Abstract
In diabetes, angiogenesis is disturbed, contributing to proliferative retinopathy, nephropathy and neuropathy. Kallistatin, a serine proteinase inhibitor, has anti-angiogenic effects. We assessed serum kallistatin in children and adolescents with type 1 diabetes as a potential marker for microvascular complications and its relation to carotid intima media thickness (CIMT). Sixty patients with type 1 diabetes were divided into two groups according to the presence of microvascular complications and compared with 30 healthy controls. High-sensitivity C-reactive protein (hs-CRP), HbA1c, urinary albumin creatinine ratio (UACR), kallistatin levels and CIMT were assessed. Kallistatin levels were significantly higher in patients with microvascular complications (9.9 ± 2.38 ng/mL) and those without complications (5.0 ± 1.5 ng/mL) than in healthy controls (1.39 ± 0.55 ng/mL; p<0.001). Kallistatin was increased in patients with microalbuminuria compared with the normoalbuminuric group (p<0.001). Positive correlations were found between kallistatin and disease duration, fasting blood glucose, HbA1c, triglycerides, total cholesterol, hs-CRP, UACR and CIMT (p<0.05). A kallistatin cut-off value at 6.1 ng/mL could differentiate patients with and without microvascular complications, with a sensitivity of 96.87% and specificity of 93.75%. Increased kallistatin levels in type 1 diabetes and its relation with CIMT may reflect vascular dysfunction and suggest a link between micro- and macro-angiopathy.
Keywords
Introduction
Diabetes mellitus (DM) is a group of metabolic disorders characterized by chronic hyperglycemia and is associated with increased prevalence of microvascular complications. Type 1 diabetes results from cellular-mediated autoimmune destruction of pancreatic β-cells in the islets of Langerhans, leading to loss of insulin production. 1 Diabetes is a risk factor for myocardial infarction and stroke. 2 The relationship between diabetes and other traditional cardiovascular risk factors (e.g. an adverse lipid profile, obesity, hypertension and physical inactivity) explain the increased risk in diabetic individuals. It has been suggested that inflammation contributes to the increase of cardiovascular diseases among diabetic patients. 3
In diabetes, angiogenesis is disturbed, contributing to proliferative retinopathy, nephropathy and neuropathy. 4 The risk of developing microvascular complications is related mainly to the duration of diabetes and degree of glycemic control achieved over time. 5 Hyperglycemia, hypertension, dyslipidemia, smoking, adiposity, inflammation and oxidative stress may promote vascular complications, 4 and some effects of these stresses may be mediated by disturbances in the levels of, or balance of pro- and anti-angiogenic factors, such as (anti-angiogenic) kallistatin. 6
Kallistatin, a serine proteinase inhibitor, was first identified as a tissue kallikrein-binding protein. 7 It has multiple biologic functions including blood pressure regulation, protection against inflammation, vasculature relaxation, and stimulation of neointima hyperplasia. 8 Kallistatin has been localized in vascular smooth muscle cells and endothelial cells of human blood vessels, suggesting that kallistatin may be involved in the regulation of vascular function. 9 Kallistatin has independent effects as a vasodilator and modulator of vascular growth, and anti-angiogenic, anti-oxidant and anti-inflammatory effects.10 –12 Kallistatin may predict and modulate diabetic angiopathy 13 and has potential for use as a therapeutic agent or target.14,15
Carotid intima media thickness (CIMT) is a simple, non-invasive, sensitive, screening tool for the assessment of atherosclerosis risk/prognosis in type 2 diabetics, 16 but there are limited studies evaluating its role in type 1 diabetes in developing countries. 17 Increased CIMT is associated with a higher prevalence of coronary artery disease and correlates with future development of myocardial infarction and stroke. 18
Only one study evaluated kallistatin in adult patients with type 1 diabetes and found elevated levels in microvascular complications, as a compensatory mechanism. 6 Therefore, we determined the circulating kallistatin levels in children and adolescents with type 1 diabetes and assessed its relation to inflammation, glycemic control and microvascular complications. In addition, CIMT was assessed as a surrogate marker of atherosclerosis and its relationship to microangiopathy and kallistatin levels.
Materials and methods
This cross-sectional study was conducted on 60 children and adolescents with type 1 diabetes attending the Pediatric Diabetes Clinic, Pediatric Hospital, Ain Shams University (Cairo, Egypt). Patients were defined according to the criteria of American Diabetes Association. 19 They included 19 males and 41 females and their ages ranged from 9 to 18 years with a mean age of 14.1 ± 2.6 years. Another group of 30 age- and sex-matched healthy volunteers were enrolled as controls. An informed consent was obtained from each patient or control subject or their legal guardians before enrollment into the study. The study was approved by the local ethical committee of Ain Shams University.
Exclusion criteria were the presence of any clinical evidence of infection, history of allergies, rheumatoid arthritis, recent trauma, surgery, physical inactivity, liver dysfunction, connective tissue disease, or other autoimmune disorders, and any other conditions that could influence high-sensitivity C-reactive protein (hs-CRP). Patients with hs-CRP>10 mg/L were excluded.
Patients were divided into two groups according to the presence of microvascular complications.
The studied patients were subjected to a detailed medical history and thorough clinical examination with special emphasis on age of onset of diabetes, disease duration, insulin therapy and chronic diabetic complications (retinopathy, neuropathy, nephropathy, or cardiovascular ischemic events). All patients were on insulin therapy using human insulin with a mean dose of 1.6 ± 0.38 IU/kg/day.
Anthropometric measurements were recorded and body mass index (BMI) was calculated. Blood pressure was measured after a 5-minute rest in the seated position using a mercury sphygmomanometer. If it was greater than the 90th percentile for age and sex, the blood pressure was repeated twice to check the validity of the reading.
Diabetic retinopathy was diagnosed by doing a complete ocular examination including visual field testing, slit-lamp biomicroscopy, Volk lens and indirect ophthalmoscopy. A fundus examination was performed through dilated pupils using a 90-diopter Volk lens and biomicroscope. The presence and grading of diabetic retinopathy was based on the International Clinical Diabetic Retinopathy and Macular Edema Disease Severity Scales. 20 The simple rapid bedside Neuropathy Disability Score (NDS) was adopted as a screening tool for diabetic peripheral neuropathy. The NDS was derived from examination of vibration perception (by means of a 128-Hz tuning fork), pin-prick and temperature perceptions in the great toe, and the presence or absence of ankle reflexes. The sensory modalities were scored and a score above two was defined as clinical diabetic peripheral neuropathy. 21 Results were confirmed by nerve conduction velocity to provide accurate diagnosis. 22
Laboratory investigations
Liver and kidney function tests, fasting lipid profile, hs-CRP, as well as mean fasting blood glucose (FBG) levels in the last 6 months prior to the study were measured using the Cobas Integra 800 (Roche Diagnostics, Mannheim, Germany). Dyslipidemia was defined if at least one of the following was present: serum total cholesterol ⩾200 mg/dL, low-density lipoprotein (LDL) cholesterol ⩾100 mg/dL, high-density lipoprotein (HDL) cholesterol <40 mg/dL, or serum triglyceride ⩾150 mg/dL. 23 Further analysis was done after controlling for age and pubertal stage to avoid differences in lipid values. 24 Assessment of mean HbA1c% in the year preceding the study was performed using the D-10 System (BioRad, France). Urinary albumin excretion (as an indicator of nephropathy) was measured in an early morning urine sample as an albumin-to-creatinine ratio by an immuno-nephelometric method. Microalbuminuria and macroalbuminuria were present if the urinary albumin excretion in at least two out of three consecutive urine samples, 2 months apart, was 30–299 mg/g creatinine and ⩾300 mg/g creatinine, respectively.25,26 Determination of serum kallistatin levels was done by enzyme-linked immunosorbent assay (ELISA) using a Human Kallistatin (KAL) ELISA Kit (Glory Science Co., Ltd, Del Rio, TX, USA) according to the manufacturer’s instructions.
Sampling
Peripheral blood (PB) samples were collected on ethylene diamine tetra-acetic acid (EDTA) (1.2 mg/mL) for analysis of HbA1c. Serum obtained from clotted samples by centrifugation for 15 minutes at 1000g was used for chemical analysis and stored at −20
Measurement of CIMT
All of the carotid scans were done using a carotid Doppler ultrasound scanner (Xario; Toshiba, Tokyo, Japan) with a 10.0-MHz linear array transducer following a predetermined standardized scanning protocol. 27 The Doppler ultrasound studies were performed after an overnight fast and before the administration of a morning insulin dose. Patients were placed in a supine position with the neck in hyperextension. The left and right common carotid arteries were imaged in a standardized magnification (2 × 2 cm) with images of the posterior wall of the distal 10 mm of the common carotid artery, just proximal to the carotid bulb. Images were captured when both the anterior and posterior wall margins were clearly seen, to ensure the images were taken perpendicular to the vessel. A minimum of four images of each of the common carotid arteries were taken. All images were taken at end-diastole, incident with the R-wave on a continuously recorded electrocardiogram and then digitally stored for later analysis. The three best quality images for each of the carotid arteries were selected and analyzed. Best quality was defined as those images that produced the most number of points for analysis. For each image, the greatest distance between the lumen–intima interface and media–adventitia interface (intima media thickness [IMT]) was measured at a minimum of 100 points. The mean and maximum IMT of each image were then averaged to give the final result for each subject. All the ultrasonographic examinations were analyzed by a single experienced radiologist blinded to the group to which the subjects belonged.
Statistical analysis
Analysis of data was done using the Statistical Program for the Social Sciences version 15 (SPSS Inc., Chicago, IL, USA). Quantitative variables were described in the form of mean, SD or median and inter-quartile range (IQR), and qualitative variables were described as number and percent. In order to compare quantitative parametric variables between the three studied groups, an analysis of variance (ANOVA) test with a post hoc test was used while Student’s t-test was applied for comparison between two groups. Comparison of non-parametric variables was carried out using Kruskal–Wallis and Mann–Whitney tests. Categorical variables were compared using the chi-squared (χ2) test. Spearman’s rank correlation coefficient and multiple linear regression analysis were employed to assess the relation between each of kallistatin and CIMT and clinical as well as laboratory variables. Logistic regression was used to examine the relation between kallistatin and microvascular complications after adjustment of other variables: age, sex, and BMI. A receiver operating characteristic (ROC) curve was used to determine the best cut-off value of kallistatin to detect microvascular complications. The area under the curve (AUC) was calculated for each plot. A p-value <0.05 was considered significant in all analyses.
Results
Clinical and laboratory characteristics among the studied groups with type 1 diabetes
The clinical and laboratory data of patients are listed in Table 1. Age, sex, and BMI were consistent among all patients compared with controls and in each subgroup of patients when compared separately with the control group (p>0.05). The most common microvascular complications encountered in the studied diabetic patients was microalbuminuria (31.7%) followed by peripheral neuropathy (13.3%) and retinopathy (10%). None of the studied patients had macroalbuminuria or macrovascular complications. As shown in Table 1, patients with microvascular complications had longer disease duration and higher blood pressure, FBG, HbA1c, hs-CRP, triglycerides, total cholesterol, LDL, and urinary albumin creatinine ratio (UACR) compared with patients without microvascular complications and with controls (p<0.001). HDL cholesterol was significantly lower in patients with complications than in the other two groups (p<0.001).
Comparison of clinicopathological characteristics among type 1 diabetic patients with and without complications and the control group.
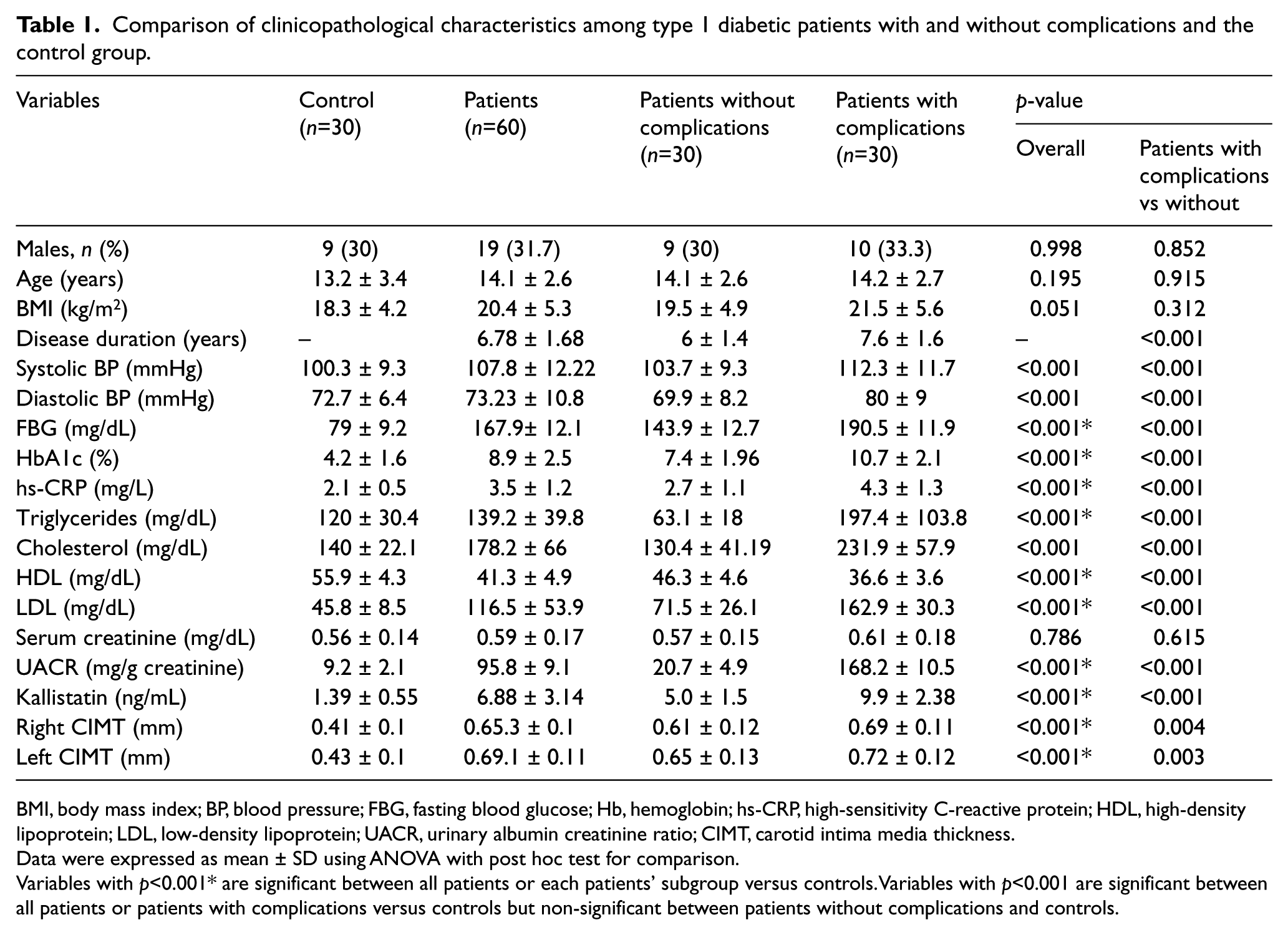
BMI, body mass index; BP, blood pressure; FBG, fasting blood glucose; Hb, hemoglobin; hs-CRP, high-sensitivity C-reactive protein; HDL, high-density lipoprotein; LDL, low-density lipoprotein; UACR, urinary albumin creatinine ratio; CIMT, carotid intima media thickness.
Data were expressed as mean ± SD using ANOVA with post hoc test for comparison.
Variables with p<0.001* are significant between all patients or each patients’ subgroup versus controls. Variables with p<0.001 are significant between all patients or patients with complications versus controls but non-significant between patients without complications and controls.
Kallistatin levels and CIMT in type 1 diabetes
Kallistatin levels were significantly higher in patients with microvascular complications (9.9 ± 2.38 ng/mL) and those without complications (5.0 ± 1.5 ng/mL) than in healthy controls (1.39 ± 0.55 ng/mL; p<0.001), with the highest levels in patients with complications (p<0.001). Serum kallistatin was significantly higher in patients with microalbuminuria (9.05 ± 2.15 vs 6.48 ± 3.15 ng/mL; p<0.001), peripheral neuropathy (10.7 ± 1.86 ng/mL vs 4.3 ± 1.54 ng/mL; p<0.001) or retinopathy (7.58 ± 1.59 vs 4.3 ± 1.54 ng/mL; p<0.001) compared with those without these complications (Figure 1). ROC curve analysis revealed that the cut-off value of serum kallistatin at 6.1 ng/ml could differentiate patients with and without microvascular complications in type 1 diabetes, with a sensitivity of 96.87%, specificity of 93.75% and AUC 0.979.
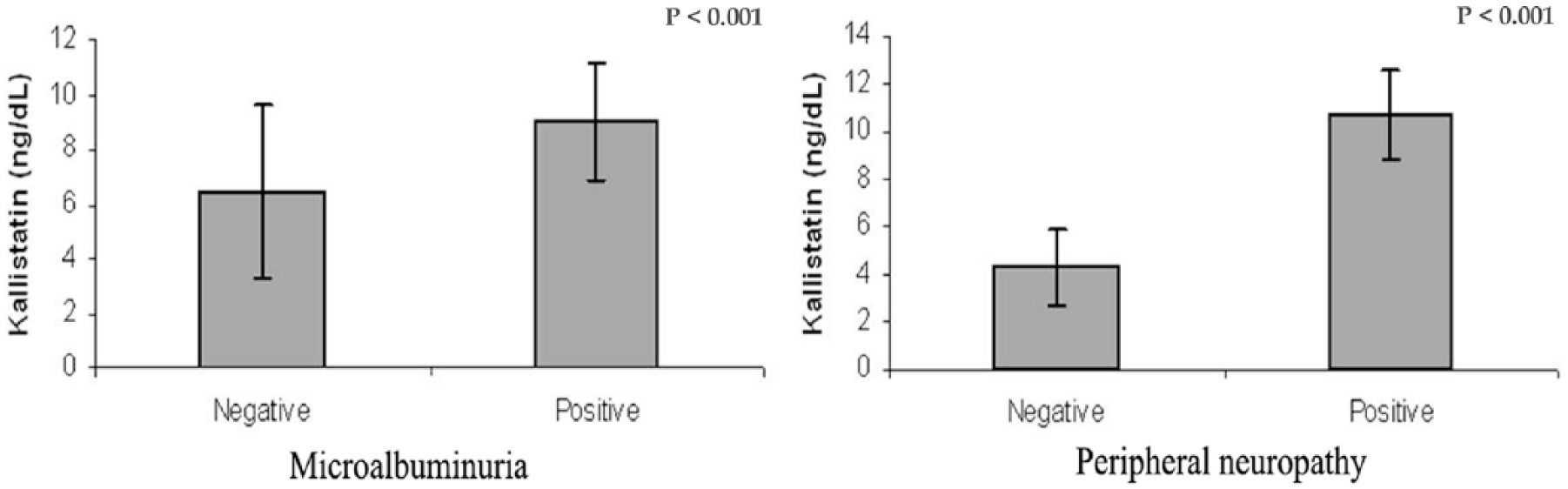
Kallistatin levels in patients with and without microalbuminuria and peripheral neuropathy.
Both right and left CIMT were significantly increased in patients with and without microvascular complications compared with the control group (Table 1). Significant positive correlations were observed between serum kallistatin and disease duration (r=0.513, p<0.001), systolic and diastolic blood pressure (r=0.623 and r=0.67, p<0.001), FBG (r=0.932, p<0.001), HbA1c (r=0.77, p<0.001), triglycerides (r=0.733, p<0.001), total cholesterol (r=0.824, p<0.001), LDL (r=0.719, p<0.001), hs-CRP (r=0.403, p<0.001), and UACR (r=0.694, p<0.001). Kallistatin was also positively correlated to both right and left CIMT (r=0.775 and r=0.881, respectively; p<0.001) (Figure 2). Multiple linear regression analysis showed that FBG, HbA1c, total cholesterol and CIMT were independently related to serum kallistatin levels (Table 2).
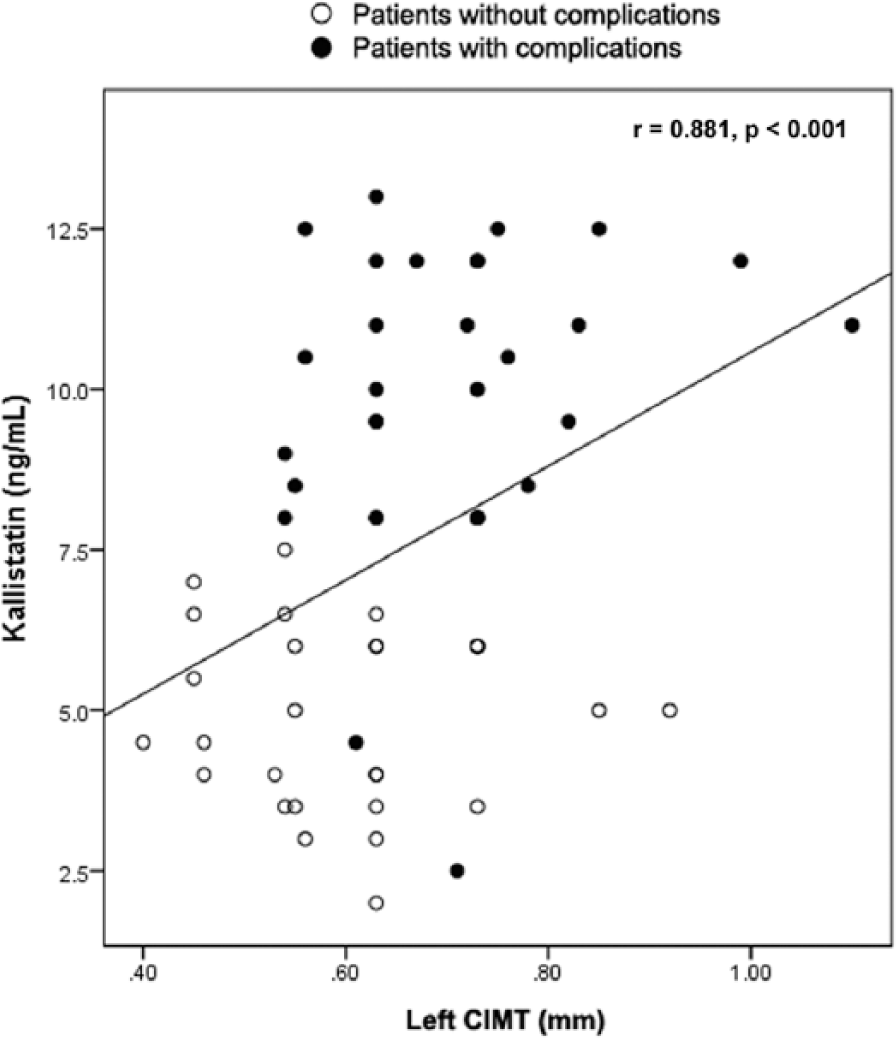
Scatter diagram showing the significant positive correlation between kallistatin levels and CIMT among type 1 diabetic patients with and without microvascular complications.
Multiple linear regression analysis of the relation of serum kallistatin to clinical and laboratory variables in type 1 diabetic patients.
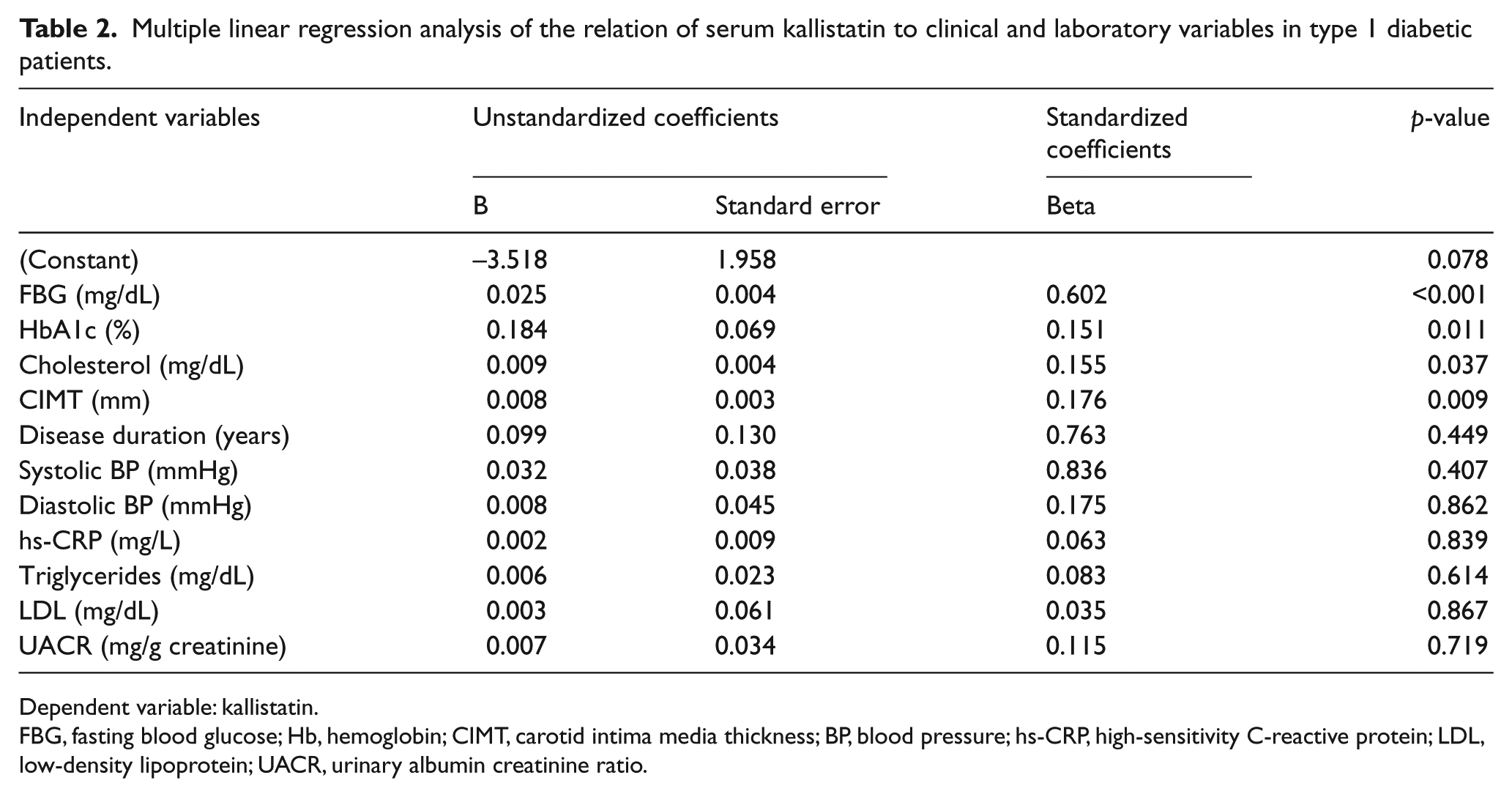
Dependent variable: kallistatin.
FBG, fasting blood glucose; Hb, hemoglobin; CIMT, carotid intima media thickness; BP, blood pressure; hs-CRP, high-sensitivity C-reactive protein; LDL, low-density lipoprotein; UACR, urinary albumin creatinine ratio.
As regards CIMT, significant positive correlations were observed with age (r=0.539, p<0.001), disease duration (r=0.649, p<0.001), systolic blood pressure (r=0.316, p=0.01), FBG (r=0.408, p=0.001), HbA1c (r=0.664, p<0.001), total cholesterol (r=0.633, p<0.001), kallistatin (r=0.881, p<0.001), and hs-CRP (r=0.534, p<0.001). On multiple regression analysis with CIMT as the dependent variable, disease duration, HbA1c, total cholesterol and kallistatin were the significant independent variables (Table 3).
Multiple linear regression analysis of the relation of CIMT to clinical and laboratory variables in type 1 diabetic patients.
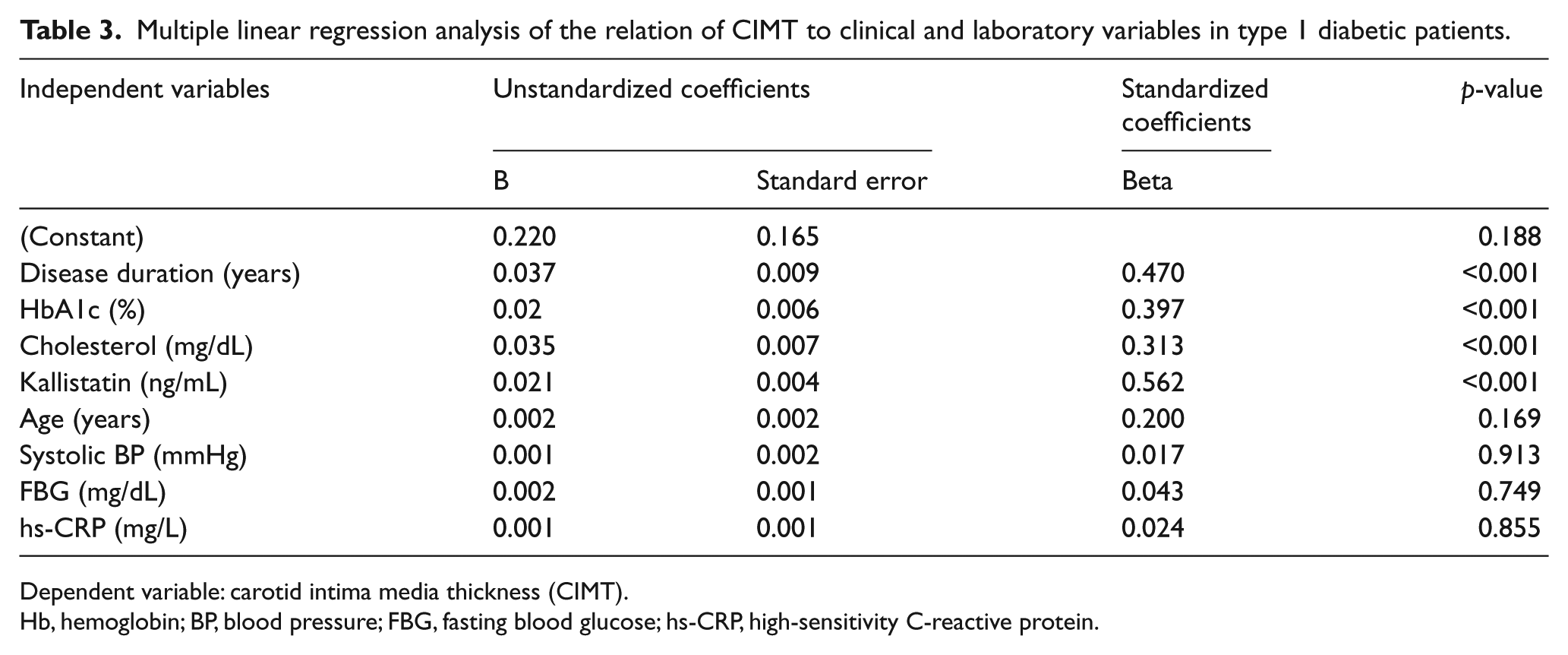
Dependent variable: carotid intima media thickness (CIMT).
Hb, hemoglobin; BP, blood pressure; FBG, fasting blood glucose; hs-CRP, high-sensitivity C-reactive protein.
Logistic regression analysis with microvascular complications as the dependent variable (Table 4) showed that kallistatin was a significant independent factor for microvascular complications (odds ratio 3.82, 95% CI 2.60–9.09; p=0.002). Other significant variables contributing to the occurrence of microvascular complications were disease duration, HbA1c, total cholesterol and CIMT.
Logistic regression analysis for factors contributing to microvascular complications among patients with type 1 diabetes.
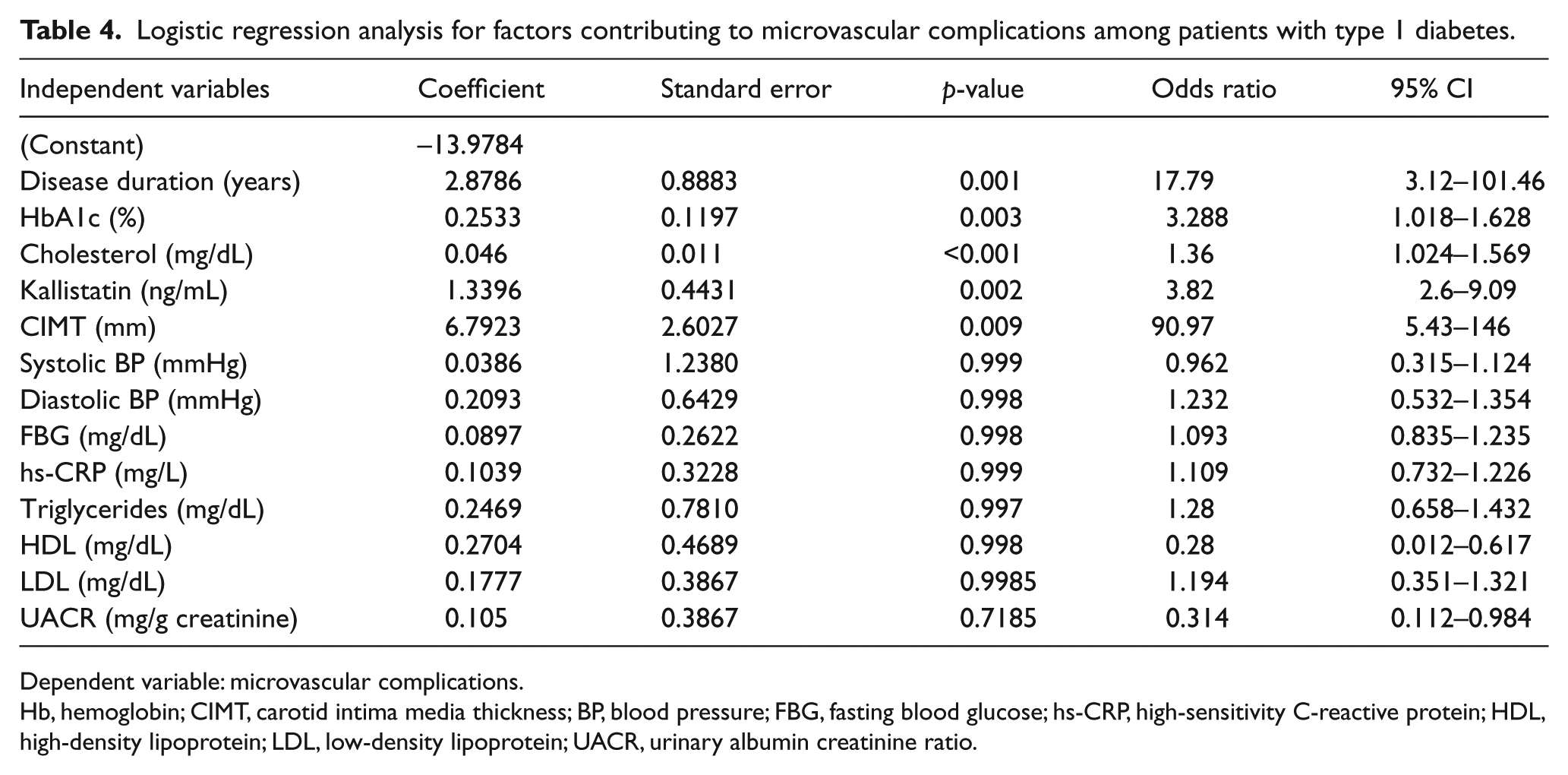
Dependent variable: microvascular complications.
Hb, hemoglobin; CIMT, carotid intima media thickness; BP, blood pressure; FBG, fasting blood glucose; hs-CRP, high-sensitivity C-reactive protein; HDL, high-density lipoprotein; LDL, low-density lipoprotein; UACR, urinary albumin creatinine ratio.
Discussion
Microvascular complications of diabetes are mediated by disturbances in the balance of pro- and anti-angiogenic factors. 6 In this study, the most common microvascular complication was microalbuminuria with an incidence of 31.7%. This was in agreement with Cohen-Bucay and Viswanathan 28 who reported that diabetic nephropathy affects approximately one-third of all people with diabetes. Increased disease duration, poor glycemic control and high blood pressure among our patients with microvascular complications coincides with Maric and Hall. 29 We found increased kallistatin levels in patients with and without microvascular complications compared with the control group, with the highest levels among patients with complications. Elevated kallistatin levels in patients with type 1 diabetes, even in those without complications including patients with normoalbuminuria, could imply the significant role of this marker as a potential predictor of diabetic vascular complications even before development of microalbuminuria. Jenkins et al. 6 showed that kallistatin levels were higher in adults with type 1 diabetes and microvascular complications than in controls, but did not differ between complication-free diabetic patients and controls. Recently, McBride et al. 30 reported that circulation levels of kallistatin were elevated in type 2 diabetic patients with diabetic vascular complications and may contribute to impaired wound healing through inhibition of Wnt/β-catenin signaling.
Kallistatin mRNA is expressed in platelets, neutrophils, lymphocytes, monocytes, endothelial cells, hepatocytes, and colon and prostate carcinoma cells. 31 Studies suggest that metabolic abnormalities of diabetes alter the angiogenic process.32–34 Hyperglycemia induces the generation of reactive oxygen species (ROS) that cause endothelial derangements, including the reduced synthesis and accelerated degradation of endothelial-derived nitric oxide (NO). The bioactivity of NO is critical for the survival, proliferation and migration of endothelial cells.35–37 In addition, inflammatory cytokines increase vascular permeability, change vasoregulatory responses, increase leukocyte adhesion to endothelium, and facilitate thrombus formation. 38 As inflammation proceeds, the risk for vascular complications increases. 39 Thus, inflammation, endothelial dysfunction and disturbed angiogenesis could trigger kallistatin release to counteract the angiogenic and inflammatory responses in type 1 diabetes.
Like other SERPIN members, kallistatin is expressed in the heart, kidney, retina, blood vessels, and many other tissues and cell types. 40 In diabetic retinopathy, unregulated angiogenesis is observed. 41 Therefore, we expected to find high serum kallistatin levels in our studied patients with diabetic retinopathy. However, Ma et al. 42 found significantly lower immunoreactive kallistatin levels in vitreous fluid from 18 patients with diabetic retinopathy compared with 17 non-diabetic subjects. This suggests that decreased vitreous levels of kallistatin may contribute to a proangiogenic and proneuroinflammatory environment in diabetic retinopathy. 42 Jenkins et al. 43 have found a similar pattern with another serpin, pigment epithelium derived factor (PEDF), with low vitreous fluid levels in diabetic retinopathy patients and high serum levels with microvascular complications. Although Matsuyama et al. 44 found that the levels of plasma PEDF increased with advances in both diabetic retinopathy and nephropathy, the level of intraocular PEDF has been shown to decrease with advancing stages of diabetic retinopathy.45,46 Because PEDF is a potent anti-angiogenic and anti-inflammatory cytokine,47,48 PEDF may be consumed in the eye with diabetic retinopathy to counteract the angiogenic and inflammatory responses of the endothelial cell, which would then lead to lower levels. 44 The same theory could be applied to kallistatin as it has potent anti-angiogenic, anti-oxidant and anti-inflammatory effects.10–12 The increase in serum kallistatin levels in diabetic patients with diabetic retinopathy may be a counteractive system that inhibits the vascular damage in type 1 diabetes mellitus.
Increased serum kallistatin in our patients with microalbuminuria may be explained by the fact that kallistatin has been localized in human urine31,49 and in kidneys,10,31,50 where it is thought to regulate salt and water handling, renal perfusion and blood pressure, and to reduce intra-renal fibrosis, inflammation, and oxidative stress.10,31,51,52
In our study, positive correlations were observed between serum kallistatin and disease duration, blood pressure, FBG, HbA1c, lipid profile, hs-CRP, and UACR. This suggests a link between poor metabolic control, low-grade inflammation, albuminuria level and elevated kallistatin levels. It appears that hyperglycemia, dyslipidemia and poor glycemic control are significant contributing factors to increased levels of kallistatin. In line with these results, McBride et al. 30 found a positive correlation between kallistatin and HbA1c in type 2 diabetes. Jenkins et al. 6 found that kallistatin levels were higher in type 1 diabetic patients with hypertension and were significantly correlated with total cholesterol and LDL as well as renal function.
The correlation between kallistatin and inflammation among the studied population supports other studies.51,53–55 In people with (inflammatory) rheumatoid arthritis, plasma and joint kallistatin levels were increased relative to osteoarthritis patients. 54 In animal studies, kallistatin gene delivery has anti-inflammatory and anti-oxidant effects.9,51,55,56 However, Jenkins et al. 6 did not support a major anti-inflammatory role where serum kallistatin levels were not strongly associated with inflammation markers. The authors attributed this to the relatively low level of inflammation in diabetes; therefore, local tissue anti-inflammatory effects are not being well reflected by circulating measures. It is worth noting that the correlation between kallistatin and hs-CRP in our study was weak and further studies using other markers of inflammation are needed to investigate the anti-inflammatory role of kallistatin in type 1 diabetes.
In our study, we found high CIMT among type 1 diabetes patients with and without microvascular complications compared with controls. Disease duration, HbA1c and total cholesterol were independently related to CIMT. Gupta et al. 17 found that CIMT was higher in type 1 diabetic patients with a higher HbA1c versus those with a normal HbA1c and correlated significantly to blood pressure, serum cholesterol and serum LDL, indicating the onset of early cardiovascular changes in adolescence. In this study, kallistatin was positively correlated with CIMT. Whilst kallistatin’s vasodilatory, potential anti-inflammatory and anti-oxidant effects may be beneficial, its potent anti-angiogenic effects may delay wound healing, impair collateral vessel formation and accelerate atherosclerosis. 6 Our findings denote the presence of a state of subclinical atherosclerosis among children and adolescents with type 1 diabetes and support the role of kallistatin in endothelial dysfunction. In agreement with our results, Miao et al. 8 showed that kallistatin plays a role in neointima hyperplasia. Other animal and isolated vascular cell experiments support the role of kallistatin in vascular and cardiac remodeling.10,11
Limitations
Limitations of our study include the presence of a relatively small number of patients in subgroups with a single complication. Although comparison of all patients with microvascular complications revealed similar results to the analyzed subgroups, further prospective analysis including larger numbers of patients for each individual complication might provide additional information. Further longitudinal studies with repetitive samples from each patient are also needed to evaluate whether increased kallistatin levels in type 1 diabetic patients would indicate microvascular damage and decrease with better glucose control or not. Urinary kallistatin excretion was not measured in this study and merits future analysis. The underlying mechanism of increased kallistatin in type 1 diabetes may be multifactorial and represents an important area for future research.
Conclusion
In conclusion, increased serum kallistatin in children and adolescents with type 1 diabetes, particularly in patients with microvascular complications, and its relation with CIMT may reflect the presence of inflammation, endothelial dysfunction and disturbed angiogenesis. This further suggests a link between diabetic micro- and macro-angiopathy. Evaluation of serum kallistatin might become a useful test in the early stages of diabetes to identify normotensive and normoalbuminuric children and adolescents with type 1 diabetes at increased risk of developing vascular complications later in life, who might benefit from a timely and appropriate intervention.
Footnotes
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
