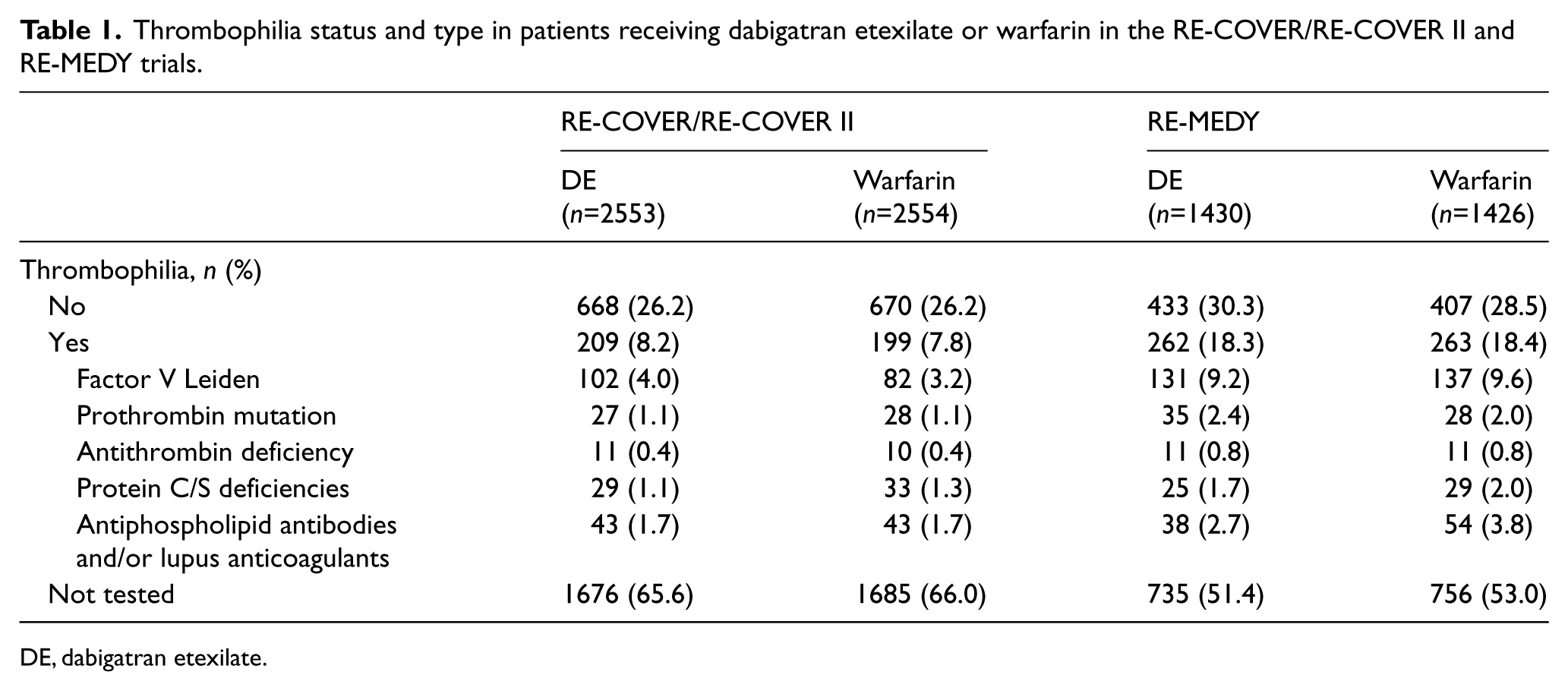
DE, dabigatran etexilate.
Demographics and baseline characteristics (Table 2) indicated differences between patients with thrombophilia and those without. Patients with thrombophilia were younger, more often male, more likely to have the combination of DVT and PE as their qualifying index event, and more likely to have suffered prior VTE.
Demographics and baseline characteristics for patients with and without thrombophilia receiving dabigatran etexilate or warfarin in the RE-COVER/RE-COVER II and RE-MEDY trials.
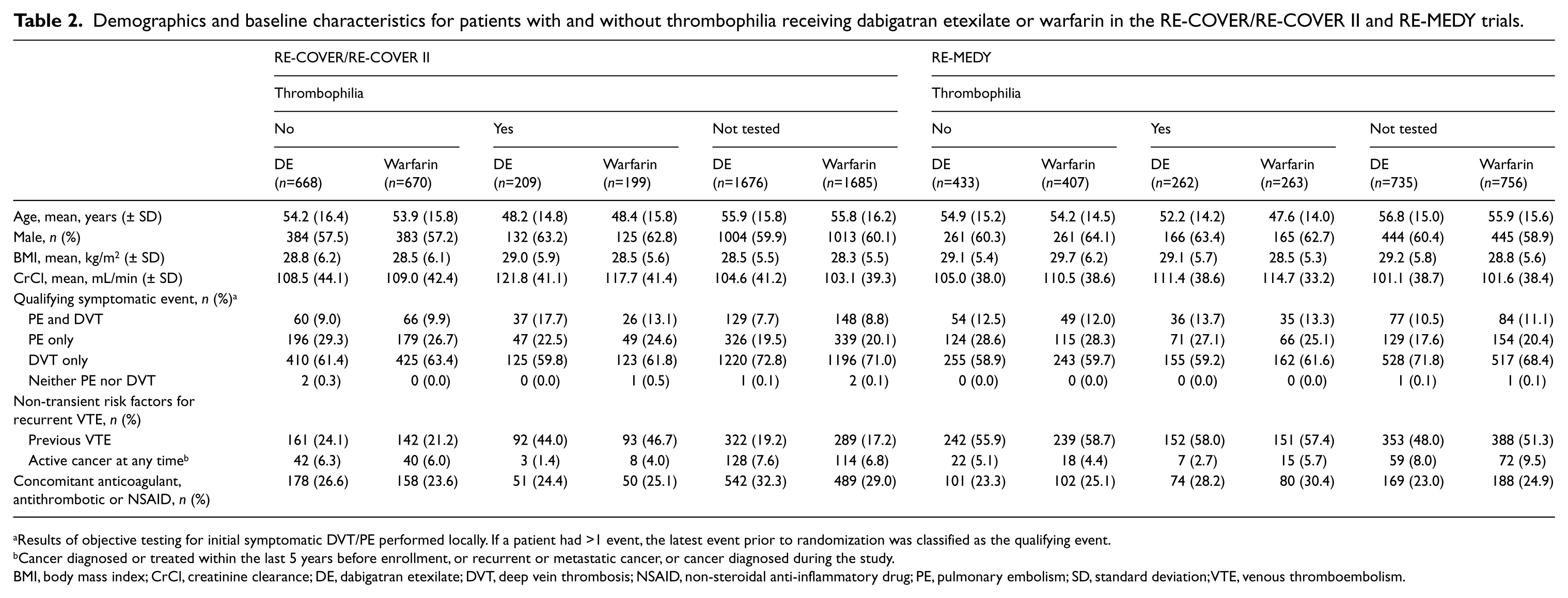
Results of objective testing for initial symptomatic DVT/PE performed locally. If a patient had >1 event, the latest event prior to randomization was classified as the qualifying event.
Cancer diagnosed or treated within the last 5 years before enrollment, or recurrent or metastatic cancer, or cancer diagnosed during the study.
BMI, body mass index; CrCl, creatinine clearance; DE, dabigatran etexilate; DVT, deep vein thrombosis; NSAID, non-steroidal anti-inflammatory drug; PE, pulmonary embolism; SD, standard deviation; VTE, venous thromboembolism.
The RE-MEDY efficacy dataset consisted of 1430 and 1426 patients who were randomized to dabigatran versus warfarin, respectively. 13 Of these, 48% of patients were tested for thrombophilia. Overall, 262/1430 (18.3%) patients randomized to dabigatran etexilate and 263/1426 (18.4%) randomized to warfarin had thrombophilia identified at baseline (Table 1). This represents 38% and 39% of the tested patients, respectively. After elimination of double counting, 711 patients (10.4%) in the three trials were found to be positive for thrombophilia.
Similar to RE-COVER and RE-COVER II, factor V Leiden thrombophilia was the most common type of thrombophilia in RE-MEDY (Table 1). Demographics and baseline characteristics (Table 2) showed several differences between patients with thrombophilia and those without. Patients who had not been tested for thrombophilia were more likely to have had DVT alone as their index event and less likely to have had prior VTE, but they had a higher incidence of cancer before/during the study.
The APS analysis was based on pooled data from 6822 treated patients from the RE-COVER, RE-COVER II, and RE-MEDY trials. Of the 151 patients diagnosed with APS at baseline, 71 were treated with dabigatran etexilate and 80 with warfarin (Table 3). Patients with APS were slightly younger than those who had no APS or had not been tested.
Demographic and baseline characteristics for patients receiving dabigatran etexilate or warfarin with and without APS (pooled RE-COVER, RE-COVER II, and RE-MEDY data).
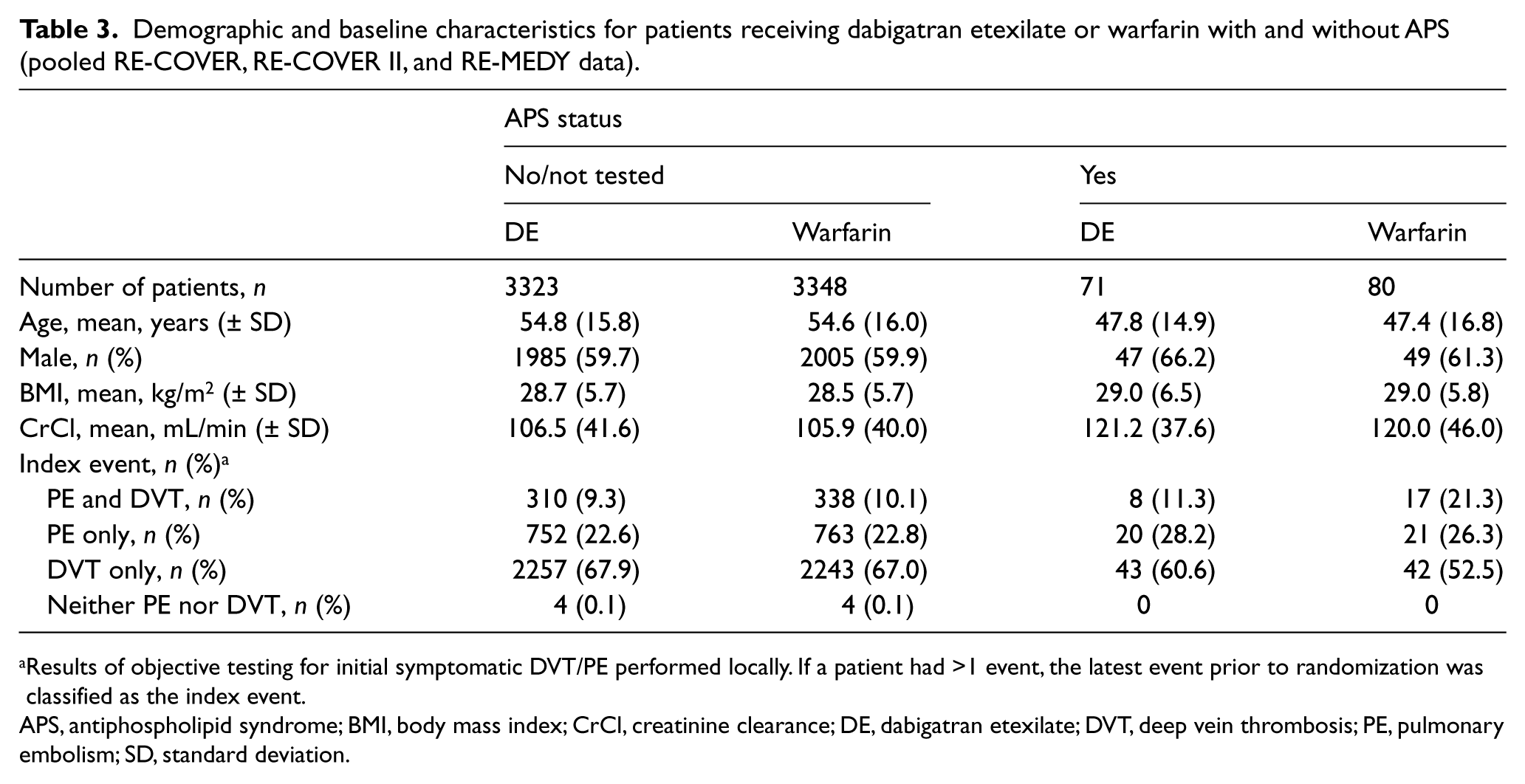
Results of objective testing for initial symptomatic DVT/PE performed locally. If a patient had >1 event, the latest event prior to randomization was classified as the index event.
APS, antiphospholipid syndrome; BMI, body mass index; CrCl, creatinine clearance; DE, dabigatran etexilate; DVT, deep vein thrombosis; PE, pulmonary embolism; SD, standard deviation.
Efficacy outcomes
RE-COVER and RE-COVER II pooled analysis
In the overall population of RE-COVER and RE-COVER II, the incidences of both VTE/VTE-related deaths and of PE were similar for dabigatran etexilate versus warfarin (2.4 vs 2.2%; HR 1.09 [95% CI 0.76, 1.57] and 0.9 vs 0.9%; HR 0.95 [95% CI 0.53, 1.71], respectively) (Table 4).
Efficacy and safety outcomes for patients with and without thrombophilia in the RE-COVER/RE-COVER II and RE-MEDY trials.
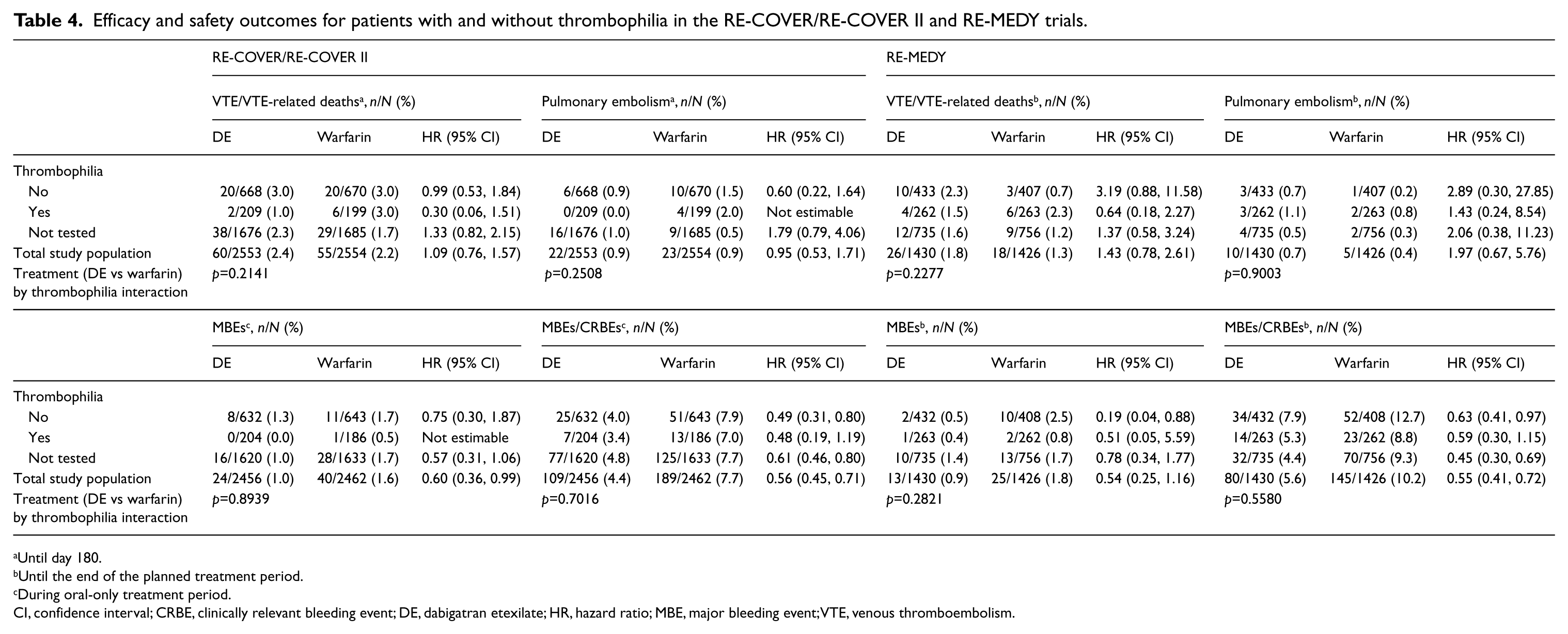
Until day 180.
Until the end of the planned treatment period.
During oral-only treatment period.
CI, confidence interval; CRBE, clinically relevant bleeding event; DE, dabigatran etexilate; HR, hazard ratio; MBE, major bleeding event; VTE, venous thromboembolism.
In the analysis by thrombophilia at baseline, the incidences of VTE/VTE-related deaths and of PE until day 180 were not significantly different for dabigatran etexilate versus warfarin in patients with thrombophilia (1.0 vs 3.0%; HR 0.30 [95% CI 0.06, 1.51] and 0.0 vs 2.0%, respectively) (Table 4; Figure 1). In patients without thrombophilia, the frequencies of both efficacy outcomes (VTE/VTE-related deaths and PE) were similar for dabigatran etexilate and warfarin (Table 4). Cox regression analysis for interaction showed that treatment efficacy (dabigatran etexilate vs warfarin) was not significantly affected by the presence or absence of thrombophilia (Table 4).
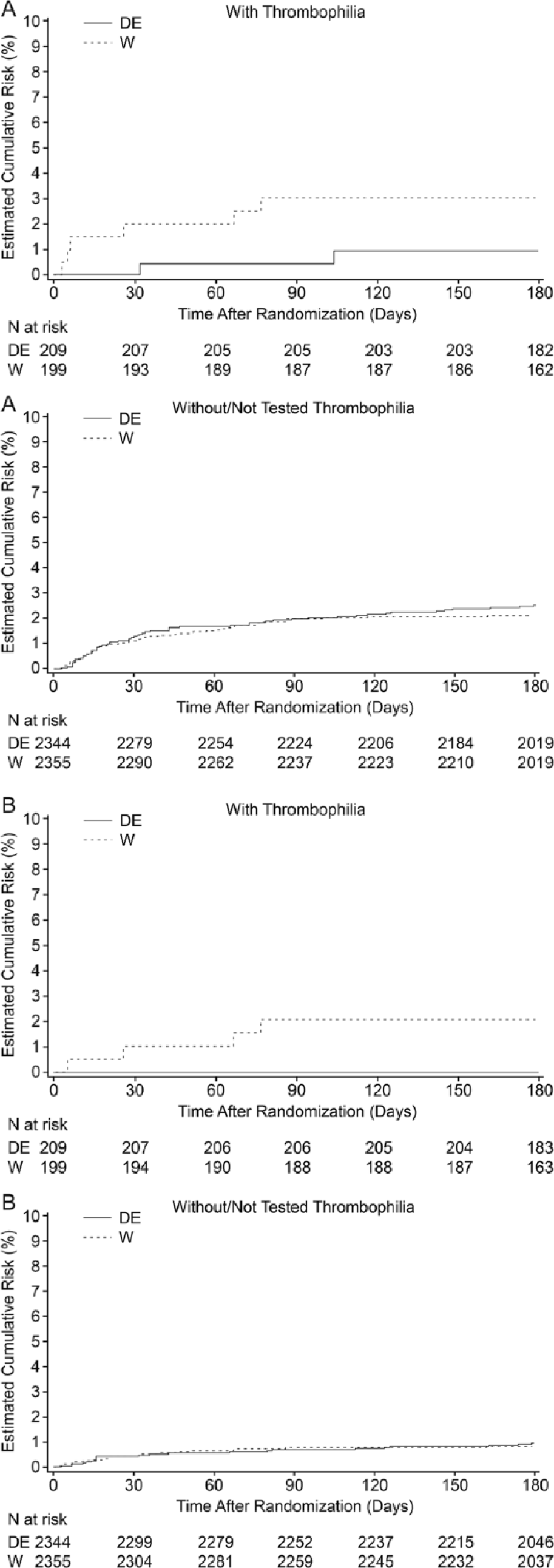
Kaplan–Meier cumulative event rates for (A) VTE and VTE-related death, and (B) PE, with dabigatran etexilate (DE) and warfarin (W) in patients with and without/not tested thrombophilia in the RE-COVER and RE-COVER II trials. (VTE, venous thromboembolism; PE, pulmonary embolism.)
RE-MEDY
In the overall efficacy analysis for the RE-MEDY trial, dabigatran etexilate was non-inferior to warfarin for the primary efficacy outcome of VTE/VTE-related death (1.8 vs 1.3%; HR 1.43 [95% CI 0.78, 2.61], respectively). The risk of PE was low and not significantly different between dabigatran etexilate and warfarin (0.7 vs 0.4%; HR 1.97 [95% CI 0.67, 5.76], respectively) (Table 4).
In patients with thrombophilia, there were no clear differences in efficacy between dabigatran etexilate and warfarin (Table 4; Figure 2). In patients without thrombophilia, VTE/VTE-related deaths trended toward more frequent with dabigatran etexilate versus warfarin, but the differences were not significant (2.3 vs 0.7%; HR 3.19 [95% CI 0.88, 11.58], respectively). The incidence of PE was also not significantly different between the dabigatran etexilate and warfarin treatment arms (0.7 vs 0.2%; HR 2.89 [95% CI 0.30, 27.85], respectively) (Table 4). The p-values for treatment by thrombophilia interaction suggested that treatment efficacy (dabigatran etexilate vs warfarin) was not significantly affected by the presence of thrombophilia for either VTE/VTE-related death (p=0.23) or for PE (p=0.9) (Table 4).
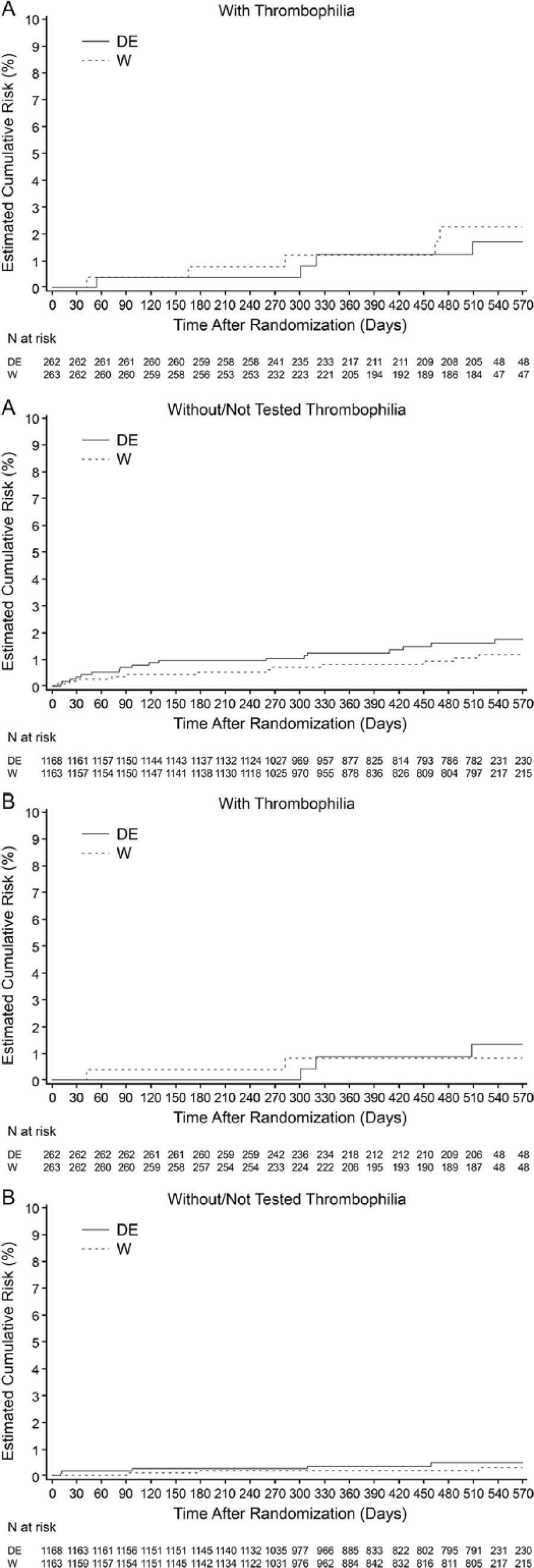
Kaplan–Meier cumulative event rates for (A) VTE and VTE-related death and (B) PE, with dabigatran etexilate (DE) and warfarin (W) in patients with and without/not tested thrombophilia in the RE-MEDY trial. (VTE, venous thromboembolism; PE, pulmonary embolism.)
Antiphospholipid syndrome analysis
The incidence of VTE/VTE-related deaths in patients with APS from the pooled population of RE-COVER, RE-COVER II, and RE-MEDY was not significantly different between dabigatran etexilate and warfarin (4.2 vs 5.0%; HR 0.43 [95% CI 0.08, 2.38]) (Table 5).
Efficacy and safety outcomes for patients with APS at baseline by anticoagulant treatment; pooled analysis of data from the RE-COVER/RE-COVER II and RE-MEDY trials.
Bleeding events were counted from the start of the double-dummy, oral-only treatment period.
For rollover patients from RE-COVER/RE-COVER II to RE-MEDY without treatment switch, the combined time period is considered for VTE/VTE-related deaths and bleeding.
Non-rollover patients in RE-COVER/RE-COVER II and rollover patients from RE-COVER/RE-COVER II to RE-MEDY with treatment switch are censored at day 180 in RE-COVER/RE-COVER II for VTE/VTE-related deaths or at the end of the post-treatment period in RE-COVER/RE-COVER II for bleeding events.
For non-rollover patients in RE-MEDY, the RE-MEDY planned treatment period is considered for VTE/VTE-related deaths and bleeding.
APS, antiphospholipid syndrome; CI, confidence interval; CRBEs, clinically relevant bleeding events; DE, dabigatran etexilate; HR, hazard ratio; MBE, major bleeding events; VTE, venous thromboembolism.
Safety outcomes
RE-COVER and RE-COVER II pooled analysis
In the total study population, the risks of MBEs, MBEs/CRBEs (Table 4), and any bleeds were significantly lower with dabigatran etexilate than with warfarin. The point estimates of the incidences of all bleeding event categories were lower with dabigatran than with warfarin, regardless of whether patients had thrombophilia, no thrombophilia, or were not tested (Table 4). These differences did not reach statistical significance in every group, but were significant for MBEs/CRBEs (Table 4) and for any bleeds (data not shown) in the no thrombophilia and the not-tested patient groups. Cox regression analysis for interaction showed that the safety profile of dabigatran versus warfarin was not significantly affected by the presence of thrombophilia.
RE-MEDY
In the overall safety analysis for RE-MEDY, the risk of MBEs (Table 4) was numerically lower with dabigatran than with warfarin (0.9% vs 1.8%), but did not reach statistical significance (p=0.06), while the rates of MBEs/CRBEs (Table 4) and any bleeds were significantly lower (p<0.001) with dabigatran than with warfarin.
The point estimates of the incidences of all bleeding event categories were lower with dabigatran than with warfarin, regardless of whether patients had thrombophilia, no thrombophilia, or were not tested (Table 4). These lower bleeding rates with dabigatran were statistically significant for MBEs in the no thrombophilia group, and were statistically significant for MBEs/CRBEs and for any bleeds (data not shown) in the no thrombophilia and the not-tested patient groups. Cox regression analysis for interaction showed that the safety profile of dabigatran versus warfarin was not significantly affected by the presence of thrombophilia.
Antiphospholipid syndrome analysis
Pooled data from patients with APS at baseline enrolled in RE-COVER, RE-COVER II, and RE-MEDY showed no significant difference in the incidence of MBEs between those treated with dabigatran etexilate versus warfarin (1.4 vs 2.6%; HR 0.46 [95% CI 0.04, 5.43]) (Table 5). MBEs/CRBEs occurred in numerically fewer patients in the dabigatran etexilate group than in the warfarin group (8.6 vs 18.2%). However, the difference was not statistically significant. Bleeding events of any type were less frequent with dabigatran, occurring in 20% of patients, compared with 40.3% with warfarin, which was statistically significant (HR 0.50 [95% CI 0.26, 0.95]) (Table 5).
Discussion
Our study suggests that the efficacy and safety of dabigatran etexilate are not significantly affected by the presence of thrombophilia. In the RE-COVER/RE-COVER II trials, 34% of patients were tested for thrombophilia, and 23% of these were positive. The overall proportion of patients in the pooled analysis with known thrombophilia was 8%. In RE-MEDY, 48% of patients were tested for thrombophilia, and 38% of these were positive. The overall proportion of patients with known thrombophilia was 18%.
It is possible that differences between treatment groups were not detected due to lack of statistical power. For example, the Kaplan–Meier curves for VTE/VTE-related death in thrombophilia versus no thrombophilia patients may imply a difference between treatments (HR of 0.30 favoring dabigatran). However, the confidence intervals are large due to the small sample size.
The overall proportion of patients with known APS in the pooled RE-COVER/RE-COVER II and RE-MEDY analysis was 2.2%. Antiphospholipid syndrome comprised 21% of the known thrombophilias. The results suggest that the efficacy of dabigatran etexilate remains similar to that of warfarin for the treatment and prevention of VTE in patients with APS. Rates of bleeding events tended to be lower with dabigatran etexilate than with warfarin in patients with APS, reaching statistical significance for any bleeding events.
In the RIETE Registry of 21,367 consecutive symptomatic VTE patients, 14 21% underwent thrombophilia testing. The three most common detected thrombophilias were factor V Leiden (26%), antiphospholipid antibodies (20%), and prothrombin gene mutation (18%). The rate of thrombophilia was similar in patients with idiopathic and with provoked VTE.
The two most common autosomal dominant genetic mutations predisposing to VTE are factor V Leiden, which causes resistance to the endogenous anticoagulant activated protein C (which inactivates coagulation factors V and VIII), and the prothrombin gene mutation, which increases the plasma prothrombin concentration. APS is the most common acquired cause of thrombophilia and is associated with both venous and arterial thrombosis. Antithrombin, protein C, and protein S are naturally occurring coagulation inhibitors. Deficiencies of these inhibitors are associated with VTE but are rare.
VTE patients likely to be tested in clinical practice often have at least one of the following characteristics: thrombosis at a young age, recurrent thrombotic events, thrombosis in unusual sites, a strong family history of thrombosis, recurrent miscarriages, or infertility. 1 However, no DOAC trials have reported the results of VTE treatment in studies specifically designed to study thrombophilia, especially in the high-risk group of patients with APS. Some investigators have stated that until DOAC trials studying APS are completed, these patients should not be managed with DOACs.15,16 Three such trials are under way: (1) Rivaroxaban for Antiphospholipid Antibody Syndrome (RAPS) (NCT02116036), (2) Rivaroxaban in Thrombotic Antiphospholipid Syndrome (TRAPS) (NCT02157272), and (3) Apixaban for the Secondary Prevention of Thromboembolism among Patients with the AntiphosPholipid Syndrome (ASTRO-APS) (NCT02295475).
In the present study, not all patients were tested for thrombophilia. Therefore, our results do not reflect the true number of patients with these conditions. However, the strengths of our trial are that RE-COVER, RE-COVER II, and RE-MEDY were randomized, double-blind studies with central adjudication of outcome events and included patients with and without thrombophilia. Another weakness is that diagnosis of thrombophilia was not centralized and was defined as reported by the investigator based on local analysis. Furthermore, the diagnosis of APS did not follow the revised Sapporo criteria with two tests at least 12 weeks apart, and it was not known whether all titers of antibodies reached the required criteria. 17 Without the Sapporo criteria being met, perhaps these patients could be considered as having the lupus anticoagulant disorder rather than APS.
Limitations of a post hoc analysis preclude us from providing precision in patients with the most severe thrombophilic disorders. Future research in DOAC administration should study prospectively and with more detailed definitions of those patients with antiphospholipid antibodies and those with severe inherited thrombophilia.
Conclusion
Based on pooled data from the RE-COVER and RE-COVER II trials and on data from the RE-MEDY trial, the presence of thrombophilia did not significantly affect the efficacy or safety of dabigatran etexilate in preventing recurrent VTE. Thus, the efficacy and safety profiles for the overall dabigatran trials appear to be maintained in the subgroup with thrombophilia.
Footnotes
Acknowledgements
Writing support was provided by Keith Day, PhD, who was contracted by Boehringer Ingelheim for these services.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Samuel Z Goldhaber receives honoraria for consulting activities from Bayer, Boehringer Ingelheim, Bristol-Myers Squibb (BMS), Daiichi Sankyo, Janssen, Portola, and Zafgen; Dr Goldhaber receives research support from BiO2 Medical, Boehringer Ingelheim, BMS, BTG EKOS, Daiichi Sankyo, the National Heart, Lung, and Blood Institute of the National Institutes of Health, Janssen, and the Thrombosis Research Institute. Henry Eriksson receives honoraria for lecturing and consulting activities from Boehringer Ingelheim, Bayer, Pfizer, and LeoPharma. Ajay Kakkar receives honoraria for consulting activities from Boehringer Ingelheim, Bayer, Jansen, Daiichi Sankyo, and Sanofi; Dr Kakkar receives research support from Bayer. Sebastian Schellong receives honoraria for lecturing and for consulting activities from Boehringer Ingelheim, Bayer, Bayer HealthCare, BMS, Daiichi Sankyo, and Pfizer. Sam Schulman receives honoraria for consulting activities from Boehringer Ingelheim, Bayer, Daiichi Sankyo, and BMS; Dr Schulman receives research grants from Boehringer Ingelheim, Baxalta, and Octapharma. Martin Feuring is an employee of Boehringer Ingelheim. Mandy Fraessdorf is an employee of Boehringer Ingelheim. Joerg Kreuzer is an employee of Boehringer Ingelheim. Elke Schueler is an employee of Accovion.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The RE-COVER and RE-MEDY studies were funded by Boehringer Ingelheim. The authors meet criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICMJE), were fully responsible for all content and editorial decisions, and were involved at all stages of manuscript development. The authors received no compensation related to the development of the manuscript.
