Abstract
Alternative treatment strategies for claudication are needed and cell-based therapies designed to induce angiogenesis are promising. The purpose of this report was to conduct a Phase I safety, dose-escalating, non-randomized, open-label study of autologous, fully differentiated venous endothelial and smooth muscle cells called MultiGeneAngio (MGA) for claudication due to peripheral artery disease. Twelve subjects, at two centers, received a single intra-arterial infusion of a suspension of equal amounts of transduced autologous venous smooth muscle cells expressing vascular endothelial growth factor (VEGF165) and endothelial cells expressing angiopoietin-1 (Ang-1) (Cohort 1: 1 × 107, Cohort 2: 2 × 107, Cohort 3: 5 × 107, Cohort 4: 7 × 107). The treatment was given unblinded and in the more symptomatic lower extremity. Transduced cells were tested for in vitro doubling time, telomerase activity, and gene expression. The main outcomes were clinical safety and tolerability. Other safety measures included ankle–brachial index (ABI) and walking time on a treadmill. All subjects were male (mean age 60 ± 5 years) including 25% with diabetes mellitus. At 1-year follow-up, there was one serious adverse event possibly related to MGA. Safety endpoints including VEGF and Ang-1 plasma protein levels were within normal ranges in all subjects. The mean maximal walking time increased from baseline to 1 year and the index limb ABI was unchanged, indicating no safety concerns. MGA, an autologous, transduced, cell-based therapy was well tolerated and safe in this Phase I study. Further evaluation is warranted in randomized human studies.
Introduction
Peripheral artery disease (PAD) affects 8–12 million Americans. 1 Symptoms associated with lower-extremity PAD include claudication and even more severe manifestations such as rest pain and ischemic ulcers and tissue loss. In addition to smoking cessation and medical therapy, the treatment of claudication includes structured exercise.2,3 Percutaneous revascularization or surgical bypass is reserved for patients with severe, lifestyle-limiting claudication unresponsive to medical or lifestyle modification treatment. 2 While percutaneous and surgical revascularization is most effective for aorto-iliac disease, below the inguinal ligament percutaneous revascularization is limited by high rates of restenosis. 4 Surgical revascularization for claudication due to PAD below the inguinal ligament is limited by possible bypass graft failure, and the risk associated with vascular surgery. 4 Hence, alternative treatment strategies are needed for lifestyle-limiting claudication.
Angiogenesis or remodeling of collaterals is a complex process that requires the timely participation of multiple genes and cell types.5,6 A potential advantage of cell therapy is that cells have the complete repertoire of genes to allow remodeling of mature arteries.7,8 Activation of mature vascular cells up-regulates and down-regulates multiple genes that are relevant to arteriogenesis and remodeling. Vascular endothelial growth factor (VEGF) is a key protein for development on blood vessels while angiopoietin-1 (Ang-1) is essential for maturation of blood vessels. 9
Augmentation of angiogenesis, the natural process that leads to collateral vessel development, is a potential therapeutic alternative for patients with severe vascular disease. The proteins and genes responsible for the angiogenic response have been identified and studied in multiple clinical trials. 5 However, promising pre-clinical data did not translate into positive clinical trials, and there are no approved therapeutic angiogenic agents. Similarly, cell therapies for therapeutic angiogenesis promising in pre-clinical and early-phase clinical study, have not been shown to be effective in late-phase randomized clinical trials for ischemic vascular disease. 10
MultiGeneAngio (MGA) is a novel, autologous, cell-based product transduced and conditioned to express growth factors known to be important in the angiogenic response. 6 Specifically, MGA contains transduced venous autologous smooth muscle cells (SMCs) expressing VEGF165 and autologous venous endothelial cells (ECs) expressing Ang-1. The combination of ECs and SMCs activated by angiogenic gene transfer was shown both in vitro and in vivo to expand existing collaterals. 6
We report here the in vitro results of the doubling time, telomerase activity, and gene expression of the transduced vascular cells to provide evidence for the safety of use of these cells and for their potential to produce or expand collateral arteries, and the results of a Phase I clinical trial of MGA for treatment of claudication in patients with PAD.
Materials and methods
In vitro studies
Isolation and culturing of human primary cells for in vitro studies
Primary human saphenous vein ECs were enzymatically isolated from lower-extremity vein segment using 0.153 PZ units/ml collagenase (Serva, Heidelberg, Germany) and maintained in an EC-G medium (MCDB 131 medium (Gibco, ThermoFisher Scientific, Philadelphia, PA, USA) containing 20% fetal bovine serum (FBS) (HyClone, Pittsburgh, PA, USA), 1400–1500 mg/l
Primary human saphenous vein SMCs were isolated from lower-extremity vein segment after EC isolation was completed. The remaining vein segment was incubated with 0.153 PZ units/ml collagenase (Serva) and sectioned into 0.5–2 mm2 segments. These segments were incubated with SMC-G medium (DMEM medium (Gibco) containing 20% FBS (HyClone), 1400–1500 mg/l
Doubling time determination
Cells from multiple human donors were thawed at passages 9–11 and cultured up to senescence. Proliferation rate was assessed every five passages; 1.5×105 cells were seeded in six-well plates in triplicates, trypsinized and counted 48 hours later. Cell proliferation rate is expressed as the number of cell splits in 24 hours. Senescence was defined as a proliferation rate of 0.
Flow cytometry assay
CD31 detection: ~2×105 ECs were trypsinized and washed with phosphate-buffered saline (PBS) (Biological Industries). After centrifugation, the cells were incubated with FITC-conjugated anti-human CD31 (eBioscience, San Diego, CA, USA) or iso-type control (FITC-IgG1). Stained cells were washed again with PBS and analyzed by flow cytometry.
SMA (smooth muscle actin) detection: ~2×105 SMC were trypsinized, washed with cold PBS (Biological Industries) and fixed with 4% paraformaldehyde (Electron Microscopy Sciences). Then, cells were washed with washing solution (0.1% Tween 20 (Sigma) in PBS), centrifuged and permeabilized with 0.5% Tween 20 in PBS. Cells were then incubated with PE-conjugated anti-human αSMC actin (R&D Systems (Minneapolis, MN)) or iso-type control (PE-IgG1). Stained cells were washed again with washing solution and finally re-suspended in PBS and analyzed by a flow cytometer.
Immunohistochemistry assay for CD31 detection: ~7×104 ECs were seeded on slides and incubated overnight with EC-G media. Then, cells were washed with cold PBS (Biological Industries), fixed with 4% paraformaldehyde (Electron Microscopy Sciences, Hatfield, PA, USA), retrieved with citrate buffer (Zymed, ThermoFisher) and blocked with CAS block (Zymed) to avoid unspecific binding. CD31 detection involves exposure of the cells to mouse anti-human CD31 antibody (DAKO, Carpinteria, CA, USA), biotinylated goat anti-mouse IgG (Chemicon, Pittsburgh, PA, USA), and HRP-conjugated streptavidin (Chemicon). A colorimetric reaction is catalyzed by the peroxidase (DAKO). Images of 10 randomly selected fields were captured and the percentage of stained cells out of the total cells was calculated by an experienced technician.
Telomerase activity assessment
Human ECs and human saphenous vein SMCs isolated from five human subjects were cultured to sub-confluence in six-well plates or in 25 cm2 flasks, trypsinized, counted and pelleted (2×105cells/reaction). Telomerase activity was detected by the telomerase polymerase chain reaction-enzyme linked immunosorbent assay (PCR-ELISA) assay (Roche Applied Science, Indianapolis, IN, USA). The assay’s major steps are: (1) elongation – telomerase adds telomeric repeats (TTAGGG) to the 3’ end of the biotin-labeled synthetic primer; (2) amplification – elongation products are amplified by PCR, generating with the telomerase-specific 6-nucleotide increments; (3) detection by PCR-ELISA products are denaturized and hybridized to a digoxigenin (DIG)-labeled, telomeric repeat-specific detection probe, the resulting products are immobilized via the biotin-labeled primer to a streptavidin-coated microplate. Finally, probe is visualized by virtue of peroxidase metabolizing TMB (tetramethylbenzidine) to form a colored reaction product. The assay procedures followed the recommendations of the manufacturer, with each sample being analyzed in duplicate. Positive control cells (supplied with the kit; Roche Applied Science), 293 cells, and human umbilical vein endothelial cells (HUVECs) served as the positive control, and heat-inactivated cells served as the negative control.
Gene expression analysis
A human 4×44K whole genome oligo DNA microarray chip (G2519F; Agilent Technologies, Palo Alto, CA, USA) was used for global gene expression analysis testing ECs and SMCs under three conditions: (1) cells transduced with index genes (Ang-1 for ECs and VEGF for SMCs); (2) cells transduced with control gene (Lac Z); and (3) non-transduced cells. Three types of controls were implemented in the experiment: biological replicate of two human subjects, technical triplicate from all cell conditions, and oligonucleotide hybridization replicates. Total RNA was isolated using standard RNA extraction protocols (NucleoSpin® RNAII; Macherey-Nagel, Bethlehem, PA, USA). For the Linear T7-based amplification, 1 µg of each RNA sample was labeled with Cy3 dye using an Agilent Quick Amp Labeling Kit (Agilent, Wilmington, DE, USA).
Hybridization and wash processes were performed according to the manufacturer’s instructions, and hybridized microarrays were scanned using an Agilent Microarray scanner (G2565BA). For the detection of significantly differentially expressed genes between control and treated samples, each slide image was processed by Agilent Feature Extraction Software (version 9.5.3.1). Briefly, (1) this program measured the dye signal intensities of whole probes; (2) dye-bias tends to be signal intensity dependent, and therefore the software selected probes using a set by rank consistency filter for dye normalization; (3) the respective samples were combined to ratios using the RosettaResolver™ Software (Rosetta Inpharmatics, Seattle, WA, USA); (4) the significance (p) value was based on the propagate error and universal error models; (5) the threshold of significance for differentially expressed genes was <0.01 (for the confidence that the feature was not differentially expressed); and (6) erroneous data generated owing to artifacts were eliminated before data analysis using the software. 11 The outputs of microarray analysis used in this study are available under the series number GSE31981 at the NCBI Gene Expression Omnibus (GEO) public functional genomics data repository (http://www.ncbi.nlm.nih.gov/geo/info/linking.html). All ratio data were additionally transformed to log2 ratio, and for each ratio the corresponding ‘fold-change’ was computed for a more intuitive understanding of the expression changes.
To identify the reporters specifically regulated by the index genes, the three technical replicates of each group were combined to one weighted value so that the effect of single outliers was mitigated. The different treatment groups were then compared by computing ratios out of the weighted group means to find reporters with differential regulation. Differentially regulated reporters were selected by fold-change (⩾1.7 fold in the up-regulated group and ⩽ 0.75 in the down-regulated group) and associated p-value (⩽ 0.01).
A functional grouping analysis was performed to provide a cursory overview of the different biological processes and pathways. As a result of a microarray data analysis, genes with interesting expression patterns (e.g. typically differentially regulated genes or members of co-expression groups) were obtained. These genes were used for database queries searching for annotations related to functional categories or pathway membership. In addition, the gene list was searched for known targets of certain signaling pathways. We have chosen to focus our further analysis on the Angiogenesis and the Cell Proliferation functional groups.
Gene ontology (GO) enrichment analysis using the DAVID (Database for Annotation, Visualization and Integrated Discovery) program (version 6.7) was performed to identify pathways and functions that have a higher percentage of up-regulated or down-regulated genes than would be statistically expected in naive conditions.12,13
Subject recruitment and clinical protocol
Twelve patients with claudication were recruited from clinical practices at the University of Pennsylvania and University of Michigan. After signing informed consent approved by each respective institution, subjects underwent an initial screening visit as outlined in the clinical protocol (Table 1). Males and females ⩾50 and ⩽80 years old with a diagnosis of PAD with exercise-limiting claudication were eligible for the study. A history of PAD and claudication was confirmed on screening with an ankle–brachial index (ABI) <0.90 and exercise treadmill protocol to confirm claudication symptoms. The major exclusion criteria were: (a) presence of significant inflow disease (defined as >50% stenosis) in the distal aorta or common or external iliac as assessed by conventional angiogram, digital subtraction angiography (DSA), or magnetic resonance angiography (MRA) performed < 1 year prior to screening; (b) critical limb ischemia, either chronic or acute ischemia manifested by rest pain, ulceration, or gangrene (Category 4 through 6 of the Society for Vascular Surgery (SVS) classification (Rutherford)); (c) history of malignant neoplasm (except curable non-melanoma skin malignancies); (d) renal failure defined as a serum creatinine >2.0 mg/dl; (e) significant hepatic disease (defined as >3-fold elevation in alanine aminotransferase/aspartate aminotransferase (ALT/AST)), hepatitis B virus (HBV) or hepatitis C virus (HCV) carriers, end-stage renal disease (requiring hemodialysis or renal replacement therapy), severe pulmonary disease (e.g. severe chronic obstructive pulmonary disease); (f) subjects with acute myocardial infarction or acute stroke within 6 months prior to the screening visit; and (g) subjects with uncontrolled diabetes mellitus.
Clinical protocol outline.
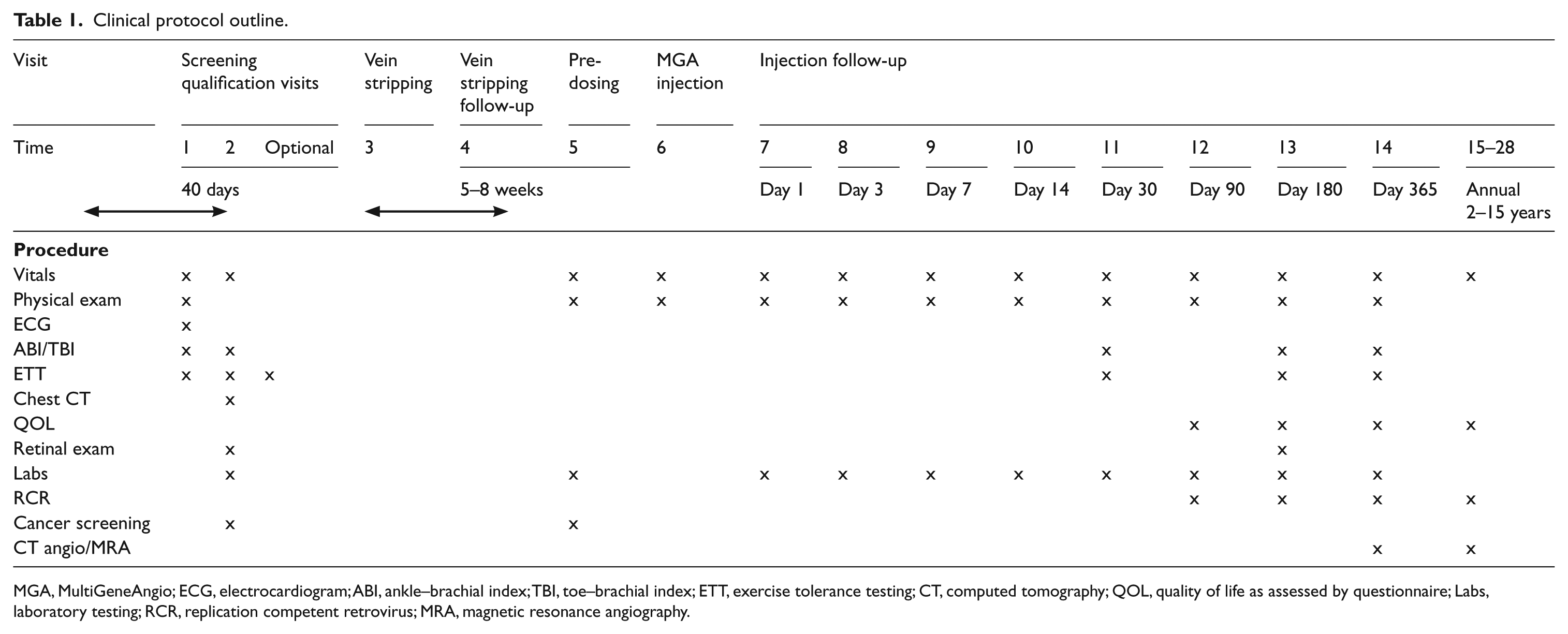
MGA, MultiGeneAngio; ECG, electrocardiogram; ABI, ankle–brachial index; TBI, toe–brachial index; ETT, exercise tolerance testing; CT, computed tomography; QOL, quality of life as assessed by questionnaire; Labs, laboratory testing; RCR, replication competent retrovirus; MRA, magnetic resonance angiography.
Subjects that qualified were enrolled and underwent collection of tissue to serve as a source of autologous cells. Specifically, a short superficial vein segment (either cephalic or basilic vein) was surgically removed from their arm under anesthesia. The MGA product was manufactured from the autologous ECs and SMCs that were isolated from the harvested vein. After cell processing and quality control testing to meet release criteria as specified in the FDA Investigational New Drug application, intra-arterial MGA was administered in the subject’s most symptomatic leg. Follow-up visits were carried out over 1 year, as detailed in Table 1, following MGA administration.
Cell transduction
Endothelial cells were transduced ex vivo by pseudo-typed retroviral vectors to express Ang-1, a protein operative in recruiting pericytes to capillaries and SMCs to newly formed arteries, thereby maturating and stabilizing them. It is also known as a survival factor, but does not induce cell proliferation. The SMCs were transduced with a retroviral vector encoding VEGF165, a protein regarded as the major switch to the process of angiogenesis. It has specific mitogenic, migratory and survival effects on ECs in vitro and in vivo. 6 The LXSN vector backbone (Genebank accession: #M28248), which was derived from Moloney Murine Leukemia Virus (MMLV), was used for the derivation of both Ang-1 and VEGF165 expression plasmids. GaLV–VEGF165 pseudo-typed retroviral vector was produced by 293-FLY-GaLV packaging cells, which express gag-pol proteins from Mo-MuLV and gibbon ape leukemia virus (GaLV) envelope glycoproteins. The 10A–Ang1 pseudo-typed retroviral vector was produced by 293-FLY-10A packaging cells, which express gag-pol proteins from Mo-MuLV and a dualtropic (or polytropic) envelope that recognizes receptors on mouse, rat, human, hamster, mink, cat, dog, and monkey cells. 6
Co-expression of the two genes in two different cell types ensures the coordination of at least two elements fundamentally involved in the angiogenic response: ECs and SMCs. Ang-1 is crucial in tempering the role of VEGF165 and appears to play an important role in vessel wall maturation through recruitment of pericytes and SMCs, as well as prevention of vascular permeability. Expression of Ang-1 with VEGF165 results in vessel wall stability while VEGF alone leads to the creation of leaky blood vessels.
MultiGeneAngio characterization and lot release testing
MGA characterization involves determination of the identity and purity of ECs and SMCs, as well as levels of transgene expression. The identity and purity of ECs and SMCs are determined by their typical morphology and by fluorescence activated cell sorter (FACS) and/or immunohistochemistry. CD31 is a specific EC marker (primary antibody: mouse anti-human CD3; DAKO, Denmark), and α-smooth muscle actin (primary antibody: mouse anti-human SMA; DAKO, Denmark), a marker of SMCs and fibroblasts are used to identify these cell populations.6,14
The EC identity-purity acceptance range was defined as when more than 85% of the cells stain positive for CD31 and less than 15% of the cells stain positive for α-smooth muscle actin. The SMC identity acceptance range is defined as when 60% of the cells or more stain positive for α-smooth muscle actin. The transgene expression was determined by immunohistochemistry and ELISA. The immunohistochemistry criteria included at least 55% of the ECs expressing Ang-1 and at least 60% of the SMC populations expressing VEGF165. The transgene expression level by ELISA was 0.05 pg for Ang-1 or VEGF165 proteins by a single cell in 24 hours. Quality control and assurance of the MGA product was performed at multiple steps during the manufacturing process and prior to release.
MultiGeneAngio formulation
Product formulation was carried out in the Clinical Cell and Vaccine Production Facility at the Hospital of the University of Pennsylvania and the University of Michigan under Good Manufacturing Practice (GMP) conditions. After completion of cell processing, both ECs and SMCs were trypsinized. Each cell suspension was separately centrifuged and gently washed three times with product injection solution to remove growth medium residues. Product injection solution consisted of Ringer’s Lactate solution supplemented with dextrose (0.1%), heparin (100 U/ml) and sodium bicarbonate 20 mM. It is used for cell washing prior to formulation, as well as a delivery solution that is injected into the subject with the cells. The cell pellet was finally re-suspended in the product injection solution and transferred to the angiography suite in a sterile container at 4–8°C.
MGA is a clear cell suspension, formulated in two separate polypropylene vials. Each vial contains 5–35×106 of ECs or SMCs suspended in 10–20 ml product injection solution (depending on the dose group; Table 2). The product stability is maintained if kept at 4–8°C immediately after formulation until dosing, for no longer than 8 hours.
Dose escalation scheme for all subjects.
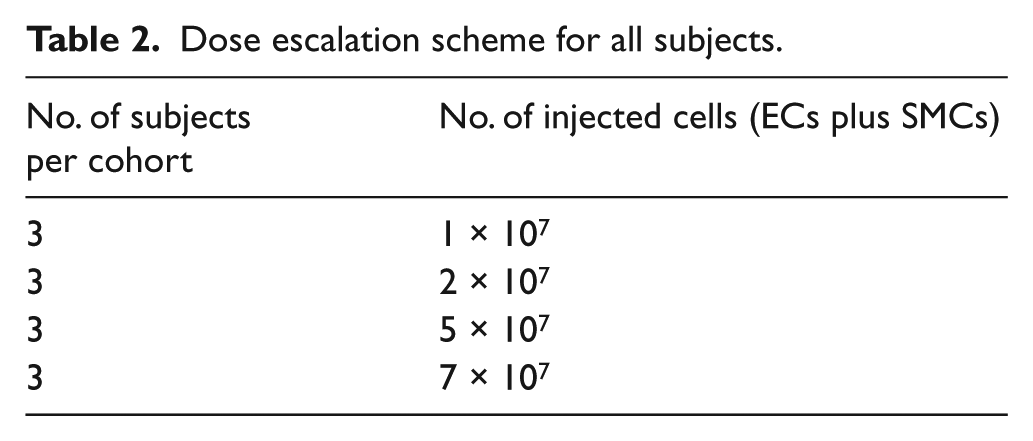
MultiGeneAngio dose
The number of subjects per cohort and the dose escalation scheme is listed in Table 2. Quality control and assurance of the MGA product was performed at multiple steps during the manufacturing process and prior to release.
MultiGeneAngio delivery
The contralateral common femoral artery (untreated) was cannulated using a 6F catheter (JR 3.5 Open End, 0 side holes Infinity™ Thrulumen catheter. 534-519T; Cordis Corp., Miami, FL, USA or equivalent). The catheter was positioned in the abdominal aorta and, using a cross-over technique, steered using a guidewire proximally to the location of the occlusion (superficial or common femoral artery). The catheter location was confirmed using injections of contrast medium. The location was recorded with the help of anatomic landmarks. A selective angiogram of the index extremity was then obtained. After performance of the selective angiogram, the catheter was flushed with at least 10 ml of normal saline in order to clear the contrast agent residuals from the catheter. The MGA EC suspension was injected initially, and subsequently the SMC suspension was injected via the catheter with a rate of 2 ml/min. The catheter was then flushed with an additional 10 ml of normal saline 0.9% NaCl. The catheter was subsequently removed and the 6F sheath was removed and hemostasis obtained with manual pressure per local institution protocols.
Safety measures
The following measures were obtained to assess disease severity at baseline and follow-up to ensure that the investigational product did not result in any deleterious effects on these parameters.
The ABI is the ratio of the highest systolic blood pressure of either the dorsalis pedis or posterior tibial arteries at the ankle, divided by the highest systolic blood pressure in the arm using a 5–7 MHz Doppler ultrasound instrument. This was performed after the subject has been lying supine for at least 10 minutes prior to the treadmill test. For subjects with an ABI of >1.3 (non-compressible calcified arteries) a toe–brachial index (TBI) in the great toe was allowed. The TBI is the ratio of the systolic blood pressure at the first toe divided by the systolic blood pressure in the arm. The TBI had to be ⩽ 0.70 for subject qualification.
A graded exercise treadmill study (Gardner protocol 15 ) with continuous electrocardiogram monitoring was performed on all subjects. The claudication onset time (COT) for the index and non-index lower extremity was recorded. The peak walking time (PWT) in the painful lower extremity (index or non-index) causing the subject to stop the treadmill study was recorded during this visit.
The walking impairment questionnaire (WIQ) was administered as it assesses the degree of walking impairment and efficacy of treatment in improving walking ability by evaluating the subject-assessed concepts of walking speed, walking distance, and claudication symptom severity associated with walking difficulty. The SF-36 version 2 questionnaire was also administered and is a generalized measure of quality of life that measures the subject’s perception of his/her health status in three physical concepts, mental health concepts, and combined physical mental health concepts.
Statistical analysis
This was an open-label, unblinded, single arm trial. The paired t-test or non-parametric signed-rank test were applied for testing the statistical significance of the changes in the ABI ratio, WIQ scores, SF-36v2 quality of life questionnaire scores, and exercise treadmill tests (COT and PWT). The Pearson correlations were applied for testing the correlations between the study following the parameters: between COT and PWT, between WIQ scores, and between SF-36v2 quality of life questionnaire sub-scores. All tests applied were two-tailed, and a p-value of 5% or less was considered statistically significant. The data were analyzed using the SAS® version 9.1 for Windows (SAS Institute, Cary, NC, USA).
Results
In vitro studies
Doubling time
The ECs and SMCs isolated from three donors were tested for proliferation capacity at high passages. As demonstrated in Figure 1, the number of cell splits per day dropped to zero in ECs by passage 30 and in SMCs by passage 25. These results are supported by our previous data that primary human venous ECs and SMCs cannot be cultured beyond 30 passages as they reach senescence (data not shown).
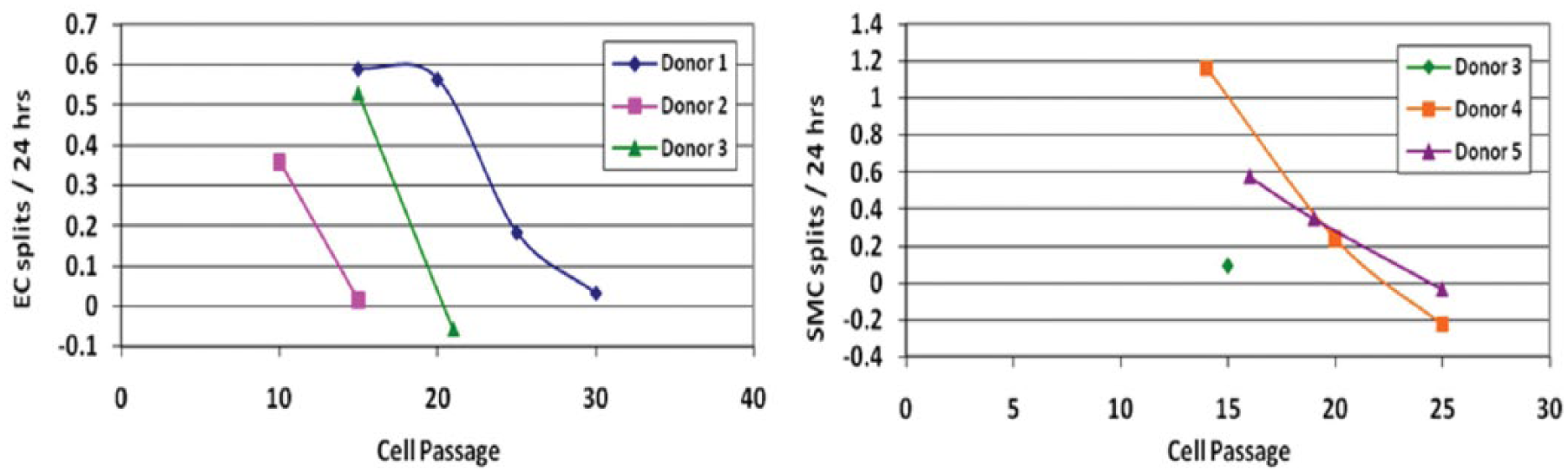
Endothelial cell (EC) and smooth muscle cell (SMC) proliferation diminishes at higher passages. (Left: EC transduced with Ang-1; Right: SMC transduced with VEGF (vascular endothelial growth factor).)
Telomerase activity
Another way to evaluate the proliferation capacity of cultured cells is by telomerase activity. Telomeres play an essential role in the stable maintenance of the eukaryotic chromosome within a cell by specifically binding to structural proteins. Telomere length is maintained by telomerase, a ribonucleoprotein that catalyzes the addition of ‘TTAGGG’ telomeric repeats onto chromosome ends, using a complementary sequence of its intrinsic RNA component as a template. It was demonstrated that telomere length was inversely correlated to passage number of primary human ECs and SMCs in culture,16–18 implicating a significant role for the telomere in vascular cell senescence.19–21 Enhanced telomerase activity is found in cancer and stem cells. 22 In this study, the telomerase activity of retrovirally transduced human saphenous vein endothelial cells (HSVEC) and smooth muscle cells (HSVSMC) was tested, using the telomerase PCR-ELISA kit (Roche Applied Science). Results (presented in Figures 2a and 2b) show that the telomerase activity levels of all human EC and SMC donors tested were significantly lower than the human embryonic kidney cell line (293 cells, ATCC (cat. no. ATCC CRL-1573)) and HUVECs (ATCC (cat. no. ATCC CRL-1730)), both known to have high proliferation potential. In addition, the telomerase activity levels of transduced cells were lower or similar to the corresponding naive cells, demonstrating that retroviral transduction had no apparent effect on cell proliferation capacity.
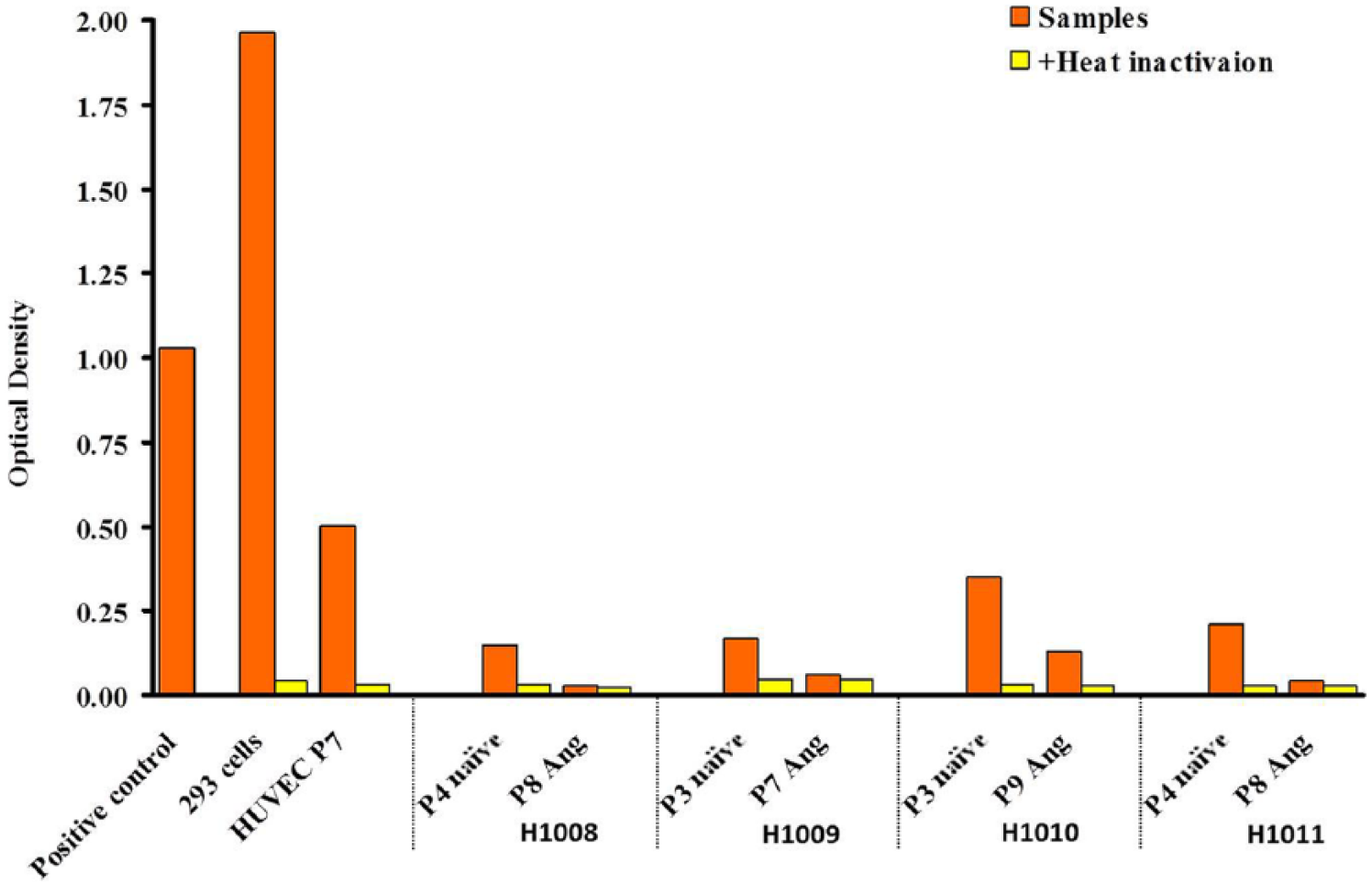
Telomerase activity in primary human lower-extremity saphenous vein endothelial cells (ECs). Telomerase activity was measured in ECs isolated from four different human donors – either non-transduced (naïve) or transduced with Ang-1(Ang). Positive control cells (supplied with the kit), 293 cells, and HUVECs (human umbilical vein endothelial cells) served as the positive control, and heat-inactivated cells served as the negative control.
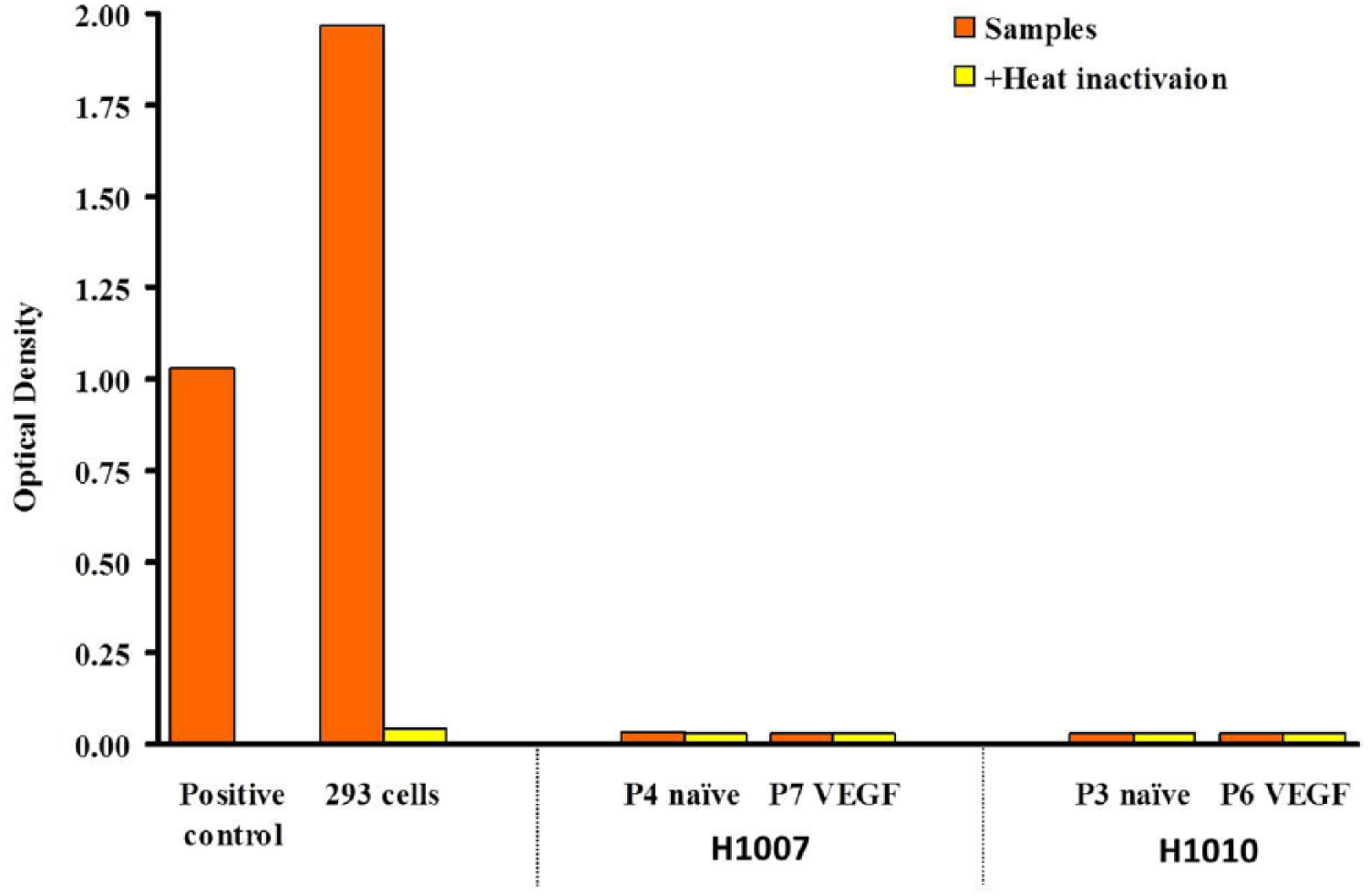
Telomerase activity in primary human lower-extremity saphenous vein smooth muscle cells (SMCs). Telomerase activity was measured in SMCs isolated from two different human donors – either non-transduced (naïve) or transduced with VEGF165 (vascular endothelial growth factor). Positive control cells (supplied with the kit) and 293 cells served as the positive control, and heat-inactivated cells served as the negative control.
Gene array
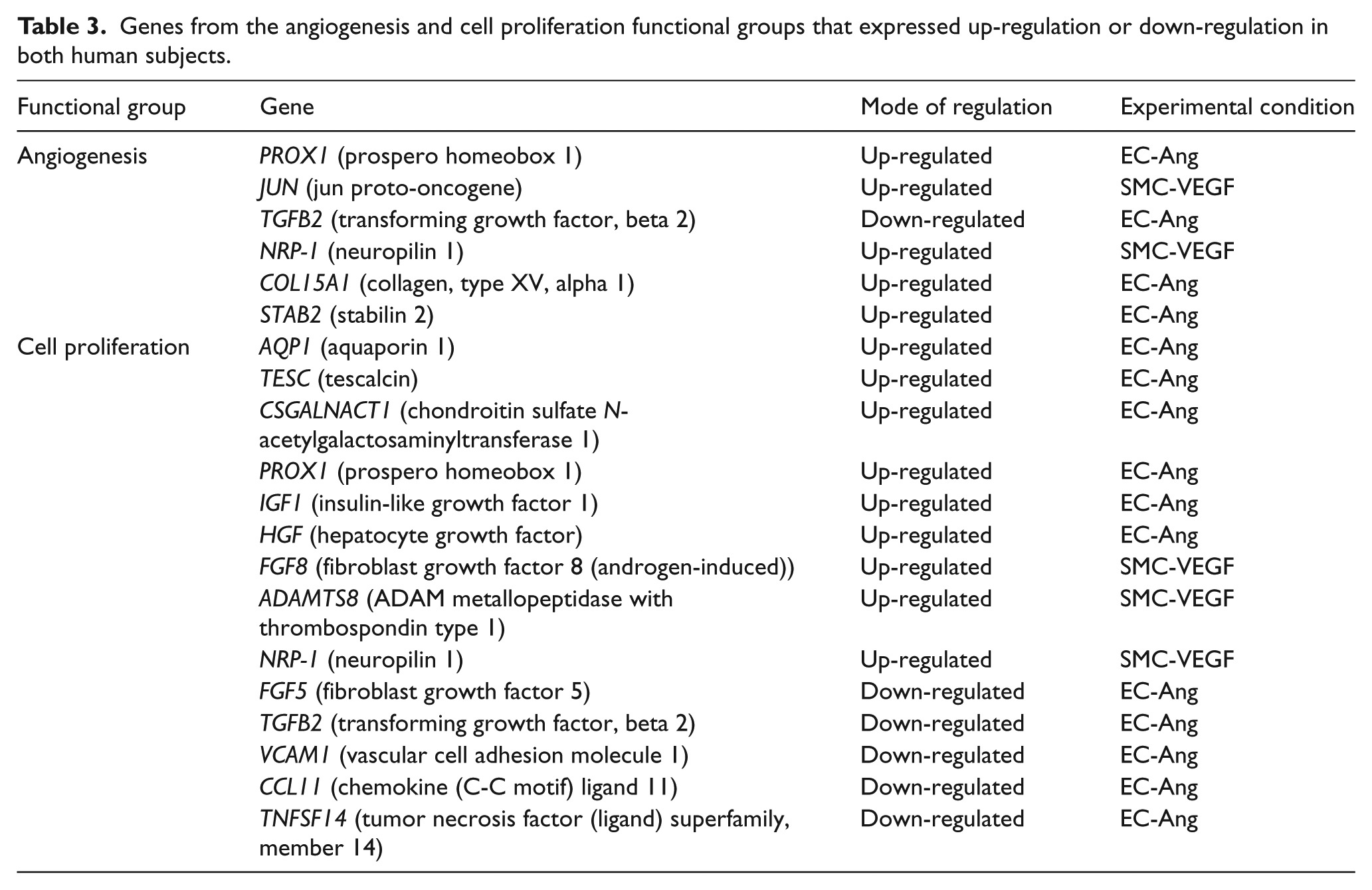
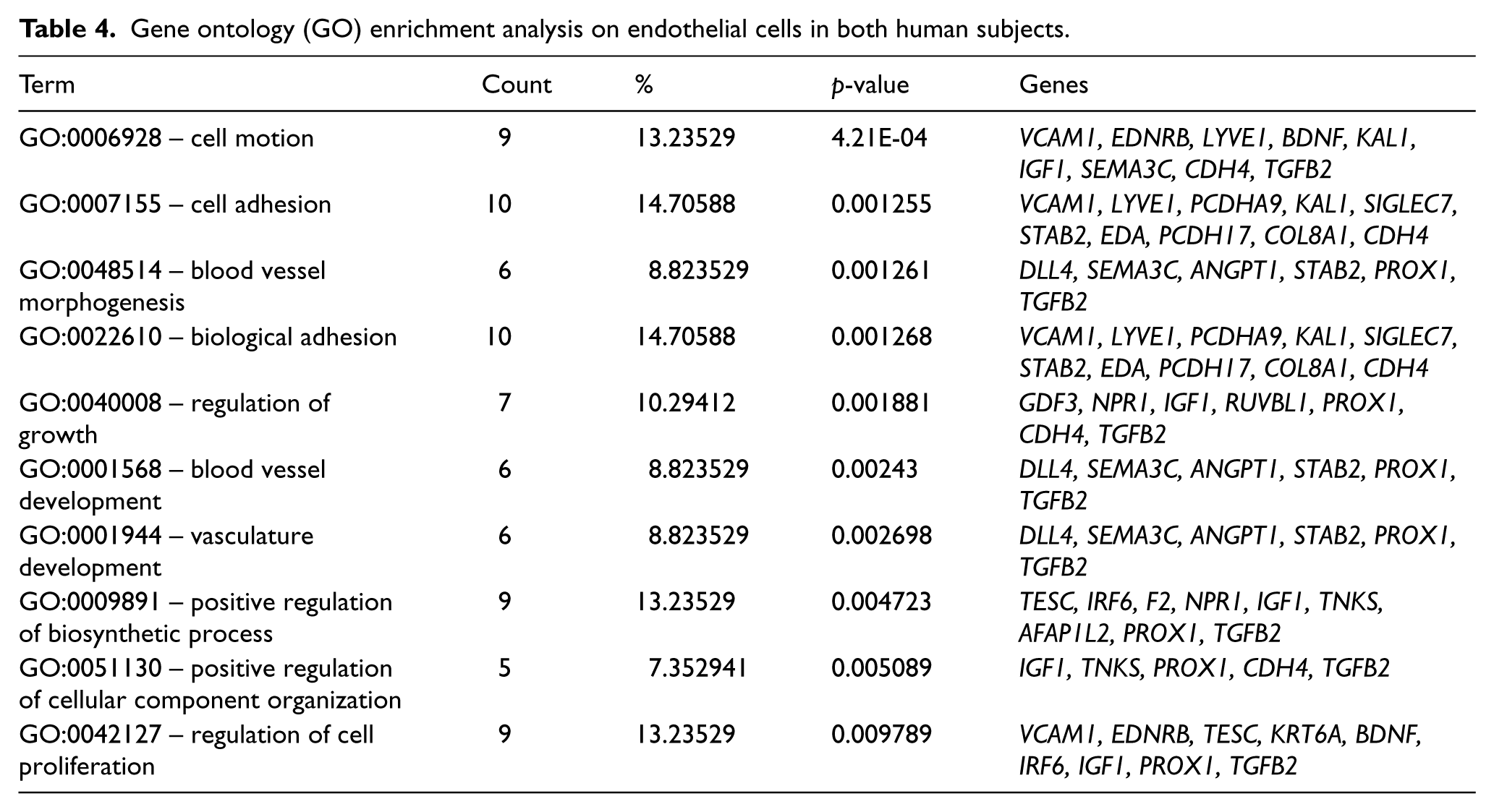
We examined the changes in gene expressions in the human ECs and SMCs transduced with therapeutic genes, Ang-1 and VEGF, respectively, and their functional categorization. Using the total cell RNA and the 4×44K human whole genome DNA microarray chip in conjunction with fold-change analysis (as explained in ‘Methods’), genome-wide global gene expression profiles were obtained for the gene-transferred cells. The results revealed 256 and 178 events of gene induction (⩾ 1.7 fold) as compared with 21 and 42 events of gene suppression (⩽ 0.75-fold) in EC-Ang-1 and SMC-VEGF cells, respectively, for Donor #1 and 166 and 143 events of gene induction as compared with 92 and 93 events of gene suppression in EC-Ang-1 and SMC-VEGF cells, respectively, for Donor #2. We next categorized the affected genes on the basis of their function under GO terms of biological process. 13 We have chosen to focus on the angiogenesis and cell proliferation functional groups and to examine closely the gene expression patterns in these two groups. Genes that were commonly up-regulated or down-regulated in both donors in each functional group were selected as candidate genes for further examination. The list of the candidate genes is provided in Table 3. Further on we have subjected the ratio profiles to enrichment analysis, which exhibited certain enrichment patterns in GO terms such as cell motion, cell adhesion, blood vessel morphogenesis, blood vessel development, regulation of growth and biological adhesion. Only terms containing five or more genes (p-value ⩽ 0.01) were considered as significant. In ECs, 10 such terms were identified, as represented in Table 4. Interestingly, several genes (such as PROX1, TGFB2, STAB2, VCAM1), which were identified as significantly regulated in the angiogenesis and cell proliferation functional grouping, also appeared to significantly enrich certain angiogenesis-related terms. Moreover, the data obtained in this study corresponds with an earlier study performed by our group using the Human Angiogenesis RT 2 ProfilerTM PCR Array. In EC-Ang-1 cells, the DLL4, IGF-1, ID-4, TGF-β and TNF superfamily representative exhibited a similar mode of regulation (data not shown). Using the GO software in SMC transduced to express VEGF we found no activated pathways.
Genes from the angiogenesis and cell proliferation functional groups that expressed up-regulation or down-regulation in both human subjects.
Gene ontology (GO) enrichment analysis on endothelial cells in both human subjects.
Clinical trial
Patient demographics
A total of 12 patients were successfully enrolled at two centers. All subjects were male, and their mean age was 60 ± 5 years. Historical factors included diabetes (25%), hyperlipidemia (92%), hypertension (92%), coronary artery disease (CAD) (75%), chronic obstructive pulmonary disease (COPD) (33%), and smoking history (92%). Seven of the subjects were classified as Rutherford 2 and five subjects as Rutherford 3 criteria. 23
Treatment safety and serious adverse events
There were four serious adverse events reported. One subject, with a prior history of left ventricular (LV) thrombus and thromboembolic event, had a recurrent transient ischemic attack related to LV thrombus 7 months after MGA treatment. A second subject had a non-index external iliac artery occlusion due to an obstructive dissection of iliac artery related to the catheterization procedure occurring 10 days after treatment. A third subject had an acute index leg ischemia due to a thrombus occluding the iliac artery 6 months after treatment. This patient had a mechanical aortic valve and it was thought the serious adverse event was related to inadequate warfarin management and embolization from the mechanical valve. A fourth subject suffered a myocardial infarction requiring a coronary artery bypass graft occurring 6 months after treatment. The second event described above was thought possibly related to treatment, as confirmed by the principal investigators, medical monitor and data safety monitoring board. The replication competent retrovirus (RCR) was negative in all patients at least 3 months after treatment (data not shown). The VEGF and Ang-1 plasma protein levels remained within normal ranges in all subjects throughout the study (Figures 3a and 3b).
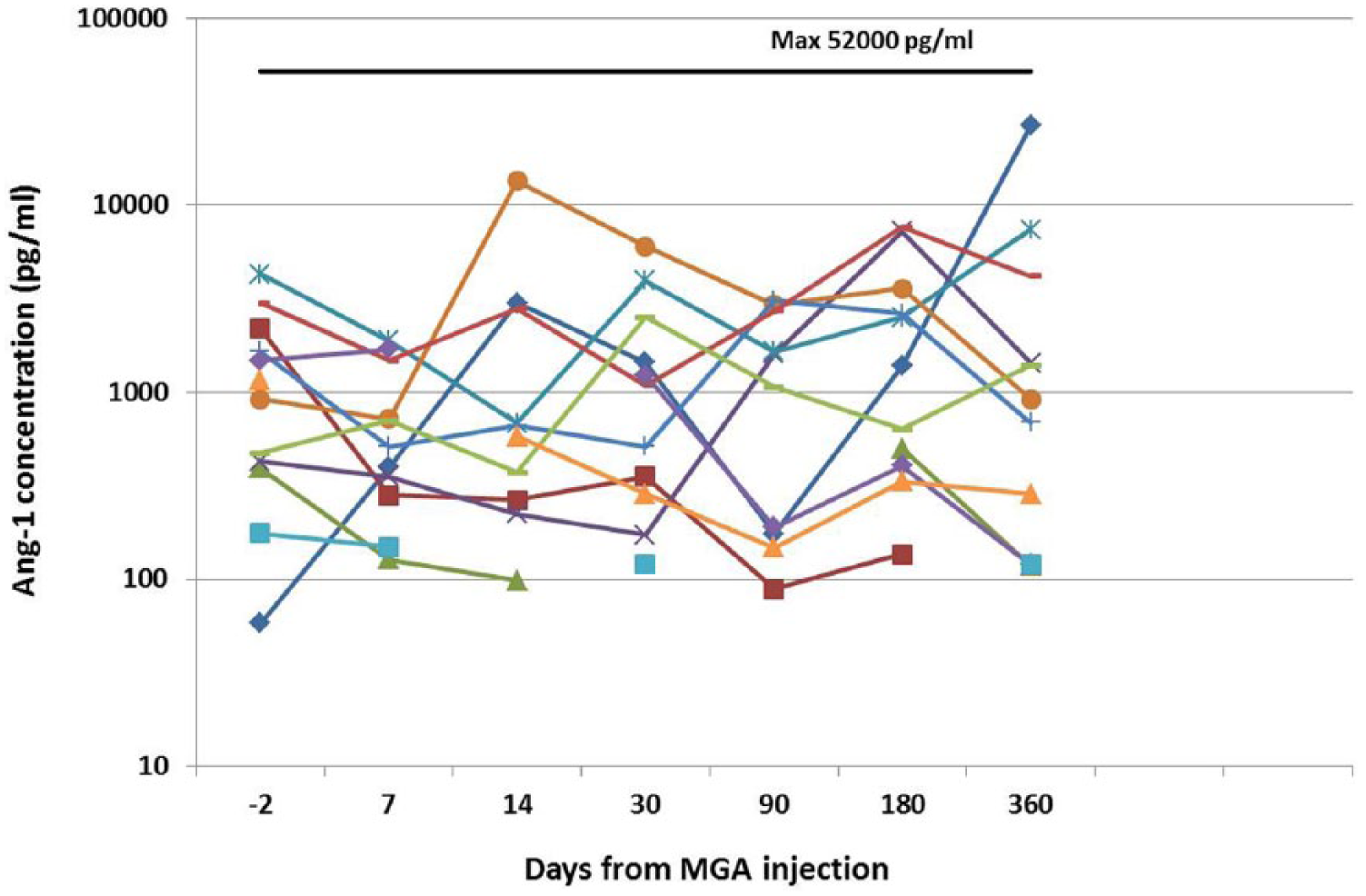
Transprotein plasma levels for each subject at baseline and at various time points after MultiGeneAngio (MGA) treatment for angiopoetin-1 (Ang-1).
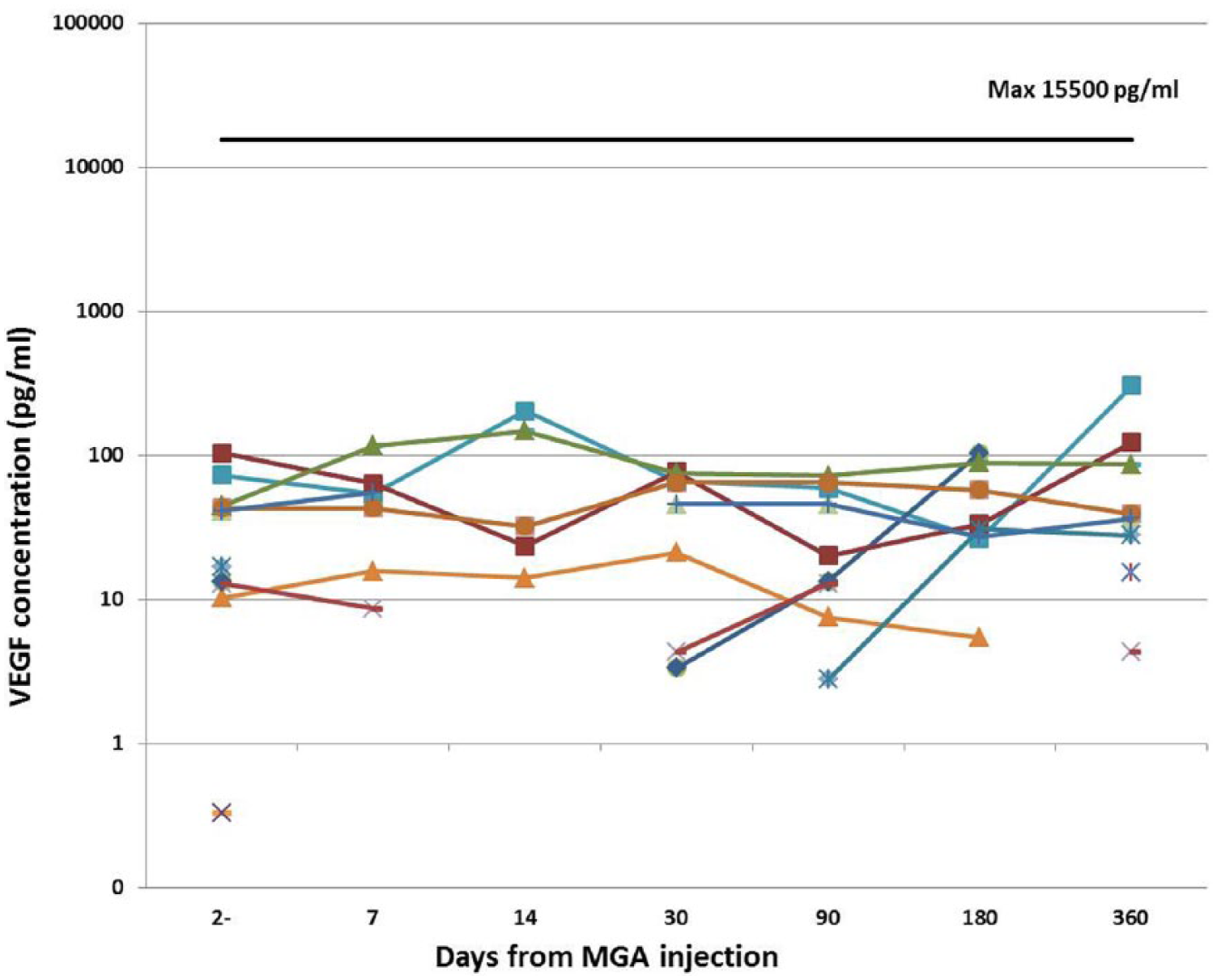
Transprotein plasma levels for each subject at baseline and at various time points after MultiGeneAngio (MGA) treatment for vascular endothelial growth factor (VEGF).
Functional safety measures
Subjects underwent a graded treadmill study (Gardner protocol) at screening, day 30, day 90, day 180 and day 365. The mean COT (Supplement Figure 1) and PWT (Supplement Figure 2) increased (p<0.05) at all of the follow-up time points. The ABI did not decrease through the course of the study (Supplement Figure 3). We did not observe a dose–response relationship between cell concentration and changes in PWT as may be expected in such a small group of patients.
The SF-36v2 quality of life questionnaire was administered to the subjects. There was no change in the mean physical health sub-score or the mean mental health sub-score over time (data not shown). However, the claudication-related quality of life was increased according to the walking impairment questionnaire (WIQ) as seen with the mean walking distance score (Supplement Figure 4, p<0.05).
Discussion
This is the first clinical study in humans evaluating MGA as treatment for claudication in patients with PAD. MGA is an example of a combination of cell types conditioned with a ‘cell activation’ approach for manufacture of an autologous therapeutic angiogenesis agent. The results of this study demonstrate the feasibility of ex vivo manufacture of this investigational combination cell and gene therapy and that MGA appeared to be safe in this small cohort. The levels of circulating Ang-1 and VEGF were not significantly changed compared to baseline, indicating there was no toxic systemic effect as a result of the therapy. The functional safety measures were included to assess safety and were not designed to assess if clinically significant improvement occurred in response to therapy. Therefore, any change in PWT must be interpreted with caution in this non-randomized, Phase I study. This is particularly true in studies related to therapy for PAD, where a significant ‘placebo effect’ may occur. 24 Given the functional safety measures did not worsen with treatment supports a favorable safety profile for MGA treatment.
The angiogenic process fundamentally involves progenitor cells, resident ECs, SMCs and growth factors. These growth factors include VEGF, FGF, placental growth factor and Ang-1 that act in a coordinated fashion to recapitulate a multitude of steps that eventually result in the formation of a blood vessel. 7 Angiogenesis theoretically can be achieved through either delivery of recombinant protein, gene transfer or cell therapy approaches. 5 Clinical trials involving systemic administration of recombinant bFGF (FGF-2) or VEGF165 protein in subjects with myocardial ischemia have not demonstrated clinical benefit. 25 Similarly, studies in PAD with locally expressed single growth factors, delivery of pro-angiogenic protein or administration of transcription factors such as hypoxia-inducible growth factor (HIF-1α) have failed to show a significant clinical benefit.26–28 Although a variety of reasons have been cited for the negative nature of these trials, one of the common themes has been reliance on delivery of a single growth factor, often with approaches that do not allow sustained expression of the protein in the area where it is most required. Cell transfer offers an alternate approach that alleviates this concern. Not only do the cells provide a repository of growth factors, but also through engraftment of cells they actively participate in the development or expansion of blood vessels. Preliminary results from cell transfer approaches in critical limb ischemia have demonstrated improvements in limb perfusion and even limb salvage. 29 Gene array study confirms the activation of multiple genes relevant to blood vessel remodeling and formation in MGA cells. The timely activation of these genes is essential for the initiation and completion of arteriogenesis.
Use of fully differentiated vascular cells with limited doubling capacity and no telomerase activity increase the safety of this mode of cell-based therapy. The lack of cell machinery that may drive cancerous transformation after gene transfer with retroviral vectors make this treatment safe even after growth factor transfer. The fact that systemic transgenes expression was within known normal levels of VEGF and Ang-1 lends additional evidence to the safety of MGA.
The pseudo-typed retroviral vectors used for transduction in the formulation of MGA are highly infective to cells expressing the CD34 antigen.30,31 With the very high transduction rates, the time-consuming process of selection with G418 is unnecessary. The transduced cells expressed high amounts of VEGF165 or Ang-1. Viral vector batches were tested and proved to be free of wild-type viruses. The oncogenic potential of these gamma retroviral vectors was implied in patients with severe immune deficiency in which bone marrow cells were transduced using these vectors. 32 Importantly, MGA is composed of fully differentiated cells with no telomerase activity and limited life span and therefore the likelihood of oncogenic transformation of the cells after gene transfer is extremely low.
In many ways, angiogenesis offers a great opportunity to validate the utility of cell transfer approaches. The advantages of using PAD for the initial testing field of angiogenesis and cell transfer approach include: (a) easy access to afflicted tissue; (b) simple delivery strategies through direct access to a peripheral vessel or intra-muscular injection; (c) amplification of the angiogenic response secondary to the ischemic up-regulation of the receptors and growth factors such as VEGF in hypoxic areas; and (d) measurable and reproducible end points such as walking time that is directly related to the severity of disease.
Owing to the complexity of the angiogenic process, use of more than one angiogenic gene for therapeutic angiogenesis may yield better induction of collaterals. 33 Dual gene transfer (VEGF and Ang-1) was shown in a rabbit hind limb ischemic model to be more effective than either gene alone in inducing angiogenesis. 34 Cell transfer or ‘activation’ approaches that focus on delivery of multiple growth factors along with cells are theoretically more likely to recapitulate the endogenous angiogenic process. This Phase I clinical trial is a first step in testing the concept of cell activation to promote angiogenesis in patients with PAD.
In conclusion, MGA, an autologous, transduced, dual cell-based therapy, was well tolerated and safe in this Phase I study. The therapy was not associated with any worsening of vascular function and safety measures. The results of this study warrant further randomized human studies of MGA for the management of severe PAD.
Footnotes
Acknowledgements
We thank Elizabeth Medenilla and the staff of the Clinical Cell and Vaccine Production Facility at the Hospital of the University of Pennsylvania and the University of Michigan for their assistance. We thank MultiGene Vascular Systems employees for their dedication and high quality work. We thank the Data and Safety Monitoring Board (DSMB) members of the study for their invaluable contribution: Eithan Galun, MD – Chair of the DSMB, Sam and Ellie Fishman Chair in Gene Therapy, and Director – Goldyne Savad Institute of Gene Therapy, Hadassah Hebrew University Hospital, Jerusalem, Israel; Bradley Strauss, MD, PhD, Sunnybrook Health Sciences Center, Toronto, Canada; and Robert J Lederman, MD, National Heart, Lung, and Blood Institute NIH, Bethesda, MD, USA.
Declaration of conflicting interest
Dr Flugelman is the co-founder and CTO of MultiGene Vascular Systems Ltd, Haifa, Israel. The other authors have nothing to disclose.
Funding
This research was supported by National Institutes of Health Grant UL1TR000433 and by MGVS.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

