Abstract
Portal vein thrombosis is an unusual thrombotic condition not frequently seen in the general population; however, it has a higher prevalence in special circumstances such as in liver cirrhosis and hepatic or pancreatic malignancy. It also can be associated with significant morbidity and mortality. In this review, we discuss the current data available to guide therapy in the setting of different associated co-morbidities, hypercoagulable states, and associated thrombosis of the remaining splanchnic circulation. We discuss indications for anticoagulation, including the choice of anticoagulants, as well as the role of conservative ‘wait and watch’ and invasive therapies, such as thrombolysis, thrombectomy, and transjugular intrahepatic portosystemic shunt.
Introduction
The portal vein is a vital vasculature providing up to 75% of blood supply to the liver. It originates posterior to the pancreas by the confluence of superior mesenteric and splenic veins, and, prior to entering into the liver, it divides into the left and right portal vein. 1 Thrombosis of the portal vein is an uncommon disorder comprising of thrombosis of the extrahepatic portion of the portal vein and/or its branches. It can occur concomitantly with mesenteric and/or splenic vein thrombosis.
Prevalence and epidemiology
Prevalence of portal vein thrombosis (PVT) ranges from 0.7 to 1/100,000 in the general population2–5; however, it is seen more commonly in certain conditions. Studies using radiographic techniques, mostly using ultrasound or computed tomography (CT), have reported the prevalence of PVT in liver cirrhosis from 2.1% to 16.2%; however, it is higher in studies evaluating the prevalence of PVT at the time of liver transplantation, ranging from 5.5% to 26%.4,6–11 One study evaluating patients listed for liver transplant found that 7.4% of patients developed de novo PVT after having an initial negative screening study. 5
Pathophysiology and etiology
Similar to venous thrombosis occurring in other locations such as the lower extremities, Virchow’s triad of hypercoagulability, endothelial injury, and stasis lead to PVT, although the etiology of these alterations is quite different. Local factors often contribute to PVT and these include liver cirrhosis, abdominal infections, abdominal inflammatory conditions (e.g. pancreatitis, cholecystitis), inflammatory bowel disease, abdominal malignancy (e.g. hepatocellular carcinoma, gastric and pancreatic adenocarcinomas), and portal vein injury (e.g. during splenectomy, surgical shunts, trauma).2,3,12–14 Inherited or acquired thrombophilia are also seen in up to 20% of individuals with PVT, and are seen more often in individuals with cirrhosis and PVT compared to those with only cirrhosis. 2 Inherited thrombophilia, such as factor V Leiden mutation and prothrombin G20210A gene mutation, have been associated with PVT. 15 Low levels of antithrombin and proteins C and S have been reported in cirrhotics; however, it is not certain if this is true inherited thrombophilia or acquired from liver disease. A meta-analysis showed that antithrombin and proteins C and S are not significantly related to the development of PVT in cirrhosis. 16 Interestingly, myeloproliferative disorders, specifically a JAK2V617F mutation, are strongly associated with splanchnic vein thrombosis.2,17 Also, antiphospholipid antibody syndrome, pregnancy, and hormonal therapies have been associated with PVT. 2 Table 1 summarizes the etiologies of PVT.
Etiology of portal vein thrombosis.
PVT, portal vein thrombosis; OR, odds ratio.
Clinical presentation
Acute PVT (Figure 1) can present variably, ranging from asymptomatic an incidental diagnosis on imaging to mild to moderate abdominal pain, nausea, and/or vomiting, to mesenteric ischemic symptoms with severe abdominal pain, hypotension, and even death. In a chronic setting (Figure 2), it can lead to signs and symptoms of portal hypertension such as esophageal varices with bleeding, splenomegaly, and ascites.
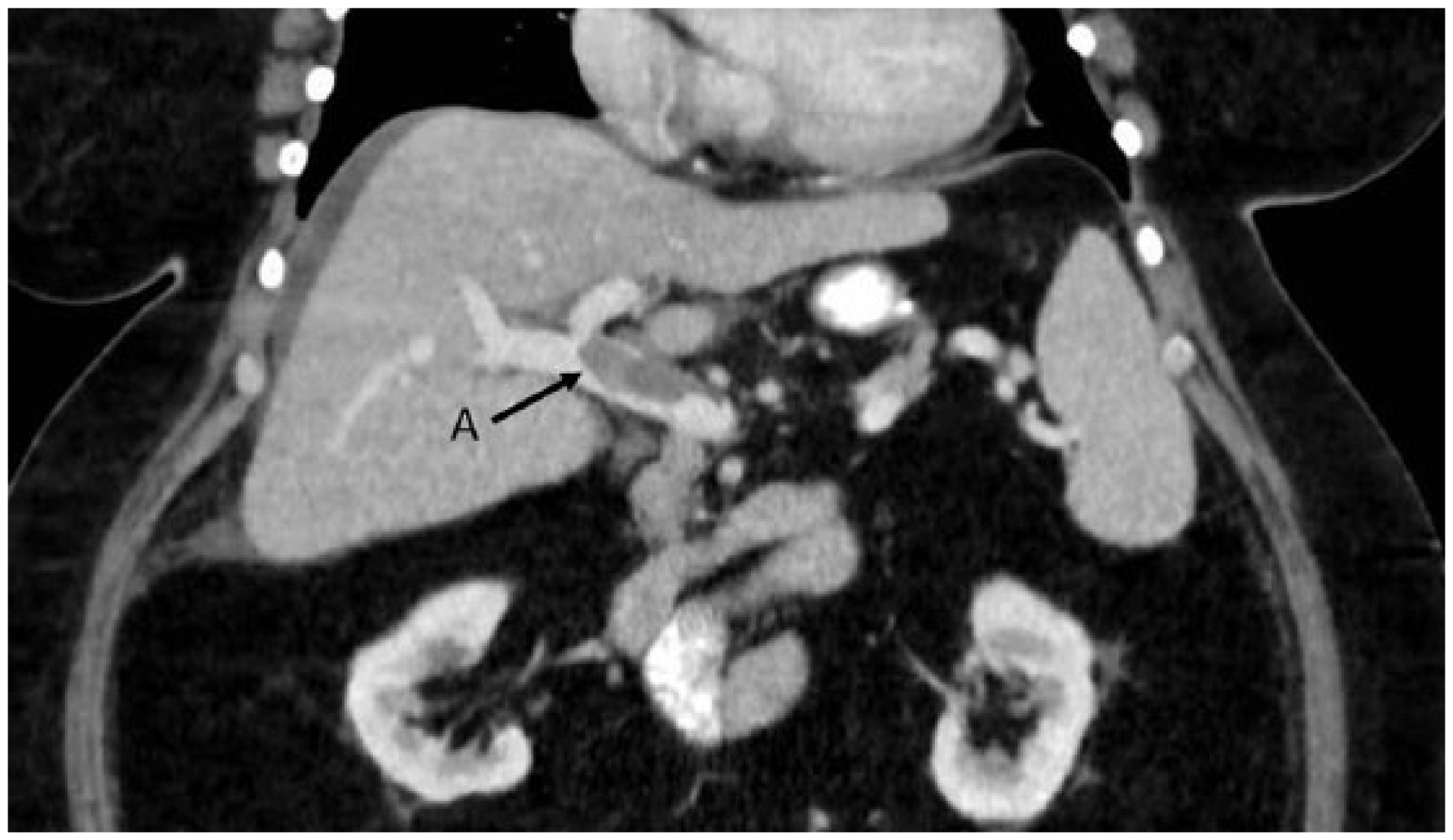
CT scan of acute portal vein thrombosis (PVT) in a 56-year-old female with non-alcoholic steatohepatitis (NASH) cirrhosis presenting with acute PVT one month after bariatric surgery. ‘
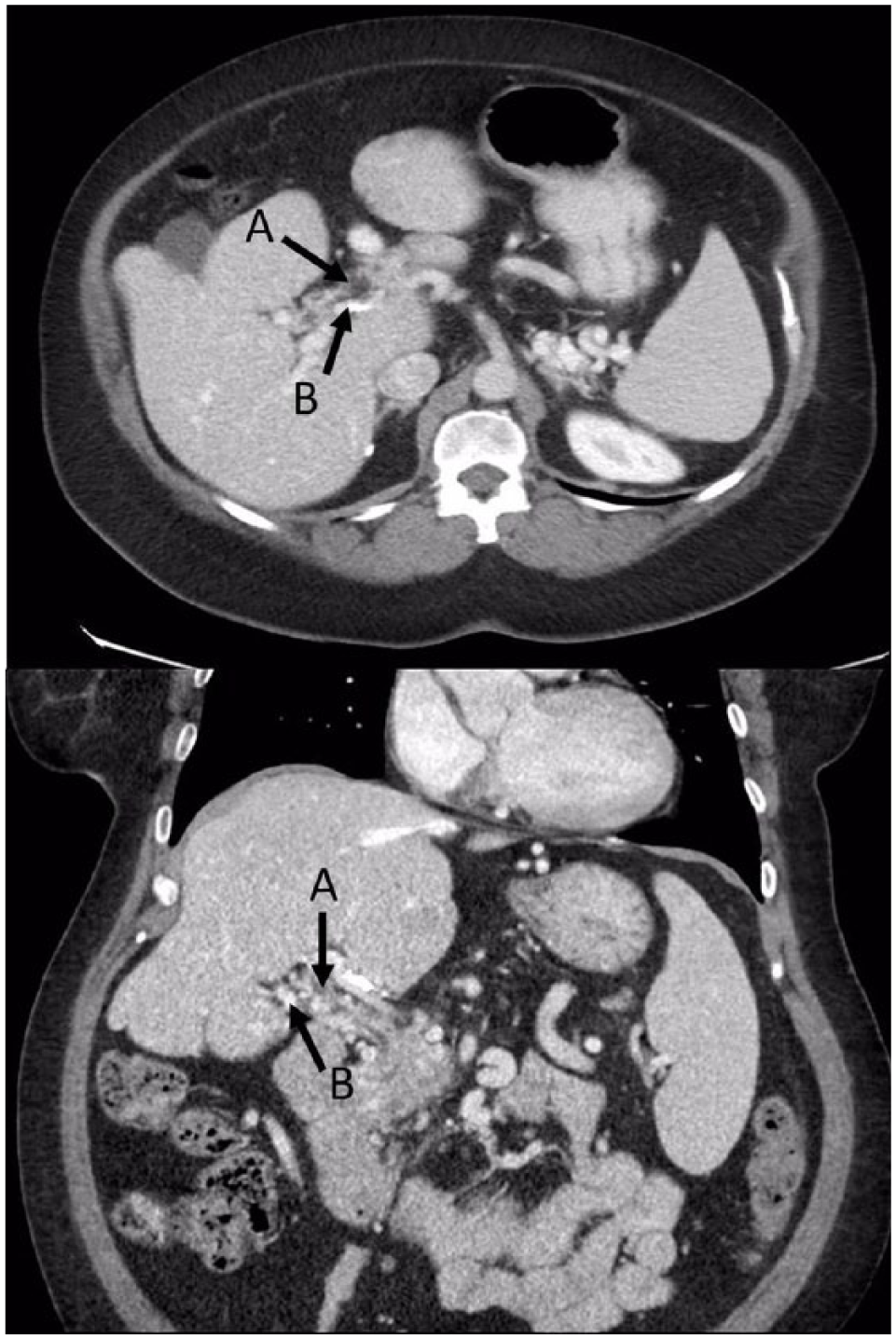
CT scan of coronal (top) and sagittal (bottom) views of chronic portal vein thrombosis (PVT) in a 56-year-old female with a history of cryptogenic cirrhosis complicated by chronic portal vein thrombosis and cavernous transformation. ‘
Treatment
Treatment of PVT currently ranges from observation with no active therapy to anticoagulation or thrombolytic therapy, thrombectomy, and transjugular intrahepatic portosystemic shunt (TIPS) placement. The goal for treatment consists of resolution of symptoms, prevention and treatment of mesenteric ischemia, and prevention of thrombus extension. Treatments and outcomes of acute PVT depend on the involvement of the remaining splanchnic circulation as well as associated factors such as liver cirrhosis or malignancy. We have further reviewed the literature on this disorder based on the presence of these factors.
Non-cirrhotic, non-malignant acute portal vein thrombosis
Currently the American College of Chest Physicians (ACCP) and American Association for the Study of Liver Disease (AASLD) have separate clinical guideline recommendations for treatment of PVT.18,19 The ACCP recommends anticoagulation for symptomatic PVT with a grade 1B level of evidence, which is a fairly scientifically robust recommendation; however, they do not suggest anticoagulation for asymptomatic, incidentally diagnosed PVT, which has a grade 2C level of evidence – a very weak recommendation given the lack of good data in this group of patients. 19 The AASLD clinical guidelines suggest anticoagulation for all acute PVT regardless of symptomatology. 18 There is clearly equipoise in the treatment of PVT, even between different thrombotic society guidelines.
PVT in non-cirrhotic and non-malignant individuals can occur in the setting of abdominal inflammatory processes such as pancreatitis, infections, and inflammatory bowel diseases, and after abdominal surgeries. Literature is very sparse regarding this group of PVT, and there are no randomized controlled trials. Baril et al. identified pylephlebitis (PVT in the setting of fever, leukocytosis, and/or positive blood cultures for micro-organisms) in 44 patients with PVT on CT scans. 20 Of these, 18 were hypercoagulable due to clotting factor deficiencies (6/18), malignancy (8/18), or AIDS (4/18). Fifteen patients had mesenteric vein thrombosis in addition to PVT. Five of the 32 cases that were not anticoagulated died compared to none in the remaining 12 patients who were anticoagulated. 20 Kanellopoulou et al. reviewed 100 case reports of pylephlebitis published since 1971, and reported that complete recanalization was higher in anticoagulated cases compared to those with no anticoagulation (25.7% vs 14.8%; p>0.05); no recanalization was also seen less commonly in those who were anticoagulated (5.7% vs 22.2%; p<0.05), with lower death rates (5.7% vs 22.2%; p<0.01). 21 All patients with pylephlebitis were treated with antibiotics. 21 In a meta-analysis of 315 patients by Hall et al., 12 were not anticoagulated and only two had spontaneous recanalization, and both of those patients had self-limiting conditions that provoked thrombosis. 22 Of the 228 cases that were anticoagulated, 38.3% had complete and 14% had partial recanalization of the portal vein. On follow-up, 20% demonstrated cavernous transformations, 47 patients developed esophageal varices, and upper gastrointestinal bleeding was seen in five cases, all of whom had not recanalized completely. Seventy-one patients underwent thrombolysis with or without thrombectomy (34 had failed a trial of anticoagulation), with a majority having an indication of increasing pain on anticoagulation or thrombus extension. 22 Complete recanalization occurred in 40.8% and partial recanalization occurred in 45.1%, and 14.1% remained occluded. 22 Procedure-related morbidity occurred in 21% of cases. Among 111 of the 315 patients in which long-term follow-up was available, 26 cases had complete recanalization and all of them had no complications of portal hypertension. Up to 50 cases had partial recanalization, of which four had varices, two had ascites, and one had a small bowel obstruction. 22
Acute portal vein thrombosis in the setting of liver cirrhosis
PVT in cirrhosis can have adverse outcomes as it is frequently associated with variceal bleeding. Liver cirrhosis in the past was considered to be a hypocoagulable state; however, this is a myth. Not only are thrombophilic disorders common in patients with cirrhosis, but there is also a unique balance of hemostasis related to cirrhosis that includes both hyper- and hypocoagulable states.8,23–25 In liver cirrhosis, not only are the procoagulant proteins (vitamin K-dependent proteins as well as factor V) reduced but the anticoagulant proteins (protein C and S) are impaired, often in equilibrium, thereby creating a hemostatic balance in the majority of cases.8,26–28 One significant difference in cirrhosis as compared to the effects of vitamin K deficiency seen with warfarin use is that factor VIII and von-Willebrand factor are elevated in patients with cirrhosis, creating a potential procoagulant milieu at times.28,29
As noted previously, patients with cirrhosis have a relatively high risk of PVT compared with the general population. Regrettably, there is a significant lack of strong evidence in treating PVT in the setting of cirrhosis. There are no randomized clinical trials, and most evidence in treatment comes from case series and observational studies. Up to half of the patients presenting with PVT in this setting can be asymptomatic and incidentally diagnosed. 30 Studies evaluating the natural history of PVT in the setting of cirrhosis noted that up to half of PVT cases in cirrhotic patients will show a reduction in thrombus burden, and the clinical outcomes do not always correlate with progression of PVT (Table 2).5,9,31–34 Studies have also shown that PVT can lead to worsening liver function and increased mortality in patients with cirrhosis, and the presence of PVT may negatively impact morbidity and mortality at the time of liver transplant.5,7,35
Case series of portal vein thrombosis in liver cirrhosis with and without anticoagulation.
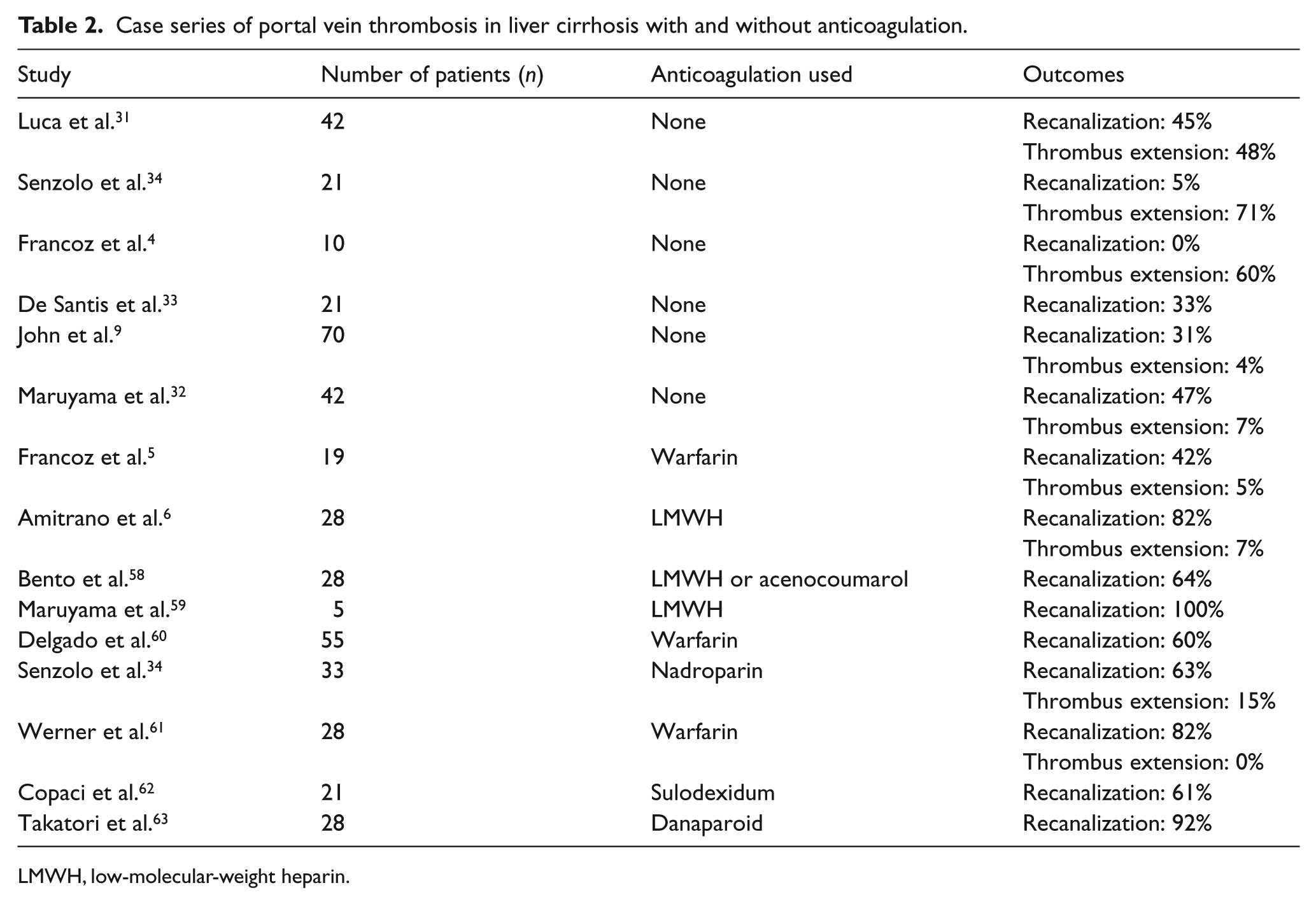
LMWH, low-molecular-weight heparin.
When considering whether or not to treat PVT in patients with cirrhosis, many factors should be taken into consideration: acuity of thrombosis, transplant candidacy, presence of an inherited coagulopathy, bleeding risk factors, and co-morbidities. There is ample data regarding the safety and efficacy of anticoagulation in patients with cirrhosis. Francoz et al. reported a higher incidence of partial or complete recanalization of thrombus burden with anticoagulation over no anticoagulation (p=0.002). 5 Amitrano et al. reported that with therapeutic doses of low-molecular-weight heparin (LMWH), complete recanalization was seen in 33% of patients and partial recanalization was seen in 50% of patients with no increased bleeding risk. This group had up to 50% of patients with variceal bleeding on presentation who received anticoagulation after appropriate treatment of their varices. 6 Several other studies have shown similar results as noted in Table 2. The hypothesis, although not yet proven, is that for patients with cirrhosis and PVT, treatment with anticoagulation and subsequent recanalization of portal vein may lead to decreased portal pressure, and thus, decreased risk of portal hypertensive bleeding. A study by Senzolo et al. utilizing therapeutic doses of LMWH for treatment of PVT showed a higher rate of variceal bleeding in patients not on anticoagulation (five) than those on anticoagulation (one). 34 Other modalities utilized in this situation include TIPS, which may be a reasonable adjunct for treatment. TIPS has shown to completely recanalize portal veins in up to 57% of cases; however, it is associated with a high incidence of low stent patency rates (bare stents: 85% in 2 years; covered stents: 29% in 2 years) as well as hepatic encephalopathy (32% in 2 years).4,36 Hence, the benefits of TIPS need to strongly weigh against the risks of hepatic decompensation, hepatic encephalopathy, and risk of embolization.30,31,34
Anticoagulation of patients with liver cirrhosis can be complicated. Warfarin is often difficult to manage since some of these patients have baseline elevated international nationalized ratio (INR) levels, and hence, the ideal INR goal to maintain a therapeutic range is difficult to assess. 27 INR also has only been validated in individuals with normal liver functions, and a study has shown 29% variation in mean INR levels in cirrhotic patients.37,38 In addition, often it is difficult to maintain stable INR levels in these patients due to a fragile hemostatic environment. LMWH is an excellent alternative to warfarin that is safe and efficacious for these patients.6,34 Direct oral anticoagulants (DOACs) such as apixaban, dabigatran, and rivaroxaban have not been studied well in this patient population. Case series using rivaroxaban and apixaban have been reported; however, larger studies are required prior to large-scale utilization of these agents in these cases given the increased gastrointestinal bleeding risk with certain DOACs and their metabolism through the cytochrome P450 3A4 pathway, which may be impaired in cirrhotic patients.39–41
Acute portal vein thrombosis in the setting of malignancy
PVT is identified in 12.5–39.7% of patients with hepatocellular carcinoma (HCC) and up to 20% of patients with pancreatic adenocarcinoma.42–47 PVT is an independent predictor of increased mortality among patients with HCC. Untreated HCC with PVT was reported to be associated with a median survival time of 2.7–4 months.48,49 Lee et al., in the CLOT trial (Randomized Comparison of Low-Molecular-Weight Heparin versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Throm-boembolism in Patients with Cancer), demonstrated that patients with malignancy have an increased risk of recurrent deep vein thrombosis or pulmonary embolism even when treated with warfarin compared to LMWH. 50 Owing to the difficulty in monitoring INR appropriately, especially in cirrhosis associated with malignancy, and extrapolating data from the CLOT trial, it is often recommended to use LMWH rather than warfarin in treatment of thrombosis in the setting of malignancy. 51 DOACs have not yet been studied in the setting of malignancy, and the American Society of Clinical Oncology clinical practice guidelines recommend against the use of DOACs in this setting. 51 Recently, Schulman et al. published pooled data from two clinical trials that included patients with acute thrombosis and malignancy using warfarin or dabigatran and found similar symptomatic recurrent VTE and related death rates between the two groups (hazard ratio 0.63; 95% confidence interval, 0.20–2.0) as well as similar bleeding rates; however, dabigatran was not compared to LMWH. 52
Portal vein thrombosis with and without involvement of other splanchnic veins
Outcomes vary significantly based on anatomic involvement of thrombosis. Thatipelli et al. reported a 10-year survival rate of 60% in patients with splanchnic vein thrombosis, with independent predictors being older age, cancer, and myeloproliferative disorders. 53 Interestingly, patients with isolated segment thrombosis compared to multisegment thrombosis had a better survival rate (68% vs 48%, respectively; p < 0.001) and lower recurrence rate (80% vs 64%). 53 Isolated portal vein thrombosis had the lowest survival rate in this case series; however, this was driven by a higher incidence of malignancy in these patients. 53
How do we treat acute portal vein thrombosis?
Management of PVT involves complex decision making, taking into consideration the involvement of other splanchnic veins apart from portal vein involvement; hypercoagulable states; presence or absence of local factors including inflammatory disorders, infections, and cirrhosis; and active malignancy and bleeding risk based on the presence of varices, platelet count (may be low in cirrhosis), or other factors. Often a multidisciplinary approach involving a vascular medicine or thrombosis specialist, gastroenterologist, interventional radiologist, and colorectal surgeon is necessary to provide optimal treatment options for these patients. We have provided recommendations and our approach to these patients below and in Tables 3 and 4.
Summary of treatment approach to acute portal vein thrombosis.
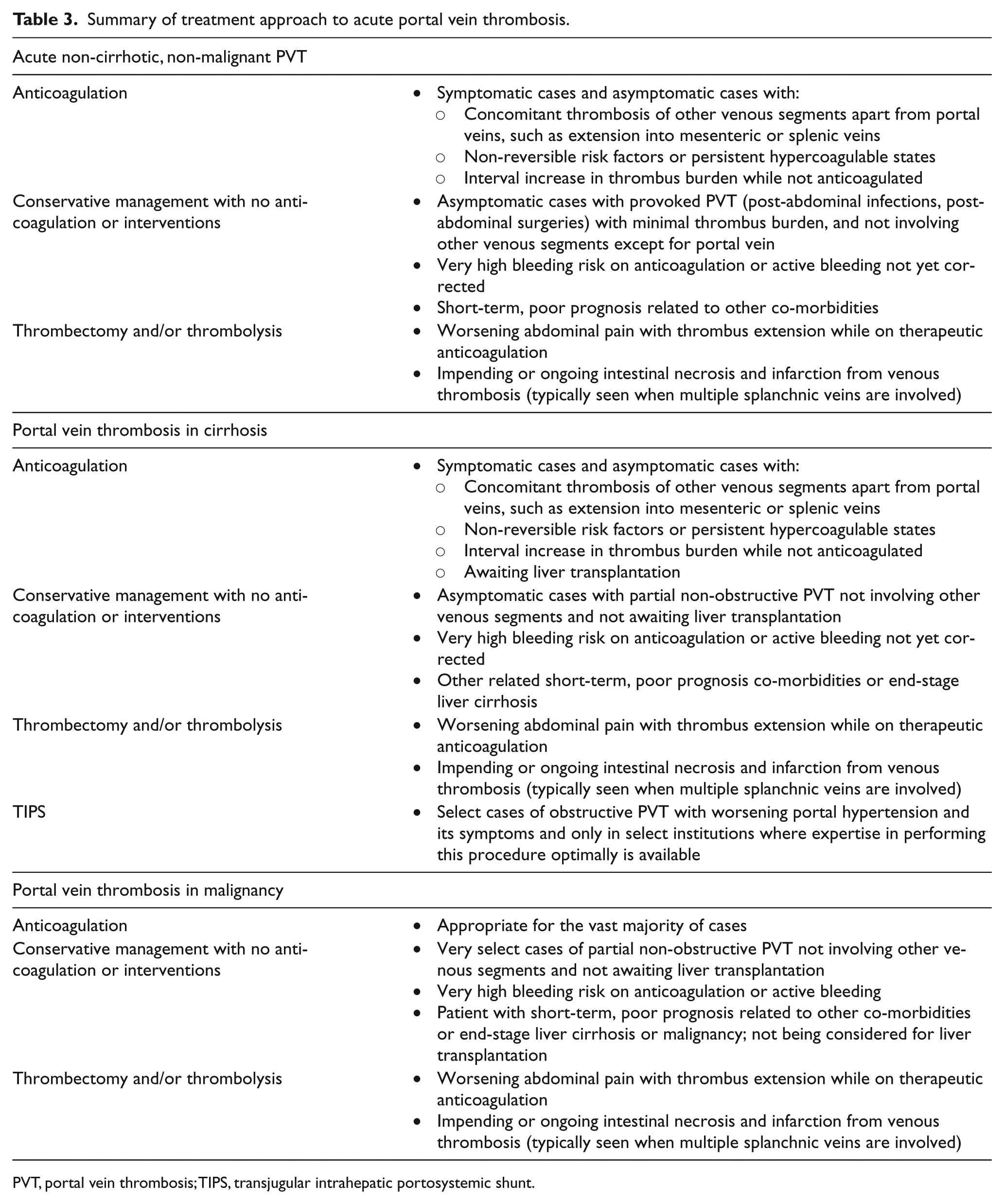
PVT, portal vein thrombosis; TIPS, transjugular intrahepatic portosystemic shunt.
Antiphospholipid antibodies, double heterozygosity for factor V Leiden mutation and G20210A mutation in prothrombin gene or single homozygosity.
PVT, portal vein thrombosis; LMWH, low-molecular-weight heparin; DOAC, direct oral anticoagulant.
In cases of non-cirrhotic, non-malignant, symptomatic PVT, anticoagulation is recommended. For asymptomatic PVT, especially with extension to the mesenteric veins, non-reversible risk factors, hypercoagulable states, and interval increase in thrombus burden, anticoagulation is reasonable to prevent or reduce symptoms such as abdominal pain, nausea or vomiting, or ischemic complications like bowel necrosis and portal hypertension-related symptoms. It is also reasonable to monitor conservatively without anticoagulation in the setting of reversible risk factors such as pancreatitis or abdominal infections in patients with minimal thrombus burden in portal veins without extension into the other splanchnic veins. Thrombolytic therapy with or without thrombectomy can be considered in those with thrombus extension or worsening pain while on therapeutic anticoagulation and in patients with impending or ongoing bowel necrosis due to thrombosis. When anticoagulation is pursued, warfarin is frequently the anticoagulant of choice, although LMWH can be safely considered as an alternative. DOACs such as rivaroxaban, apixaban, or dabigatran have been well studied for use in deep venous thrombosis involving the lower extremities and pulmonary embolism; however, their use has not been studied in this patient population.54–56 Extrapolating the data from those trials, these agents can be considered in these patients in the absence of any liver disorders.
In cases of PVT in the setting of liver cirrhosis, it would be reasonable to anticoagulate the majority of these patients. Anticoagulation should be considered strongly when a thrombus extends or is also present in other splanchnic veins, when there is a persistent hypercoagulable state, or for patients listed for transplant. Typically, LMWH is the anticoagulant of choice given its overall safety and efficacy in these patients; however, it can be difficult to administer with concerns of distribution in patients with severe edema and ascites since absorption subcutaneously may not be optimal in sites with interstitial edema. 57 In such cases, careful attention to the location of administration of LMWH and monitoring of anti-Xa levels may be considered. Warfarin is also an alternative; however, measurement and goal INRs can be unreliable for reasons noted above.37,38 DOACs are generally not recommended due to a lack of large-scale studies and should only be considered under the guidance of a thrombosis or vascular medicine specialist. Conversely, observation with no anticoagulation may be reasonable for an incidental diagnosis of mild partial non-obstructive PVT with no mesenteric or splenic vein involvement in a patient not on the transplant list or in those with active bleeding or a high bleeding risk. Lastly, TIPS can be considered in cases with occlusive PVT with worsening portal hypertension and esophageal varices in centers where expertise is available. Given the high incidence of hepatic encephalopathy and low patency rates, a decision to pursue TIPS should be well evaluated in a multidisciplinary fashion.
In patients with PVT in the setting of malignancy, anticoagulation is recommended in the majority of cases with the exception of cases of minimal thrombus burden, active bleeding, or severe bleeding risk. When anticoagulation is pursued, LMWH is the preferred agent of choice given the data of superiority in preventing recurrent thrombosis compared to warfarin. Recently, a study comparing warfarin to dabigatran in cancer patients showed equivalent clinical benefits from both agents. 51 Hence, in patients who are unable to be on LMWH, warfarin would be the next choice of agent; however, if unable to take warfarin, a DOAC can be considered.
Conclusion
Portal vein thrombosis continues to be a complicated and difficult condition with significant morbidity and mortality. Outcomes are more severe in cirrhotic patients and those with malignancy. Although there has been some progress in understanding the pathophysiology and treatment options, there is still a lack of randomized clinical data to guide therapy. For the majority of individuals with acute PVT, anticoagulation is the preferred method of treatment, with fewer cases managed conservatively (‘wait and watch’) or aggressively with TIPS, thrombolysis, or thrombectomy. With the availability of DOACs, there is an increasing need for randomized clinical data to guide treatment of this condition.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.