Abstract
The introduction of fenestrated stent grafts (SGs) to treat abdominal aortic aneurysms (AAAs) with short proximal necks began in 1999. Nowadays, the whole visceral aorta can be treated totally by endovascular means. The established use of fenestrated devices to treat complex AAAs as a first-line management option has been previously reported. An up-to-date evaluation of the literature was performed including all types of publications regarding the use of fenestrated technology to repair complex AAAs. Fenestrated repair is now an established alternative to hybrid/chimney/snorkel repairs. However, specific criteria and prerequisites are required for the use and improvement of this method. Multiple device morphologies have been used incorporating the visceral arteries in various combinations. This modular strategy connects different devices (bridging covered stents and bifurcated SGs) with the aortic main body, thus excluding the aneurysm from the circulation. Precise deployment of the fenestrated SG is mandatory for successful visceral vessel revascularization. Accurate SG sizing and customization, a high level of technical skill, and facilities with modern imaging techniques including 3D road mapping and dedicated hybrid rooms are required. Most experience has been with the custom-made Zenith Cook platform, although off-the-shelf devices have been recently implanted. More complex repairs have been performed over the last few years, but device complexity has also increased. Perioperative, mid-term, and a few recently reported long-term results are encouraging. Secondary interventions remain the main problem, similar to that observed after traditional endovascular abdominal aortic aneurysm repair (EVAR).
Evolution of the fenestrated technology: from EVAR to stent grafts with holes and scallops
Endovascular abdominal aortic aneurysm repair (EVAR) is an established alternative to open surgery for the treatment of infrarenal abdominal aortic aneurysms (AAAs). 1
Overall, its suitability is primarily affected by poor proximal and distal sealing zones and limited iliac access. 2 Short, wide or severely angulated proximal aortic necks are found to be too unfavorable to be used as a landing zone and are thought to be the most likely causes for ineligibility of EVAR.3,4 Should these circumstances be ignored, or attempts made to extend the use of commercial infrarenal stent grafts (SGs) (Table 1) into complex anatomy, problems related to early and late failure will eventually arise. 5 For example, the shorter the non-dilated segment between the aneurysm and the lowest renal artery (what we call proximal neck), the less contact there is between an entirely infrarenal SG device, and the higher the likelihood of a type I endoleak or SG migration. 6
Available EVAR devices. a
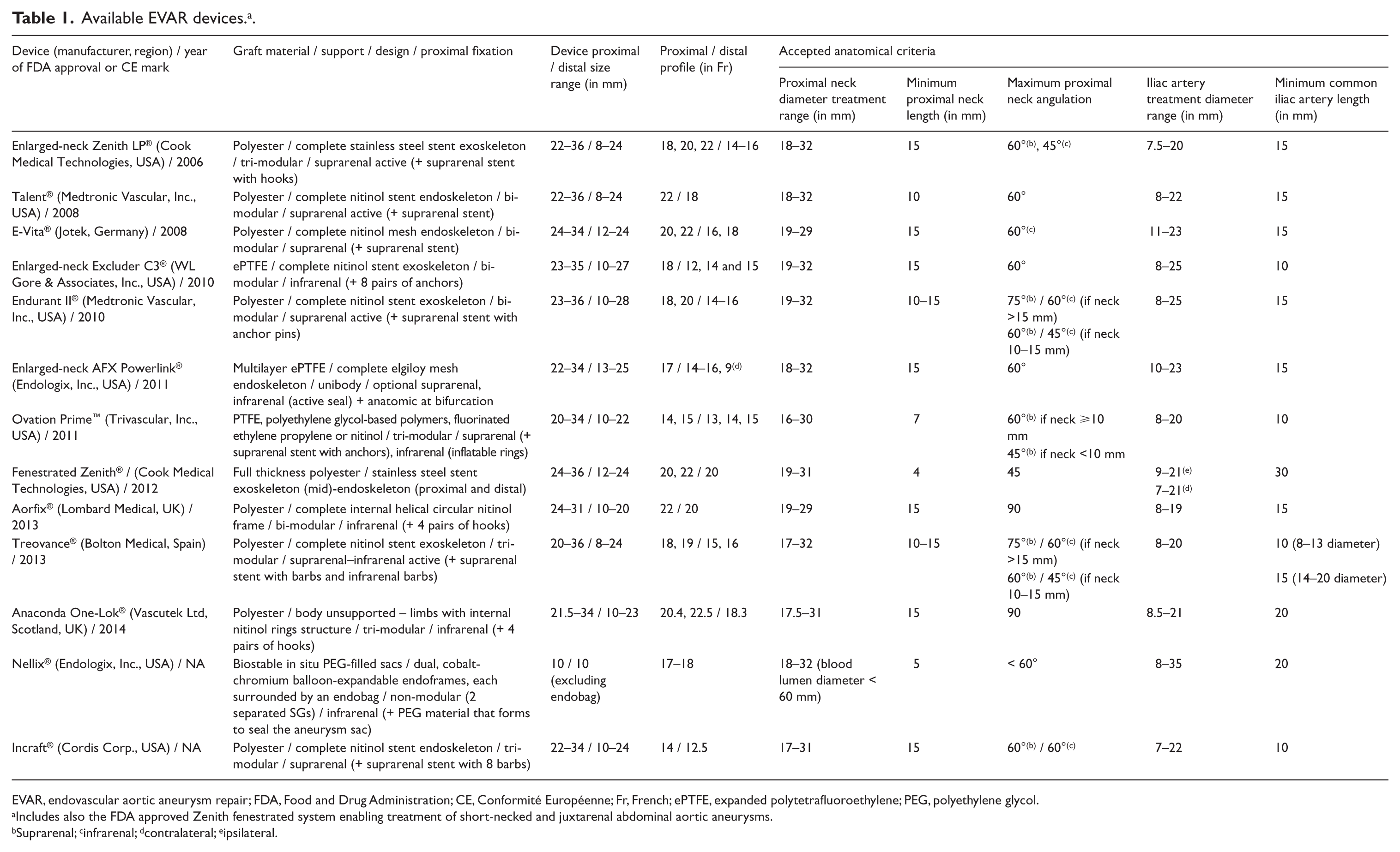
EVAR, endovascular aortic aneurysm repair; FDA, Food and Drug Administration; CE, Conformité Européenne; Fr, French; ePTFE, expanded polytetrafluoroethylene; PEG, polyethylene glycol.
Includes also the FDA approved Zenith fenestrated system enabling treatment of short-necked and juxtarenal abdominal aortic aneurysms.
Suprarenal; cinfrarenal; dcontralateral; eipsilateral.
Besides the above-described unsuitable infrarenal neck anatomy, many patients possess aortic aneurysm changes that encroach upon the visceral region and might be unsuitable candidates for infrarenal aortic devices. 7 Furthermore, a high percentage of them have severe comorbidities precluding traditional open repair.8–10 Thus, the unmet medical needs to preserve the endovascular management of complex aortic aneurysms urged clinicians and manufactures to develop SGs with fenestrations (holes) or scallops (gaps or valleys in the upper graft fabric margin) to access visceral arteries, called fenestrated SGs, simply to allow extension of the sealing zone indefinitely, limited only by the desire to limit the number of visceral vessels incorporated into the repair. 11 When successful, fenestrated EVAR (FEVAR) leads to complete exclusion of the aneurysm while maintaining antegrade renovisceral perfusion in vital organs. Some designs are without fenestrations but with branches (pre-attached limbs or cuffs) targeted for the visceral aortic vessels, suitable for the endovascular treatment of thoracoabdominal aortic aneurysms (TAAAs). At least for short-necked and juxtarenal aortic aneurysms (JRAs), fenestrated stent-grafting technology has been shown to be feasible,12–20 with published results of high-volume experienced single centers21–31 and a few recent multicenter trials32,33 sharing remarkable short- and mid-term results, with the durability of the technique being satisfactory (technical success > 95%, low mortality rates averaging 1–2% and low rates of mid-term branch vessel occlusion averaging 4–9%).25–29,34 Other imaginative procedures have also been developed or modified to allow for the segmental or complete coverage of the aorta in the region of vital branches, including the ‘chimney’, ‘snorkel’, ‘periscope’35–37 and combined endovascular exclusion and extra-anatomic mesenteric/renal bypasses (hybrid visceral revascularization). 38
The objective of this paper was to provide an up-to-date review on the elective repair of perirenal and TAAAs via the FEVAR approach.
Criteria and prerequisites for use and improvement of the fenestrated technology
General criteria
The most important indications for fenestrated SGs are listed in Table 2.2,22,24,27,28,30,34,39–50 Owing to the normal suprarenal aortic lumen, it is generally accepted that fenestrated devices are preferably used for pararenal aneurysms (PRAs).
Patient selection/anatomical considerations and prerequisites for use of fenestrated technology. a
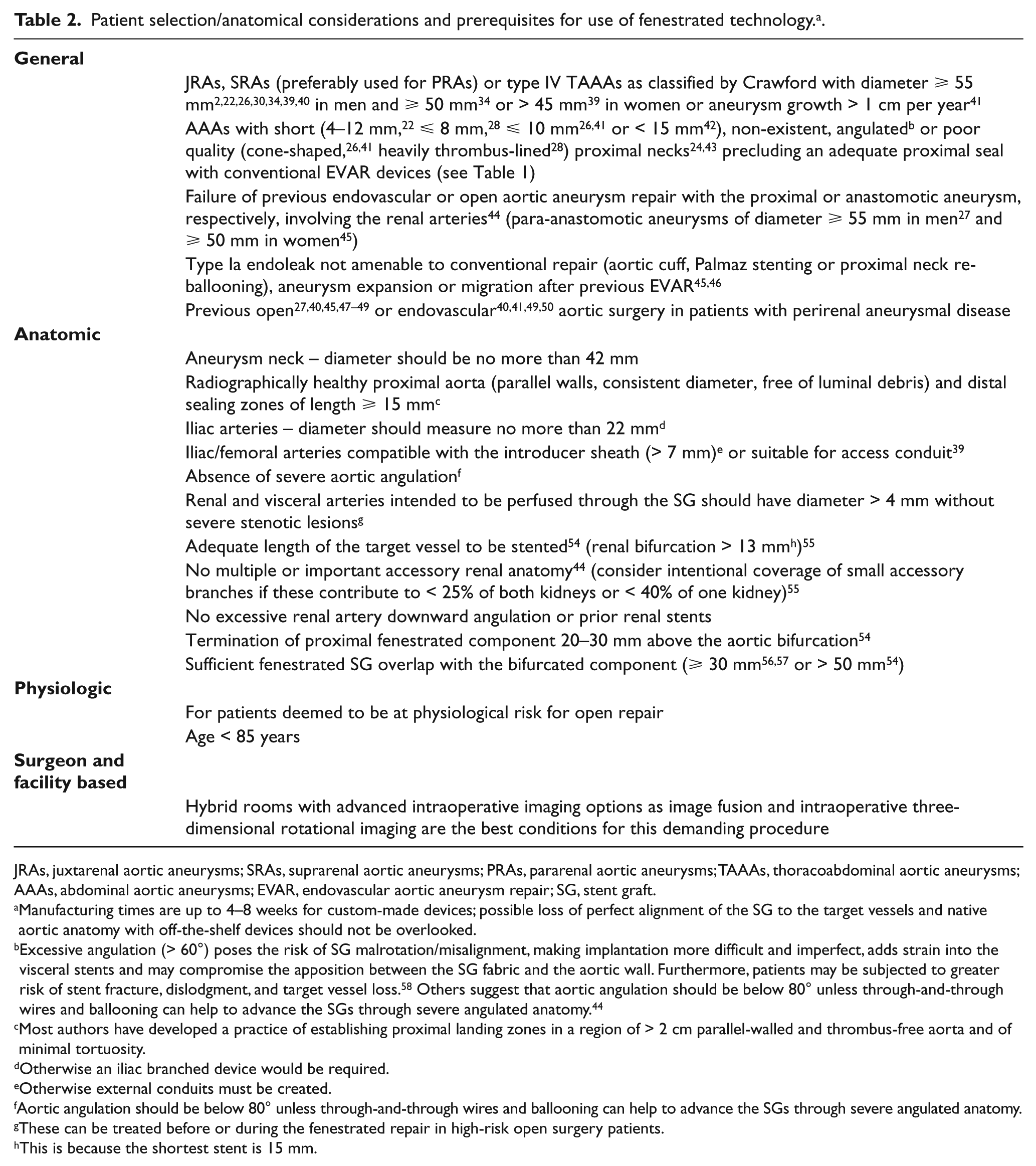
JRAs, juxtarenal aortic aneurysms; SRAs, suprarenal aortic aneurysms; PRAs, pararenal aortic aneurysms; TAAAs, thoracoabdominal aortic aneurysms; AAAs, abdominal aortic aneurysms; EVAR, endovascular aortic aneurysm repair; SG, stent graft.
Manufacturing times are up to 4–8 weeks for custom-made devices; possible loss of perfect alignment of the SG to the target vessels and native aortic anatomy with off-the-shelf devices should not be overlooked.
Excessive angulation (> 60°) poses the risk of SG malrotation/misalignment, making implantation more difficult and imperfect, adds strain into the visceral stents and may compromise the apposition between the SG fabric and the aortic wall. Furthermore, patients may be subjected to greater risk of stent fracture, dislodgment, and target vessel loss. 58 Others suggest that aortic angulation should be below 80° unless through-and-through wires and ballooning can help to advance the SGs through severe angulated anatomy. 44
Most authors have developed a practice of establishing proximal landing zones in a region of > 2 cm parallel-walled and thrombus-free aorta and of minimal tortuosity.
Otherwise an iliac branched device would be required.
Otherwise external conduits must be created.
Aortic angulation should be below 80° unless through-and-through wires and ballooning can help to advance the SGs through severe angulated anatomy.
These can be treated before or during the fenestrated repair in high-risk open surgery patients.
This is because the shortest stent is 15 mm.
Appropriate terminology is important when referring to FEVAR to fully understand the repair. Aortic aneurysms in the region of the renal arteries are named PRAs and further classified into juxtarenal and suprarenal types. 51 A JRA is defined as an aneurysm with a < 10 mm infrarenal aortic neck (i.e. with involvement of the aneurysmal dilatation up to the level of the renal arteries) that originates from normal aorta. 52 Although a JRA by definition is that in which the surgeon is able to place an infrarenal cross-clamp safely, a clear definition or a clear stratified classification system for JRAs does not exist, 40 especially in FEVAR studies. Donas et al. described a primary JRA as that with a short infrarenal neck < 9 mm or an aneurysmal extension to the inter-renal aorta. 40 Depending on the definition, JRAs represent almost 15% of all AAAs. 20
SRAs involve the origin of at least one renal artery and include the PRAs, which involve the renal arteries but do not extend into the superior mesenteric artery (SMA), and paravisceral aortic aneurysms, which involve the renal arteries and the SMA, but not the celiac axis. 52 Type IV TAAAs involve the origin of all four visceral arteries including the celiac axis, extending up to the diaphragmatic hiatus.17,52,53 Other authors prefer to classify repairs based on the postoperative degree of aortic coverage. 31
Anatomical criteria
Basic anatomic conditions
Generally, patients will have a survival benefit if the size of their AAA exceeds 5–5.5 cm. 1 Slightly larger diameters are universally accepted for fenestrated repair of JRAs and paravisceral aneurysms. Indications for treatment with FEVAR include short-necked AAAs (Figure 1), JRAs, or SRAs with diameters ⩾ 55 mm2,22,30,34,39,40 in men and ⩾ 50 mm 34 or > 45 mm 39 in women. Other described indications are listed in Table 1.
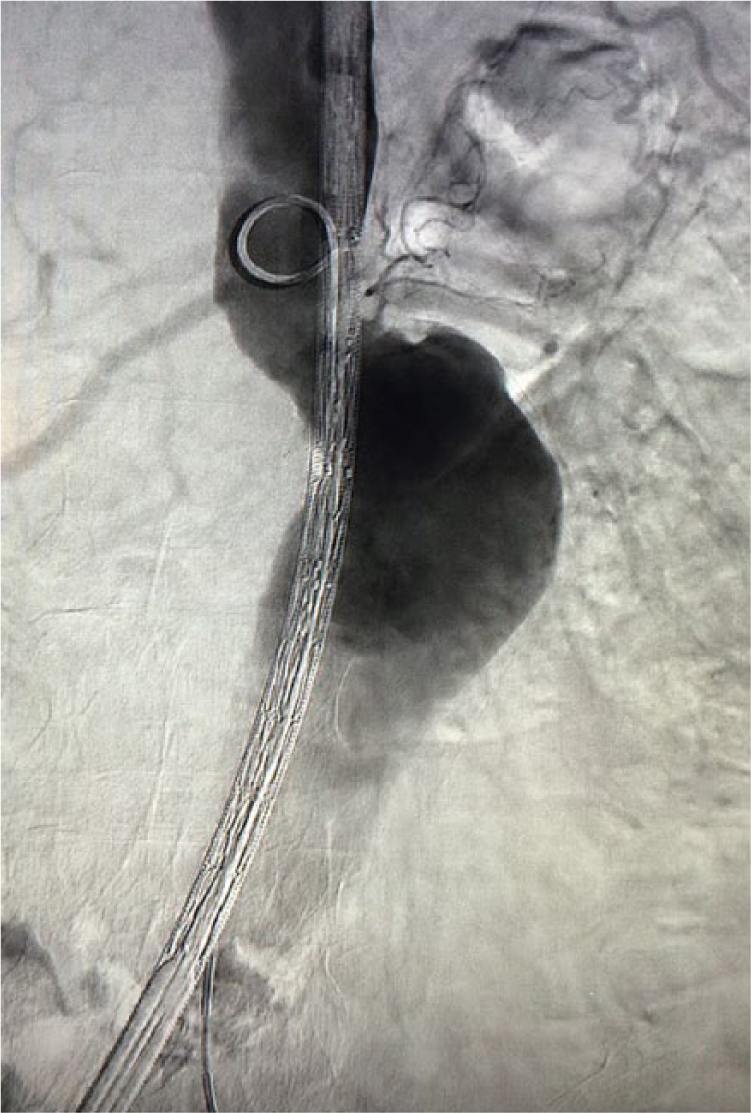
Angiography of a high-risk patient with a very short-necked infrarenal abdominal aortic aneurysm, which is unsuitable for standard endovascular repair (EVAR) but suitable for fenestrated repair.
Specific anatomic conditions
Extended basic anatomic criteria and technical issues for application of FEVAR with custom-made endografts are summarized in Table 2.39,44,54–58 Despite the use of fenestrations which are necessary to incorporate visceral vessels bearing the desired sealing zone, a segment of relatively normal suprarenal aorta that constitutes the proximal neck is still required in fenestrated grafts. Most authors have developed a practice of establishing proximal landing zones in a region of > 2 cm parallel-walled and thrombus-free aorta and of minimal tortuosity. This is conceptually different to landing zone choice in conventional infrarenal repair, given the lack of confinement to the non-branched aorta. However, with FEVAR there is likely additional risk incurred by incorporating the entire visceral segment. Traveling up the supravisceral segment, there is concern about whether the incidence of spinal cord ischemia markedly rises. 31
Physiologic criteria
Given the complexity of open aortic aneurysm repair and its complications, the application of a less invasive aortic intervention, like FEVAR, for patients deemed to be at physiological risk for open repair is intuitive. 33 The terms ‘unfit’ or ‘at high risk for open aneurysm repair’ patients, combined with unfavorable anatomic criteria for standard EVAR, were used as inclusion criteria in several FEVAR studies.23,27,28,30 Significant comorbidities which were analyzed are based on Societies’ or National reporting guidelines. 59 The French counterpart of the US Food and Drug Administration (FDA) determined the following high-risk patient characteristics for open repair: age > 80 years; coronary artery bypass graft (CABG)/coronary stent or previous myocardial infarction (MI) with no possible coronary revascularization; clinical symptoms of congestive heart failure (CHF); left ventricular ejection fraction (LVEF) < 40%; severe chronic obstructive lung disease (COPD), renal insufficiency (creatinine > 120 µmol/L) and hostile abdomen. 56 In other reports on FEVAR, medical high risk was also complementary based on patients anticipated to be unable to tolerate aortic cross-clamping 60 or redo surgery (abdominal 30 and/or iliac aortic repair), 40 patients with LVEF < 25%,30,43 patients with an American Society of Anaesthesiologists (ASA) score more than 3,40,61 or evidence of non-reconstructable cardiac ischemia. 30
Surgeon and facility-based criteria
In general, FEVAR is indicated for elective cases due to the customization required for the SGs, although surgeon-modified SGs and fixed-design standardized off-the-shelf devices have been used in the acute setting 62 or for specific anatomies, respectively.42,57,63 The surgeon/interventionalist and his tools (armamentarium) and imaging facility (operating theatre environment) are important for the FEVAR procedure. Current developments in fixed X-ray platforms enable advanced intraoperative imaging options such as image fusion and intraoperative three-dimensional (3D) rotational imaging.
Device planning and implantation are complex, requiring advanced training and a steep learning curve. Owing to the demanding nature of the procedure, considerable debate still exists about whether fenestrated endografts should be limited to a small number of specialized centers. 64 Compelling evidence suggests that FEVAR requires appropriately dedicated teams, experience, technical infrastructure and definitely a minimum annual enrollment rate (institutional and individual practitioner rate) in order to offer acceptable results for these fragile patients. An annual threshold of 15–20 complex aneurysms performed with FEVAR technology, preferably treating JRAs or PRAs with one or two fenestrations and a SMA scallop, including a few cases with proper proctoring, are necessary until a requisite level of competency is achieved. 64
Alternative options to FEVAR
Off-label use of EVAR outside instructions for use
Newer and more flexible SGs have marginally extended their instructions for use (IFU): proximal neck length > 10 mm when straight, or angulation < 90° where neck length is > 20 mm.16,65 Although not considered an alternative option, in some instances the use of standard EVAR for suboptimal necks, and outside IFU of commercially available devices, has been advocated;66,67 however, late re-intervention rates are likely to be higher. 66 In a large cohort of patients (> 10,000) who underwent EVAR with commercial devices in the US between 1999 and 2008, the incidence of AAA sac enlargement after EVAR was 41% at 5 years, a rate that increased over the study period. As shown in pre-EVAR anatomic computed tomography (CT) imaging data, liberalization of the anatomic characteristics deemed suitable for EVAR has occurred, and several of these factors, including aortic neck diameter and aortic neck angle, were independently associated with aortic aneurysm sac enlargement, 68 which is a clinical parameter of increased risk of rupture. Patients with low compliance with EVAR guidelines had an incidence of endoleak and sac enlargement at 5 years: 32% and 41%, respectively. 68 Recently, a greater incidence of type I endoleak was documented in an off-label group of patients versus an IFU group of patients receiving the new generation Endurant II device (Medtronic Inc, Minneapolis, MN). This endoleak, identified at 1 year, persisted in this patient population up to 5 years, although there was no difference between the IFU and off-label groups as regards survival, graft thrombosis, or re-intervention rates. 69 These mid- and long-term data support a recommendation for cautious application of standard EVAR in patients outside the device-specific IFU. 69
Hybrid, chimney and snorkel repairs
A variety of alternative approaches have been used to extend the indications of EVAR to patients with unfavorable anatomy.70,71 Retrograde revascularization of the visceral vessels (surgical bypass) followed by endovascular exclusion of the aortic aneurysmal site (open debranching technique) has been used, 72 but complication rates remain similar to that of open repair.73,74
This approach, which is considered as another off-the-shelf solution, can also be used as a bailout from accidental coverage of vital side branches during deployments requiring close approximation of the main body to the branch artery in question.36,77,78 Elective indications include JRAs or aneurysms with severely angulated necks, when alternative techniques and/or devices are not options. 57 The chimney technique fundamentally differs from fenestrated and branched EVAR with regards to the relationship of the visceral stents to the aortic SG. While fenestrated and branched visceral stents are mated and sealed within integral constructs of the SG, chimney stents are positioned along the outside of the SG. Thus, the technique relies on the close conformation of the SG and the aortic wall around the chimney stent. The sandwich technique involves the use of CVs placed in the abdominal aorta branches, like chimneys, that are ‘sandwiched’ between two aortic endografts. 75 This concept of parallel endografting, although off-label, has been popularized, but the additional seal achieved is imperfect. As such, concern remains about the effects of type I endoleak through the ‘gutters’ created by the multiple side-by-side CVs outside the main endograft on long-term durability as well as branched vessel patency.70,71 Potential strategies to nullify the gutter effect include the achievement of a ring of seal, or ‘gasket’ seal, in the neck of the aneurysm (JRAs) outside the area of the parallel endografts, the use of long overlap zones (no neck aneurysms) or, more attractively, direct obliteration of the gutters (‘eye of the tiger maneuver’). These adjuncts might help to improve outcomes with parallel grafts, not for only bailout situations. 79
Although technical success is high, two systematic reviews have shown that operative mortality (6% and 4.3%) and rate of type I endoleak (10% and 14%) are unquestionably high.70,71 In anatomically matched populations undergoing open or chimney repair of JRAs or SRAs, operative mortality did not differ between groups (4.8%); however, acute kidney injury was higher in the endovascular arm (29% vs 4.8%), possibly due to planned renal artery coverage in some patients. Nonetheless, permanent dialysis occurred only in open surgery patients (9.5%). 78 In a recent comparable study with a population of 90 consecutive JRA patients, early mortality and permanent hemodialysis have been reported as higher in the endovascular arm (chimney and fenestrated repair) versus open repair arm (0% vs 6.4% and 0% vs 6.5%, respectively). 40 In another similar study (50 consecutive JRA patients undergoing fenestrated or chimney repair), early mortality did not differ between groups (8%); however, renal complications including deterioration of renal function (> 50% rise in serum creatinine) and renal infarction were considerably higher in the chimney group (25% vs 13% and 25% vs 8%, respectively). 80 In the latter study, the occurrence of early postoperative high-flow type I/III endoleaks was in favor of the fenestrated option (7.9% vs 25%). 80 Although the potential for this type of endoleak is reduced by creating two chimneys with a 20 mm seal zone length, 7 such a proximal segment endoleak is difficult to repair, and proximal extremity ballooning of the endograft with a compliant balloon or placement of a Palmaz stent may not be sufficient in the long term. 80 The high rate of complications like persistent dialysis and type I or III endoleak were also confirmed in other single-center studies for the chimney procedure (7.1% dialysis and 14% endoleak rate, respectively). 81
Available devices
Fenestrated SGs are usually custom-made and significantly more expensive than standard SGs. Manufacturing that is based on precise plans of the location of the visceral arteries from CT datasets is time-consuming and may take up to 4–8 weeks.
Currently, fenestrated or branched endografts are not commercially available. The Zenith Cook device (Cook Medical Inc, Bloomington, IN) formed the foundation of the fenestrated grafts in nearly all the cases, now with over 15 years of experience. It is composed of woven polyester fabric and Gianturco stainless steel stents. The top of the fabric is attached to a barbed anchor stent, while the stent below this is the high radial force sealing stent. The abdominal fenestrated Zenith SG has a composite body with an individual customized tubular proximal body (which typically contains from one to four fenestrations, depending on patient characteristics, for preservation of flow to both renals, SMA and celiac axis), a distal bifurcated body and iliac limb components. 82 The required length of the SG portion placed above the renal arteries will dictate which vessels must be incorporated in the repair as fenestrations. 83
Proximal SG components have holes in the fabric or scallops at the end of the fabric that are positioned precisely adjacent to the aortic branch artery orifices. Fenestrations are nitinol reinforced and can be constructed of varying sizes. Small fenestrations (6×6 mm, 6×8 mm) are preferably designed for the renal arteries, while larger (8, 10, or 12 mm) strut-free ones are used preferably for the SMA and celiac axis. 51 Scallops used for the celiac artery (or combined with the SMA), represent a U-shaped gap within the proximal fabric of the SG and are provided in 10–40 mm widths and 6–12 mm heights depending on the device manufacturer and design. These fenestrations can be fixed in position over the ostia by deploying CVs (side branches) through the fenestrations and into the arterial branches. CVs are inserted in places where the aorta is aneurysmal, and ordinary stents are inserted in places where the aorta is of normal caliber. 84 Since 2005, more and more bare metal stents have been replaced by CVs. Almost all bridging stents used in recently published reports are balloon-expandable CVs. CVs have enhanced the applicability of endovascular options, such as the use of side branches and fenestrations apart from aortic wall, and the use of SGs without stent-supported proximal components (PCs). Scallops are intended to preserve vessel perfusion with aneurysm exclusion without the use of further bridging devices. In contrast to fenestrated SGs, branched SGs incorporate pre-attached limbs or cuffs targeted for the visceral aortic branches. 27 In fenestrated endografting, the seal occurs between the CVs and the nitinol ring of the reinforced fenestration but the seal in a branched repair is obtained between the outer wall of the CVs and the inner wall of the branch artery. 85
The Zenith system has been commercially available in the European Union (CE-marked) since late 2005 and was approved by the FDA in April 2012. Consequently, an explosive increase in FEVAR may be expected also in the USA. However, several manufacturers are in the process of developing fenestrated devices for JRAs based on their corresponding infrarenal SG platform. Results on the use of the custom-made human implant Anaconda™ (Vascutek, Paisley, Scotland) have been reported recently. 42
As increasingly complex aortic pathology is treated via endovascular means, innovation has been driven through the use of novel techniques and devices customized in consultation with manufacturers. 75 For example, the combination of fenestrations and branches in the same proximal SG component has been used in narrow or angulated reno-visceral aortic segments, 86 or internal/external cuffs (axially oriented or caudally directed) for TAAAs and PRAs. 87 Moreover, to overcome the necessity of aortic main body customization, off-the-shelf devices have become apparent. These are fixed SG configurations, depending on the manufacturer, developed to accommodate in a wide range of aortic anatomies owing to the fact that recurrent patterns of similar arterial morphologies exist. Thus, off-the-shelf devices are applicable in predicted but not all possible anatomies (~54–88% in the literature). Currently, the main reason for their use is to treat patients with symptomatic or ruptured complex aortic aneurysms who are poor candidates for open surgical repair or have limited or no other immediate endovascular options. 88 Off-the-shelf designs are intended to treat patients with JRAs or PRAs, provided that a healthy neck exists below the SMA. Additionally, two other anatomical parameters are fundamental for the high applicability of the device: an adequate proximal neck, which is required for sealing and fixation, and the proper geometric relationship of the visceral vessels in order to provide a means of accommodating a large range of anatomies. Standard off-the-shelf devices developed for the treatment of patients with complex AAAs include the Ventana (Endologix, Irvine, CA, USA) (no longer clinically available) and p-Branch (Cook Inc., Bloomington, IN, USA) systems, which can be used for the treatment of JRAs/PRAs, or for aneurysms that limit just below the SMA, respectively. The p-Branch standardized device is a modular system intended to incorporate the renals with two conical pivot fenestrations and the SMA with one fixed fenestration and maintain patency to the celiac axis with a scallop. Recent reports describe up to 97% and up to 75% of neck and visceral vessel applicability using this SG. 62
Several authors have attempted to modify existing endografts to offer this technique to some patients who have no other options for aneurysm repair. 89 Of interest, 30-day mortality has been quoted as low (2%) in a recent report, suggesting that physician-modified off-the-shelf devices will definitely play a future role in the management of these complex patients. 89 Lack of quality control, inconsistent use of modifications, the potential risk of device contamination, and integrity issues that could originate from structural changes in the device are the most significant drawbacks of this practice. 88
Procedure planning
Planning procedures that extend the proximal sealing zone above the renal arteries are challenging since there is little margin for error. FEVAR work-up consists of clinical examination and high-resolution contrast-enhanced spiral CT scans (slice thickness < 2 mm), which are then reconstructed in a vascular 3D workstation. Custom-made SGs are then designed on multiplanar (axial, sagittal and coronal) reconstructions and centerline of flow calculations. Essential information required includes the relationship of the aneurysm to the aortic branches and the spatial relationships between visceral vessels (including the presence and orientation of supernumerary or aberrant vessels). An appreciation of the angulations and the tortuosity, calcification, diameter and coexisting occlusive disease of the access vessels is also required. Diameters and lengths of the aorta and iliac arteries (sealing zone cross-sectional diameters, total aorta length to be covered by the SG, diameter of the aortic lumen at the level of the target vessels), and visceral vessel morphology length between the ostium of each target vessel, diameter and ‘clock position’ (refers to the circumferential position related to the clock face, recorded to the quarter hour) of the origin of each target vessel are used to measure the relative positions of visceral arteries to match the fenestrations. Small, large or scalloped fenestrations (located at the uppermost portion of the fabric) are incorporated in the device design according to individualized patient anatomy.2,56 The required length of the SG portion placed above renal arteries will dictate which vessels must be incorporated in the repair as fenestrations. 83
Details of the technique
SG implantation technique has been described in more detail previously,25,30,90 but has undergone several modifications over time. In brief (basic steps), open or percutaneous arterial access is required via both femoral arteries. In case of small and calcified external iliac arteries, a retroperitoneal approach and an iliac conduit may be required. The PC is introduced in one side (the delivery sheath of the PC has a diameter of 22–24 Fr and a large (20–24 Fr) sheath is inserted on the contralateral side, each over a stiff wire. The latter is positioned just above the aortic bifurcation and after retrieval of the stiff wire three or four smaller sheaths are introduced through the large sheath valve. Through one of these sheaths, a pig-tail angiography catheter is then advanced into the aneurysm, proximal to the target vessels. 56 In some institutions, to reduce the amount of contrast medium required, branch vessels are pre-cannulated at this point to serve as markers for easier alignment of the device. 4 The PC is then advanced to the pre-planned aneurysm position and oriented with respect to branch arteries prior to unsheathing. To match the intended target vessels, not only is precise longitudinal positioning required, but correct rotational torque control of the delivery system must be applied. 46 When accurate positioning of the unsheathed SG is confirmed using small injections of contrast volume in repeated angiograms, implantation of the PC starts slowly (first two stents are deployed). This slow deployment and the partially constrained PC allows for last-minute repositioning if required, 56 taking into account that this facility to reposition the device is compromised in adverse neck angles. 82 The lumen of the distal end of the PC is then catheterized from the contralateral side and, accordingly, after over-the-wire side branch access sheath insertion (7 or 8 Fr depending on the stent diameter to be used), each target vessel is selectively cannulated through its corresponding fenestration (Figure 2). Successful catheterization is confirmed with a small amount of contrast medium injection.
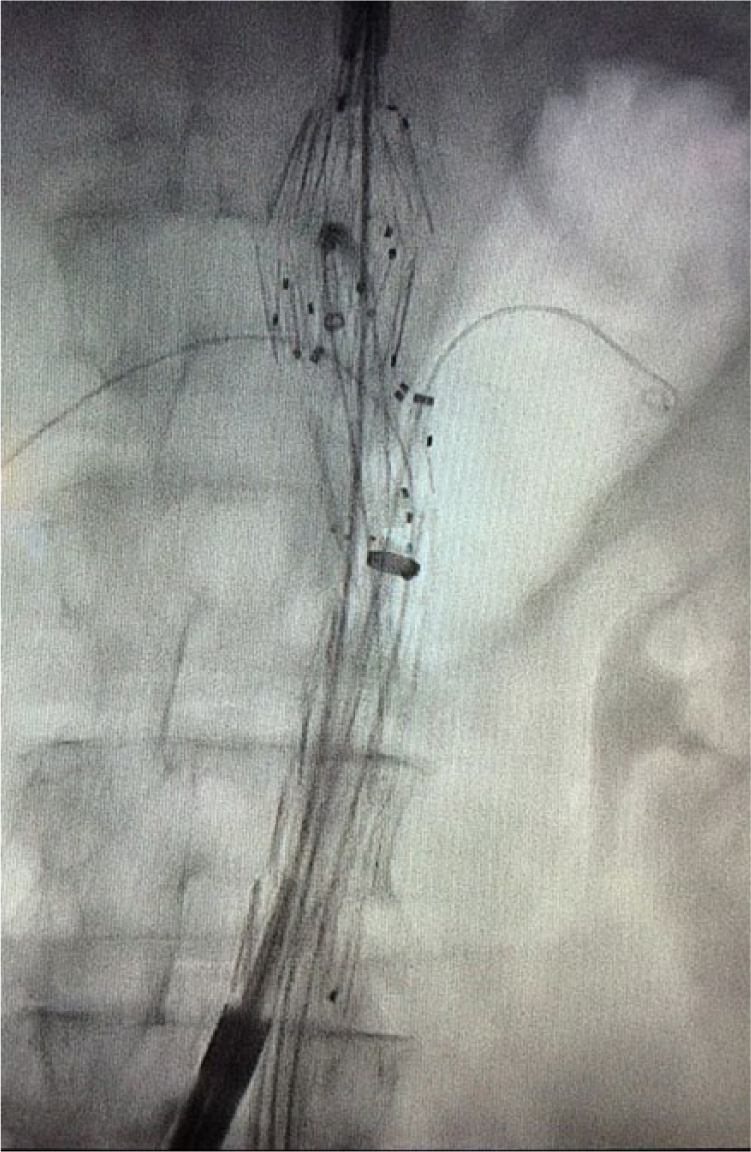
With the proximal component still partially constrained, the superior mesenteric artery (SMA) and renal arteries are cannulated through the dedicated fenestrations (in this patient the stent graft has a scallop fenestration for the celiac axis).
After all access sheaths are sequentially advanced to target arteries over stiff wires, appropriately sized CVs are inserted in places where the aorta is aneurysmal, and ordinary stents are inserted in places where the aorta is of normal caliber (Figure 3). 84
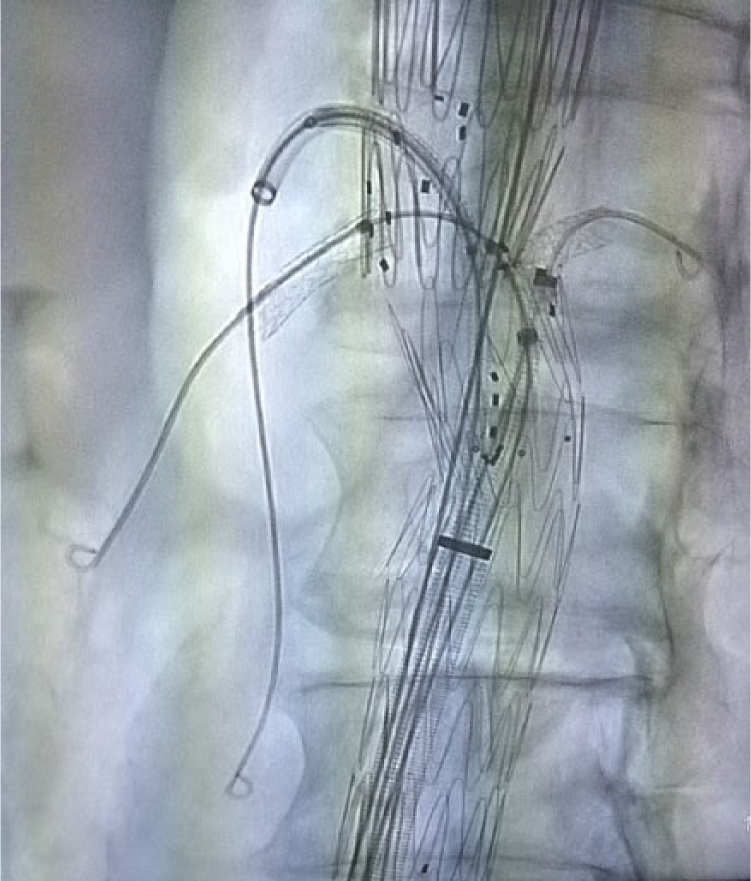
Fluoroscopy image of the proximal component of the stent graft: through a 7 Fr sheath, BeGraft balloon-expandable covered stents are advanced and deployed into the renal arteries. The superior mesenteric artery covered stent is in place but not yet expanded.
When all bridging stents/CVs are in place, the constrained PC is fully deployed. Prior to CVs deployment, the PC is dilated with a large compliant balloon. Using C-arm tube projections perpendicular to the fenestrations, the CVs are then deployed to their profile and flared proximally (the segment protruding into the SG lumen beyond the fenestration, usually 3–5 mm) with a balloon (10–12 mm); all maneuvers were performed after partial access sheath withdrawal (Figure 4). The procedure ends with cannulation of the PC gate permitting its connection with the bifurcated and other distal aortic components as planned (iliac extensions and/or iliac side branch devices) (Figure 5). A final angiogram confirms the absence of endoleak and patency of all SG components (Figure 6). The last steps are similar to the standard EVAR technique. Large sheaths in both femoral arteries must be removed as quickly as possible in order to avoid the occurrence of leg ischemia.
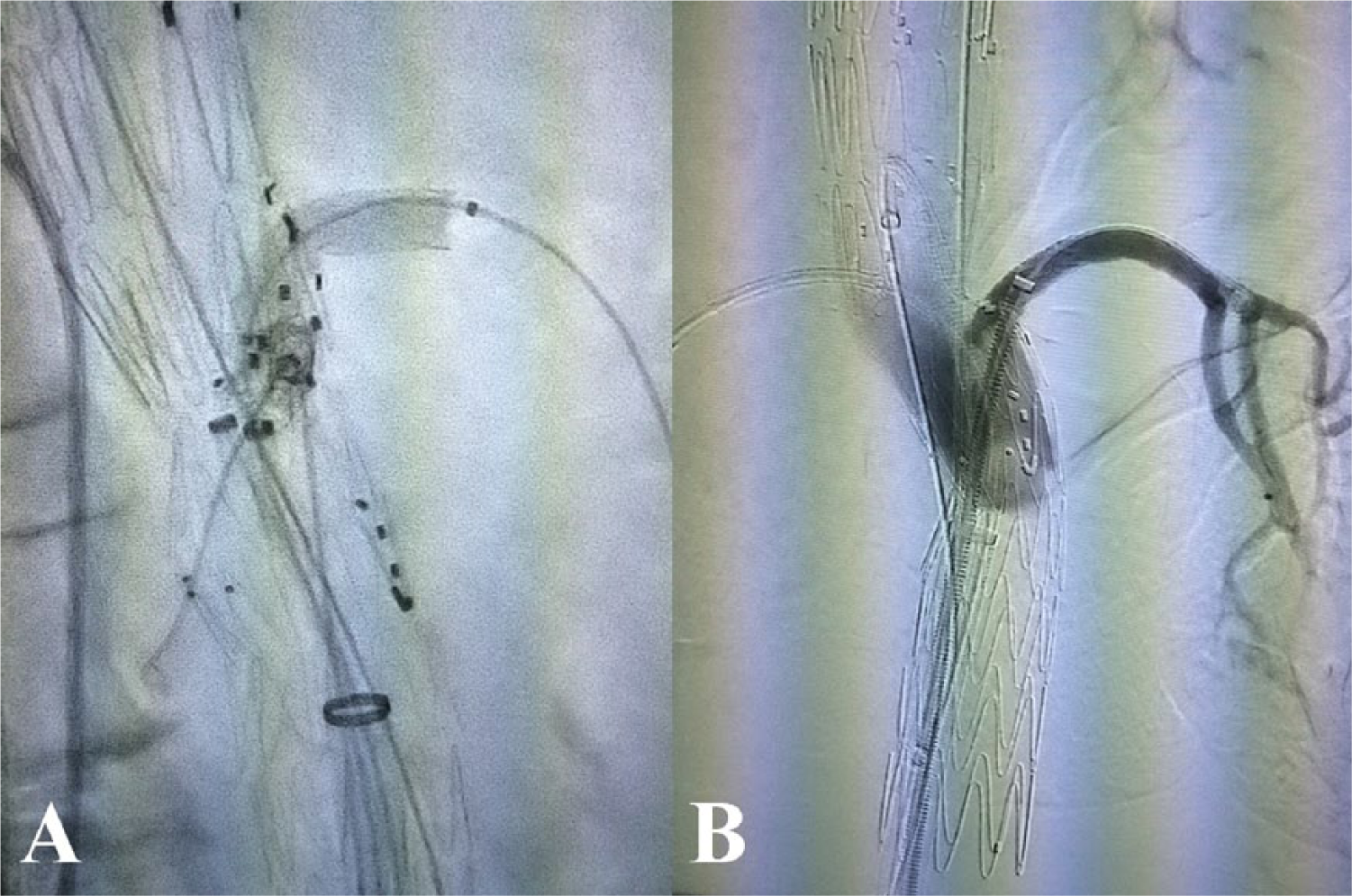
Flaring of the aortic portion of the left renal artery bridging covered stent is performed using the tip of a large (10–12 mm diameter) balloon catheter (A). Selective angiography of the left renal artery after covered stent placement showing good patency of the target vessel and no signs of type I or type III endoleak at this level (B).
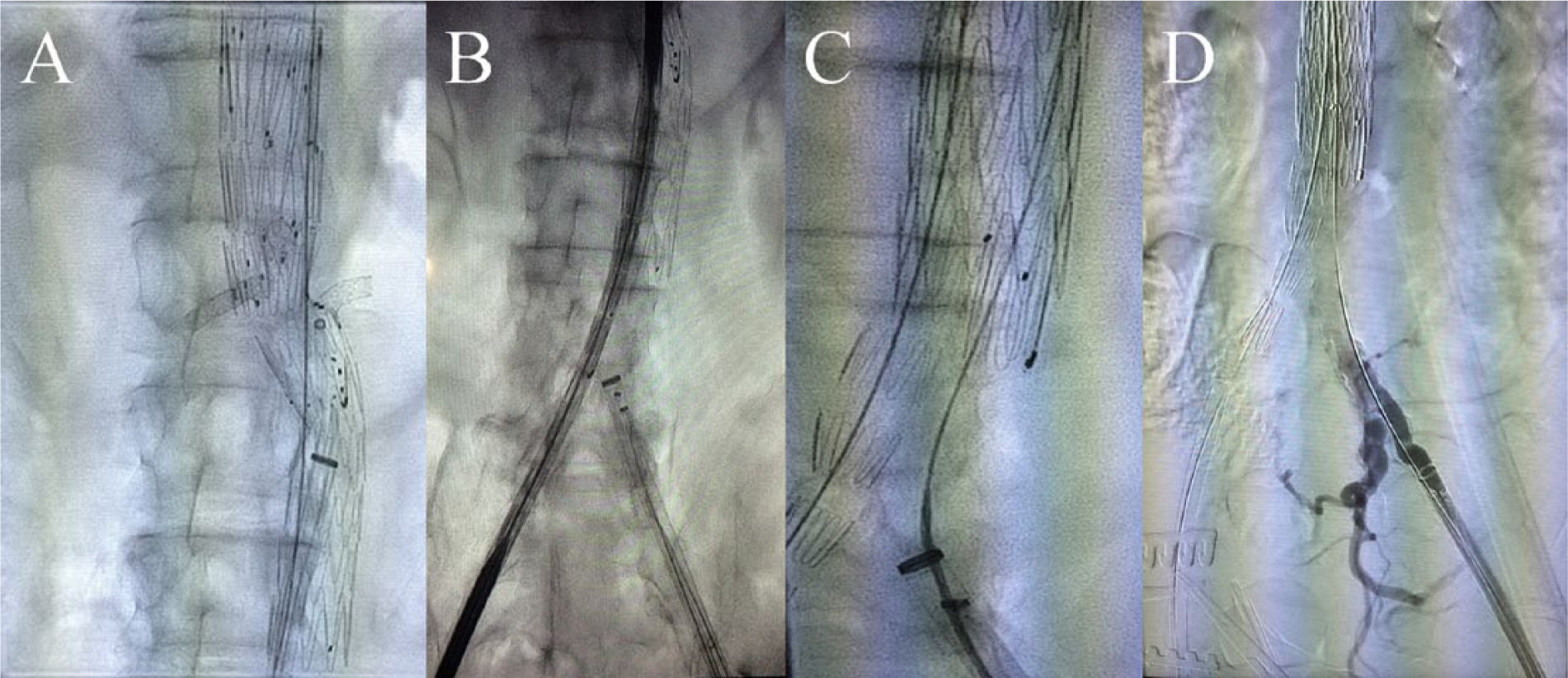
After all the sheaths and covered stents are in the correct place, the proximal component is fully deployed (A). Insertion of the bifurcated distal component of the stent graft from the right side (B), which is then deployed after adjustment for correct overlapping with the proximal component (C). Cannulation of the contralateral limb (C) and retrograde angiogram through the left-sided sheath showing the iliac bifurcation (D) for the correct length measurement and placement of the left iliac limb.
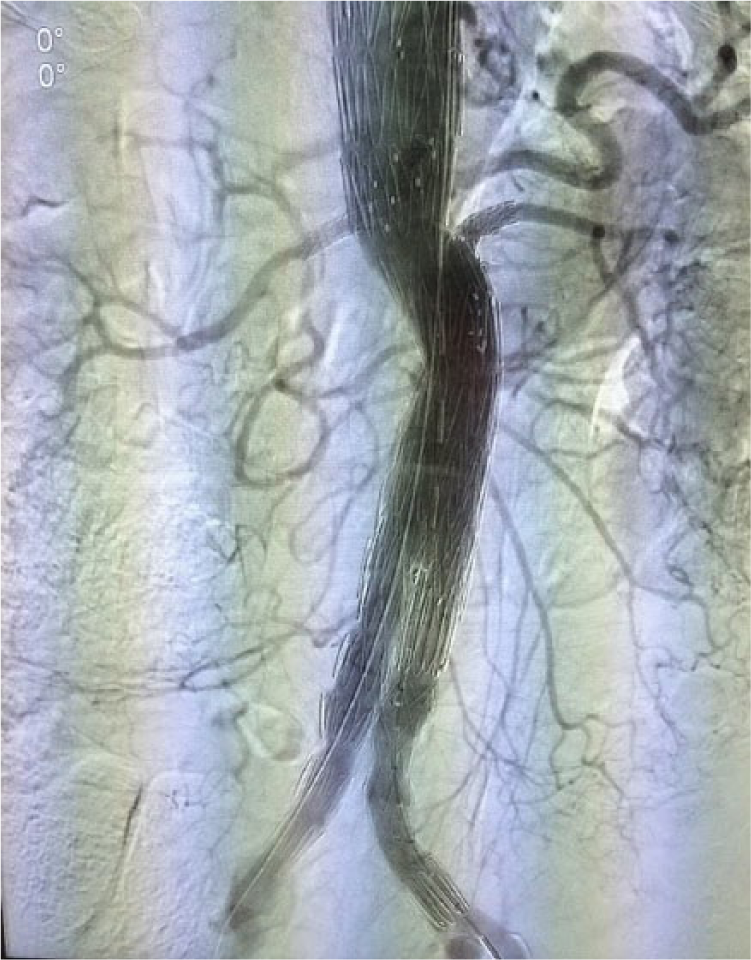
Completion angiogram after the fenestrated repair (fenestrations for the renal arteries and the SMA; scallop fenestration for the celiac axis). All visceral and renal vessels are nicely patent and no endoleak is detected.
Outcome measures
Perioperative and late mortality
Despite the theoretical advantages of FEVAR over open surgery, like the minimal invasive technique and avoidance of laparotomy and suprarenal aortic cross-clamping, investigations revealed comparable perioperative mortality rates9,20 (with some studies reporting higher mortality with open repair).12,91 In specialized centers, FEVAR was associated with even lower 30-day mortality rates (1% to 2.6%).22,25,26,30,31 This compares favorably with that of open surgical repair of short-necked aneurysms, which has operative mortality rates of 2.5% to 5.8%.92,93 Notably, in a recent meta-analysis, the mortality rate in open repair and FEVAR studies (21 and 14 studies, respectively) were exemplary and equivalent (4.1%), although a higher rate of postoperative major complications and re-interventions due to bleeding and embolic events were observed with open repair. 9 Noteworthy, greater rates of postoperative complications such as renal impairment (18.5% vs 9.8%), new onset of dialysis (3.9% vs 1.5%) and cardiac complications (11.3% vs 3.7%) were reported for open repair. 20 In a large UK registry not including less experienced centers, perioperative mortality was marginally higher (4.1%) than that reported in single-center or single-operator studies, suggesting that patient comorbidities should be taken into account. 10
Despite the fear of target vessel loss, ischemic consequences of end-organs appear to be more frequent with open repair in the early postoperative period, as shown by instances of mesenteric ischemia necessitating bowel resection and acute renal failure requiring permanent dialysis. 91 In a recent large national database, 94 FEVAR had comparable mortality to EVAR, but FEVAR was associated with an increased risk of overall complications attributed to the higher morphological complexity of an operation utilizing, for example, visceral vessel cannulation and renal stenting with attendant risks of embolization and thrombosis, lasts longer, and requires a learning curve with new technology. Pooled estimated perioperative mortality for patients undergoing fenestrated repair is 2.3%, 31 which is lower than that reported for open or endovascular repair of standard infrarenal aneurysms.
Deaths because of technical errors or intraoperative complications, fortunately at the early phases of the learning curve, are not uncommon.27,34,95 An increased duration of surgery (> 260 minutes) and postoperative events (postoperative complications, the need for dialysis and redo procedures) are strongly associated with a greater risk of 30-day and in-hospital death. 39 Mortality has also been correlated with adverse renal events, as shown in several studies.34,96 Patients experiencing spinal cord ischemia, for example, have a 10 times greater risk of death and notably the majority of those not fully recovered die rapidly. 39
There is also evidence that outcomes are strongly correlated to the level and to the proximal extension of aneurysm disease. In a multicenter study, in-hospital mortality, combined (30-day, in-hospital) mortality and/or severe complications were strongly associated (more than double risk of death) with the aneurysm disease extension (JRAS and PRAs vs SRAs and all types of TAAAs). 39 Besides the prognostication in endovascular repair of TAAAs depending in the classical Crawford classification, there is also a trend for poorer long-term outcomes for patients treated with a device that requires coverage above the celiac artery. 31 Also, the extent of the aneurysm repair, as determined by the proximal sealing zone of the SG, is also associated with outcome. As the proximal zone of SG coverage ascends, mortality significantly increases. 97
Worse 5-year survival rates were seen with FEVAR compared to open repair (55% vs 73%) in a large meta-analysis. As stated, selection bias, higher-risks patients offered FEVAR and significantly higher secondary re-interventions rates of FEVAR might explain the decreased long-term survival. 9 Data emerged from a 12-year experience center reported decreased, survival (20%) after a mean follow-up of 8 years. This survival was negatively associated with increased age, congestive heart failure, cancer and previous aneurysm repair. Only 2% of these deaths were aortic aneurysm-related. 31 In a similar study, overall survival was 76% at 3 years and 60% at 5 years. During the 10-year study period, 54% of the patients died (9% of aneurysm related causes), an overall mortality rate which is in line with that observed in patients with peripheral vascular disease, indicating severe comorbidities in this patient group. 95
Target vessel stenosis/occlusion
Target vessel occlusion still represents one of the most main complications of FEVAR. 85 Series from referral centers have included between 45 and 607 patients, with 117–1907 target vessels.5,10,29,32–34,39,80,97 Up to July 2012, a total of 2463 target vessels have been accessed through fenestrations and/or scallops, all within the use of the fenestrated Zenith SG. 20 Overall, early target vessel patency rate ranges from 90.5% to 99.7%. 33 The published 1-year target vessel patency rates ranges from 90.5% to 97%. 12 Recently pooled estimates of vessel patency at completion angiogram and in the long-term (5 years) during follow-up were reported 98.2% and 95.8%, respectively. 9 Verhoeven et al., reported a cumulative visceral target vessel patency (275 target vessels in 100 patients) of 93.3% at 5 years. 22 However, stent patency of target vessels must be differentiated from target vessel patency and was reported 95.7% at 1 year and 88.6% at 4 years. 34
In-stent-visceral stenosis/occlusion can be catastrophic with serious clinical sequelae (death, lifelong dialysis, bowel ischemia). Stenting of a fenestration is mandatory to avoid early visceral artery occlusion. Scallops are generally not stented. 22 Cannulating target vessels followed by tracking sheaths and CVs can be challenging when doing a FEVAR. Unsuccessful catheterization/stenting and visceral vessel loose is reported rather low despite the more complexity of FEVAR compared to standard EVAR. Important factors contributing to technical failure include the inadequate device design and suboptimal procedure planning. 86 Intraoperative technical failure was reported only 1.1% and 2.1% by Verhoeven et al. 22 and by Mastracci et al., 31 suggesting that expertise is of paramount importance.
Renal issues
One concern regarding FEVAR is the potential of negative effect on renal function. Postoperative renal impairment was not rare in a multicenter trial (18%) 39 and in a literature review (22.2%), 17 and was mostly observed in patients with PAOD, long-lasting operations and complex cases. 39 Moreover, the risk for adverse renal events was 16% in patients without preoperative renal dysfunction and 39% in patients with preoperative renal dysfunction. 96 In other studies, transient as well as permanent post-procedural renal impairment was not influenced by preoperative renal status. 34 If the high prevalence of renal insufficiency of the patient population prior to treatment of aneurysms that involve or abut the renal arteries is a stronger contributing factor, remains to be determined. Notably, Rao et al., reported that preoperative renal dysfunction is rather common in patients with JRAs scheduled for FEVAR (6–44% in all 13 included studies, > 10% in 8/13 studies). 9 High incidence of renal insufficiency has been reported in patients with AAAs (up to 25%); however, no correlation with greater risk of failure of primary EVAR has been found. In contrast, an increased tendency for FEVAR failure in those patients with renal insufficiency has been reported. 46
The primary reason for renal deterioration or transient/permanent dialysis is renal artery thrombosis after failed catheterization of this target vessel. 33 The renal artery as a target vessel was strongly correlated with perioperative vessel related events (technical failure, vessel injuries and early occlusions). 86 Renal insults not requiring hemodialysis, such as iatrogenic arteriovenous fistula, perinephric hemorrhages and small infarcts to the renal pole, are not uncommon.21,82,98 Postoperative evolution of estimated glomerular filtration rate (eGFR) has been reported in a few studies. In the French multicenter trial (n = 134 patients), renal function showed a significant improvement or remained stable in 90% of patients and hemodialysis, either temporary or permanent, was required in 4.5%. 33
Renal dysfunction is prevalent also after open repair techniques. Long-term survival after open repair of infrarenal AAAs decreases from 51% to only 12% at 10 years in the setting of the presence or absence of post-procedural renal dysfunction. 96 Short-term mean endovascular postoperative eGFR and serum creatinine values were presented significantly lower compared to those obtained after open surgical repair. 40 The avoidance of suprarenal clamping and ischemia possibly explains the excellent results of the endovascular arm (FEVAR and chimney EVAR) in comparable studies with open surgical repair (0 vs 6.5% requirement of persistent dialysis). 40 In contrast to these results, a recent well-conducted meta-analysis reported similar perioperative mortality rates (30-day and in-hospital) between open and endovascular repair of juxtarenal aneurysms (4.1%). Similarly, pooled proportions for postoperative renal insufficiency and permanent dialysis were not statistically different (13.9% vs 11.4% and 2.8% vs 1.9%, respectively). However, secondary interventions were more and long-term survival was lower (at a statistically significant level) in FEVAR compared to open repair (pooled event rate 12.7% vs 4.9% and survival rate 55% vs 73%). Furthermore, more FEVAR patients developed renal impairment during follow-up than open repair patients (19.7% vs 7.7%, p < 0.0001), suggesting that apart from possible higher preoperative comorbidities and heterogenous reporting of renal outcomes, repeat interventions and contrast nephropathy from utilization of CT angiography surveillance at short intervals might play a role in this progressive renal damage. 9
Type I and type III endoleaks
Like standard EVAR, successful aneurysm exclusion should follow any fenestrated endograft implanted in the perirenal position without any type I or III endoleaks (failure of graft/aorta or branch stent/branch artery wall interface (e.g. branch stent dislocation from poor apposition of branch walls) or failure of device–device interface, respectively (either from PC/branch stent, branch stent/branch stent, PC/bifurcated device or bifurcated device/limb extension component separation or from branch stent/branch stent fracture)) at completion angiogram. Failure to obtain an adequate seal between the various SG modular components and the arterial wall may lead to continuous pressurization of the aneurysm sac and subsequent rupture. Sealing zone failure in EVAR has been related to morphologic factors such as neck angulation, short neck, conical geometry, excessive calcium burden, 66 and non-adherence to device instructions for repairs. 68 However, within FEVAR, the physician has the opportunity to customize more proximal and longer sealing zones, thus removing the anatomical barrier of the visceral aorta. 99 Although more complex, higher than juxtarenal repairs to achieve healthier sealing zones correspond to lower endoleak rates, 99 despite the greater risk of spinal cord ischemia with increasing graft coverage above the celiac artery, and the greater risk of celiac occlusion with supraceliac landing zones. 31 Parallel-walled aortas are more protective against proximal endoleaks than non-cylindrical shaped aortas. 99
Proximal endoleaks detected at the operative room can be corrected with ancillary procedures, usually with the insertion of a Palmaz stent. However, their occurrence is considered a bad prognostic sign for the durability of the repair, as many of them, regardless of the immediate success, usually recur during follow-up. 99
The rates of early types I and III endoleak were 5.8% and 2.6%, respectively, and the rates of late endoleaks were 2.2% and 3.2% in the latest meta-analysis on FEVAR versus open repair. 9 Overall, and in a recent large data (607 patients receiving four design devices), the rates of types I, II and III endoleak through the entire follow-up period (mean = 8 years) were 3%, 16% and 4.6%, respectively. Notably, a higher incidence of type I endoleak was observed in patients receiving device configurations with only renal fenestrations, suggesting that sealing in the mid-visceral aorta, with a shorter length of aorta coverage, jeopardizes the stability of the proximal SG. 31 A high percentage of endoleaks eventually require redo operations, which puts into question the longevity of the repair.28,31
Regular CT examinations are required to adjudicate the absence of endoleaks. Not all patients demonstrate shrinkage of the aneurysm sac during follow-up. Aneurysm diameter changes after FEVAR are considered significant when ⩾ 5 mm. Aneurysm diameter decreased ⩾ 5 mm in 39% of patients at 12 months, 64% at 36 months and 71% at 60 months in a recent study with a mean follow-up of 67 months. 95
Endograft migration
Migration in infrarenal EVAR has been classified as any movement > 10 mm or that caused symptoms or required re-intervention. There is little detailed information regarding migration in fenestrated SGs in the literature. Early identification of any migration is crucial. This early recognition of device instability gives the physician the opportunity to intervene early and avert any serious clinical sequelae. 100 The main concerns are blood flow compromise to target vessels and loss of an aortic seal leading to aneurysm reperfusion. Noteworthy, only those cases complicated by serious clinical events or requiring re-intervention are reported in the literature.
Migration rates between 1% and 7% have been reported,25,29 or higher in other studies, all requiring re-interventions. 85 Previous evaluation confirmed that insufficient landing zone or imprecise planning of the procedure could explain most of the adverse events, including migrations/type I endoleak with or without visceral stent fractures. 85 England et al. 100 pointed out that protective contributing factors to a low incidence of migration rates in FEVAR include barbs, higher fixation of SG fabric, bare proximal stent struts and a separate proximal component. Technical (proximal SG oversizing, length of common iliac artery landing zone) and other acquired anatomical issues (proximal neck enlargement, angulation) may predict SG migration. As such, cranial progression of aortic disease with further dilation of the aortic neck in the fenestrated segment results in loss of fixation and possible migration. 32
Migration can also occur at any time between the two components of the SG. Component separation is defined as any movement between the proximal fenestrated component and the distal bifurcated part. Iliac limb migration has a very low incidence. 100
In another series, migration has been defined as cranial or caudal movement of the device relative to a vascular landmark of ⩾ 4 mm. 100 This does not mean that subtle levels of migration preclude clinical events. Target vessel stent crushing and subsequent occlusion have been reported even with small (< 5 mm) movements. 32 Applying the above definition, 14% of a patient cohort (n = 55, mean fenestration ratio = 2.02) receiving fenestrated SGs had an estimated probability of proximal migration at 12 months; by 36 months, this had increased to 22% (mean: 8% per year). Iliac limb migrations were less frequent with 12- and 36-month probabilities of 3% and 8%, respectively. 100
Re-interventions
Re-interventions are usually required for endoleak, target vessel event (stenosis/occlusion) and graft limb complications. Kristmundsson et al., among 31 re-interventions performed in 20 (37%) patients undergoing FEVAR, reported that 29% of re-interventions were endoleak related, 26% target-vessel related and 13% graft-limb related. Most of them were based on complications detected within the regular follow-up. 95
Troisi et al. documented after a mean follow-up of 25 months that the incidence of early and late complications requiring a secondary procedure after utilization of fenestrated and branched devices is not negligible. Overall, 34 repeat procedures were performed in 28 patients (26.2%) but the majority of them were carried out within 1 year postoperatively (mean follow-up 2 years). 85 Re-interventions were performed primarily for type III endoleaks. Compared to open surgery, FEVAR requires more re-interventions in the long-term (12.7% vs 4.9%, p < 0.0001). 12 Most of the re-interventions were for bleeding during surgery (⩽ 30 days) or for endoleak or vessel occlusion/stenosis during follow-up (most occurred between 6 and 12 months) after FEVAR. Noteworthy, some FEVAR redo procedures, including SG components destruction, endoleaks, graft kinking and renal artery occlusion, led to worse mortality and morbidity compared to open repair re-interventions. 9
In the largest fenestrated report (607 patients), long-term survival in patients classified as having JRAs or type IV TAAAs has been reported 20% after a mean of 8 years of follow-up. During that period, only 2% of deaths were aortic related. Moreover, patients treated with a device requiring coverage above the celiac artery (supraceliac landing zone) experience a greater branch re-intervention rate compared to patients treated with other configurations. 31 In other studies, freedom from secondary interventions was 89% 58 and 63% 32 at 5 years, respectively, after fenestrated and branched endografting.
Given the high rate of re-interventions required after endovascular repair of complex aortic aneurysms and the need for aggressive follow-up in the long-term, it is alarming to see that FEVAR is applied to a similar cohort of patients as open repair.
All basic outcome measures are summarized in Table 3.
Early (< 30 days) results of the use of fenestrated/branched technology.
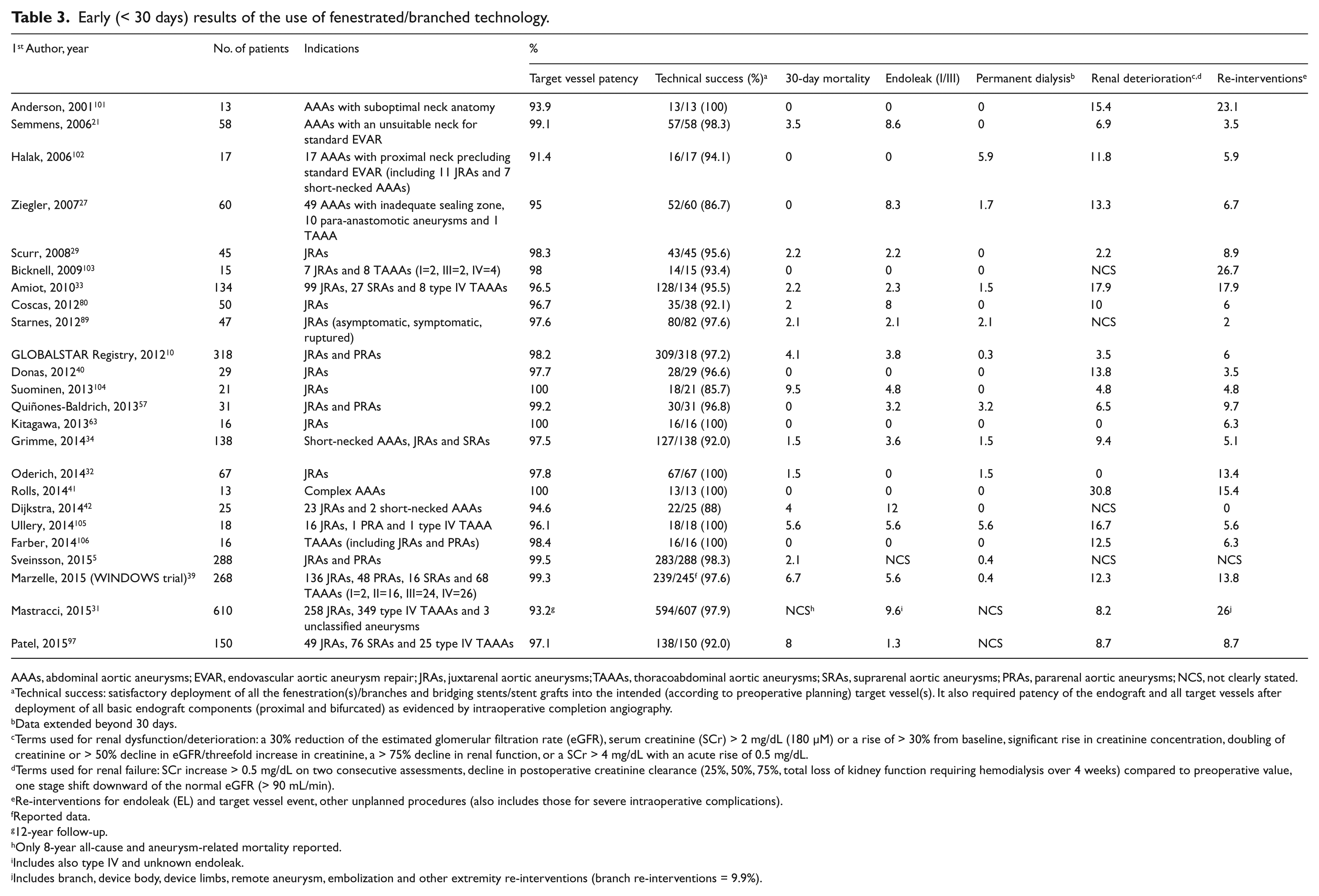
AAAs, abdominal aortic aneurysms; EVAR, endovascular aortic aneurysm repair; JRAs, juxtarenal aortic aneurysms; TAAAs, thoracoabdominal aortic aneurysms; SRAs, suprarenal aortic aneurysms; PRAs, pararenal aortic aneurysms; NCS, not clearly stated.
Technical success: satisfactory deployment of all the fenestration(s)/branches and bridging stents/stent grafts into the intended (according to preoperative planning) target vessel(s). It also required patency of the endograft and all target vessels after deployment of all basic endograft components (proximal and bifurcated) as evidenced by intraoperative completion angiography.
Data extended beyond 30 days.
Terms used for renal dysfunction/deterioration: a 30% reduction of the estimated glomerular filtration rate (eGFR), serum creatinine (SCr) > 2 mg/dL (180 µM) or a rise of > 30% from baseline, significant rise in creatinine concentration, doubling of creatinine or > 50% decline in eGFR/threefold increase in creatinine, a > 75% decline in renal function, or a SCr > 4 mg/dL with an acute rise of 0.5 mg/dL.
Terms used for renal failure: SCr increase > 0.5 mg/dL on two consecutive assessments, decline in postoperative creatinine clearance (25%, 50%, 75%, total loss of kidney function requiring hemodialysis over 4 weeks) compared to preoperative value, one stage shift downward of the normal eGFR (> 90 mL/min).
Re-interventions for endoleak (EL) and target vessel event, other unplanned procedures (also includes those for severe intraoperative complications).
Reported data.
12-year follow-up.
Only 8-year all-cause and aneurysm-related mortality reported.
Includes also type IV and unknown endoleak.
Includes branch, device body, device limbs, remote aneurysm, embolization and other extremity re-interventions (branch re-interventions = 9.9%).
Conclusions
The evolution of fenestrated and branched EVAR has opened up the visceral aorta to totally endovascular repair. The use of fenestrated SGs for complex aneurysm disease has grown enormously the last years and has been proved safe and effective in the short- and mid-term follow-up. Stent grafts with fenestrations, scallops and occasionally branches have to be customized to each patient’s anatomy and precisely deployed in vivo. Bridging covered stents between the main graft and the target vessels eventually exclude the aneurysm preserving blood flow to vital organs. Outcomes depend mostly on the level of expertise and organizing facilities, correct planning but also on proximal extension of the disease which increases also the complexity of the repair. Remaining issues are within the range of those reported for EVAR.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
