Abstract
We evaluated the impact of the prescription of evidence-based medical therapy (EBMT) including aspirin (ASA), beta-blockers (BB), ACE-inhibitors or angiotensin receptor blockade (ACE/ARB), and statins prior to discharge after peripheral vascular intervention (PVI) on long-term medication utilization in a large multi-specialty, multicenter quality improvement collaborative. Among patients undergoing coronary revascularization, use of the component medications of EBMT at hospital discharge is a major predictor of long-term utilization. Predictors of EBMT use after PVI are largely unknown. A total of 10,169 patients undergoing PVI between 1 January 2008 and 31 December 2011 were included. Post-PVI discharge and 6-month medication utilization in patients without contra-indications to ASA, BB, ACE/ARB, and statins were compared. ASA was prescribed at discharge to 9345 (92%) patients, BB to 7012 (69%), ACE/ARB to 6424 (63%), and statins to 8342 (82%), and all four component drugs of EBMT in 3953 (39%). Compared with patients not discharged on the appropriate medications, post-procedural use was associated (all p<0.001) with reported 6-month use: ASA (84.5% vs 39.2%), BB (82.5% vs 11.1%), ACE/ARB (78.2% vs 11.8%), statins (84.6% vs 21.8%). Multivariable analysis revealed that prescription of EBMT at the time of discharge remained strongly associated with use at 6 months for each of the individual component drugs as well as for the combination of all four EBMT medications. In conclusion, prescription of the component medications of EBMT at the time of PVI is associated with excellent utilization at 6 months, while failure to prescribe EBMT at discharge is associated with low use of these medications 6 months later. These data suggest that the time of a PVI is a therapeutic window in which to prescribe EBMT in this high-risk cohort and represents an opportunity for quality improvement.
Keywords
Introduction
Lower-extremity peripheral artery disease (PAD) results from narrowing of the blood vessels of the lower limbs, predominately secondary to atherosclerotic vascular disease. 1 The prevalence of lower-extremity (LE) PAD is increasing. 2 Patients with PAD are at increased risk of cardiovascular morbidity and mortality. 2 Risk factors associated with LE PAD are similar to those for atherosclerotic vascular disease elsewhere, and include age, smoking, diabetes mellitus, hyperlipidemia, and hypertension. 3 Evidence-based medical therapy (EBMT) is of crucial importance in reducing disease burden and limiting cardiovascular morbidity and mortality in this patient population.2,4,5
Although EBMT is proven to lower cardiovascular morbidity and mortality and improve vascular outcome, use of this medical therapy is often suboptimal.1,5 Reasons for lack of utilization are multifactorial. Previous literature has suggested three important factors for lack of utilization: patient-related, physician-related, and healthcare system-related. 1 Patient-related factors include patient understanding and acceptance of the seriousness of their disease, especially in the asymptomatic patient. Patient adherence can be increased through regular office visits, education, and the growing use of the electronic medical record that directly involves the patient in his or her care. Physicians from multiple disciplines are involved in the care of patients with PAD, and medical education and comfort with risk factor reduction and EBMT is necessary. In addition, healthcare systems can improve prescription of EBMT through organized systems of care and patient education. The purpose of this study was to evaluate the prescription of EBMT, including aspirin (ASA), beta-blocker (BB), ACE-inhibitor or angiotensin receptor blocker (ACE/ARB), and statin medications, after LE peripheral vascular intervention (PVI) procedures in a contemporary, real-world population of patients with symptomatic LE PAD. In addition to prescription at the time of PVI, we explored factors that impact utilization of EBMT 6 months after the PVI procedure.
Methods
Our study population comprised patients who underwent LE PVI for symptomatic PAD between 1 January 2008 and 31 December 2011 at 47 hospitals participating in the Blue Cross Blue Shield of Michigan Cardiovascular Consortium Peripheral Vascular Intervention (BMC2 PVI) Registry. Data regarding EBMT use 6 months post-discharge were included in the analysis. Patient-reported medication utilization at 6 months was compared between those patients discharged on each drug (ASA or another antiplatelet, BB, either an ACE or ARB and statins) and patients not receiving the drug at discharge. The dose of medical therapy was not recorded, and, therefore, was not part of this analysis. Patients with contraindications documented in the medical record to ASA, BB or statins were excluded from the analysis; however, contraindications to both ACE and ARB were not identifiable. Contraindications to medications were not prescribed by the registry, but were required to be recorded in the medical record. These contraindications may have included medication allergy or intolerance.
Statistical analysis
Variability in EBMT prescription at discharge between hospitals was investigated using hierarchical logistic generalized linear mixed effects regression (GLMER) models allowing for a normally distributed hospital random effect and adjusting for baseline patient clinical and demographic characteristics as fixed effects. Posterior random effects mode and 95% confidence intervals are presented in a caterpillar plot to visually assess the extent of variability between facilities. Potential associations between individual patient clinical and demographic factors and EBMT prescription at discharge on patient-reported EBMT adherence at 6 months post-discharge were assessed using Student’s t-tests for continuous variables and Pearson chi-squared tests for categorical variables, as well as through inclusion as predictors in multivariable logistic regression models. R version 2.14 6 was used for all analysis, with the lme4 package 7 used for fitting hierarchical GLMER models.
Blue Cross Blue Shield of Michigan Cardiovascular Consortium Peripheral Vascular Intervention
Details of the construct, data collection, and data quality assurance for BMC2 PVI have been described previously. 8 BMC2 PVI is a prospective, multicenter registry of patients undergoing PVI by multiple operators at participating hospitals. It is designed to collect information on patients undergoing PVI in an effort to evaluate evidence-based disease management and to support collaborative improvement in quality of care and outcomes. BMC2 PVI is a physician-run collaborative that is supported by, but operates independently of, the funding agency – Blue Cross Blue Shield of Michigan™. A physician advisory committee is responsible for setting the quality goals and developing quality improvement efforts without any input from or sharing of data with the study sponsor. A data form is compiled for each PVI procedure, including demographic information, medical history, lab results (pre- and post-procedure), presenting symptoms, procedural indications, patient medications, PVI type, procedural details (including complications), and in-hospital outcomes. Data regarding patient status and medication use 6 months post-PVI is collected via review of the medical record and/or direct patient interview either in the outpatient clinic or via telephone. Data quality and the inclusion of consecutive procedures are contractually required and assured by ad hoc queries, random chart review, and a series of diagnostic routines. A list of standard definitions outlined by the American College of Cardiology Data Standards Committee has been used as a reference. The registry has been approved or the need for approval waived by the institutional review board of each participating hospital.
Data quality control
All data undergo a three-step validation process, including manual review for completeness and face validity, review of rejected data forms during the import process, and review of forms that fail diagnostic inquiries. All site data collection agents participate in ongoing education regarding the data definitions, and all sites are audited twice yearly by a nurse investigator from the coordinating center. In addition to examining cases in which an adverse event was recorded (including death, stroke, amputation, and transfusion), a random 5% sample of cases is audited for completeness and accuracy. The compliance and accuracy of data in the audits is >95%. Data that are found to be inaccurate or incomplete are corrected and feedback is provided to the responsible centers.
Results
Of 15,305 PVI procedures performed between 1 January 2008 and 31 December 2011, 1201 (7.8%) were performed in patients with documented contraindications to ASA, BB or statins and were excluded from this analysis. Of the remaining 14,104 procedures, follow-up data on medical adherence at 6 months post-PVI was available for 10,169 patients (72.1%), and this subset was utilized for analysis. Clinical characteristics are shown in Table 1. Of the 10,169 PVI patients in the analysis, 9345 (91.9%) were prescribed post-procedural ASA, 7012 (68.9%) were prescribed post-procedural BB, 6424 (63.2%) were prescribed ACE/ARB, 8342 (82.0%) were prescribed statins, and 3953 (38.9%) were prescribed all four prior to discharge (all EBMT). Table 2 compares baseline characteristics for patients prescribed and not prescribed all EBMT prior to discharge. Patients prescribed all EBMT were less likely to be current smokers and more likely to have a number of comorbidities including hypertension, diabetes, congestive heart failure (CHF), valve disease, cerebrovascular disease, atrial fibrillation, and renal failure.
Baseline characteristics of patients at the time of peripheral vascular intervention (PVI) and at 6-month follow-up.
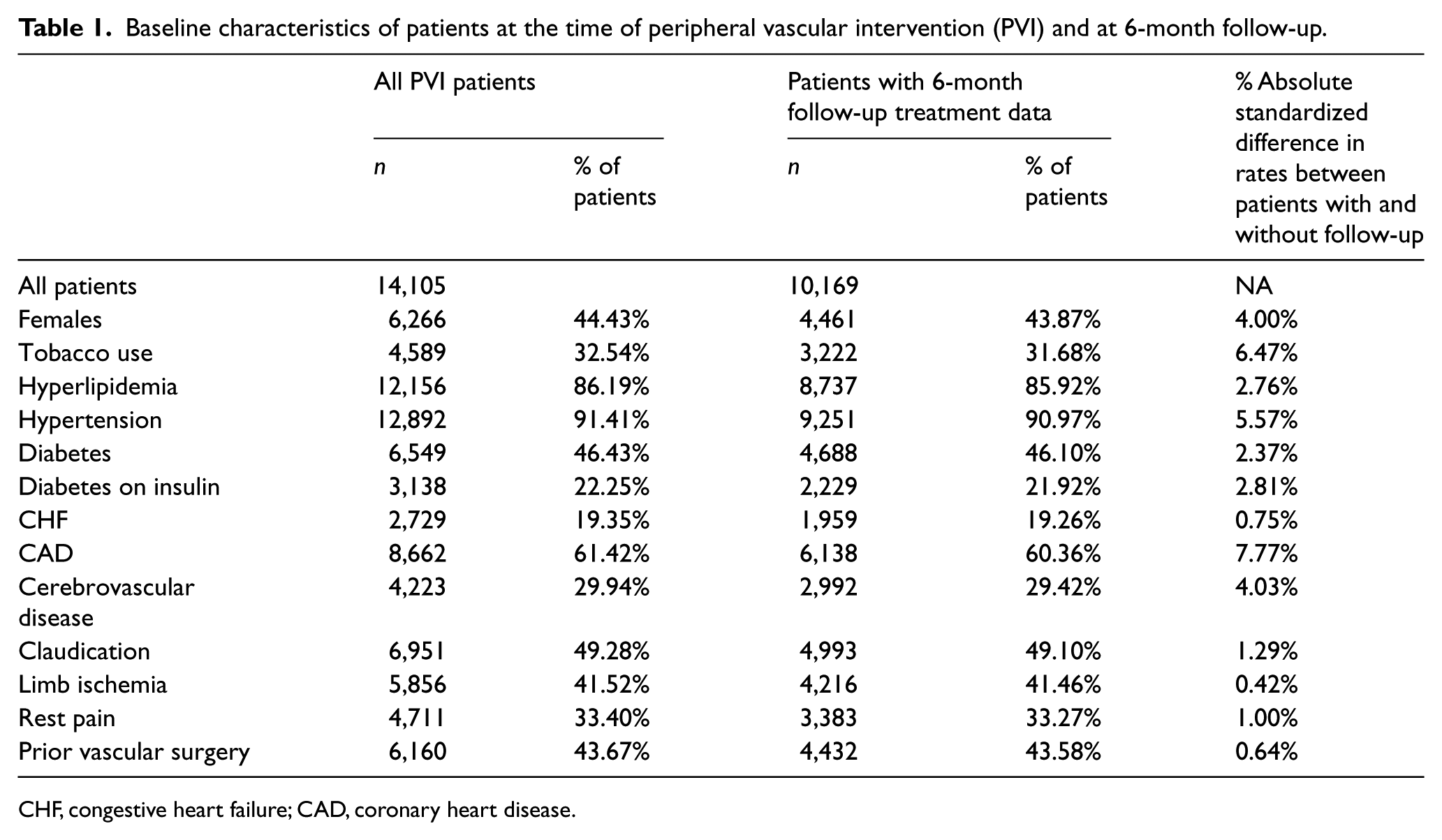
CHF, congestive heart failure; CAD, coronary heart disease.
Baseline characteristics for patients prescribed and not prescribed evidence-based medical therapy (EBMT) prior to discharge.
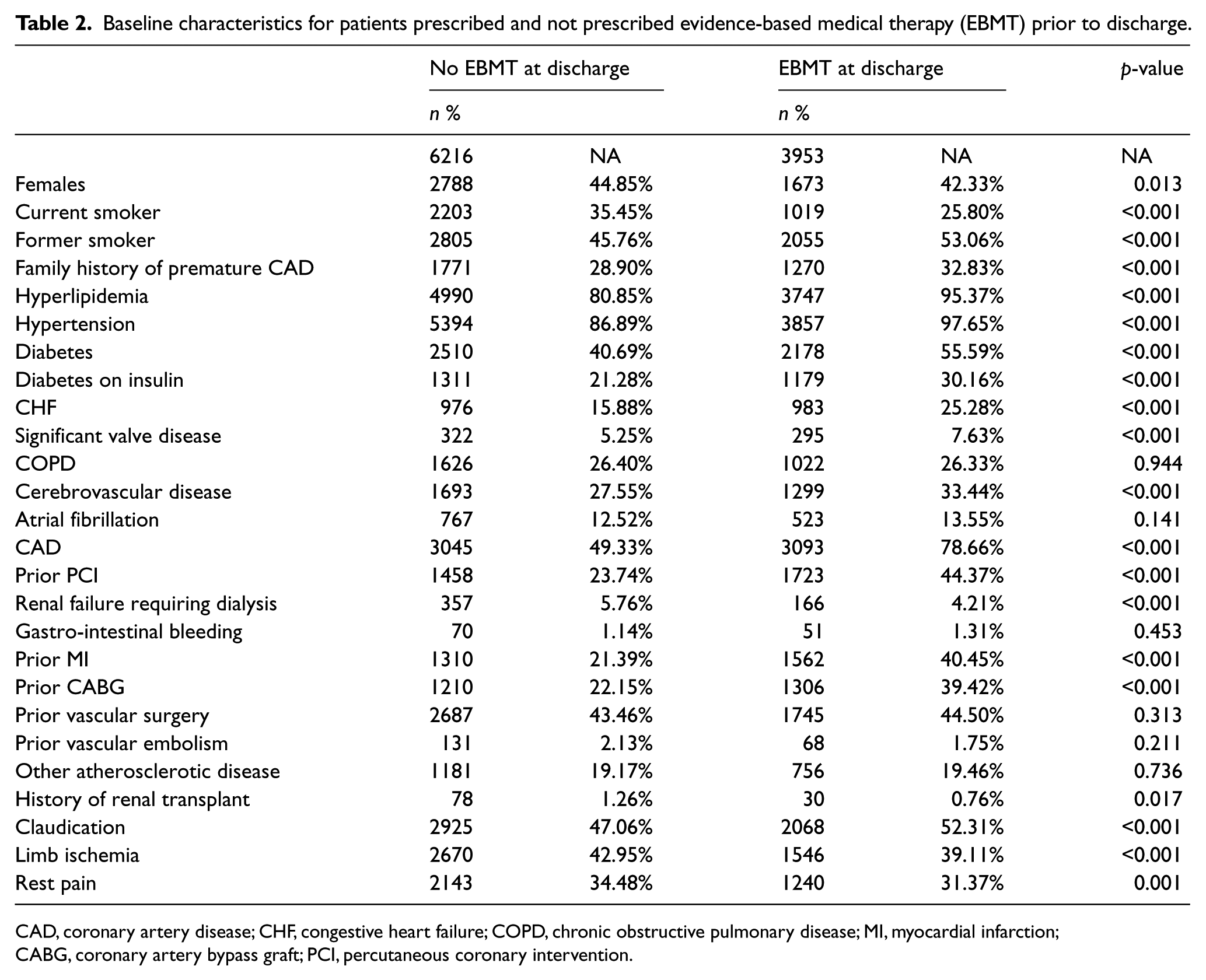
CAD, coronary artery disease; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; MI, myocardial infarction; CABG, coronary artery bypass graft; PCI, percutaneous coronary intervention.
Rates of prescription of EBMT prior to PVI, at discharge post-PVI and at 6 months are displayed in Table 3. Post-PVI discharge use was strongly associated with reported 6-month use for ASA (p < 0.01), BB (p < 0.01), ACE/ARB (p < 0.01), statins (p < 0.01), and all EBMT (p < 0.01) (Figure 1). Six months after the PVI procedure, ASA was used by 7901 (84.5%) patients prescribed the drug post-procedurally, while only 323 (39.2%) of those not prescribed ASA post-PVI were taking it 6 months later. For BB, 5788 (82.5%) patients prescribed the drug post-PVI were taking it at 6 months, and only 350 (11.1%) patients not prescribed BB post-PVI were taking this medication 6 months after the procedure. Six months after the PVI procedure, for ACE/ARB, 5022 (78.2%) patients prescribed the drug post-PVI were taking the medications, and only 443 (11.8%) of those not prescribed it post-PVI were taking them. Six-month post-PVI statin use was ongoing in 7058 (84.6%) patients prescribed the drug post-PVI versus 398 (21.8%) patients not prescribed it post-PVI. When all four drugs were prescribed at discharge, 63.5% (2510/3953) of patients reported taking all EBMT at 6 months post-PVI versus only 7.8% (486/6216) of patients who did not receive all of the drugs at discharge.
Patients receiving evidence-based medical therapy (EBMT) prior to PVI, at discharge and 6 months post-PVI.
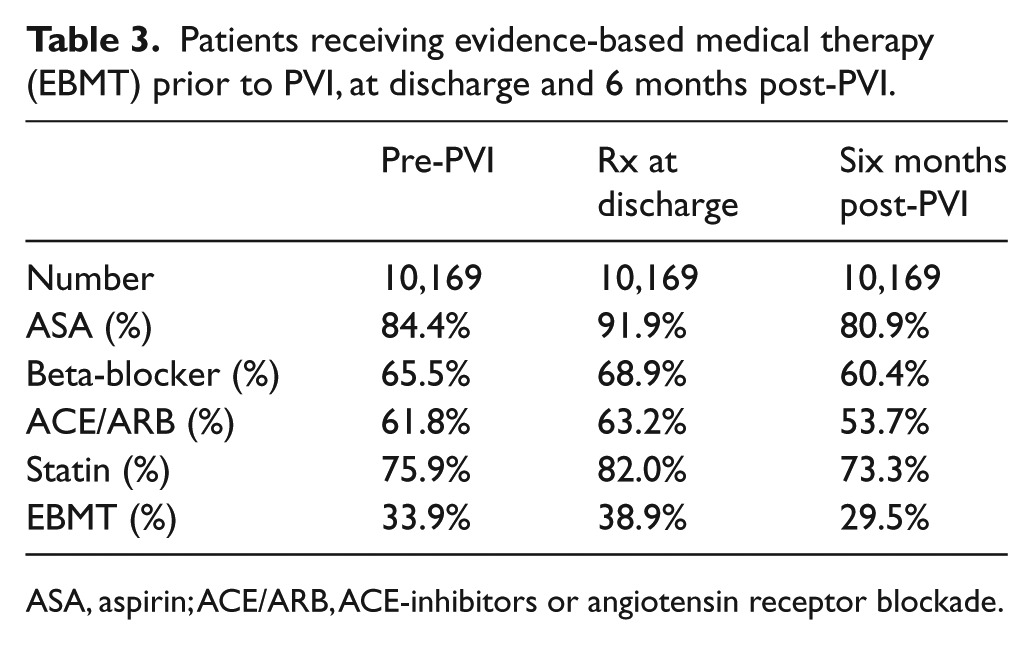
ASA, aspirin; ACE/ARB, ACE-inhibitors or angiotensin receptor blockade.
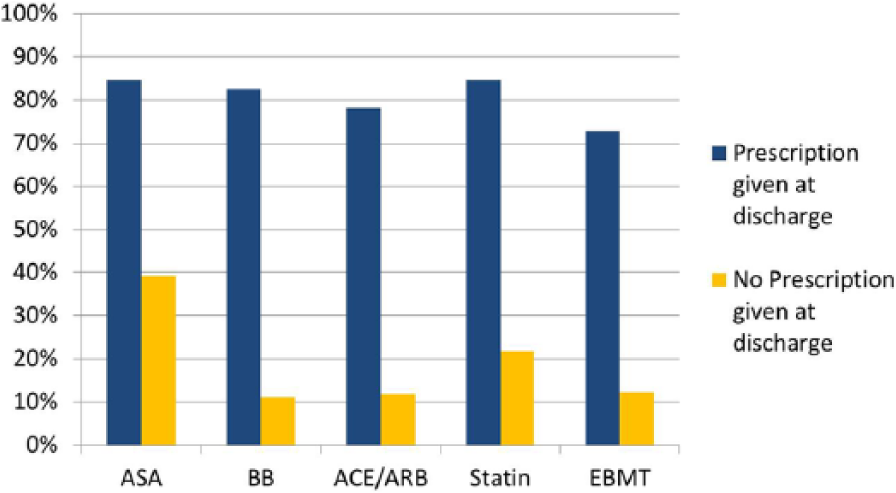
Medication utilization 6 months post-PVI for those patients prescribed these medications at the time of discharge. Evidence-based medical therapy (EBMT) is the combination of all four medications (aspirin (ASA), beta-blockers (BB), ACE-inhibitors or angiotensin receptor blockade (ACE/ARB), and statins). Post-procedural use was associated (all p<0.001) with reported 6-month use.
After adjusting for the baseline clinical variables included in Table 2 using multivariable logistic regression models, prescription of EBMT at the time of discharge remained strongly associated with ongoing use at 6 months for each of the individual component drugs as well as for the combination of all three EBMT medications (Figure 2). Odds ratios (OR) for ASA were 9.1 (95% confidence interval (CI) 7.7–10.8), BB was 40.8 (95% CI 35.0–47.6), ACE/ARB was 30.2 (95% CI 26.4–34.6), and statins were 19.3 (95% CI 16.5–22.5). Other variables with a statistically significant independent association with ASA usage included hyperlipidemia (OR 1.31, 95% CI 1.08–1.58), family history of CAD (OR 1.29, 95% CI 1.13–1.47), and prior vascular interventions (OR 1.24, 95% CI 1.06–1.46). Variables associated with use at 6 months for BB included hypertension (OR 1.42, 95% CI 1.12–1.81) and other vascular disease (OR 1.28, 95% CI 1.21–1.81). Variables significantly independently associated with utilization at 6 months for ACE/ARB included female gender (OR 0.85, 95% CI 0.75–0.96, p = 0.010), family history of PAD (OR 1.21, 95% CI 1.07–1.38, p = 0.003), history of hypertension (OR 1.77, 95% CI 1.40–2.23, p < 0.001), history of CHF (OR 0.79, 95% CI 0.67–0.93, p = 0.004), history of cerebrovascular disease (OR 0.87, 95% CI 0.76–0.99, p = 0.032), history of atrial fibrillation (OR 0.70, 95% CI 0.59–0.83), history of renal failure requiring dialysis (OR 0.56, 95% CI 0.42–0.74, p < 0.001), and limb ischemia (OR 0.73, 95% CI 0.55–0.96, p = 0.024). Variables associated with statin usage at 6 months were a history of hyperlipidemia prior to PVI (OR 1.66, 95% CI 1.38–1.99) and family history of CAD (OR 1.28, 95% CI 1.12–1.46).
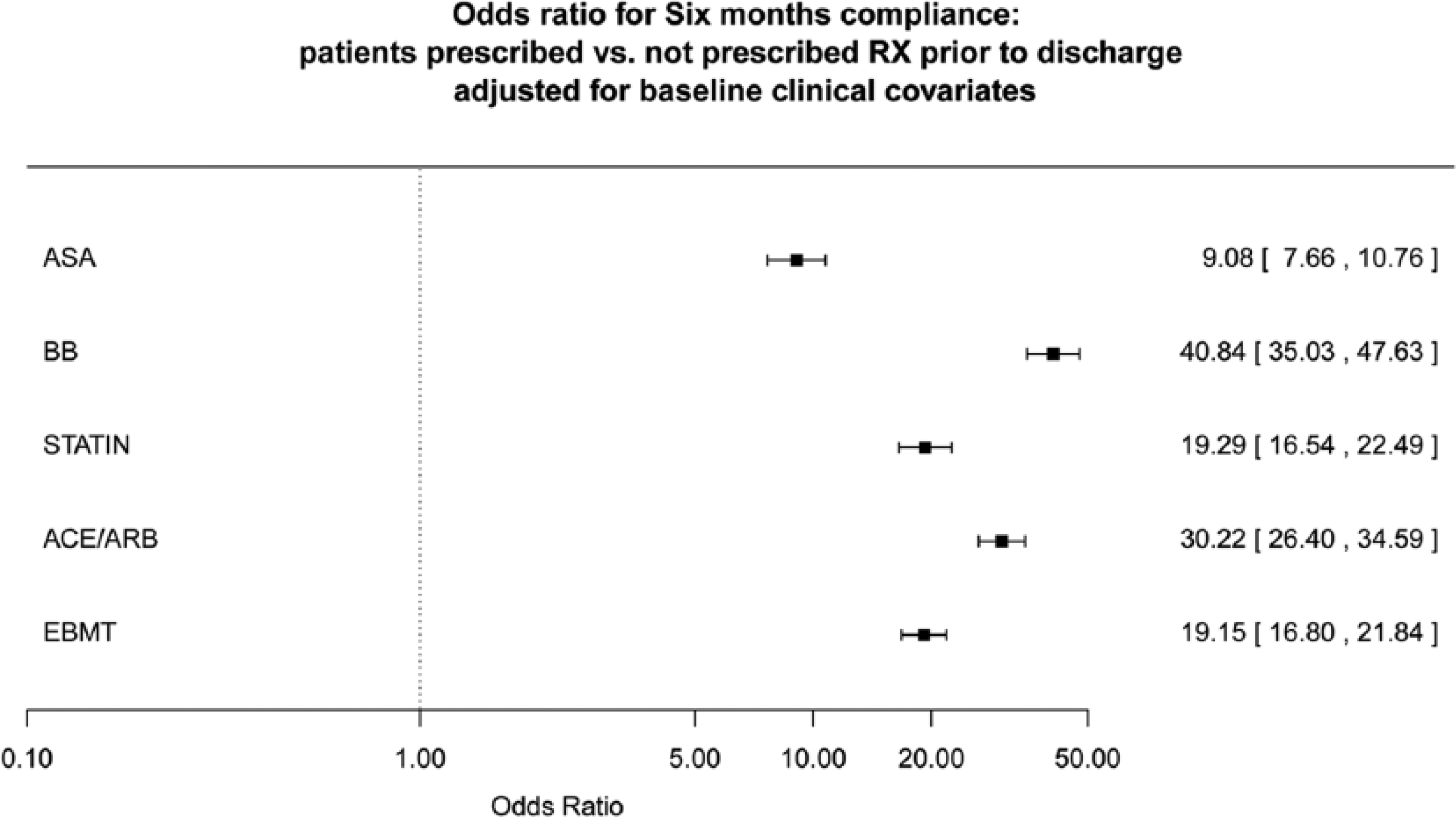
Adjusted likelihood of being on medical therapy 6 months after PVI. Odds ratio for 6 months’ compliance: patients prescribed versus not prescribed treatment prior to discharge adjusted for baseline clinical covariates. (ASA, aspirin; BB, beta-blockers; ACE/ARB, ACE-inhibitors or angiotensin receptor blockade; EBMT, evidence-based medical therapy.)
An additional sub-analysis was performed for patients who were not on EBMT pre-procedurally (and had discharge and 6-month follow-up data available). Follow-up 6-month EBMT rates were 50.5% for patients who were on EBMT at discharge and 6.5% for patients who were not on EBMT at discharge (OR 14.7, p < 0.001).
In addition, each hospital in the BMC2 PVI registry was analyzed for patient use at 6 months. Median and interquartile ranges for hospital post-procedure use of ASA [median (IQR)] was 87.6% (81.6–95.0%), BB use 66.9% (62.9–72.4%), ACE/ARB use 60.6% (56.5–66.8%), and statin use 76.5% (73.2–85.4%). The overall median and interquartile range of the hospital prescription of all four drugs was 34.9% (25.7–43.6%). After adjusting for patient baseline characteristics and evaluating hospital clustering through a hierarchical random effects model, the hospital random effects conditional modes demonstrate substantial unexplained hospital variability in EBMT at discharge following PVI procedures annually (Figure 3).
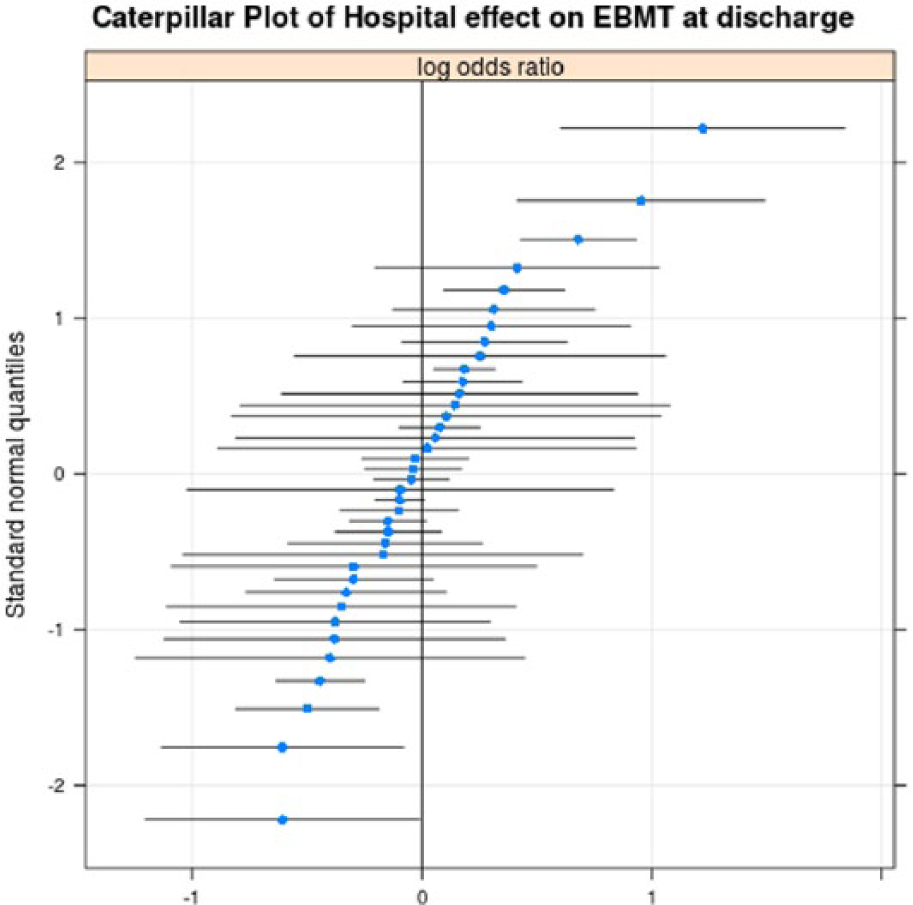
Hospital effect on variability of prescribing evidence-based medical therapy (EBMT). Caterpillar plot of 38 facilities with more than 300 procedures showing the variability of prescribing EBMT at the time of discharge when controlling for baseline characteristics. Facilities on the right of the graph are above the average percentage for prescribing EBMT, while those to the left are below the overall average.
Discussion
In this study, we demonstrated that the prescription of EBMT at the time of PVI is associated with ongoing utilization at 6 months, while failure to prescribe EBMT at discharge is associated with low use of these medications 6 months later. In fact, in this contemporary population of patients with symptomatic PAD, the strongest predictor of patient compliance with EBMT was the prescription of these drugs after their PVI procedure. Additionally, in a sub-analysis of patients who were not on EBMT pre-PVI, the prescription of EBMT at discharge was associated with a greater than 14-fold increased utilization of these medications at 6 months post-PVI. Notably, we found a significant and unexplained variability in the prescription of EBMT, even in hospitals that perform a relatively larger number of PVI procedures.
In the therapeutic window of the procedure, healthcare providers have the opportunity to prescribe medications that are proven to decrease overall cardiovascular morbidity and mortality. The most common cause of death in patients with PAD is ischemic heart disease (IHD). 9 In IHD patients, prescribing EBMT at discharge significantly increases long-term treatment rates and utilization. 10 Non-adherence to BB, statins, and ACE-inhibitors was associated with a 10–40% relative increase in cardiovascular hospitalization and a 50–80% relative increase in risk of mortality for patients with known IHD. 11 However, similar data are lacking in patients with PAD. Studies have shown that atherosclerotic risk factors are prevalent in patients with PAD, but these patients are treated less intensively than patients with IHD using statins, anti-platelet therapy and anti-hypertensives. 12 In addition, a small study has shown a reduction in death, myocardial infarction, and stroke at 6 months using EBMT in patients with PAD. 13 In patients undergoing vascular surgery, use of statins decreases perioperative mortality. 14 In light of these data, our own findings suggest that at the time of discharge after PVI, healthcare providers have an opportunity to reduce long-term morbidity and mortality by prescribing EBMT.
Outcome after the PVI procedure may also be improved with EBMT. We have previously shown that prescription of EBMT prior to percutaneous revascularization is associated with fewer peripheral vascular events at 6 months, including fewer repeat PVI procedures. 15
Programs such as the American College of Cardiology’s Guidelines Applied in Practice (GAP) project and Get with the Guidelines (GWTG) program have been shown to improve adherence to EBMT in CAD patients. The GAP project is a multifaceted intervention that created systems and guideline-based tools to increase adherence to EBMT for patients with an acute myocardial infarction. 16 The GWTG program demonstrated increased utilization of statins, ASA, BB, ACE-inhibitors, and smoking cessation in patients with CAD. 17 Similar programs are lacking in patients with PAD, but multi-specialty society-endorsed performance measures related to medical therapy for patients with PAD have been published. 18
Medication utilization is a large area of active research in all areas of medicine, including cardiovascular medicine. Research on medication compliance in specific areas of cardiovascular medicine, such as acute myocardial infarction and heart failure, have shown decreased compliance in the post-discharge period. 19 For example, one study on patients with acute myocardial infarction discharged on ASA, BB, and statin medications, found that 34% stopped taking at least one medication and 12% stopped all three medications at 1 month after hospital discharge. 20 Another study of patients diagnosed with CAD by angiography demonstrated that between 6 and 12 months 71% of patients reported taking ASA, 46% were taking BB, and 44% were taking statins; 21% of patients reported taking all three medications. 21
There have been several proposed strategies in order to improve utilization of EBMT at the time of discharge and in the 6-month post-PVI period. One such strategy is to reconcile medications in the discharge summary. Unfortunately, studies have shown that discharge summaries often lack crucial information, and that many do not arrive to the primary care provider in a timely manner. 22 The time at discharge allows for patient education and discharge counseling. Studies have shown that discharge counseling was associated with improved adherence after hospital discharge for myocardial infarction. 10 Additionally, applying ‘hard-stops’ for EBMT on the discharge summary can potentially increase prescriptions at the time of discharge. Also, a multidisciplinary approach involving the physician, nurse case manager, social worker, and pharmacist may allow for improvement adherence. For outpatients with clinically stable heart failure, intensive pharmacist-led interventions versus usual care led to a 10.9% improvement in adherence of cardiovascular medications. 23
Lastly, appropriate follow-up in a timely manner could potentially increase patient medication compliance. Telephone follow-up within a few days from discharge is an effective way to communicate in the early discharge period. Data indicate that telephone follow-up increases patient satisfaction, increases medication adherence, decreases preventable adverse drug reactions, and decreases the number of emergency room visits and hospital readmissions. 22 In addition, appropriate follow-up visits with the specialist and/or PCP within a reasonable time frame may increase medical compliance. Patients are more likely to show up for appointments if it is made prior to their discharge, rather than having them call to make an appointment. 22 These appointments allow the physician to continue to educate and encourage compliance with EBMT.
Limitations
Several potential limitations of this study are worth considering. This is an observational study of real-world clinical practice, and confounding variables that may have impacted medication utilization and outcome may not have been captured. Medication use is based upon self-reported patient questionnaires and review of the medical record. Reasons for not being prescribed the medication at 6 months were unavailable but might have included such as financial issues, side effects, polypharmacy, or failure to continue the medication by treating physicians post-PVI.
Conclusion
In this study, the prescription of EBMT at discharge was strongly associated with patient use of these medications 6 months after the PVI procedure. Conversely, patients without contraindications to EBMT who are not prescribed these medications at the time of the PVI procedure are extremely unlikely to be taking these medications 6 months later. In addition, variability between institutions in the prescription of EBMT was observed. These data suggest that the time of a PVI procedure opens a therapeutic window where medical therapy that has been shown to improve morbidity and mortality in this high-risk population can be prescribed with associated excellent compliance. Therefore, the time of these procedures may provide an opportunity to positively impact overall outcome. The variability in prescription of EBMT suggests that there is an opportunity for quality improvement efforts aimed at improving peri-procedural EBMT using a systematic approach.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
