Abstract
Objective:
The objective of the study is to describe a novel bifurcated in-stent in situ technique (BIS2T) for creation of a bifurcated bridging stent-graft system to target 2 vessels using a single directional branch during complex endovascular aortic aneurysm.
Technique:
The technique uses 3 balloon-expandable stent-grafts. The first supporting stent-graft is deployed from the directional branch into the first target vessel or aneurysm sac. A reentry catheter is used to create an in situ fenestration in the first stent-graft, which is predilatated with a 3 mm 0.014 inch angioplasty balloon. This is exchanged for a 0.035 inch 5 Fr catheter and guidewire which is used for catheterization of the second target vessel via the in situ fenestration. The docking area of the first stent-graft is predilated. The second stent-graft is deployed into the second target with >1 cm overlap into the first stent-graft, followed by a matching third stent-graft in a kissing stent fashion to reinforce the bifurcation and achieve a “D shape” stent-graft configuration.
Results:
The BIS2T was used to treat 2 patients. Neither experienced complications or endoleaks, and both had patent branches on follow-up computed tomography angiography.
Conclusions:
The BIS2T is a feasible option to incorporate 2 target vessels via a single directional branch.
Clinical Impact
Target vessel anatomy is a frequent limitation for successful endovascular incorporation using fenestrations or directional branches during complex endovascular aortic aneurysm repair. Numerous anatomical variations such as aberrant or accessory vessels, common celio-mesenteric trunks, or extension of the aneurysm/dissection into a target vessel may preclude successful incorporation using standard endovascular techniques. This technical note describes a novel bifurcated in-stent in situ technique (BIS2T) for the creation of a bifurcated bridging stent-graft system to target 2 vessels using a single directional branch. This approach may enable target vessel incorporation in challenging anatomical situations that might otherwise preclude endovascular treatment or require intentional artery coverage.
Keywords
Introduction
Endovascular aortic aneurysm repair (EVAR) has been increasingly utilized to treat patients with complex aortic aneurysms using fenestrations, directional branches or parallel grafts. 1 These techniques have expanded the indications of EVAR to include patients with aortic arch, thoracoabdominal aortic aneurysms (TAAA) and aortoiliac artery aneurysms.1 –3 An anatomical requirement is the presence of suitable target vessels with adequate diameter and length, free of dissection and early vessel bifurcation. 4 Numerous anatomical variations such as aberrant or accessory vessels, common celio-mesenteric trunks or extension of the aneurysm/ dissection into a target vessel (eg, internal iliac artery, innominate artery) may preclude successful incorporation using standard endovascular techniques and off-the-shelf stent-grafts.4 –8 In these patients, sacrificing one of the target vessels while preserving the largest and most suitable vessel is a frequent strategy. 5
This article describes a novel bifurcated in-stent in situ technique (BIS2T) to create a bifurcated bridging stent-graft system to target 2 vessels via a single directional branch. The BIS2T was used in 2 patients to incorporate a total of 6 target vessels during endovascular repair of TAAA using an off-the-shelf multibranch stent-graft and bilateral internal iliac artery aneurysms (IIAAs) using iliac branch devices.
Technique
This technical note was reported in accordance with the CARE (CAse REport) guidelines. 9 It was approved by the Institutional Review Board, and all participants provided written informed consent prior to participating.
The BIS2T is applicable to patients who need incorporation of 2 target vessels via a single directional branch and in whom an off-the-shelf device or a patient-specific stent-graft that addresses the specific anatomical variation is not available. This technique represents an off-label modification of an approved bridging stent-graft. Potential indications include the following clinical scenarios (Figure 1):
a. Variations or the normal 4-vessel renal and mesenteric anatomy in patients with complex abdominal aortic aneurysms (CAAAs) and TAAAs who present with large accessory renal arteries, accessory and/or aberrant hepatic arteries, early branch bifurcations or common celio-mesenteric trunks.
b. Large IIAAs with unsuitable sealing zones in the main trunk of the internal iliac artery (IIA) requiring extension of the bridging stent-graft into either the posterior and anterior divisional branches using iliac branch devices or parallel grafts.
c. Common iliac artery aneurysms with a short or unsuitable landing zone in the main trunk of the IIA.
d. Aortic arch aneurysms requiring repair into zone 0 with unsuitable innominate artery anatomy due to aneurysm involvement or dissection and requiring placement of bridging stent-graft into the right common carotid artery.
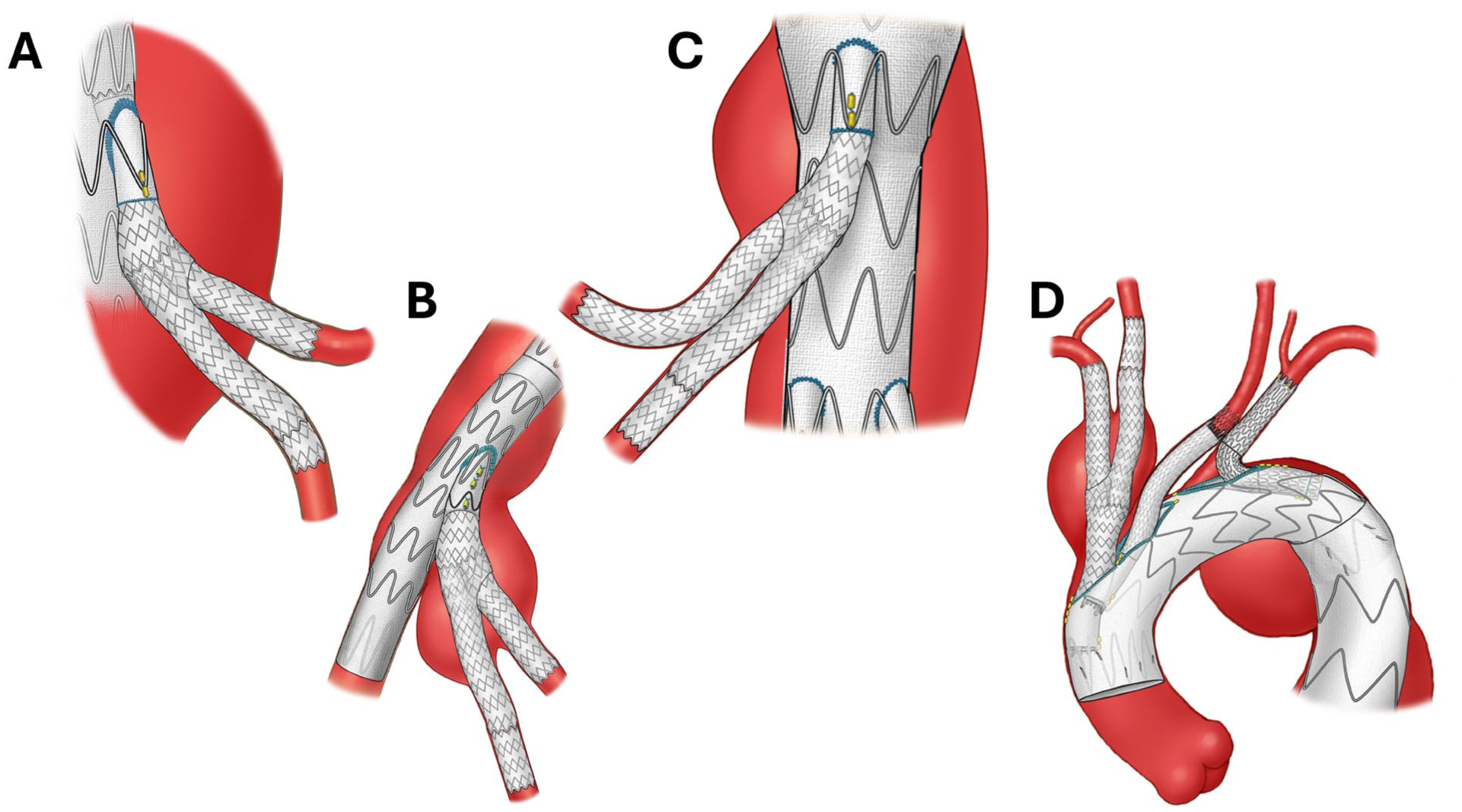
Artistic illustration of the applications of the bifurcated in-stent in situ technique (BIS2T) in addressing challenging anatomical variations to incorporate 2 vessels via a single directional branch.
Construction of the BIS2T requires co-axial access via a single directional branch using 1 sheath with “buddy system” or 2 separate sheaths. The directional branch is usually deployed well above the intended target vessels to allow construction of the bifurcated stent-graft system using balloon-expandable stent-grafts with double PTFE (polytetrafluoroethylene) layer and open-cell stent structure. The use of the Gore VBX stent-graft is recommended due to its doubled PTFE layer and sandwiched open-cell stent structure, although the procedure may be performed using other bridging stent-grafts with similar characteristics. A minimum of 3 balloon-expandable stent-grafts is needed depending on the length between the directional branch and target vessels.
The first stent-graft is deployed from the directional branch to the first intended target vessel or into the aneurysm sac with its distal end above the target vessels (Figure 2). It is critical that the first stent-graft be well attached to the directional branch to prevent disconnection, which may require postdilatation and flaring. Use of an 8Large VBX stent-graft (WL Gore, Flagstaff, Arizona) for the first stent-graft offers the potential advantage of postdilatation in the BIS2T segment up to 16 mm. If this stent-graft is too oversized to the first intended target vessel a distal taper may be created using a smaller diameter stent-graft. Anchoring the first stent-graft into the directional branch and target offers added stability.
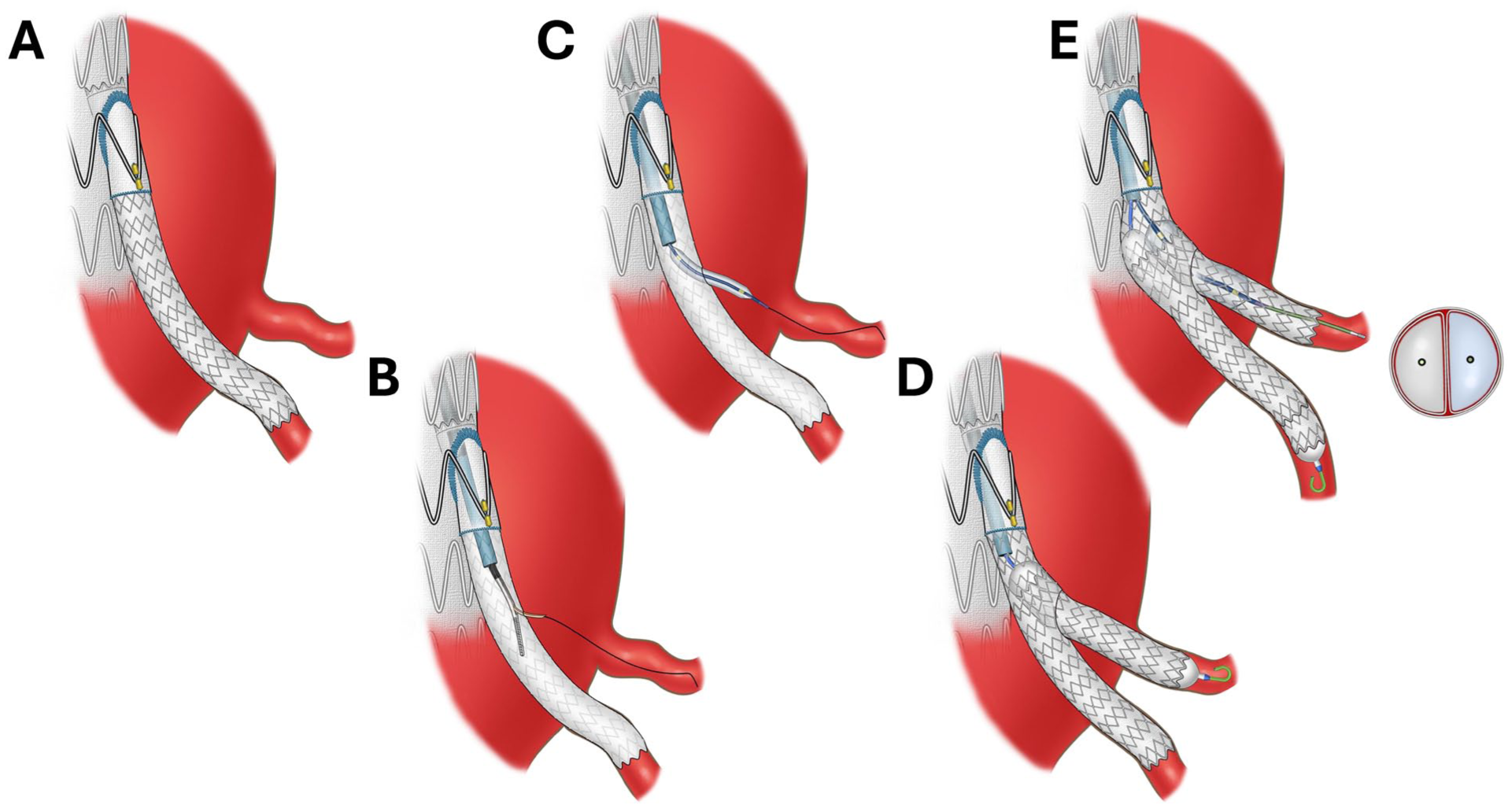
Artistic illustration depicting the steps of the bifurcated in-stent in situ technique (BIS2T).
To guide in situ fenestration, preoperative assessment of the target vessels’ clock position (ie, their circumferential orientation relative to the aortic wall) is used to facilitate vector alignment between the reentry catheter and the target vessel under fluoroscopic guidance. The in-stent in situ fenestration is created at least 2 cm distal to the directional branch using Outback Elite reentry catheter (Cordis, Santa Clara, California) advanced over a 0.014 inch guidewire. The reentry catheter is exchanged over the 0.014 inch guidewire for a 3 mm angioplasty balloon, which is used to predilate the in situ fenestration. The balloon is exchanged for a 4 to 5 Fr catheter and a 0.035 inch guidewire, which are used for selective catheterization of the second target vessel. Prior to the deployment of the second bridging stent-graft the overlapping area of the BIS2T may be predilated with a 10×20 mm angioplasty balloon which will facilitate advancement of a buddy wire during the kissing stent placement.
The second target vessel is stented using a balloon-expandable stent-graft and the guidewire is exchanged for a 0.018 guidewire and 5 to 6 mm angioplasty balloon, which is positioned in the BIS2T overlapping area.
Finally, the third balloon-expandable stent-graft is positioned for deployment in the overlapping BIS2T area and deployed using kissing balloon technique to reinforce the bifurcation zone. The overlapping bifurcated segment should be postdilated using kissing balloon technique to achieve a “D-shaped” or “eye-of-the-tiger” configuration as described by Minion. 10 This may be confirmed by noncontrast cone-beam computed tomography (CBCT) prior to removal of both balloons until the ideal shape is obtained. A final selective completion angiography of the bifurcated stent-graft system is performed. Although the technique may be performed using 2 instead of 3 stents, this is not recommended due to potential risks of dislodgement, movement, disconnection, and luminal compromise in the BIS2T overlapping area.
An in-vitro demonstration highlighting the key steps of the BIS2T along with imaging findings illustrating the double “D-shape” conformation of the bifurcated stent-graft system are presented in Figures 3 to 5.

Phantom model setup for technical evaluation of the BIS2T procedure. The setup consists of a perforated plastic plate, serving as the anchoring structure. Attached to it are plastic hoses of varying diameters: a 30 mm diameter plastic hose cranially, representing the distal thoracic aorta into which the main body of the branched stent-graft device is deployed. An 18 mm hose distally secures the stent-graft in place, while two 6 mm plastic hoses are closely positioned and fixed laterally to simulate the target vessels. This phantom model is used for the technical assessment of the procedure, ensuring proper deployment and stability of the stent-graft system in different vessel sizes.
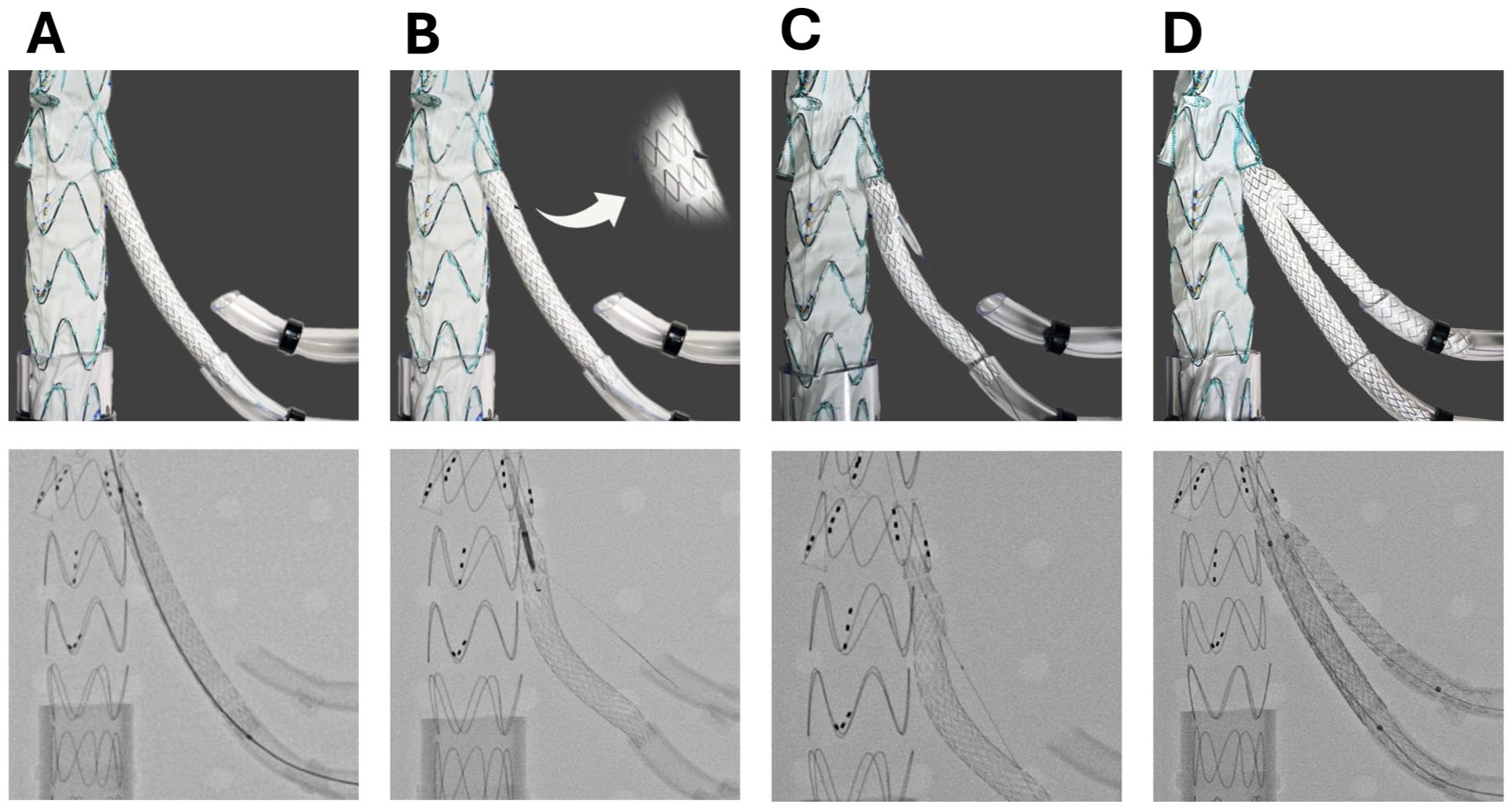
Demonstration of the BIS2T technique using a phantom model under direct visualization (
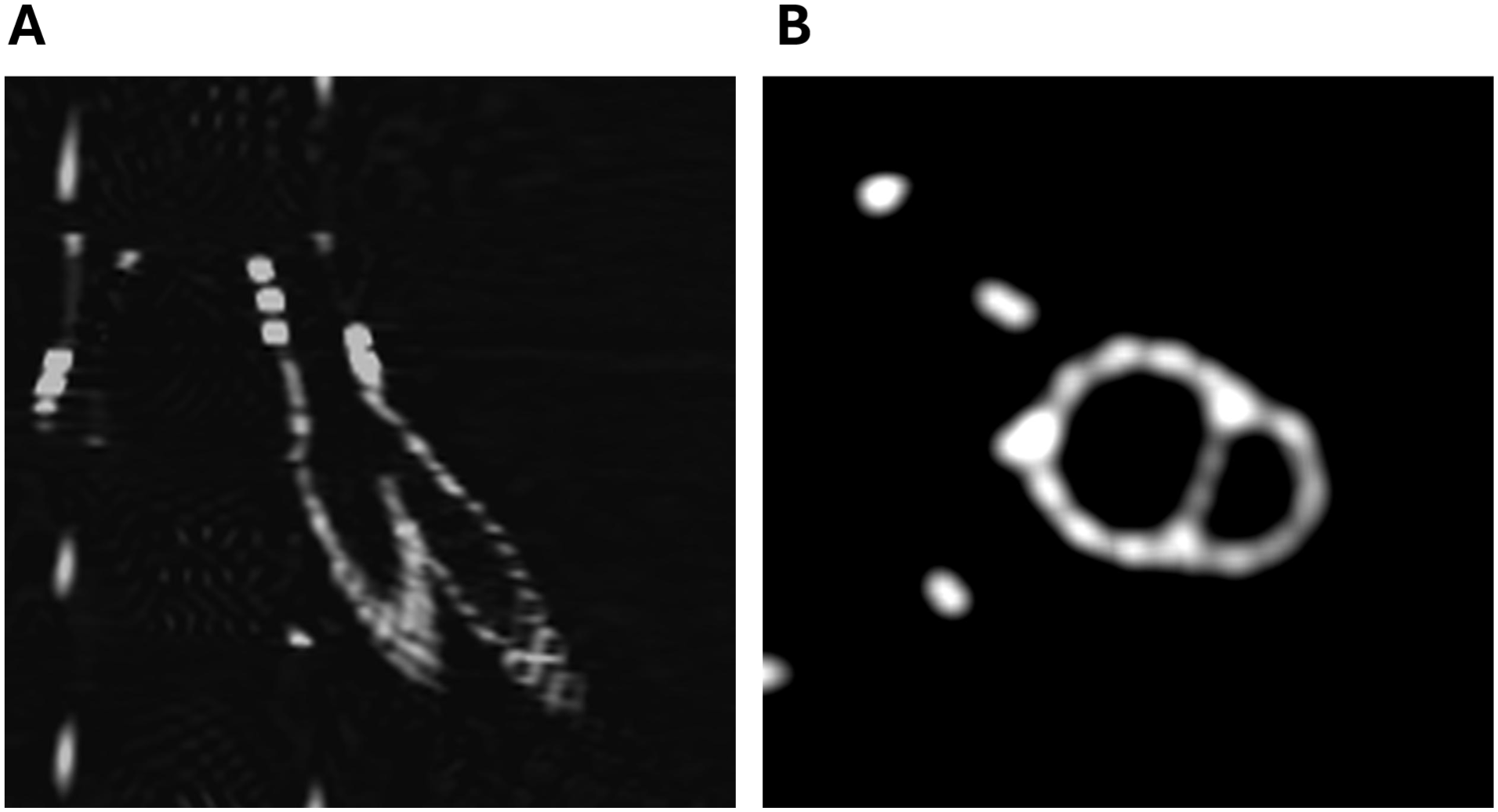
Computed tomography (CT) images showing the final disposition of the phantom model after the BIS2T procedure.
Clinical Utilization
The BIS2T was used clinically in 2 patients to incorporate a total of 6 target vessels including 1 patient with accessory renal arteries and 1 patient with bilateral IIAAs. Both patients were treated by the senior author (G.P.) at their primary institution and consented for endovascular repair using the off-label stent-graft modification.
Case 1. Incorporation of 2 left-sided renal arteries using 4-vessel off-the-shelf multibranch stent-graft via a single renal directional branch
A 70-year-old male patient presented with symptomatic 6.1 cm Extent IV TAAA. The patient’s medical history was notable for hypertension, cigarette smoking, hyperlipidemia and morbid obesity. The patient complained of increasing mid-abdominal pain for several days and had discomfort upon palpation of a large pulsatile abdominal mass. A computed tomography angiography (CTA) revealed a 6.1 cm TAAA involving 5 target vessels with origin from the aneurysmal segment, including celiac axis (CA), superior mesenteric artery (SMA), 1 right renal artery (RA) and 2 left-sided RAs measuring 4.5 and 4 mm in diameter. The patient had chronic occlusion of the right common iliac artery with reconstitution at the iliac bifurcation and collateral flow via the contralateral internal iliac and profunda femoris artery. The patient had no symptoms of claudication. Due to the presence of a large symptomatic TAAA, endovascular repair using an off-the-shelf multibranch stent-graft was recommended.
The procedure was performed under general endotracheal anesthesia in a hybrid operating room with fixed GE Discovery imaging system (GE Healthcare, Buc, France) using on-lay fusion, digital zoom, and high-definition cone-beam computed tomography (CBCT). Unilateral percutaneous left trans-femoral access was achieved using preclosure technique in standard fashion and left axillary artery access was obtained via open surgical exposure. After systemic heparinization, an off-the-shelf t-Branch multibranch stent-graft (Cook Medical Inc, Denmark) was oriented extra-corporeally, advanced via the left femoral approach and deployed in the distal thoracic aorta with each of the directional branch 1 to 2 cm proximal to its intended target vessel. This was followed by placement of a universal distal bifurcated stent-graft and ipsilateral left iliac limb extension. The contralateral gate of the bifurcated device was occluded using an Amplatzer vascular plug (Abbott Cardiovascular, Minneapolis, Minnesota). The left femoral sheath was downsized to a small 6F sheath restoring flow to the lower extremity.
Sequential renal and mesenteric stenting was performed via left axillary artery approach using an 8 Fr flexor sheath (Cook Medical Inc, Bloomington, Indiana). The CA was stented using a 10×57 mm iCover balloon-expandable stent-graft (iVascular, Barcelona, Spain), the SMA using 9×50 mm self-expandable Viabahn stent-graft (WL Gore) with reinforcement of the proximal cuff using an 8×27 mm iCover balloon-expandable stent-graft and the right RA was stented using 5×50 mm Viabahn stent-graft with reinforcement of the proximal cuff using 6×27 mm iCover stent-graft.
The 2 left-sided renal arteries were incorporated using BIS2T. First, a 5×39 mm VBX stent-graft was deployed from the directional branch to the inferiorly located left renal artery, with distal extension using a 5×30 mm self-expanding Pulsar stent-graft (Biotronik, Lake Oswego, Oregon). An Outback Elite reentry device (Cordis) was used for creation of an in situ fenestration in the VBX stent-graft segment. A 0.014″ PT2 guidewire (Boston Scientific, Minneapolis, Minnesota) was advanced into the second superiorly located left renal artery, followed by predilatation of the in situ fenestration with a 3 mm NC Trek balloon (Abbott Cardiovascular). After guidewire exchange, a PK Papyrus (Biotronik; Lake Oswego) 3.5×23 mm balloon-expandable stent-graft was deployed into the renal artery. Then, a third stent-graft (PK Papyrus 4.5×26 mm) was deployed inside the VBX in the BIS2T overlapping segment. Final angiography demonstrated patent bifurcated stent-graft with no endoleak, dissection or embolization. The patient had no postoperative complications. Clinical and imaging follow-up at 6 months revealed patent stent-grafts with no evidence of endoleak, aneurysm sac expansion or stent-graft integrity issues (Figure 6).
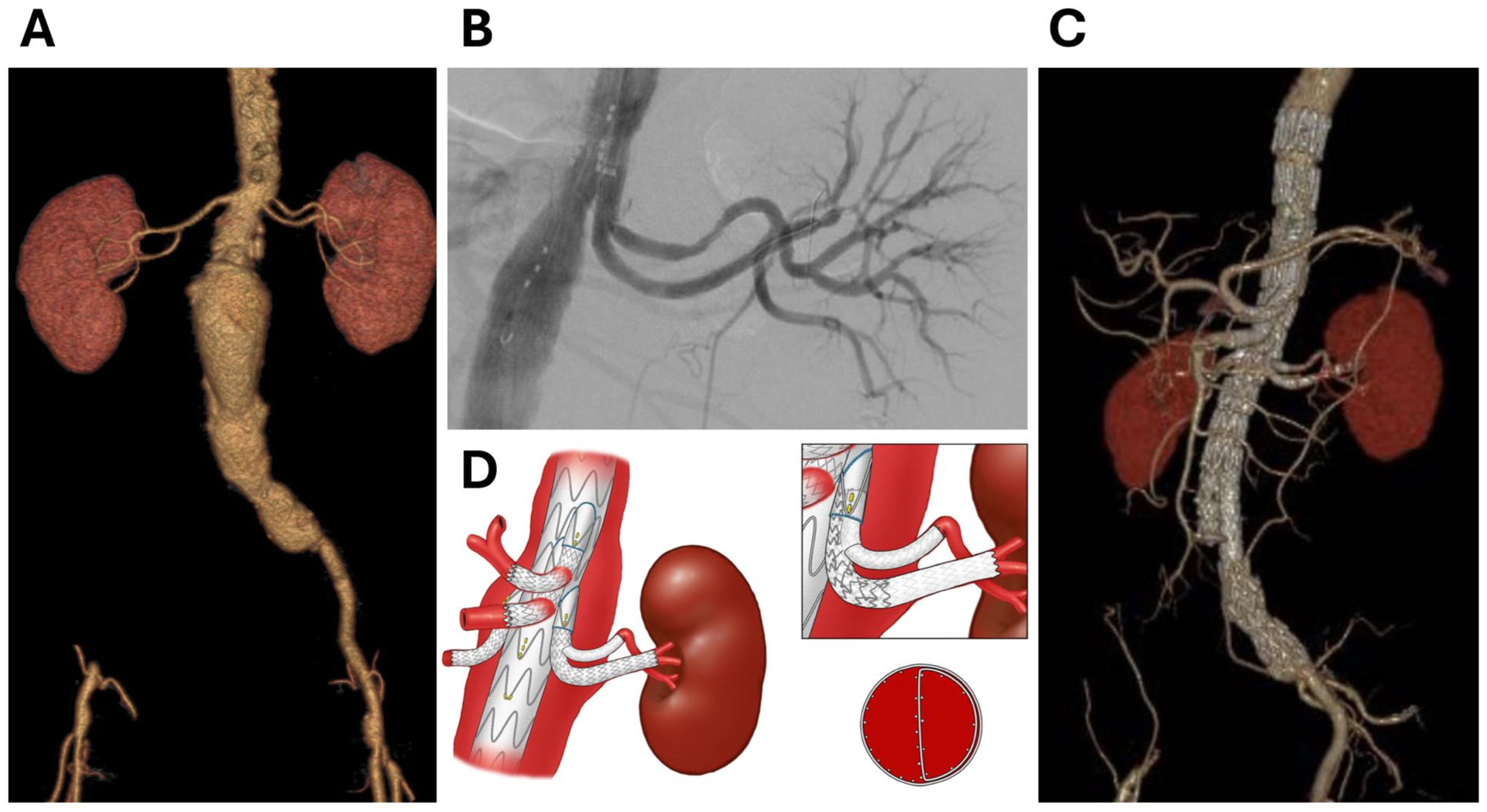
Demonstration of Case-1 incorporation of 2 left renal arteries using the BIS2T technique.
Case 2. Incorporation of the bilateral anterior and posterior divisional branches of the IIA using bilateral iliac branch devices via single internal iliac directional branch
A 78-year-old male patient presented with aortoiliac and large bilateral IIAAs. The patient’s medical history was notable for hypertension, cigarette smoking, and ischemic cardiomyopathy with no active symptoms. A CTA revealed a 3.7 cm infrarenal abdominal aortic aneurysm, bilateral 3 cm common iliac artery aneurysms, 4.9 cm right and 4.1 cm left IIAAs. The anterior and posterior division branches of the IIAs were patent bilaterally with diameters of 4 and 8 mm, respectively. Endovascular repair was recommended using E-tegra (Artivion, CryoLife Jotec, Atlanta, Georgia) bifurcated stent-graft and bilateral E-iliac Iliac Branch devices (Artivion CryoLife Jotec) with preservation of bilateral anterior and posterior divisional branches using BIS2T.
The patient was treated under general endotracheal anesthesia in a hybrid operating room with fixed GE Discovery system. Bilateral percutaneous femoral access was established in standard fashion using preclosure technique. Through-and-through femoral-femoral access was established. The left E-iliac was loaded in the aortic guidewire and the femoral-femoral guidewire, advanced into the proximal left common iliac artery and deployed with the directional branch 1 cm proximal to the origin of the aneurysmal left IIA. A 12 Fr flexible Dryseal sheath (WL Gore) was advanced up and over the aortic bifurcation and into the portal, followed by selective catheterization of the left IIAA. The first balloon-expandable VBX stent-graft (8Large×79 mm) was deployed from the directional branch to the aneurysm sac. The proximal portion of the stent-graft was postdilated with a 10 mm angioplasty balloon to stabilize the stent-graft at the directional branch. An in situ fenestration was created in the VBX stent-graft using Outback Elite reentry catheter and a 0.014 inch PT2 guidewire (Boston Scientific) was advanced into the aneurysm sac, followed by removal of the reentry catheter and predilatation of the fenestration with a 3 mm angioplasty balloon. A 4 Fr vertebral catheter was advanced via the in situ fenestration and used for selective catheterization of the anterior divisional branch. The second balloon-expandable VBX stent-graft was advanced via a Rosen guidewire (Cook Medical Inc) and deployed via the in situ fenestration into the anterior divisional branch with approximately 1 cm overlap into the first VBX stent-graft. The posterior divisional branch was selectively catheterized via the first VBX stent-graft and a third VBX stent-graft was deployed into the posterior divisional branch overlapping into the first VBX stent-graft. Kissing balloon angioplasty of the overlapping area of the BIS2T was performed using 5 and 6 mm angioplasty balloons. Selective left IIA branch completion angiography was performed which showed a widely patent bifurcated stent-graft system with no dissection, endoleak or embolization. The right contralateral IIAA was treated in identical fashion, followed by placement of the bifurcated aortic stent-graft and connecting iliac limb extensions. Follow-up CTA revealed no evidence of endoleak and widely patent stent-grafts (Figure 7).
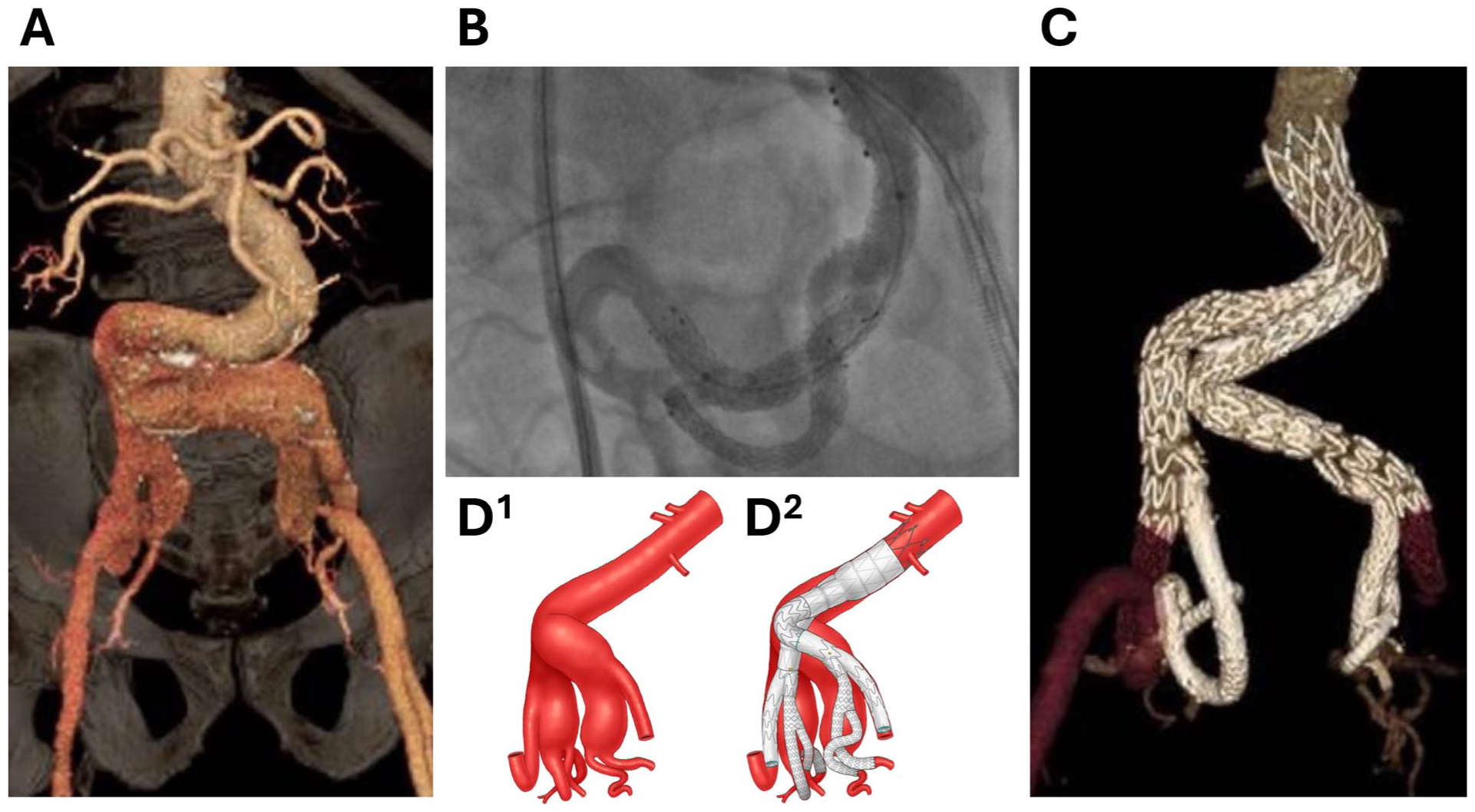
Demonstration of Case-2 incorporation of bilateral anterior and posterior divisional branches of the internal iliac arteries (IIAs) using the BIS2T technique.
Discussion
This report describes the use of BIS2T as an adjunctive technique for incorporation of 2 target vessels via a single directional branch. Potential clinical indications for this technique are variations of the normal anatomy in patients undergoing endovascular repair of aortic arch, thoracoabdominal and iliac artery aneurysms. Although this report demonstrates the technical feasibility in 2 patients with accessory renal arteries and large internal iliac artery aneurysms, additional ex vivo laboratory testing, larger clinical experience and longer follow-up are needed prior to its widespread utilization. Although we have not observed fabric tears in the initial benchtop deployments, the use of BIS2T may compromise the integrity of the bridging stent-graft and/or result in vessel occlusion or stent disconnection.
Abnormalities of target vessel anatomy represent one of the most frequent impediments for complex endovascular repair using fenestrated, branched or parallel grafts.4,6,7 Because off-the-shelf TAAA devices allow incorporation of a maximum of 4 target vessels in the renal and mesenteric segment, patients with more vessels or aneurysmal involvement of the target vessel up to large secondary branches are typically managed with intentional sacrifice of one of the targets.3,6 Mendes and associates reported renal artery issues in 19% of patients undergoing repair of complex abdominal aortic aneurysms, including large accessory renal arteries in 5% and early vessel bifurcation in 9%. 4 Other frequent variations are replaced right hepatic artery origin from the superior mesenteric artery in 12%, aberrant or accessory hepatic artery origin from the aorta in 1% to 2% and a common celio-mesenteric trunk in 0.5% to 3%.7,11 While variations may be potentially managed by custom or patient-specific stent-grafts, these devices are not always available and may not be feasible in patients who present with rapidly expanding, symptomatic or ruptured aneurysms. Among patients undergoing endovascular repair of iliac aneurysms with iliac branch devices, the most frequent anatomical limitation is lack of a suitable landing zone in the main trunk of the internal iliac artery due to aneurysm involvement in nearly 60% of patients. 12 In these cases, the posterior divisional branch is most frequently targeted due to large size and preservation of flow into the gluteal vessels and spinal collaterals. 3 However, sacrificing the anterior division branch (particularly bilaterally) may compromise flow into the pudendal and penile branch collaterals causing arterial erectile dysfunction. 3 Lastly, during endovascular arch repair into zone 0, involvement of the innominate artery by aneurysm or dissection may preclude placement of a bridging stent-graft into this target requiring cervical debranching procedures (bypass or transposition) to create a suitable landing zone in the right common carotid artery. 2
A potential technique to manage these anatomical variations is the use of parallel stent-grafts.10,13 When used within a single directional branch, parallel grafts may be associated with risk of gutter endoleaks or bridging stent compression/ thrombosis. Although this issue is also pertinent to BIS2T, initial ex vivo experimentation with in situ fenestration of an open-cell double-layered PTFE bridging stent-graft demonstrates absence of fabric tears with post dilatation and enough space to dilate both stents in a kissing fashion. The in situ fenestration coupled with the overlapping “D shaped” configuration recommended by Minion may minimize the risk of endoleak and stent thrombosis. 10
Another creative solution for managing patients with accessory vessel anatomy or early bifurcations is the use of bidirectional double-cuff inner branches. 14 Initial experience with this custom-made device has demonstrated promising results. 14 However, as acknowledged by the authors, the technique has several limitations, including limited applicability in urgent settings due to the time required for device manufacturing, the need to occlude the unused cuff when only 1 artery is incorporated, and the potential compromise of the distal landing zone in the infrarenal aorta when the lowest renal artery is targeted. 14
The BIS2T allows a bifurcated system to be created within a single directional branch using a minimum of 3 balloon-expandable stent-grafts. Although the technique may also be performed with 2 stent-grafts and may be reproduced with multiple bridging stent-graft manufacturers, a few stent-graft characteristics are recommended to potentially prevent disconnection and integrity issues. First, the quality and tensile strength of the PTFE is of paramount importance to avoid tears or disruption with postdilatation. 15 Ideally the use of a doubled layer of PTFE and an open-cell stent structure sandwiched between the layers is recommended for durability and to avoid disconnection between the metal cage and fabric. Stent-grafts with wider range of postdilatation, such as the 8Large VBX stent-graft, offer the ability of postdilatation up to 16 mm within the BIS2T overlapping segment, which may minimize the risk of stent disconnection or compression. This is especially important when the BIS2T is bridging a longer gap between the directional branch and the target and in vascular territories with significant hemodynamic forces or motion.
The in situ fenestration technique using the Outback Elite catheter was selected due to the surgical team’s familiarity with the device, its wide availability, and its ability to provide directional needle deployment. 16 Future studies may explore other in situ fenestration techniques in adaptations of the BIS2T procedure. However, each alternative method presents specific limitations that must be carefully considered. Other reentry devices, such as the Pioneer Plus catheter (Philips Volcano, San Diego, California), which integrates intravascular ultrasound (IVUS) for real-time guidance, may enhance targeting accuracy but is also associated with higher technical complexity and procedural costs. 16 In situ laser fenestration, although effective, carries the risk of thermal injury to adjacent structures, the potential liberation of toxic materials during thermal damage, and requires specialized equipment with high cost and limited availability. 17
The BIS2T has some limitations. First, the creation of the in situ fenestration can be technically challenging in anatomically complex cases, such as those with significant aortic tortuosity, which may compromise needle stability and orientation. In addition, when applied to cases involving a replaced right hepatic artery or early renal bifurcation, BIS2T can present additional challenges due to the limited intraluminal workspace. Therefore, this technique may be reserved for high-volume centers with substantial endovascular expertise.
Second, this new technique represents an off-label modification of an approved stent-graft and has not yet been extensively tested in ex vivo fatigue models for potential risk of fabric tears, fracture, or stent disconnection. Therefore, it is critical that these testing be completed and that larger clinical experience with different stent-graft combinations, and longer follow-up be obtained prior to its widespread use.
Conclusion
The BIS2T provides a versatile and feasible off-the-shelf solution for management of anatomical variations in patients undergoing endovascular repair with fenestrated, branched, or parallel stent-grafts. This report describes its initial clinical use for preservation of 2 left-sided renal arteries and bilateral internal iliac artery divisional branches, but a wide variety of other potential anatomical applications is described. Laboratory testing, increasing clinical experience and longer follow-up is needed to determine vessel patency and the incidence of stent-graft disconnection or other integrity issues.
Footnotes
Acknowledgements
None
ORCID iDs
Consent to Participate
All participants provided written informed consent prior to participating.
Consent for Publication
Informed consent for publication was provided by the participants.
Ethical Considerations
This study was approved by the Ethics Committee of the Hospital Santa Lucia (Ethics Code: 88768525.5.0000.5058) on May 16, 2025. All participants provided written informed consent prior to enrollment in the study. This research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.S.O.: Consulting agreement and scientific advisory board, Cook Medical Inc., WL Gore, GE Healthcare, Centerline Biomedical, Terumo Aortic; Research Grants: GE Healthcare (paid to UT Health Houston). The other authors (G.P., L.M., B.P.S., L.R.K., D.B., and T.A.M.) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
