Abstract
Brain-derived neurotrophic factor (BDNF) is essential for neurocognitive function. This study aims at establishing a plausible link between level of serum BDNF, functional capacity (FC), and vascular function in 181 young (age 25.5±9.1 years old), apparently healthy adults. Fasting blood samples were drawn from participants’ antecubital veins into plain glass tubes while they were in a sitting position to evaluate serum BDNF using enzyme-linked immunosorbent assay (ELISA). Mercury-in-silastic strain-gauge plethysmography was used to determine arterial function indices, blood flow and vascular resistance at rest and following 5 minutes of arterial ischemia. The 6-minute walk distance (6MWD) test was used to determine FC, according to the American Thoracic Society Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories guidelines. It was conducted in an enclosed corridor on a flat surface with a circular track 33 meters long. The walking course was demarcated with bright colored cones. The 6MWD correlated with BDNF (r=0.3, p=0.000), as well as with forearm blood inflow (r=0.5, p=0.000) and vascular resistance (r = −0.4, p=0.000). Subsequent comparison showed that BDNF and blood inflow were greater (p<0.05) while vascular resistance was less (p<0.05) in participants who achieved a longer 6MWD. Similarly, BDNF correlated with forearm blood inflow (r=0.4, p=0.000) and vascular resistance (r = −0.4, p=0.000). Subsequent comparison showed improved vascular function (p<0.05) in the participants with greater BDNF. In conclusion, these findings might suggest that improved vascular function in individuals with greater FC is mediated, at least partially, by an enhanced serum BDNF level.
Keywords
Introduction
Brain-derived neurotrophic factor (BDNF) is found and functions in the hippocampus, cerebral cortex, basal forebrain, striatum, hypothalamus, brainstem, and cerebellum. 1 Its function includes promoting growth and survival of neurons, synaptic plasticity, efficacy, and modulation. As a consequence, the protein is involved profoundly in learning and memory. 1
Though the exact role is yet to be clarified, BDNF is also expressed and found throughout the cardiovascular system, including the heart, 2 vascular smooth muscle,3,4 platelets, 5 and in the systemic circulation. 6 It promotes angiogenesis, and enhances the capillary density of ischemic limb 7 and nitric oxide (NO) production, 8 and thus facilitates vasodilation and tissue perfusion, and accelerates blood flow recovery after ischemia.7,8 A few recent studies have also reported that decreased circulating BDNF can induce endothelial and vascular dysfunction and atherosclerosis attributed to increased oxidative stress and inflammation. 9 These evidences suggest a cardiotrophic importance and a role in the risk, development, and manifestations of cardiovascular diseases. 9 In fact, in a 9-year prospective study, elevated circulatory BDNF levels were associated with reduced cardiovascular disease risk and mortality. 10
Exercise increases BDNF expressions and levels in animals 11 and humans. 12 Additionally, vascular adaptations to exercise are well documented. These adaptations have been attributed to changes in the central (i.e. neurohormonal)13,14 and peripheral (i.e. endothelium)15–18 control of the vasculature. Despite the accumulating evidence describing the effect of exercise on neurological and vascular functions, few examined the interaction between the three: functional capacity (FC) with vascular and neural functions. Therefore, the current study will examine the relationship of the 6-minute walk distance (6MWD) test, as a measure of FC level, with circulating BDNF and vascular function parameters (resting and post-occlusion blood inflow and vascular resistance) in healthy individuals. Given previous findings, the 6MWD is expected to be related to BDNF, blood flow and vascular resistance. Additionally, BDNF should correlate with vascular measures. Examining these relationships will certainly help with understanding the contribution of BDNF to these exercise-induced vascular changes.
Methodology
Design and recruitment
The study was cross-sectional to examine the role of BDNF in mediating FC-induced improvement in vascular function. Serum BDNF, FC, and vascular function were assessed from apparently healthy young (18–35 years) individuals. Advertisements, flyers, and mouth-to-mouth communications were used for recruitment from the local community. Upon arrival volunteers were stratified for inclusion and exclusion criteria. Individuals with acute medical conditions, cardiovascular diseases, diabetes mellitus, psychiatric, stress-related, and mood disorders were excluded from the study. Additionally, individuals taking drugs that might affect cardiovascular function were not included in the study. All eligible participants signed an informed consent approved by the Institutional Review Board of Jordan University of Science and Technology.
BDNF measurement
Fasting blood samples were drawn from participants’ antecubital veins into plain glass tubes while they were in a sitting position. After collection, samples were spun for 8–10 minutes at 1500 g to obtain serum for BDNF. Serum samples were then divided into several aliquots and immediately stored at −80°C. Subsequently, the serum BDNF level was determined using the enzyme-linked immunosorbent assay (ELISA), specific for BDNF and as described in the kit manual (Human BDNF DuoSet ELISA Kit; R&D Systems, Minneapolis, MN, USA). In brief, the samples were added to anti-human BDNF antibody-coated ELISA wells and allowed to incubate for 2 hours at room temperature. Subsequently, biotinylated anti-human BDNF antibody was introduced and allowed to bind to the captured BDNF for 1 hour at the same temperature. Horseradish-peroxidase (HRP)-streptavidin conjugate was afterwards added to the reaction mixture and then incubated at room temperature for 45 minutes. Finally, tetramethylbenzidine (TMB) was added to each well followed by additional incubation for 30 minutes at room temperature in the dark. The color developed after incubation and was read at 450 nm immediately, and levels of serum BDNF were deducted from the standard provided by the kit.19,20 The participants were divided into low and high BDNF serum levels according to the BDNF mean of the cohort. The inter- and intra-assay coefficients of variability (CV) were 8.9 and 5.1, respectively.
Vascular function measurements
Mercury-in-silastic strain-gauge plethysmography was used to determine arterial function indices, blood flow and vascular resistance at rest and following ischemia. A pneumatic cuff was placed directly above the elbow and another distally around the wrist. Additionally, a strain-gauge probe 2–3 cm smaller than forearm circumference was wrapped ~10 cm distal to the olecranon process.
After resting for 15–20 minutes in a supine position, the distal cuff was pumped to a pressure of 240 mmHg for 1 minute. Subsequently, blood inflow was measured and cuff pressure was removed. Hyperemic blood flow was measured once immediately after 5 minutes of arterial occlusion using suprasystolic pressure with a pneumatic cuff placed above the elbow. Subsequently, procedures during resting measures were repeated. 21
Analysis of vascular parameters
Forearm blood inflow (FBF) at rest and after occlusion were recorded as a graph at a speed of 5 and 25 mm/s, respectively, by plethysmography recorder (EC5R; Hokanson, Belleview, WA, USA). Slope was determined from a tangent drawn on record of the first three pulses for the resting FBF and the first two pulses for FBF after occlusion. Resting FBF was computed by dividing 60 seconds by the horizontal distance (in millimeters) covered by the slope from the baseline to the top of the recording paper, and the value was multiplied by the chart range. Post-occlusion FBF was obtained by multiplying 60 seconds by the paper speed (25 mm/s), then dividing the value by the horizontal distance (in millimeters) covered by the slope to a point 20 mm vertically from the baseline. Mean arterial pressure (MAP) was determined from the equation MAP = diastolic blood pressure (BP) + [(systolic BP – diastolic BP)/3]. Forearm vascular resistance (FVR) was determined by dividing MAP by FBF.21,22
Functional capacity measurements
The 6MWD test was used to determine FC, 23 according to the American Thoracic Society Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories guidelines.24,25 It was conducted in an enclosed corridor on a flat surface with a circular track 33 meters long. 26 The walking course was demarcated with bright colored cones. During the test, participants were encouraged at intervals to make sure they progressed at a constant pace. 27 Number of circuits covered by the participants was then multiplied by the circuit distance (33 m).
Statistical analysis
All statistical analyses were performed using SPSS software for Windows (version 19.0; Chicago, IL, USA). Group data are expressed as means ± SD, and α was set a priori at p < 0.05. All primary study measures were tested for normality. A measure with a skewness score (Sk) less than 1.0 is considered normally distributed. Subsequent statistical tests (i.e. partial correlation and analysis of covariance (ANCOVA)) were performed after transforming skewed variables. The relationships between 6MWD, BDNF, and vascular function indices were examined with partial correlations after controlling for possible confounding factors including age, gender, body mass index (BMI), serum glucose, cholesterol, low-density lipoprotein (LDL), high-density lipoprotein (HDL), triglyceride, and systolic and diastolic blood pressure. In general, r-values <0.10 are considered to be a small effect, >0.10 to <0.50 a moderate effect, and >0.50 a large effect. 28
ANCOVA tests were used to compare BDNF and vascular function in the participants who achieved above (Hi6MWD) versus below (Lw6MWD) the mean (μ = 563.8±93.7 m) 6MWD score, as well as vascular function in the participants with above (HiBDNF) versus below (LwBDNF) the mean (μ = 26.637±13.0 μg/dL) serum BDNF level. Additionally, a 2 (low and high BDNF) * 2 (low and high 6MWD) ANCOVA test was performed. The ANCOVA was used to covariate for possible confounding factors such as age, gender, BMI, serum glucose, cholesterol, LDL, HDL, triglyceride, and systolic and diastolic blood pressure. Cohen’s d was used as a measure of effect size; in general, <0.20 is considered to be a small effect, >0.20 to <0.50 a moderate effect, and >0.50 a large effect. 29
Results
Participant characteristics
A total of 181 (77 females and 104 males) healthy subjects agreed to participate in the study. The mean age, weight, height, BMI, and percent body fat were 25.50±9.1 years old, 70.9±19.8 kg, 167.8±9.9 cm, 25.0±6.0 kg/m2, and 24.7±9.1%, respectively. The mean 6MWD and BDNF for the participants were 563.8±93.7 m and 26.637±13.0 μg/dL. After dividing the participants according to the maximum distance achieved in the 6MWD test, Table 1 shows significant differences only in the participant heights.
Participant characteristics.
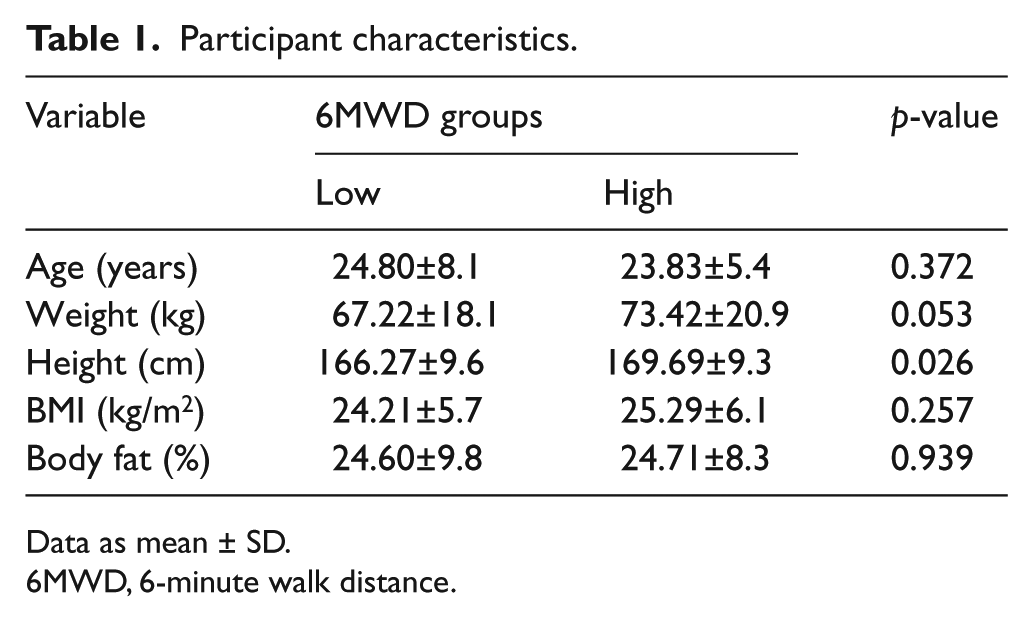
Data as mean ± SD.
6MWD, 6-minute walk distance.
Verifying normality
All primary study outcomes, including 6MWD, resting and post-occlusion FBF and FVR, and serum BDNF, were tested for normality (Sk = 1.0). Resting FBF (Sk = 3.9), resting FVR (Sk = 2.1), and post-occlusion FVR (Sk = 1.5) were skewed. Subsequently, these measures were transformed using log10, and became normally (Sk <1.0) distributed.
Relationship between 6MWD and serum BDNF
As in Figure 1, the partial correlation test showed a medium relationship (r=0.35, p=0.000) of the 6MWD test with serum BDNF. Subsequent ANCOVA in Figure 2 showed that BDNF was greater (p=0.000) in the Hi6MWD versus Lw6MWD group.
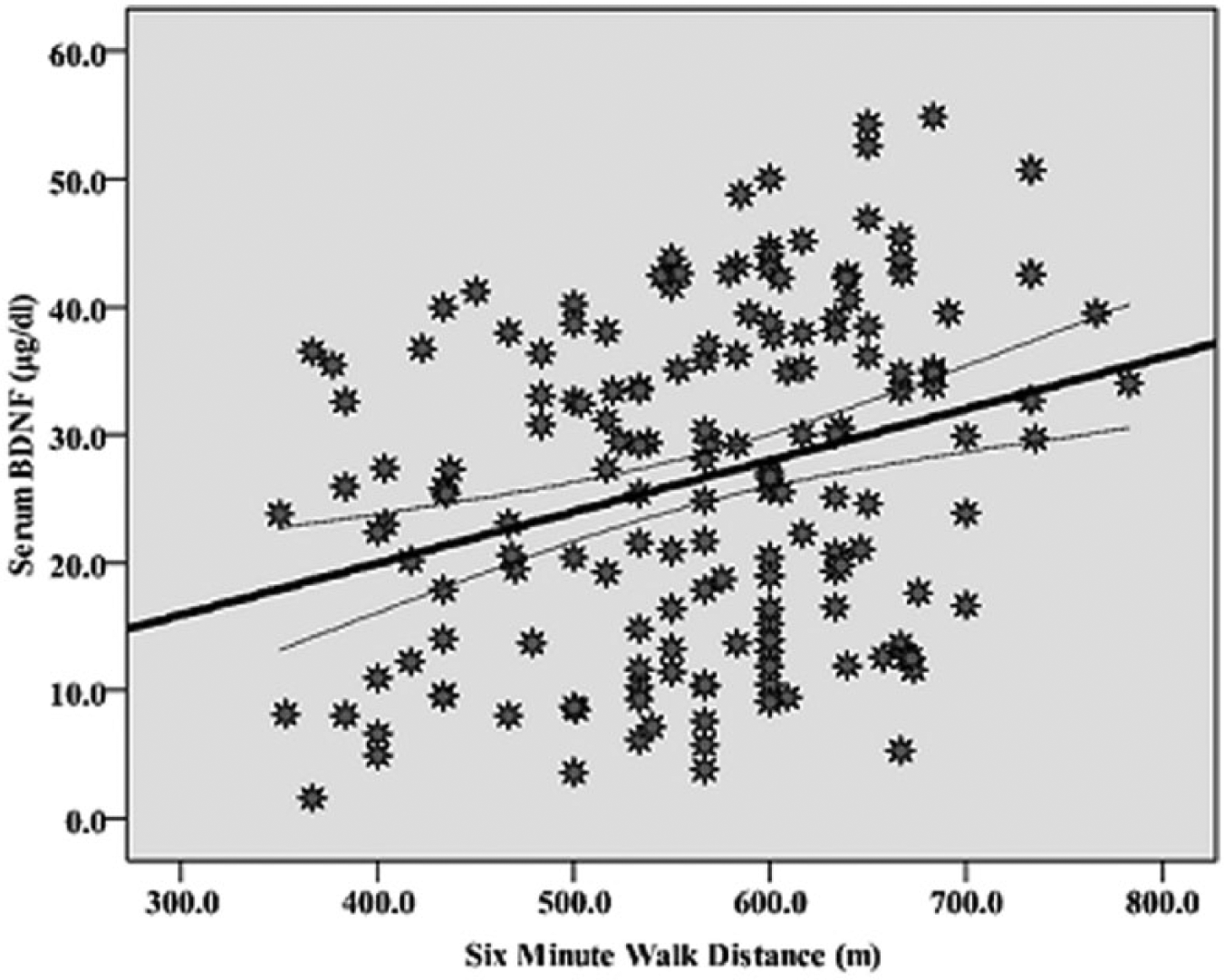
Relationship of distance walked in 6 minutes with serum BDNF (r=0.3, p=0.000). Curves for the 95% confidence intervals around the mean are presented.
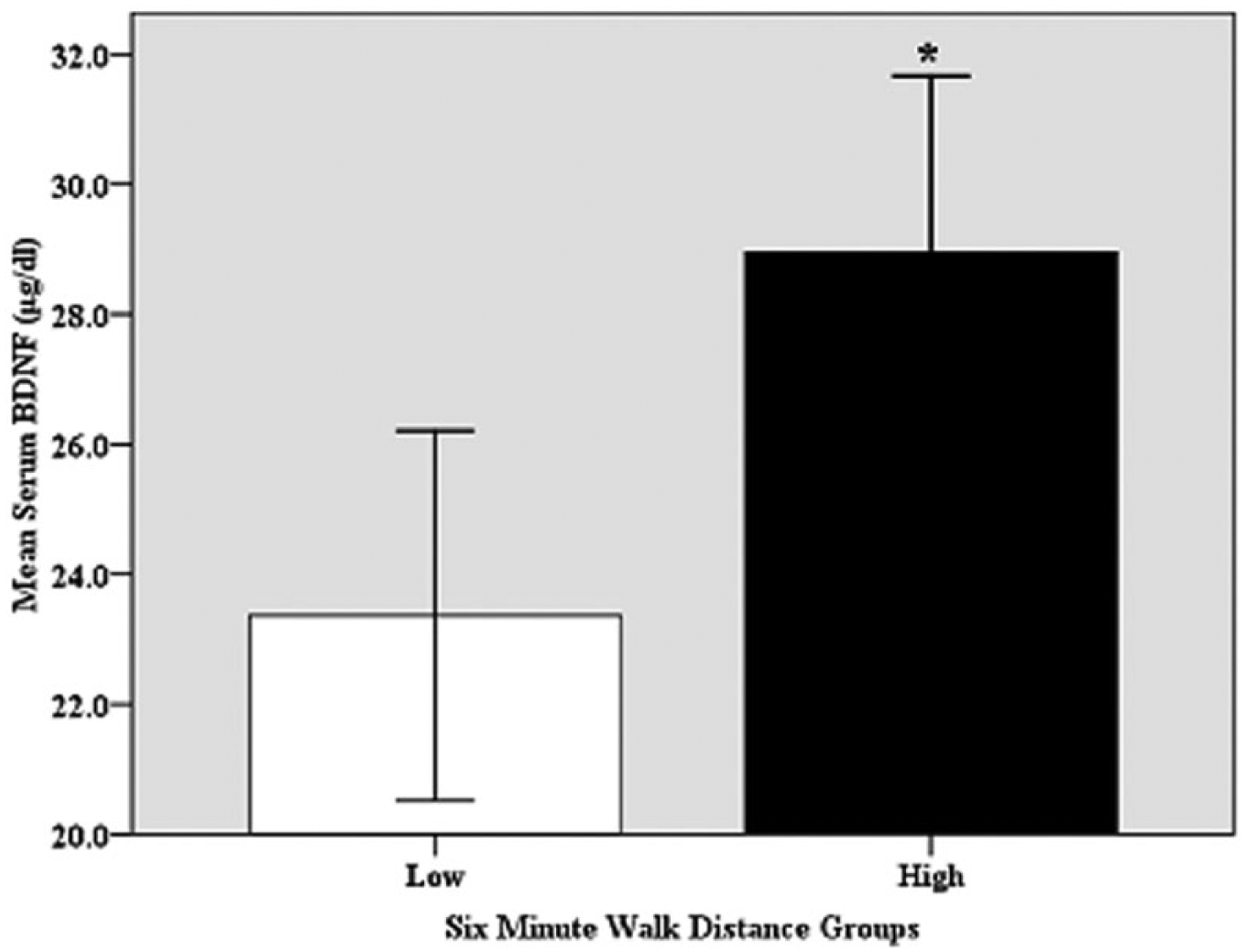
Comparison of the serum BDNF level in HiBDNF (High) versus LwBDNF (Low) 6-minute walk distance groups (*p=0.005).
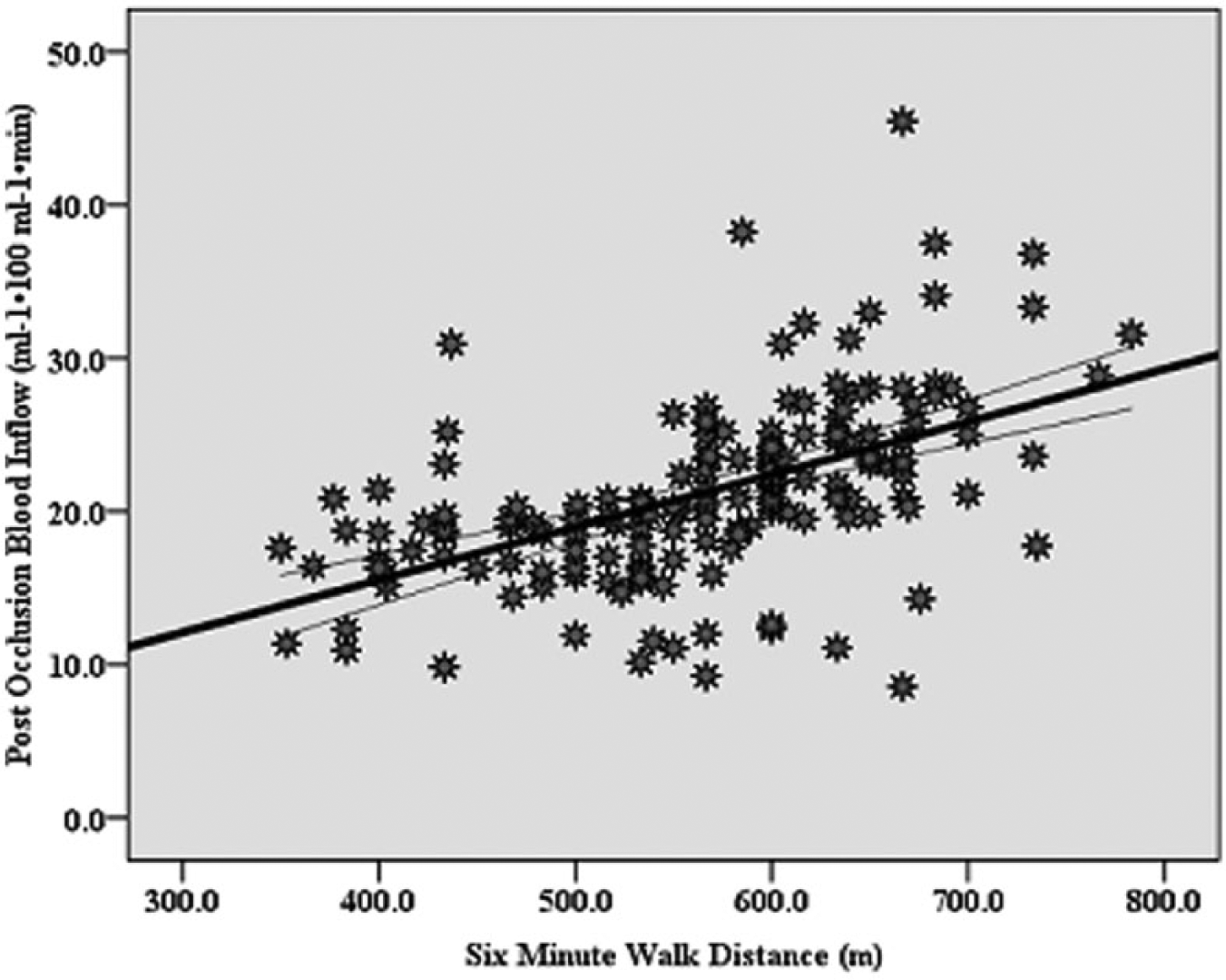
Correlation between the 6-minute walk and post-occlusion forearm blood inflow distance (r=0.5, p=0.000). Curves for the 95% confidence intervals around the mean are presented.
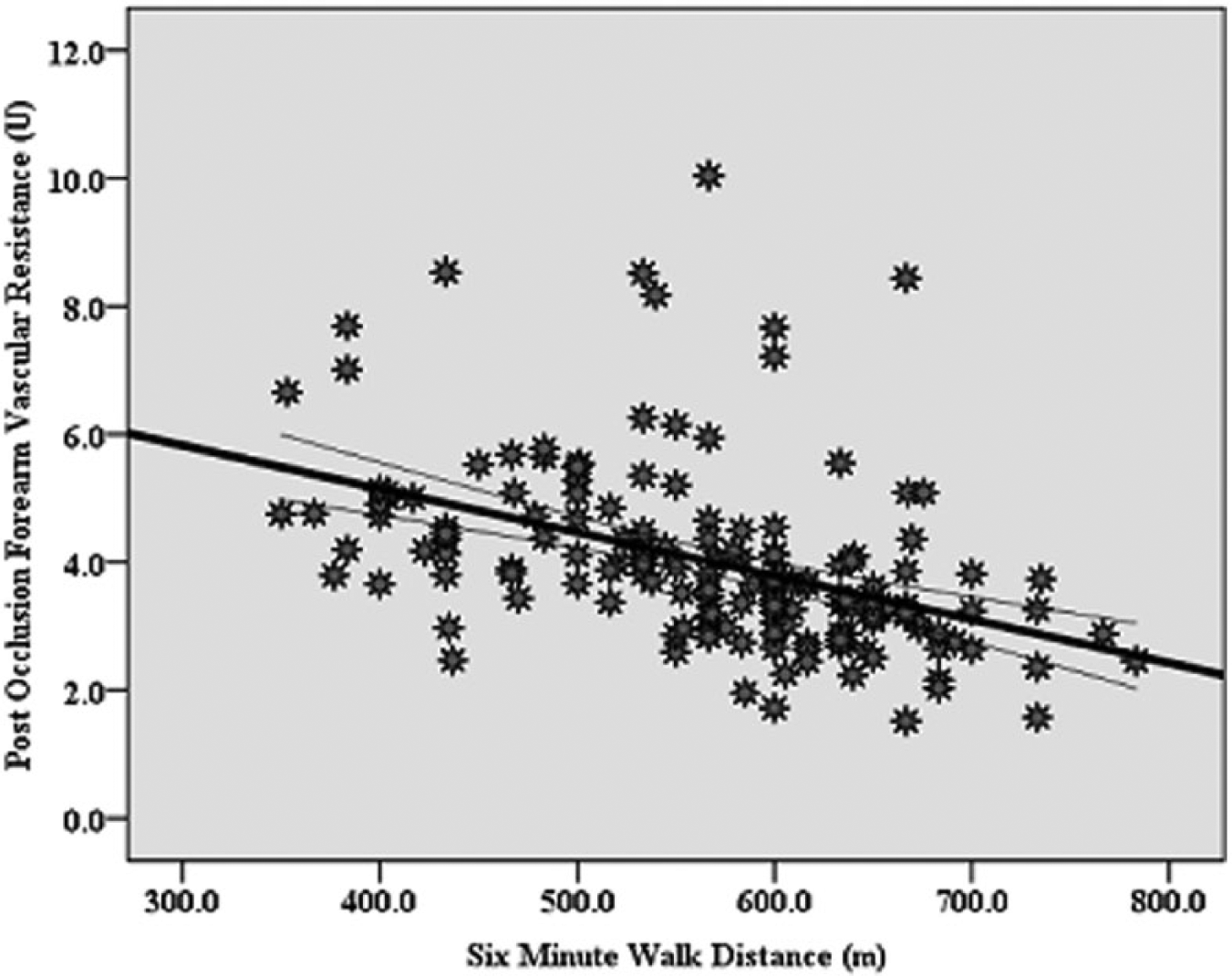
Correlation between the 6-minute walk and post-occlusion forearm vascular resistance (r = −0.4, p=0.000). Curves for the 95% confidence intervals around the mean are presented.
Relationship between 6MWD and vascular parameters
The partial correlation, shown in Figures 2A and 2B, revealed a medium–strong correlation of the 6MWD with post-occlusion FBF (r=0.5, p=0.000) and FVR (r = −0.4, p=0.000), respectively. Table 2 shows, in a follow-up ANCOVA test, that resting (p<0.01; Cohen’s d=0.3) and post-occlusion (p<0.000; Cohen’s d=0.9) FBF were greater in the Hi6MWD versus Lw6MWD group, whereas forearm resting (p<0.000; Cohen’s d=0.3) and post-occlusion FVR (p<0.000; Cohen’s d=0.5) were less in the Hi6MWD versus Lw6MWD group.
Arterial measures in the low versus high 6MWD groups.
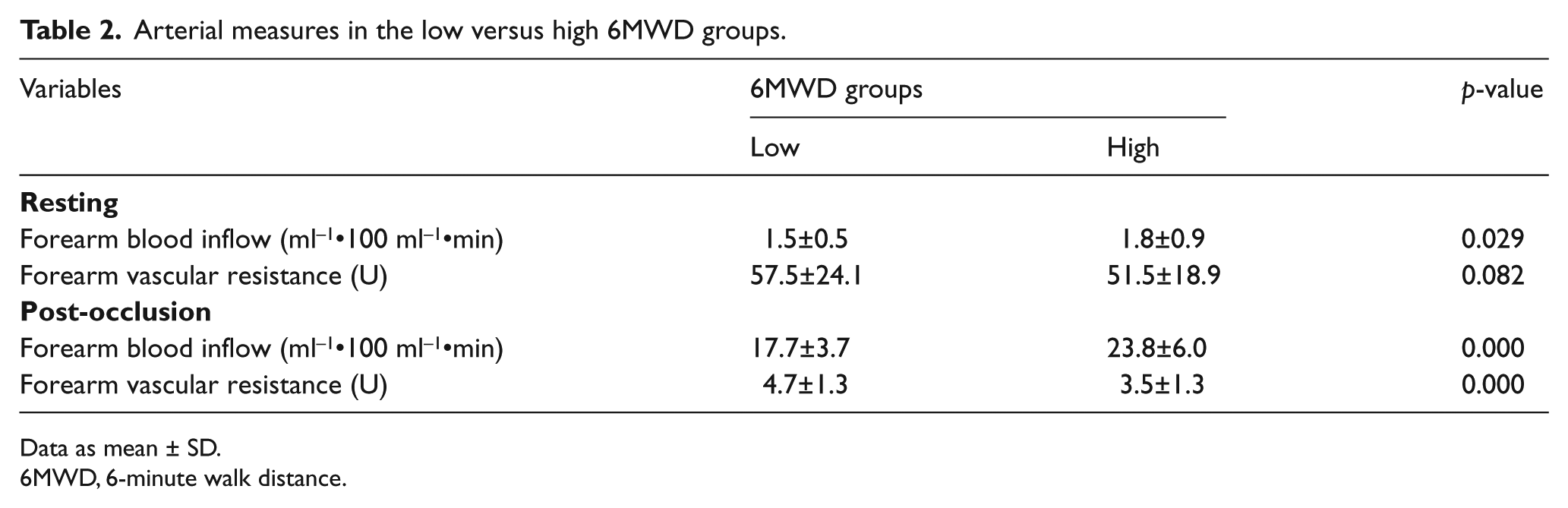
Data as mean ± SD.
6MWD, 6-minute walk distance.
Relationships between BDNF and vascular function parameters
As in Figures 3A and 3B, the partial correlation showed medium relationships of serum BDNF with post-occlusion forearm blood inflow (r=0.45, p=0.000) and vascular resistance (r = −0.43, p=0.000). Subsequent ANCOVA, shown in Table 3, reveals greater post-occlusion forearm blood inflow (p<0.000; Cohen’s d=0.7) and less forearm vascular resistance (p<0.000; Cohen’s d=0.7) in participants with HiBDNF versus LwBDNF. Additional ANCOVA for post-occlusion blood flow that incorporated HiBDNF and LwBDNF*Hi6MWD and Lw6MWD, while accounting the potential confounding factors, revealed a main effect for BDNF (p<0.001), 6MWD (p<0.000), and interaction of BDNF*6MWD (p<0.05).
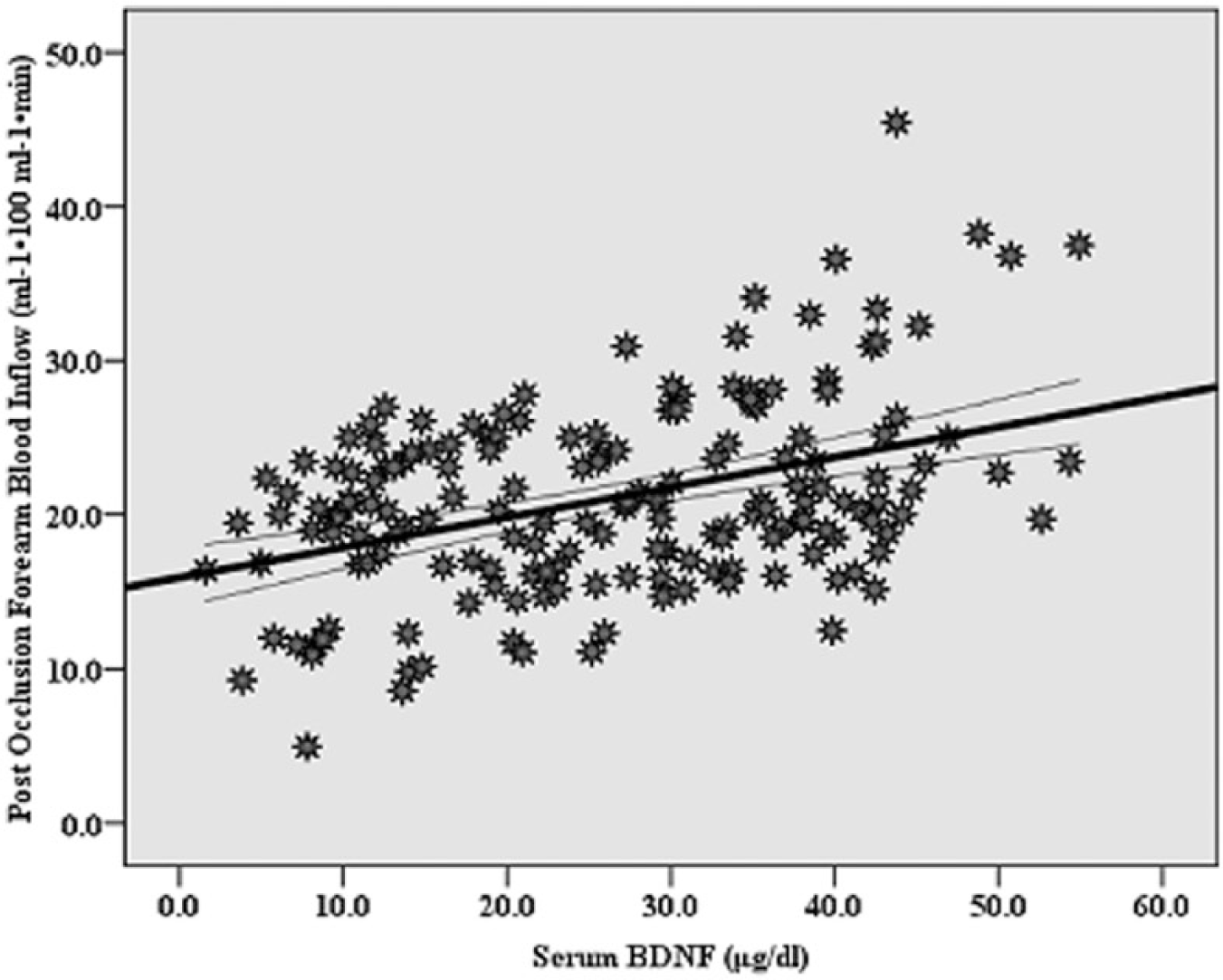
Correlation between serum BDNF and post-occlusion forearm blood inflow (r=0.45, p=0.000). Curves for the 95% confidence intervals around the mean are presented.
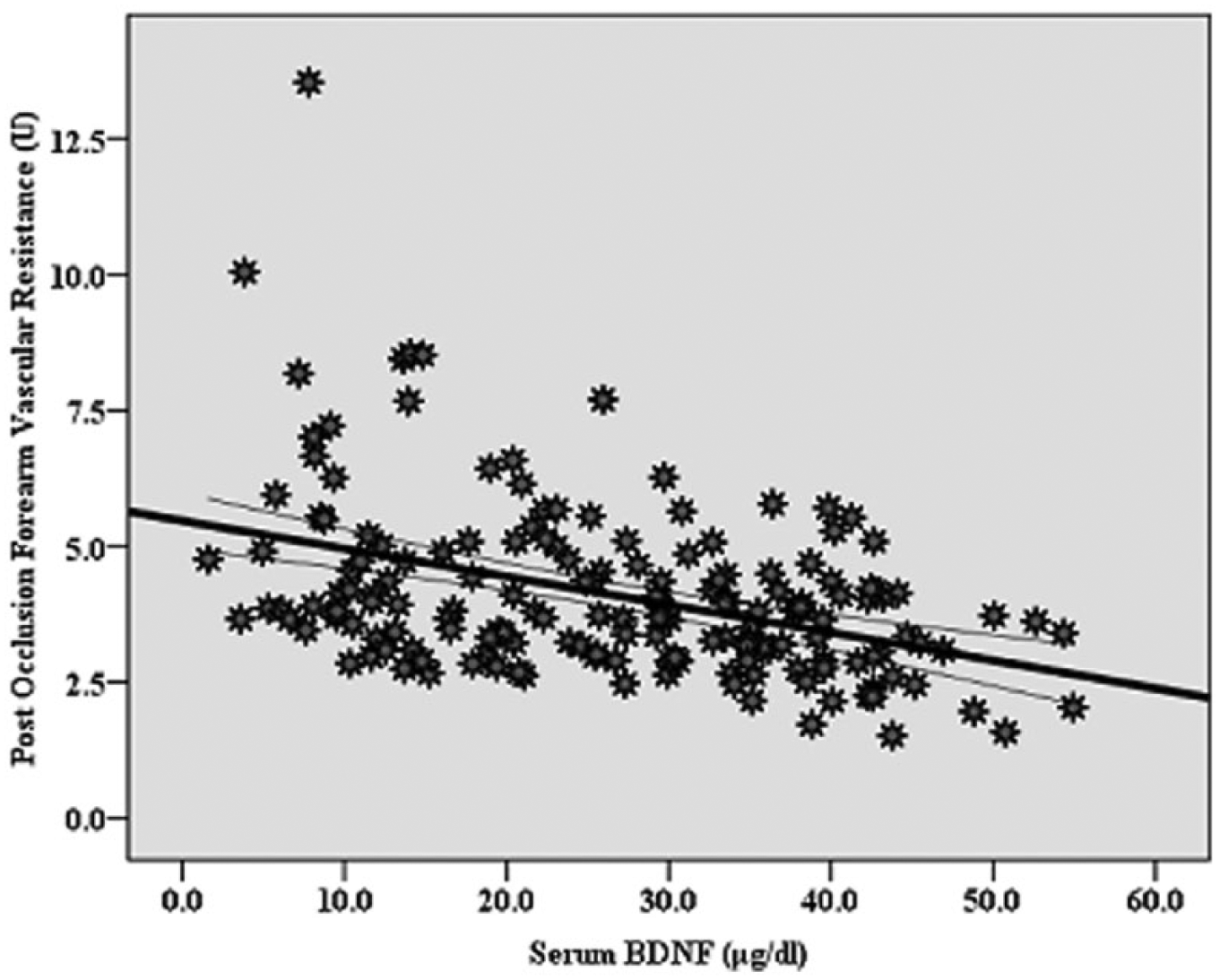
Correlation between serum BDNF and post-occlusion forearm vascular resistance (r = −0.43, p=0.000). Curves for the 95% confidence intervals around the mean are presented.
Arterial measures in the low versus high serum BDNF groups.
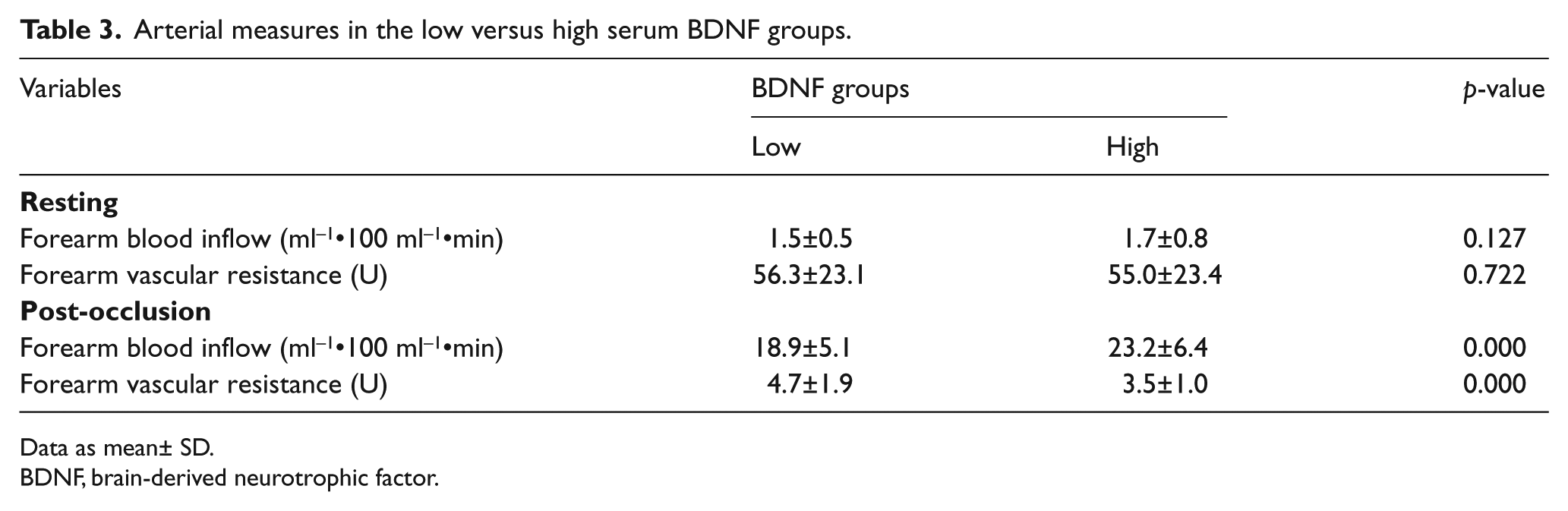
Data as mean± SD.
BDNF, brain-derived neurotrophic factor.
Discussion
The current study was designed to investigate the relationships between FC, circulating BDNF, and vascular function in healthy individuals. Serum BDNF and vascular function indices correlated with 6MWD and were greater in the participants who walked longer distance. Additionally, arterial indices correlated with serum BDNF and were enhanced in the participants with elevated BDNF.
The mean serum BDNF in the current study is 26.637.0±13.0 μg/dL, which is similar to the ranges previously reported.30–32 The mean 6MWD (μ=563.8±93.7 m) is greater than found in other Arab populations from Saudi Arabia (µ=409±51 m); 26 however, it is similar to that reported from Singaporeans (µ=560±105 m) 33 and slightly less than Brazilians (µ=571±74 m) 34 and Caucasians (µ=593±57). 35
The relationships of FC with BDNF36,37 and vascular function15,16,38,39 are consistent with previous findings. However, this is the first study to show a relationship of BDNF with vascular function. Increased FC usually reflects, and is associated with, regular participation in exercise. 40 This participation is documented to increase circulating levels of BDNF among healthy individuals.36,37 It seems to induce a genomic effect on BDNF, as it increases BDNF mRNA transcription and BDNF protein in several brain compartments.19,20,41,42 Increased brain BDNF synthesis seems to increase release of the protein from the brain12,43 store44–46 into the systemic circulation. The elevated BDNF level in the circulation appears to be maintained for a substantial amount of time following exercise.47–49 Improved vascular function among regular exercisers is also documented.15,38,39 Exercise participation improves endothelial-dependent and -independent dilation. 50 During exercise, many vasodilatory autacoids are produced, one of which is nitric oxide (NO), 51 generated as a result of increased expression and phosphorylation of endothelial NO synthase (eNOS). 52
No studies reported relationships of serum BDNF with blood flow and vascular resistance. However, previous studies have shown relationship of BDNF with endothelium factors. For example, BDNF induces phosphorylation of Akt protein kinases,53,54 an enzyme that activates eNOS. This activation leads to the production of NO, a prominent vasodilator.8,55,56 Additionally, BDNF stimulates production of prostacyclin, 57 another endothelium-derived vasodilator58,59 through the improvement of arachidonic acid metabolism. BDNF also seems to promote capillarization. 7 These effects can result in decreased vascular resistance, improved blood flow, and, subsequently, tissue perfusion. Conversely, deficiency in BDNF can induce reduced endothelial cell–cell communication, endothelial and vascular dysfunction, and atherosclerosis subsequent to increased oxidative stress and inflammation. 60 These roles might suggest a protective effect of BDNF from cardiovascular diseases. 60
The mutual effect of FC on circulating BDNF and vascular function, and the relationship of BDNF with vascular function might suggest that exercise-induced improvement in vascular function is mediated, at least in part, by BDNF. We postulate that improving FC enhances vascular function subsequent to increasing BDNF. However, we acknowledge the inherited limitations of a cross-sectional study including these relationships are not cause–effect. Additionally, the current findings are confined to a young healthy population. Therefore, future interventional studies are needed to further confirm these speculations, especially in diseases such as cardiovascular disease, dementia, and Alzheimer’s and Parkinson’s disease. Examining the relationships between changes in vascular function and BDNF following an exercise program would confirm the postulations of the current study. Additionally, examining the relationship of vascular function with FC while interfering with BDNF might give more insight into the role of BDNF in mediating this relationship. These speculations, if proven correct, mean recombinant human BDNF would be a vital cardiovascular therapeutic agent in diseased populations.
Conclusions
The current findings confirm the beneficial effect of improved FC on neural and vascular functions. Additionally, the study shows, for the first time, relationships of BDNF with vascular function indices. Thus, the study provides a positive link between FC, BDNF, and vascular function. Given the role of BDNF in endothelium function and capillarization, exercise-induced improvements in vascular function might be mediated, at least partially, by increased BDNF. However, more longitudinal studies are certainly needed in health and disease to precisely understand the nature of these relationships.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
Mahmoud Alomari received funding support from the Deanship of Research at Jordan University of Science and Technology, Grant No. 155/2014.
