Abstract
Current intra-arterial catheter-directed thrombolysis (CDT) protocols recommend treatment with small doses of a thrombolytic agent, which excludes patients in need of urgent revascularization. We evaluated the effects of accelerated thrombolysis utilizing increased recombinant tissue plasminogen activator (rt-PA) doses. Forty-one patients with acute, thrombotic limb ischemia (ALI) were treated using accelerated CDT. The treatment consisted of an initial dose of 10 mg rt-PA for 30 minutes followed by a 3-hour course of a continuous intra-arterial 10 mg/hour rt-PA infusion. Simultaneously, intravenous unfractionated heparin (500 IU/hour) was administered. No deaths occurred. Satisfactory lysis was achieved in 37 of the 41 patients (90.2%). All significant underlying lesions were corrected (89.2%). Complications developed in nine patients (22%); the most frequent complication (four patients, 9.8%) was puncture site hematoma. The reintervention rate was 2.6% and 15.4% at the 1 and 6-month follow-ups, respectively. The major amputation rate was 10.3% and 12.8% at the 1 and 6-month follow-ups, respectively. Outflow compromise was adversely related to successful outcome at the 6-month follow-up (p=0.01). In conclusion, this study confirms the effectiveness and safety of the accelerated CDT regimen for treatment of thrombotic ALI at a single center, but requires confirmation in further studies.
Keywords
Introduction
Acute lower limb ischemia (ALI) is a dramatic and often life-threatening event. The major cause of ALI is currently arterial thrombosis, which accounts for approximately 85% of all ALI cases. 1 Since the mid-1970s (after the pioneering works of Dotter et al.), intra-arterial catheter-directed thrombolysis (CDT) is one of the most widely available and effective treatments for patients with ALI 2 (confirmed in randomized controlled trials and meta-analyses3–6).
Owing to the fear of bleeding complications, most authors recommend small doses of thrombolytic agent infused over a longer period of time (even up to 48 hours).7,8 This regimen, however, precludes the treatment of patients with severe ischemia (classes IIb and III according to the Rutherford classification) who require prompt revascularization.1,9 In contrast, large doses of recombinant tissue plasminogen activator (rt-PA) administered over a short period of time are not related to an increased frequency of significant bleeding, and this finding was confirmed in patients with massive and submassive pulmonary embolisms. 10 Some studies regarding the use of high rt-PA doses (up to 17 mg/hour) for the treatment of ALI yielded satisfactory results. 11 Here, we analyze the results of an accelerated CDT protocol using increased rt-PA doses in patients with ALI of thrombotic origin treated at our department.
Methods
This study was a prospective, observational analysis of patients with ALI who were treated at our department with an accelerated CDT protocol between May 2012 and March 2015. Upon admission, the following data were collected: symptom duration, severity and type of ischemia, comorbidities, and smoking status. Further data were collected during treatment, including the location and length of thrombus, outflow compromise, vascular access site, dose of thrombolytic used, immediate clinical and angiographic results, adjuvant vascular procedures and complications. Results, including survival status, amputations, and reintervention rate, were evaluated at 1 and 6 months.
Study group
All of the patients with ALI, defined as ischemia symptoms with onset/worsening within 14 days prior to the procedure, were examined to determine the optimal treatment protocol. The severity of ischemia was assessed according to the Rutherford classification. CDT was always considered in patients under 80 years old with ischemia classes I and IIa. Patients with ischemia class IIb were considered for the procedure only if the angiography suite was readily accessible to avoid treatment delay. Patients with profound ischemia (class III) or with contra-indications to angiography and/or thrombolysis were not offered CDT (Table 1).
Contraindications to angiography and thrombolysis.
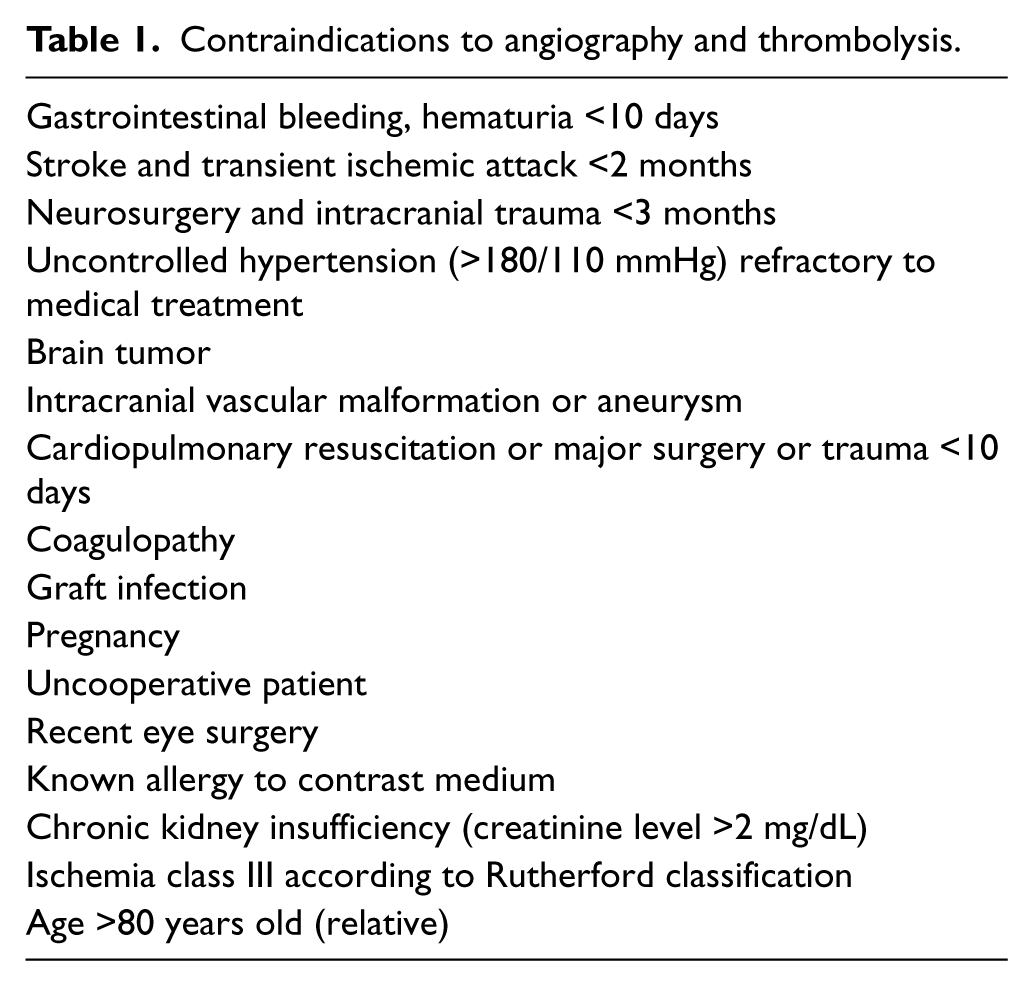
The etiology of ischemia was assessed. The following information was collected: history of arrhythmias (especially atrial fibrillation), previous coronary artery disease (CAD), previous limb claudication, peripheral emboli; physical evaluation (arrhythmia, presence of cardiac murmurs and peripheral pulses on the index and contralateral limb); electrocardiogram (ECG) and duplex ultrasound. Patients with a high probability of peripheral embolism were not offered CDT. Organized embolic material may be less prone to pharmacologic thrombolysis. Thus, surgical embolectomy/pharmacomechanical thrombectomy are the recommended treatment options for these patients.4,9,12
Data, including complete blood count, prothrombin time (PT), activated partial thromboplastin time (APTT), international normalized ratio (INR), creatinine level and serum electrolyte level, were collected upon admission. Continuous wave Doppler ultrasonography was performed to detect arterial/venous flow and duplex ultrasound to exclude embolic events.
All of the patients who qualified for CDT provided informed consent and were treated according to the good clinical practice (GCP) rules and Helsinki declaration. The CDT protocol was previously approved by the hospital’s scientific board.
Treatment protocol
Upon admission, a 50 IU/kg bolus of unfractionated heparin (UFH) was administered with a subsequent continuous infusion of 1000 IU/hour (i.v.) with adequate modifications to achieve 1.5–2.5 times the elongation of APTT.
All of the CDT procedures were performed in the angiography suite. Contralateral femoral artery access and a single-wall puncture technique were used whenever possible to minimize the risk of bleeding. First, a 5-Fr sheath was inserted, and a standard diagnostic 5-Fr pig-tail catheter was advanced into the abdominal aorta over a standard 0.035″ J-wire. Digital subtraction angiography of the abdominal aorta and lower limb arteries was obtained to assess the lesion type, location and extension (Allura Xper FD 20; Phillips, Amsterdam, The Netherlands). After crossing the aortic bifurcation, the 5-Fr sheath was replaced with a 40–55 cm 6-Fr therapeutic sheath, straight or contralateral depending on the operator’s discretion (Flexor™; Cook, Bloomington, IN, USA). A guidewire traversal test using hydrophilic 0.035″ guidewire (Aquawire™; Merit Medical Systems Inc., South Jordan, UT, USA) was performed to assess the lesion and to assure the possibility of targeted thrombolysis. If the lesion could not be crossed, thrombolysis was not conducted. After the positive guidewire test, a multiple side-hole thrombolytic catheter (Multi-Sideport Catheter Infusion Set™) was inserted into the proximal part of the thrombus so that a few proximal holes of the infusion segment of the catheter were adjacent to the proximal end of the thrombus to assure adequate lysis. The sheath and catheter were secured with a skin suture, and the patient was transferred to our intensive care unit for further treatment.
The thrombolysis was performed according to a standardized protocol, which was similar to a previously developed protocol.13,14 A continuous infusion of 10 mg rt-PA (Actilyse®; Boehringer Ingelheim, Ingelheim, Germany) in a saline solution (50 ml) was injected (30-minute bolus) through the catheter. Then, rt-PA infusion (10 mg/hour) continued for an additional 3 hours until the total dose of 40 mg was administered. Simultaneously, 500 IU/hour of unfractionated heparin was infused into the sheath.
During CDT, the patients were constantly monitored in our step-down unit for continuous heart rate, blood pressure measurement (every 10 minutes), frequent puncture site evaluation, and serial hematocrit. If hemodynamic instability and/or excessive puncture site bleeding occurred, the complete blood count and fibrinogen level were additionally tested. Because most patients suffered from transient pain due to the temporary perfusion worsening, opiate injections (pethidine 50–100 mg i.v.) were liberally administered at the attending physician’s discretion. At the end of rt-PA infusion, the treated limb was always examined for pulse, distal capillary refill, tenderness of calf muscles and sensorimotor deficits. Control angiography was performed to assess the thrombolysis result. The degree of lysis was defined according to the observed degree of thrombus lysis (Table 2). Both second and third-degree lysis indicated successful treatment. If first-degree lysis was observed and no adverse events occurred during infusion, a second course of thrombolysis (according to the same protocol) was offered to the patient. An unsatisfactory result of the second treatment course was considered a procedure failure. If no effect of thrombolysis was observed after administration of 40 mg rt-PA (grade 0), the CDT was abandoned. Both grade 0 and grade 1 were considered a clinical failure.
Classification of CDT angiographic result.

CDT, catheter-directed thrombolysis.
Outflow compromise, defined as long, chronic occlusions of at least two tibial arteries, was assessed during the initial angiography procedure and was confirmed during the completion angiography procedure.
If found, underlying lesions were always corrected (endovascular and/or surgical procedures). After successful treatment, the patients were always prescribed acetylsalicylic acid (75 mg/day) and a statin (atorvastatin, 20 mg once a day). Clopidogrel (75 mg once per day for 6 weeks) was added if additional endovascular procedures were performed. The patients were routinely monitored at discharge and at 1 and 6-month follow-ups at our outpatient clinic. If the patient missed his/her appointment, phone contact was attempted to assess the outcome.
Statistical analysis
Continuous variables with normal distributions (Shapiro-Wilk test) are presented as means and standard deviations (SD). For variables without normal distributions, the median and range values are presented. Categorical variables are expressed as frequencies and percentages of patients. The two-tailed Fisher’s exact test was used for statistical analysis. A p-value <0.05 was considered significant.
Results
Between May 2012 and March 2015, 160 patients with ALI were presented to our department. After screening, CDT was further considered in 56 patients (35%). The major reasons for screening failure were: age over 80 years old, 36 patients (22.5%); arterial embolism, 21 patients (13.1%); and ischemia severity, 20 patients (12.5%). Guidewire test failure occurred in 15 patients, and these patients were immediately revascularized (either endovascular or operative procedures). The remaining 41 patients (40 men; average age: 60.5, range 44–78, SD ± 8.1 years) were treated with accelerated CDT. The demographic details and medical comorbidities are presented in Table 3.
Baseline patient characteristics and comorbidities.
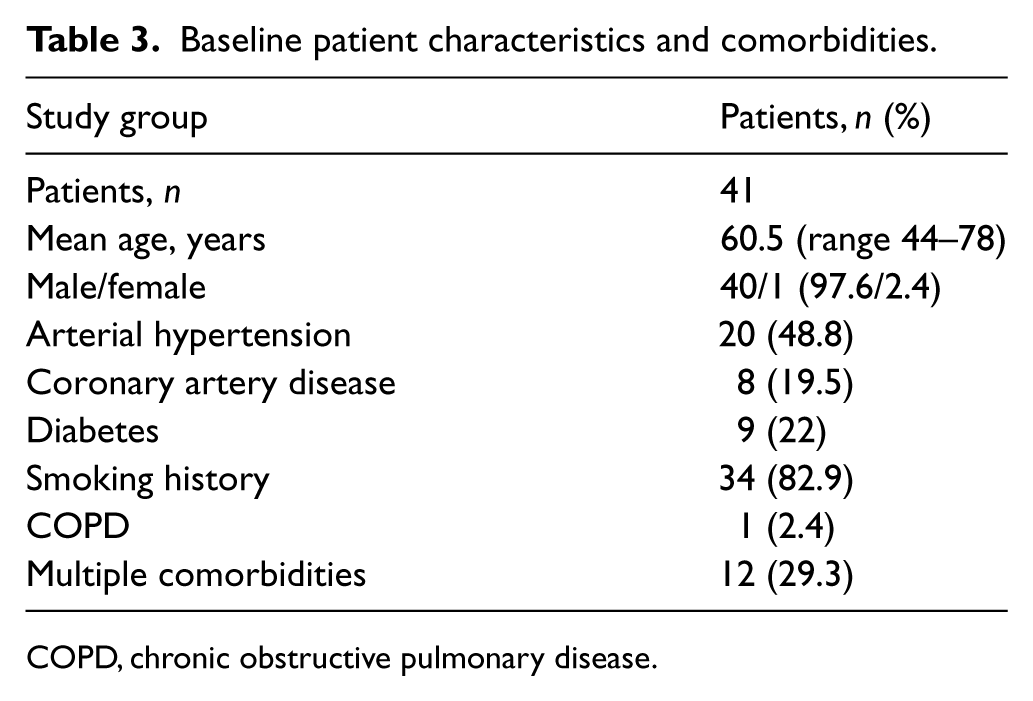
COPD, chronic obstructive pulmonary disease.
The median ischemia duration amounted to 6 days (range 0–14). The following distribution of ischemia severity was observed: class I = one patient (2.4%), class IIa = 35 patients (85.4%), and IIb class = five patients (12.2%).
Thrombosis was the cause of ischemia in 40 patients: native artery = 17 (41.5%), bypass = 14 (34.1%), and stent = 9 (22%). In one patient, embolus in the common femoral artery was identified during angiography. Iliac arteries were involved in six patients (14.6%). Infrainguinal arterial lesions were observed in the remaining 35 patients (85.4%). The median thrombus length was 19 cm (range 4–47).
Successful thrombolysis (grades 2 and 3) was observed in 37 patients (90.2%). Successful thrombolysis occurred in 30 (73.2%) patients after receiving 40 mg rt-PA and in seven (17.1%) patients after an additional course of rt-PA (total 80 mg rt-PA).
Thrombolysis failed in four patients (9.8%). In two patients, significant outflow compromise was observed during angiography. We considered further vascular procedures futile in these patients, and revascularization was no longer offered. One patient was then lost to follow-up, and the other patient underwent amputation above the knee 3 weeks later. In the two remaining cases, despite ineffective thrombolysis, satisfactory outcomes were achieved after an additional endovascular procedure (angioplasty and stenting) and surgical operation (femoropopliteal bypass) (each in one patient).
No peri-procedural mortality occurred (0%). In-hospital complications affected nine patients (22%). The most frequent complication was puncture site hematoma, which occurred in four patients (9.8%). All four patients were successfully treated with prolonged compression (Table 4).
Treatment results.
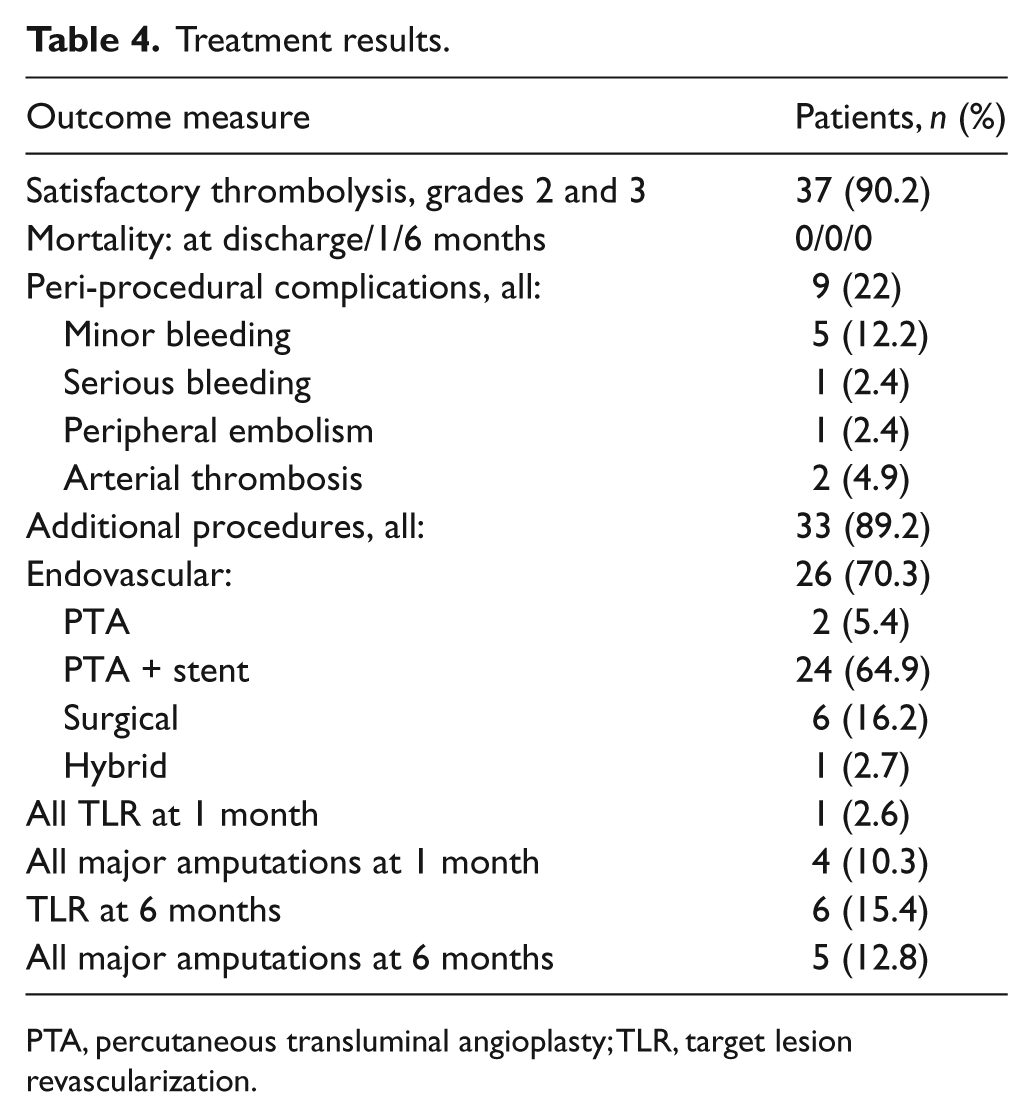
PTA, percutaneous transluminal angioplasty; TLR, target lesion revascularization.
One patient suffered an ipsilateral calf hematoma causing compartment syndrome with severe pain and sensorimotor deficits. Surgical decompression was urgently performed with a successful clinical outcome.
Peripheral embolism was observed in one patient. After an initially successful thrombolysis of an occluded femoro-popliteal bypass, the fragments of the thrombus dislodged to the P3 segment of the popliteal artery. Further thrombolysis (two courses) yielded only a partial effect, and surgical thrombectomy with patch angioplasty was performed to achieve a satisfactory outcome.
One of the two patients with thrombosis of arteries peripheral to the initial occlusion was successfully treated with another thrombolysis course, and in the other major amputation was performed.
Procedures to correct underlying lesions after successful thrombolysis were performed in all but four patients (: 89.2%): angioplasty alone in two (5.4%), angioplasty with stenting in 24 (64.9%), surgery in six (16.2%) and hybrid treatment in one (2.7%) patient(s).
Two patients were lost to follow-up (4.9%). The 1 and 6-month follow-up data were collected for the remaining 39 patients.
No mortality was recorded during the follow-up period. The reintervention rate at 1 and 6 months was 2.6% (one patient) and 15.4% (six patients), respectively. The major amputation rate at 1 and 6 months was 10.3% (four patients) and 12.8% (five patients), respectively. 5
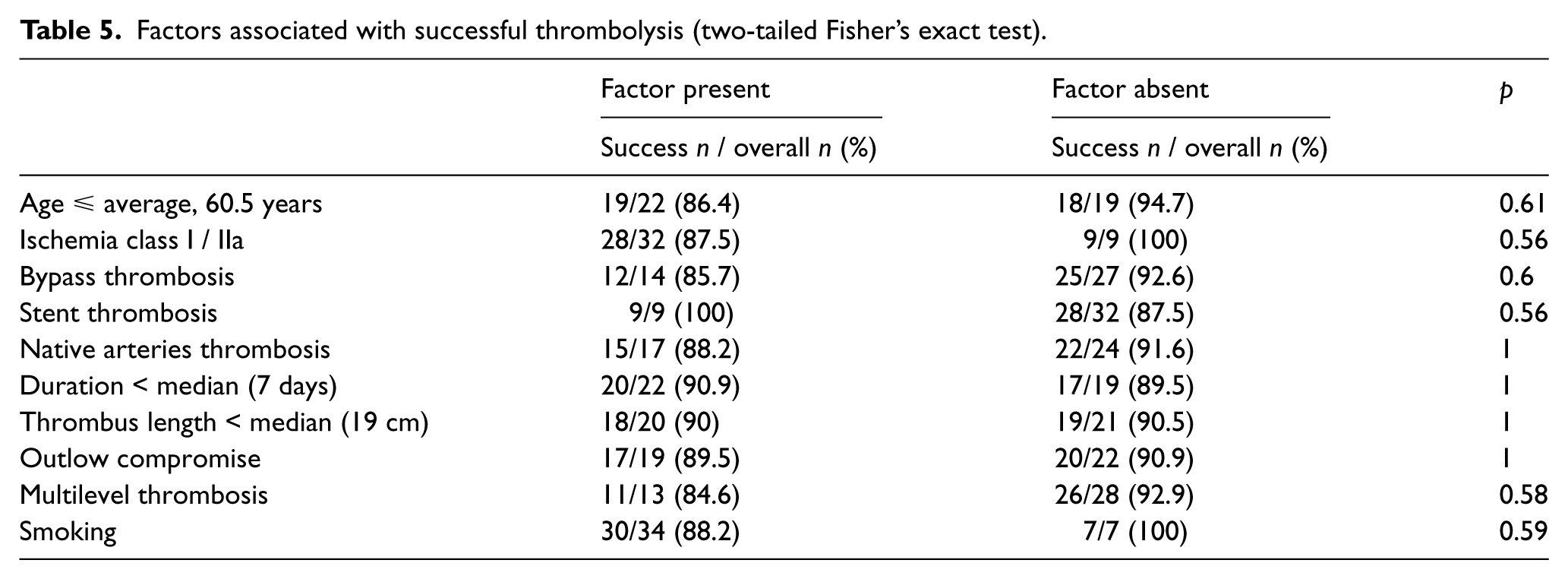
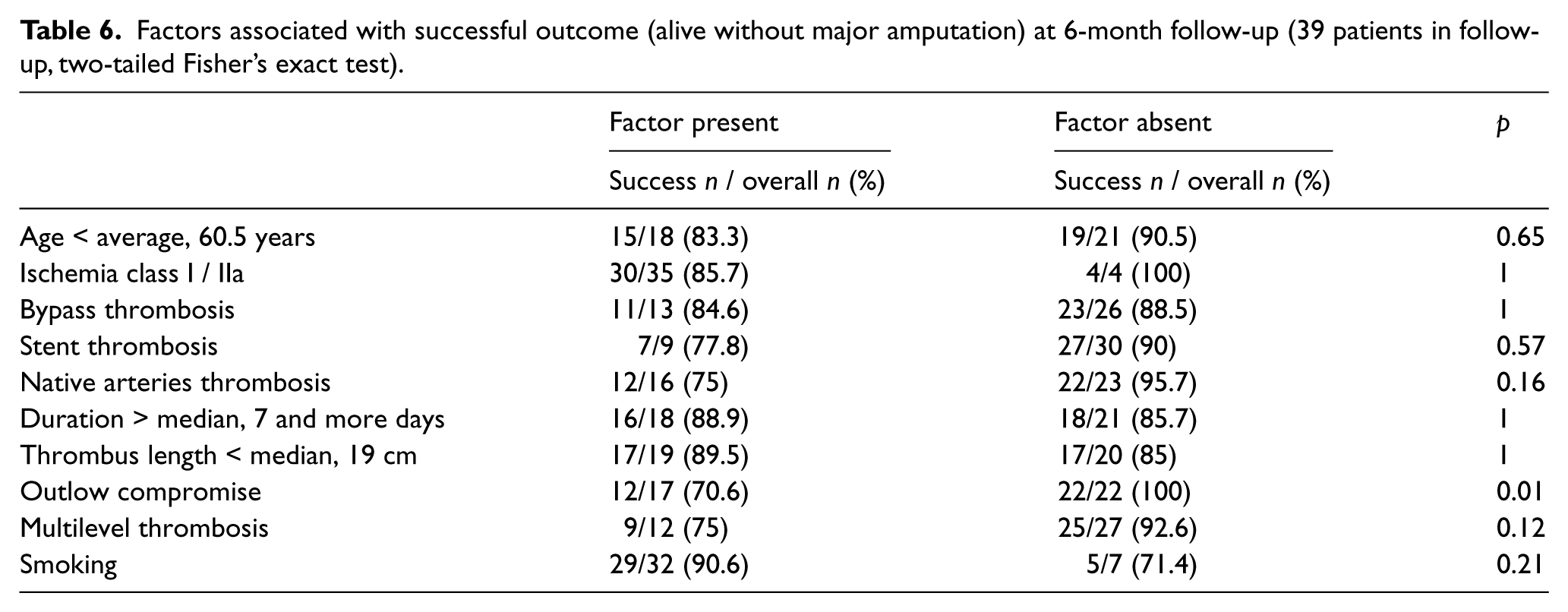
We analyzed (Fisher’s exact test) the relationships between an initially successful thrombolysis and satisfactory 6-month outcome (alive without major amputation) with the following factors: age, ischemia class, occluded vessel type (bypass, stent, native artery), ischemia duration, thrombus length, presence of multilevel thrombosis, outflow compromise and smoking status.
No relationship was observed between successful thrombolysis and any of the above factors (Table 5). An adverse relationship between success at 6 months and outflow compromise (p=0.01, Fisher’s exact test) was identified. Additionally, trends toward inferior outcomes in patients with native artery thrombosis and multilevel thrombosis were observed. These relationships did not reach significance (p=0.14 and p=0.12, respectively; Fisher’s exact test) (Table 6).
Factors associated with successful thrombolysis (two-tailed Fisher’s exact test).
Factors associated with successful outcome (alive without major amputation) at 6-month follow-up (39 patients in follow-up, two-tailed Fisher’s exact test).
Discussion
During the last 20 years, we have observed significant progress in the treatment of ALI. Endovascular techniques have gained widespread acceptance. CDT is now the best documented, minimally invasive technique used for the treatment of ALI.
The standard technique of low-dose, prolonged thrombolysis excludes patients with ischemia requiring expeditious treatment. Considering the proven safety of high-dose thrombolysis in patients with acute coronary syndromes and massive pulmonary edema, we administered higher than standard doses of rt-PA (40 mg in 3.5 hours, i.a.) in a selected group of patients with acute arterial thrombosis. Clinically successful thrombus lysis was achieved in >90% of our patients with acceptable morbidity and no mortality. Only one patient (2.4%) experienced bleeding requiring intervention. This bleeding was not life-threatening. In four other patients, puncture site bleeding required only prolonged local pressure application. Our results (in terms of efficacy and safety) are at least as good as those reported in two recently published series.11,15
The major advantage of the accelerated thrombolysis protocol was that we were able to treat patients within a period of time comparable to surgery with excellent results. We treated five patients with class IIb ischemia (12.2%), and the success rate in this group was 100%. Even higher doses of rt-PA (up to 17 mg/hour i.a.) have been used by other clinicians. However, this dose was accompanied by fatal intracranial hemorrhage (1%) and peripheral emboli (6.2%) (almost twice as high as in our experience). 11
Distal embolism was also our concern. We observed signs of perfusion deterioration during thrombolysis in most patients. Sometimes, the deterioration was severe and accompanied by aggravated pain requiring high doses of opiates. In all patients, the symptoms were relieved during ongoing thrombolysis. One patient with peripheral embolization experienced attenuation of ischemia symptoms at the end of the procedure. We believe that our technique of alteplase infusion into the proximal portion, not through the entire thrombus, is justified for two reasons. First, the thrombus is constantly exposed to the high level of plasmin (fresh plasminogen constantly delivered with flowing blood and activated at the catheter tip). Second, the distal part of the thrombus provides a form of natural embolic protection. Because the thrombus dissolves gradually at the proximal part and collateral flow through the previously occluded side branches occurs, small emboli tend to migrate to these branches. Delivering the plasminogen activator through the entire length of the thrombus may cause uncontrolled distal disruption and possible embolism.
During drug infusion, all patients were constantly monitored in the ICU. Continuous blood pressure and heart rate monitoring and frequent (every 15–20 minutes) puncture site observations were performed. We did not monitor the serum level of fibrinogen at the end of thrombolysis because the evidence to support this regimen is weak and the guidelines do not support routine fibrinogen monitoring.16,17 Larger studies are likely required to elucidate this aspect of the thrombolysis safety protocol.
We analyzed the relationships between the pre-procedural factors and the initial (thrombolysis grades 2 and 3) and final (freedom from major amputation at the 6-month observation) success rates.
Although no significant factors influenced the initial success rate, patients with at least 2 patent tibial arteries had better outcomes at 6 months than patients with outflow compromise (p=0.01). Our study confirmed that poor circulation negatively influences the results of both operative and endovascular revascularization.18–20 Another important clinical observation is that when reocclusion occurs in the presence of significant tibial vessel disease, the extent of the thrombus is increased and often precludes any revascularization. Therefore, in our opinion, the presence of poor circulation in patients with ALI warrants aggressive treatment to improve outflow.
We also noticed that amputations at 6 months occurred more frequently in patients with native artery and multilevel thrombosis, but the relationships did not reach significance (p=0.16 and p=0.12, respectively). Although it is intuitive that treatment of more extensive disease will produce less favorable results, these findings require confirmation in larger series.
Limitations
There were some limitations in our study. Despite the relatively large number of patients with ALI who presented to our department, thrombolysis was feasible in approximately 25% of them. The relatively small number of treated patients resulted in a lack of statistical power to confirm the significance of relationships between some factors and the treatment outcome. Because we used only a single-dose strategy, these results cannot be directly compared to different thrombolysis protocols, although our previous unpublished experience with standard (1 mg/hour) CDT indicates that this protocol yields better outcomes with fewer complications than the standard treatment.
In our opinion, a large, randomized, multicenter, multi-dose trial is required to determine the optimal thrombolysis protocol. Considering emerging endovascular thrombectomy strategies for the treatment of ALI, the value of high-dose CDT should be carefully assessed.
Conclusions
Thrombolysis, according to the protocol utilized at our department, provided excellent treatment results within a timeframe comparable to surgery and with an acceptable complication rate. Thus, CDT might be considered the treatment of choice in patients with ALI of thrombotic origin classes I–IIb if adequate peri-procedural care and 24/7 vascular surgery unit services are provided. We found a significant correlation between outflow compromise and treatment failure at the 6-month follow-up.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
