Abstract
The Notch pathway is definitely required for normal vascular development. Although the contribution of Notch in postnatal angiogenesis is the focus of intense investigation, the implication of Notch in reparative neovascularization in the skin remains unexplored. In this study, we investigated Notch changes using a skin model of ischemia. Thirty Sprague-Dawley rats were divided into two groups. In the surgery group (n = 24), a caudally based dorsal skin flap was raised and sutured back into its initial position. In the control group, no surgical procedure was performed. Tissue biopsies were obtained at different time intervals. Tissue specimens were assessed for Delta-like ligand 4 (DLL4) and vascular endothelial growth factor (VEGF) gene expression by real-time polymerase chain reaction (PCR). Immunohistochemical staining was used for detection of DLL4 in tissue materials. Quantitative assessment of skin flap microvasculature was made. Compared with normoperfused tissue, VEGF and DLL4 expressions increased significantly (p < 0.01). Immunohistochemical analysis revealed weak and patchy expression of DLL4 in microvascular endothelial cells of normoperfused tissues. Conversely, DLL4 expression was upregulated in capillary endothelial cells after ischemia. In conclusion, in this study we have shown that the Notch ligand DLL4 is upregulated in skin tissue after ischemia. A deeper understanding of these fundamental principles will aid in the development of new avenues for the treatment of blood vessel-related skin pathologies.
Introduction
Peripheral vascular disease (PVD) is a nearly pandemic condition that is associated with elevated morbidity and mortality. 1 Critical limb ischemia represents the terminal consequences of severe PVD, including chronic ulceration and gangrene. 2 Impaired wound healing in these situations is secondary to inadequate blood supply, and the extent of recovery is dependent on the residual ability of the organism to build up a feeding vascular tree. 3 Ischemia is generally considered to represent fundamental stimuli for angiogenesis by promoting a combination of overlapping processes including anatomic, physiologic, and molecular factors. 4 The concept of therapeutic angiogenesis in ischemic diseases relies on supporting endogenous vascular growth to improve the tissue perfusion. 5 Most of these strategies involved the delivery of growth factors such as vascular endothelial growth factor (VEGF). However, very little success with these strategies has been demonstrated. 6 One of the reasons accounting for this failure is that delivery of single growth factors may represent a simplification of the cooperative molecular interaction necessary for the establishment of a functional vascular network. 7 This underlines the importance of cross talk between the Notch pathway and VEGF in building up a proper neovascularization.
The Notch pathway is an evolutionary conserved signaling system controlling cell fate specification, tissue patterning and morphogenesis. 8 Over the past years, it has become clear that the Notch ligand Delta-like ligand 4 (DLL4) has an essential role in coordinating endothelial cell (EC) behavior during sprouting angiogenesis.9,10 DLL4 signals from tip ECs to neighboring stalk ECs to restrict the emergence of excessive sprouting through repression of VEGF receptor 2 (VEGFR2). This helps in restricting the emergence of excessive sprouting via reduction of responsiveness to VEGF. 11
Chronic skin wounds present a major problem in clinical medicine; therefore novel strategies that can promote angiogenesis in ischemic skin are urgently required. For this reason, deeper insights into the mechanisms underlying ischemia-induced angiogenesis are required. Although the contribution of Notch in postnatal angiogenesis is the focus of intense investigation, the implication of Notch in reparative neovascularization in the skin remains unexplored. In this study, we investigated Notch changes using a skin model of ischemia.
Materials and methods
This study was approved by Başkent University Ethical Committee for Experimental Research on Animals. Thirty male Sprague-Dawley rats weighting between 390 g and 430 g were used. Animals were divided randomly into two groups. In the surgery group (n = 24), a caudally based dorsal skin flap was raised and sutured back into its initial position. Tissue collections were performed postoperatively after 24 hours, 3 days, 5 days and 10 days (n = 6 at each time point) for real-time polymerase chain reaction (PCR) and immunohistochemical analysis at each time interval; animals were killed after sampling procedures. In the control group (n = 6), no surgical procedure was performed.
Surgical procedure
We used a modified McFarlane skin flap model for the surgical portion of this study. 12 This flap was shown to be a reliable model in the study of skin ischemia with predictable necrosis patterns. 13 The dorsa of the rats were shaved and a caudally based 9 × 3 cm flap was drawn on the animals. The palpable hip joints were used as anatomic landmarks to define the base of the flap. Under sterile conditions, incisions were made and the entire flap was undermined below the level of the panniculus carnosus. The flap was elevated and inspected for direct axial or muscular perforating cutaneous blood vessels supplying the base. These vessels, if found, were cauterized to ensure a random skin flap. A sterile silicone interposition sheet was secured with absorbable sutures in the wound bed below the flap to prevent survival of the flap as a graft. The skin flap was returned to its native position and the skin closed with 4-0 sutures (Figure 1).
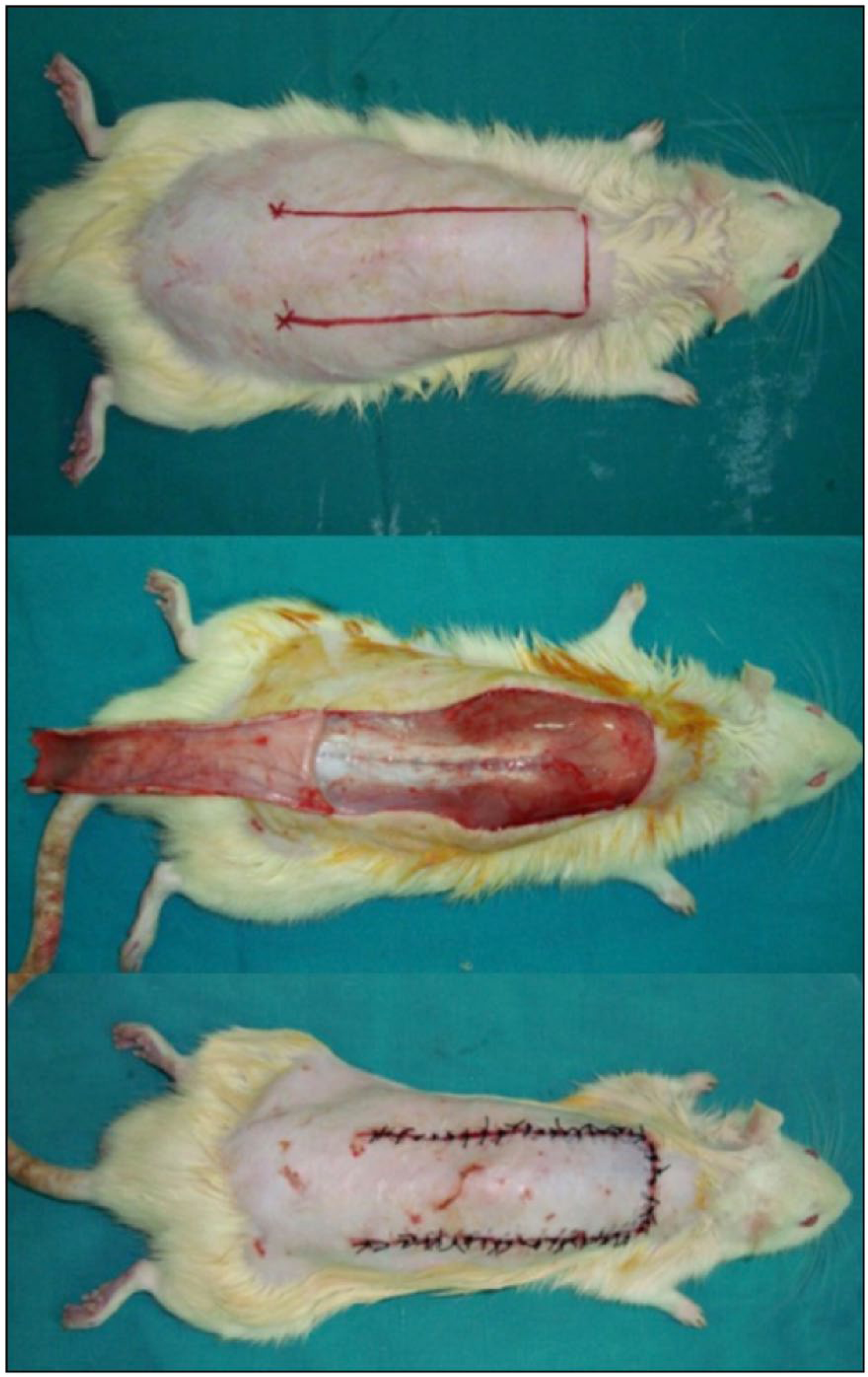
A caudally based dorsal skin flap measuring 9 × 3 cm was raised and sutured back into its initial position with a silicone sheet placed underneath to avoid vascular ingrowth from the wound bed.
Gene expression
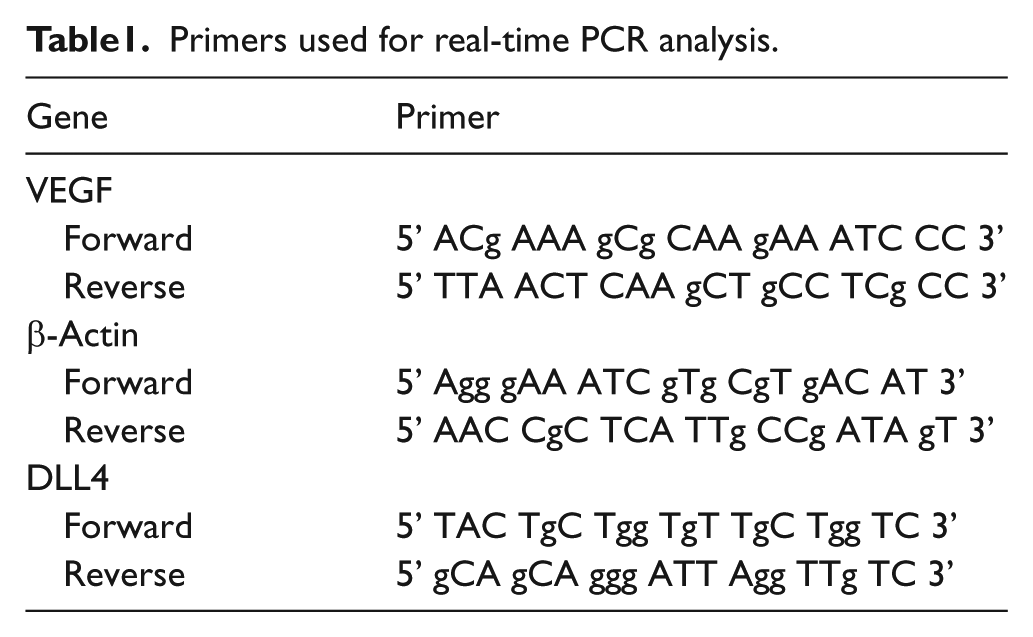
To investigate the effect of ischemia on VEGF and DLL4 expressions, VEGF and DLL4 messenger ribonucleic acid (mRNA) levels were measured at different time intervals using real-time PCR. In the surgery group, full-thickness 5-mm punch biopsies were taken from skin at the center of the outlined flap territory. These animals were divided into four subgroups (six rats for each subgroup) according to the time of biopsies: 1, 3, 5 and 10 days postoperatively. In the control group, skin biopsies were taken from cephalic dorsal skin. Expression of VEGF and DLL4 mRNAs from tissue samples of each group at various time intervals was tested by real-time PCR. Gene expressions were expressed as a fraction of VEGF or DLL4 to β-actin. Whole RNA was extracted from tissue samples using the High Pure RNA Tissue Kit (Roche Diagnostics, GmbH, Mannheim, Germany). The extracted RNA was reverse transcripted to complementary DNA (cDNA) using the Transcriptor High Fidelity cDNA Synthesis Kit (Roche Diagnostics). Real-time PCR was carried out using the LightCycler ® FastStart DNA Master SYBR Green I Kit (Roche Diagnostics). The housekeeping gene β-actin served as an internal control. Relative gene expression data were analyzed using the 2–ΔΔCT method. 14 Primer sequences of the target genes are listed in Table 1.
Primers used for real-time PCR analysis.
Whole-mount immunohistochemistry
Whole-mount immunohistochemical analysis using specific anti-DLL4 antibodies was used to analyze the expression of endothelial DLL4 in tissue samples of each group at various time intervals. Sections (5 μm) of formalin-fixed paraffin-embedded tissues were processed using standard methods. Sections were incubated with primary antibody (Abcam, Cambridge, MA, USA) overnight at 4°C and appropriate secondary antibody (Abcam) for 1 hour at room temperature. All immunostainings were analyzed using a standard light microscope (×400 magnification).
Assessment of microvessel density
Quantitative assessment of skin flap microvasculature was made using sections obtained for immunohistochemical analysis. Microvessel density (×400 magnification) was assessed in five fields for each specimen in a blinded fashion.
Statistics
Compliance with the normal distribution of continuous variables was checked with the Shapiro–Wilk test. Homogeneity of groups’ variances was checked by Levene’s test. Groups’ variances were not homogeneous in terms of VEGF and DLL4 gene expression variables so the Kruskal–Wallis test and then the Dunn test were used for comparisons. Data analyses were performed using the Statistical Package for the Social Sciences, version 17.0 (SPSS Inc., Chicago, IL, USA). A p-value <0.05 was considered statistically significant. The results of statistical analysis were expressed as number of observations (n), mean ± standard deviation (), median and minimum–maximum values [M (min–max)].
Results
Ischemia upregulates the expression of VEGF
Because VEGF is considered the main transacting factor in controlling the rate of transcription of the DLL4 gene, 15 VEGF transcription rates were measured at different time intervals to investigate the possible relationship between the expression patterns of VEGF and DLL4. Recent studies showed that ischemia enhances the expression of VEGF and that VEGF induces angiogenesis in ischemic tissues.16,17 Consistent with these findings, the present study showed that ischemia enhanced the expression of VEGF mRNA in skin tissue. VEGF mRNA levels measured in the control group represent our baseline. All postoperative levels were above the baseline (p < 0.01). Maximum levels were measured on the fifth postoperative day. Expression levels are shown in Figure 2.
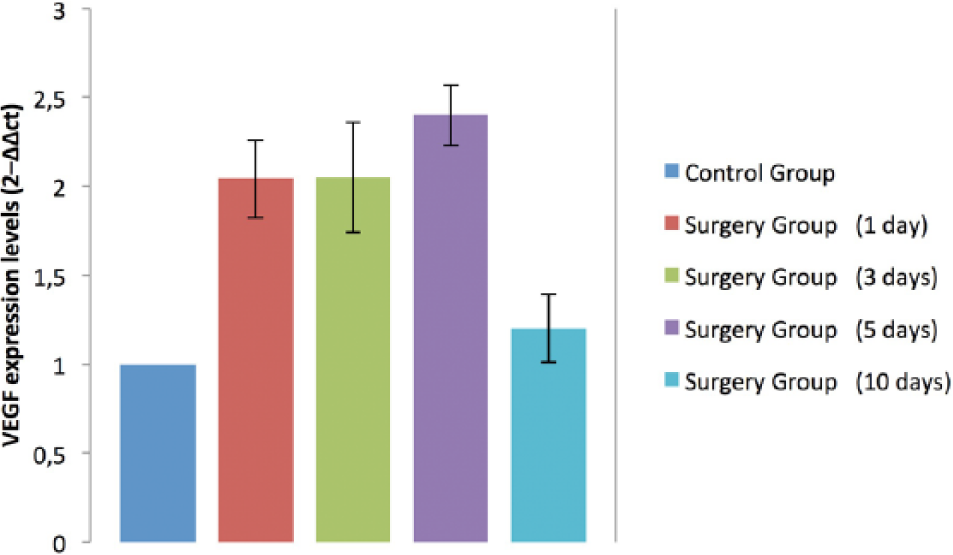
VEGF mRNA levels measured in the control group represent our baseline. All postoperative levels were above the baseline (p < 0.01).
DLL4 is expressed in newly forming vessels after ischemia
Previous studies reported DLL4 to be expressed in embryonic and tumor vasculature.18,19 Less is known about the implication of Notch in the field of ischemia-induced angiogenesis. A study by Al Haj Zen has shown that DLL4 is upregulated in muscle tissue after ischemia. 16 We have shown, for the first time, that DLL4 is expressed in skin tissue after ischemia. The sub-lethal ischemia created in skin flaps has induced the transcription of DLL4. Compared with the baseline levels measured in non-ischemic tissues of the control group, all postoperative levels of DLL4 mRNA were higher (p < 0.01). All DLL4 expression levels are shown in Figure 3. Patterns of increased DLL4 transcription are similar to those observed in VEGF transcription with maximum levels measured on the fifth postoperative day. The co-expression of VEGF and DLL4 after ischemia is coincident with the time and distribution of neovascularization in ischemic skin, suggesting that these factors are temporally and spatially correlated with angiogenesis. VEGF is mitogenic for ECs and functions as during the earliest stages of vascular development, whereas DLL4 alone does not induce endothelial proliferation but plays a role in the regulation of sprouting, indicating that VEGF and DLL4 have complementary roles to form new vessels. Immunohistochemical analysis using specific anti-DLL4 antibodies revealed weak and patchy expression of DLL4 in microvascular ECs of normoperfused tissues. Conversely, DLL4 expression was upregulated in capillary ECs after ischemia (Figure 4).
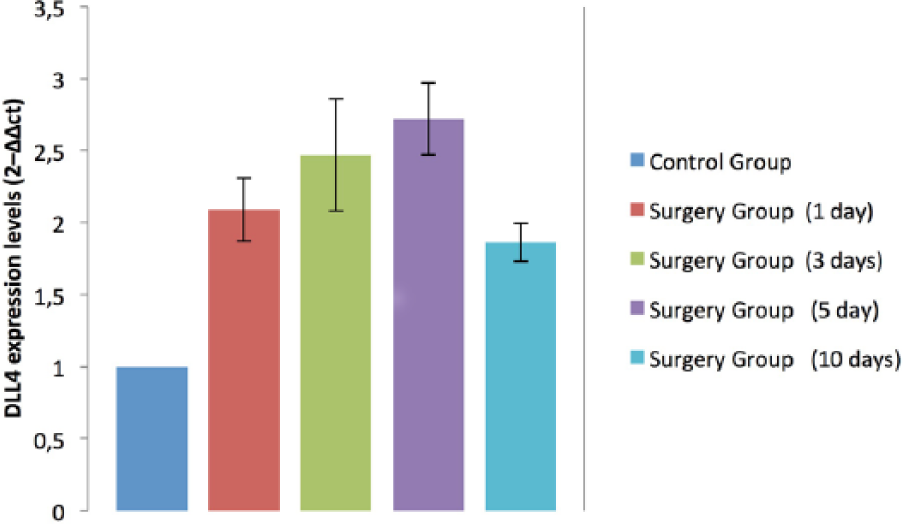
DLL4 mRNA levels measured in the control group represent our baseline. All postoperative levels were above the baseline (p < 0.01).
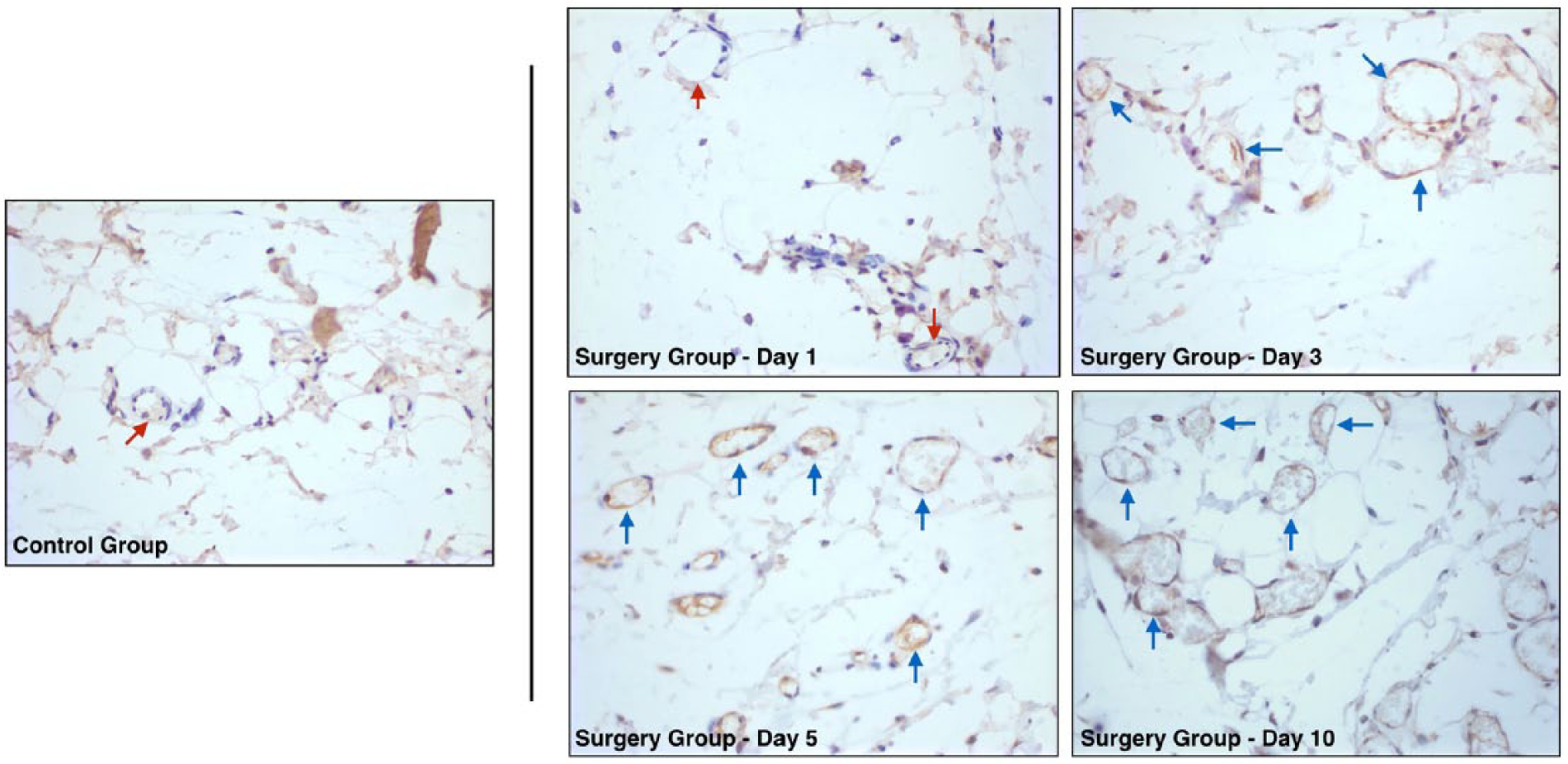
The darker/brown color indicates positive staining for anti-DLL4 antibodies. Immunohistochemical analysis using specific anti-DLL4 antibodies revealed weak and patchy expression of DLL4 in microvascular endothelial cells of normoperfused tissues of the control group and early phases of the surgery group (arrows: Control Group and Surgery Group – Day 1). Conversely, DLL4 expression was upregulated in capillary endothelial cells after ischemia at different time intervals (arrows: Surgery Group – Days 3, 5 and 10).
Microvessel density is increased in skin flaps after ischemia
Histologic examination of the flap specimens revealed no significant increase in microvessel density on day 1 when compared with the control group. On days 3 and 5 postoperatively, new capillary formation and capillary density in the ischemia group were increased when compared with the control group. On day 10, the number of new vessels was greatly increased and they all had obvious lumen and blood cell perfusion in the treatment group. The mean microvessel density was (11.6 ± 2.4) in the treatment group and (4.7 ± 1.3) in the control group, respectively, with a statistically significant difference (p < 0.01).
Discussion
The contribution of Notch in postnatal angiogenesis is the focus of intense investigation. Most of these studies are in the field of tumor vascularization. In various models, strong DLL4 expression was observed in the majority of tumor vessels, contrasting with significantly lower vascular expression in adjacent normal tissues.20,21 In humans, DLL4 expression was analyzed in tumors from the kidney, bladder, colon, brain and breast.22–24
In this study, we provided evidence supporting the involvement of the Notch through its ligand DLL4 in the regulation of ischemia-induced angiogenesis in a skin model. VEGF and DLL4 expressions were found to be increased in ischemic skin compared with normoperfused tissues. Immunohistochemical analysis confirmed that the upregulated DLL4 molecules are involved in the process of sprouting angiogenesis. We found weak and patchy expression of DLL4 in the microvascular ECs of normoperfused skins. Conversely, DLL4 expression was upregulated in capillary ECs after ischemia. Taken together, the above data provide evidence for the integration of VEGF and DLL4 signaling pathways as key coordinated regulators that together control sprouting angiogenesis. VEGF serves as the primary initiator of angiogenic sprouting.25–27 Coincidentally, with initiating angiogenesis, VEGF also upregulated DLL4 expression within the endothelium of angiogenic vessels. Activated Notch acts in feedback fashion as a ‘brake’ or negative regulator of VEGF-induced angiogenesis, modulating this process to control over exuberant vascular sprouting, thereby promoting the timely formation of a productive and well-differentiated vascular network. 15
A recent study by Al Haj Zen has demonstrated that DLL4 signaling is fundamental for the formation of a functional vascular network in a model of ischemic muscle. 16 It has been shown that DLL4 transcription is induced in adductor muscles after femoral artery ligation, with peak mRNA expression at 3 days after ischemia. Immunohistochemical staining revealed that DLL4 is mainly expressed in capillary sprouts. However, DLL4 inhibition resulted in the formation of an inefficient neovessel network unable to support post-ischemic muscle recovery. We believe that Notch inhibition has a dose-dependent manner such that it may be possible to modulate vascular response to achieve pro-angiogenic outcome. Recent studies have shown that the low dose of DLL4 inhibitor might have the therapeutic potential for promoting the revascularization and reperfusion of ischemic tissues.28,29
Besides its regulator function in angiogenic sprouting, DLL4 expression has been found to be a fundamental step in endothelial tip/stalk cell specification. In a region of hypoxia, all ECs become activated by VEGF simulation to express the Notch ligand DLL4. This gives rise to a non-uniform population of ECs. Endothelial cells that produce more DLL4 than its neighbors will eventually acquire the tip cell phenotype, whereas the others remain in the stalk phenotype.11,15,18,21 Effective vascular function requires a balance of tip and stalk cell numbers, which is coordinated by Notch.
Immunohistochemical staining showed a weak expression of DLL4 in ECs of normoperfused tissues. This shows that the Notch pathway is active in a basal level even in the absence of ischemia. This observation indicates that the Notch pathway may have critical post-angiogenic functions. It has been found that the Notch pathway controls post-angiogenic remodeling by modulating vasoconstriction and blood flow. 30 This function of the Notch appears to be largely independent of VEGF signaling.
In this study, we have shown that the Notch ligand DLL4 was upregulated in skin vessels after ischemia. Immunochemical staining in ischemic tissues showed a similar immunoreactivity pattern against DLL4 antibodies in all vascular structures. This shows that the notch ligand DLL4 is expressed in both pre-existing and newly forming vessels after ischemia. However, our method does not provide quantitative information about DLL4 expression in both vessel types. A detailed study about the spatial relationship between DLL4 expression and sprouting angiogenesis showed that DLL4 is mainly expressed in new forming sprouts. 16 This is compatible with the possibility that the Notch ligand DLL4 plays a role in regulation of tip EC formation.
The Notch appears to function as an anti-angiogenic factor, negatively regulating the sprouting activity of VEGF. Thus, blocking Notch function genetically or pharmacologically will result in increased vascular density. It might be possible to achieve a functional pro-angiogenic effect via anti-Notch therapy to treat conditions where the formation of new blood vessels is essential and beneficial. The mechanism of action of anti-Notch therapy is strikingly different to that of classic pro-angiogenic therapies that utilize exogenous growth factors.6,31 Notch inhibition augments the angiogenic effect of local VEGF molecules. This provides a promising field for investigations in building up a proper neovascularization.
Recently, several groups have investigated whether inhibition of the Notch pathway might affect tumor angiogenesis and growth. Inhibition of Notch signaling caused increased vascular sprouting in tumors. Surprisingly, this vascular overgrowth phenotype resulted in tumor growth inhibition.19,32 Perfusion studies demonstrated that the hypersprouting tumor vasculature was non-functional and, consequently, anti-Notch-treated tumors exhibited increased levels of hypoxia with resultant growth inhibitory effects on tumors.19,21,32,33 It appears that excessive branching results in a highly chaotic vascular network that lacks the hierarchy essential for efficient directional blood flow.19,21,32 In contrast to tumoral vasculature, we think that Notch inhibition in ischemic skin tissue may result in the formation of a functional vascular network. A possible explanation of the non-functional nature of tumoral neo-vasculature observed in anti-Notch therapy is the fact that blood vessels in a given solid tumor may, in fact, be mosaic vessels, composed of ECs and tumor cells. 34 Thus, inducing sprouting on these abnormal vessels will likely lead to an abnormal vascular network. In addition, very high doses of Notch inhibitors are used in these studies. By modulating therapy dosage, a functional pro-angiogenic effect may be achieved. DLL4 inhibition accelerated wound regeneration by creating a neo-vasculature that has a slight increase in vascular density. 17
Conclusion
In this study, we have shown that the Notch ligand DLL4 is upregulated in skin tissue after ischemia. More studies are needed to document the functional importance of the Notch pathway in the regulation of angiogenesis in ischemic skin tissue. A deeper understanding of these fundamental principles will aid in the development of new avenues for the treatment of blood vessel-related skin pathologies.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was approved by Başkent University Ethical Committee for Experimental Research on Animals and supported by Başkent University Research Fund.
The authors have no financial interest or other relationship with the manufacturers of any products or providers of any service mentioned in this article.
