Abstract
Although there have been advances in coronary artery bypass grafting and percutaneous coronary intervention, some patients who have ischemic coronary artery disease (CAD) are ineligible for revascularization due to suboptimal anatomy. Cardiac angiogenesis is not only a physiological response to ischemia or hypoxia but also a potential target of therapeutic strategies. Preclinical studies have shown a great enthusiasm on therapeutic angiogenesis for ischemic CAD. However, the latest trials provided the limited evidence on its efficacy. This article aims to discuss the physiological process of angiogenesis, the characteristic of angiogenic growth factors, delivery system, and clinical and preclinical studies, which can provide a novel insight into the therapeutic angiogenesis for CAD.
Introduction
Cardiovascular disease is the primary cause of death in human beings and has resulted in over 30% of the global deaths (World Health Organization fact sheet for cardiovascular disease, 2012). During past decades, coronary artery bypass grafting (CABG) and percutaneous coronary intervention (PCI) became the common treatments for acute myocardial infarction (AMI) and angina pectoris, which significantly led to the reduction in coronary artery disease (CAD) mortality. However, approximately 30% of patients with CAD are unsuitable for treatment with PCI or CABG due to suboptimal anatomy or complicated vascular conditions. 1 Patients with coronary heart disease (CHD) find it significantly challenging to look for a novel approach. Cardiac angiogenesis with angiogenic growth factors (AGFs) has been demonstrated to potentially generate new vessels for restoring blood flow in ischemic myocardium and improve the heart’s functionality. 2 Therefore, taking an insight into the process and mechanisms of angiogenesis is significant for improving therapeutic angiogenesis in CAD.
Angiogenesis is a multicellular morphogenetic process, in which new blood vessels spring from a preexisting vasculature controlled by a complicated interplay of antiangiogenic and proangiogenic molecules. The angiogenic process occurs in a highly orchestrated order, comprising the interaction among AGFs, smooth muscle cells (SMCs), endothelial cells (ECs), pericytes, the extracellular matrix (ECM), and fibroblasts. 3 -5 Although bolus injection of AGFs at the ischemic area of the heart had shown to be nonbenefit on angiogenesis at undesired area generation, 6,7 the latest targeted delivery system of AGFs has brought about a new promise. 8,9 In this article, we focus on the AGFs in physiological angiogenesis, AGFs delivery system, and recent preclinical, and clinical studies to provide a novel insight into therapeutic angiogenesis in a clinical setting.
Process of Angiogenesis
The angiogenic process consists of 4 steps. First, ECs are stimulated by AGFs, such as basic fibroblast growth factor (bFGF), transforming growth factor beta (TGF-β), vascular endothelial growth factor (VEGF), and platelet-derived growth factors (PDGFs). Second, the basement membrane of the blood vessel is degraded by activated ECs via extracellular proteinases such as matrix metalloproteinases, ECs loosen their junction, and the vessel dilates. One type of ECs, known as tip cells, forms a growth cone and navigate the extending vessels, which is arbitrated by adhesion molecules (such as integrins and ephrins; Figure 1). Meanwhile, the neighbors of the tip cells assume subsidiary positions as stalk cells, which divide to elongate the stalk and form the vessel lumen (Figure 2). Finally, the vessel becomes mature and stable for transmitting blood and oxygen. Pericytes and SMCs in the angiogenic process also signal to the endothelium to stabilize the new vessels. 4,10 Moreover, ECM remodeling together with interactions between ECs and mural cells accommodate the nascent vascular network 11,12 (Figure 3).
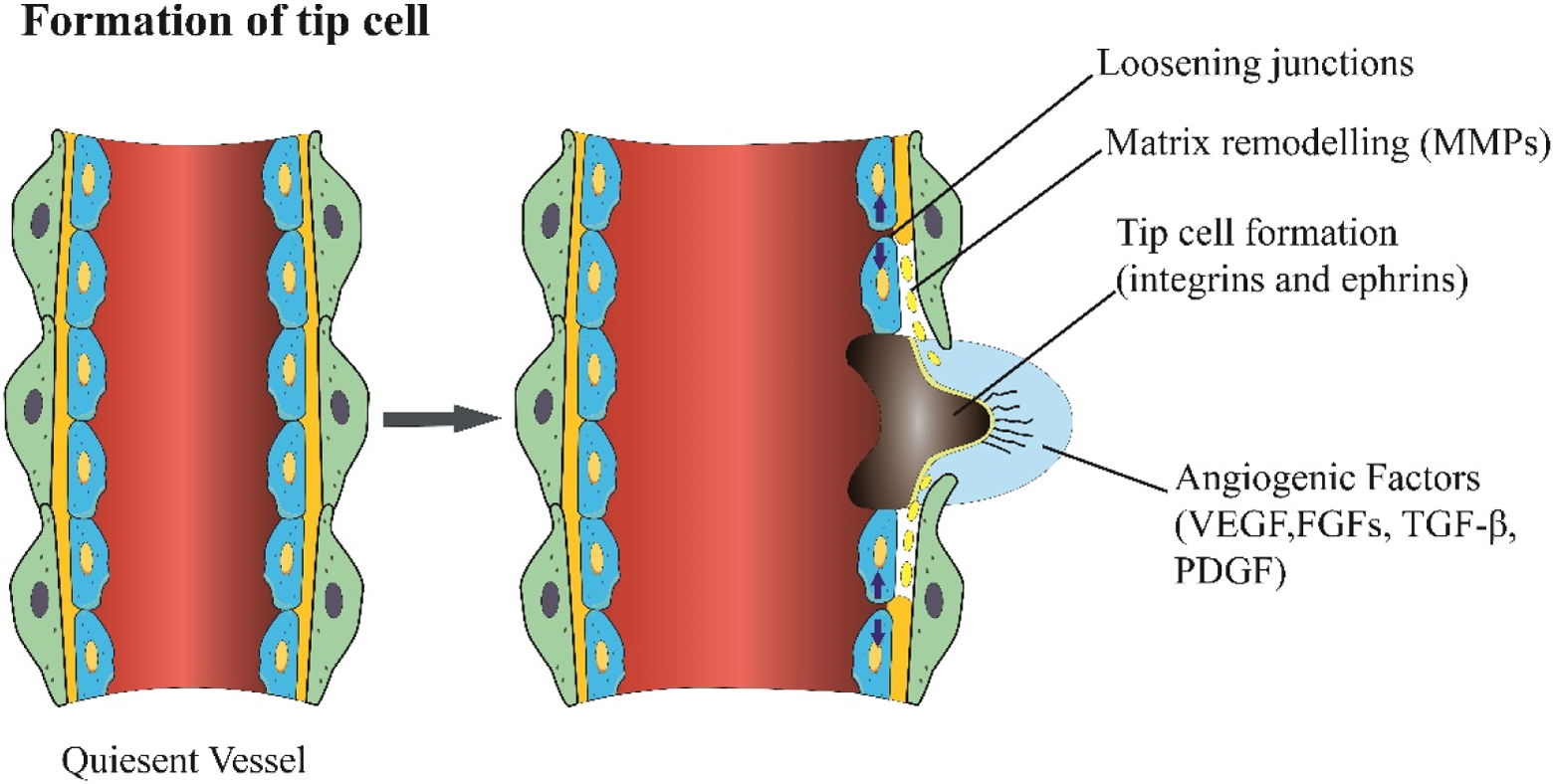
When stimulation was conducted using the angiogenic factors, endothelial cells and the quiescent vessel dilates and resulted in the expansion of their junction. Tip cell formation needs degradation of the pericyte detachment and the basement of the membrane. Increased permeability permits proteases, such as MMPs, remodel interstitial matrix which enables cell migration. MMP indicates metalloproteinase.

The proliferation of stalk cell behind the tip cell elongates to form a lumen. Stalk cells attract pericytes and deposit basement membranes to become stabilized.

After fusion of neighboring vessels, neovessels are formed and resumes stabilization through establishing the junctions, maturation of pericytes, and removal of the basement membrane, enabling the blood flow perfusion.
Angiogenic Growth Factors
Vascular Endothelial Growth Factor Family
The VEGFs are the important managers of angiogenesis. 13,14 The VEGFA, B, C, D, E, and placenta growth factor are part of the VEGF family. The VEGFA concentrations are deficient in healthy humans (1-3 pM in plasma and tissue interstitial fluid), with increasing production due to hypoxia (eg, 1-2× in diseased tissue, 5-10× in exercising a skeletal muscle, and 10× in wounds, comparative to the normal tissue). 15 -17 The VEGFs spread through interstitial space to bind the ECM and cell surface receptors, stimulating endothelial sprouts to hypoxic regions. 18 Although there are 5 VEGF ligands and 3 VEGF receptor tyrosine kinases, studies have focused on the binding of VEGFA to VEGF receptor 2 on ECs. Every VEGF ligand gene can be spliced into many isoforms, such as VEGFA165, VEGFA189, VEGFA121, and so on. The VEGFA165 and VEGFA189 are longer VEGFA splice isoforms that contain heparin-binding domains that bind to the coreceptor neuropilin 1, 19 the shorter isoform VEGFA121 does not bind to the ECM. The longer VEGFA molecules form steep concentration gradients in extracellular space, which remains near the site of vascular formation, 20 whereas VEGF121 forms shallow concentration gradients. Consequently, VEGFA165 and VEGFA189 produce large amounts of thin, highly branched vessels in mice and tumors, while non-ECM binding of VEGFA121 produces few of leaky, wide-diameter blood vessels. 20 -22
Fibroblast Growth Factor Family
The FGF1 and FGF2, expressed by ECs and many other cells, 23 are the main factors in angiogenesis and bind to FGF receptor 1 (FGFR1). The FGFs are usually at a concentration of 0.6 pM in the blood of healthy individuals and increases to 6 pM in individuals with chronic liver disease. 24 The FGF2 increased 2-fold due to hypoxia. 25,26 The FGFs played a major role in the regulation of the angiogenetic process, including the degradation of the basement membrane, integrin expression, proliferation, and migration of EC and vessel maturation. 27 The FGF-FGFR complexes amplify the angiogenic signal by cell-surface heparan sulfate proteoglycans. 28,29 Moreover, the crosstalk of FGFs and VEGF system were discovered in gene expression and signal transduction. 30
Platelet-Derived Growth Factor Family
The PDGF family composes of several homodimers of A-, B-, C-, and D-polypeptide chains and heterodimers (PDGF-AB), which mainly bind to 2 types of receptors (PDGF-α and PDGF-β belong to class III and tyrosine kinase receptor families, respectively). 31 The PDGF concentration in the plasma is comparable to the concentrations of VEGFs and FGFs in plasma. 32 Of the PDGFs, PDGF-B attracts the interest of researchers recently. The PDGF-BB and PDGF receptor-F-(PDGFR-β PDGFR are involved in vascular remodeling. 33 The PDGF-BB, produced by vascular ECs, is induced by hypoxia and binds to PDGFR-β expressed in vascular SMCs and pericytes. The EC-secreted PDGF-BB in the vascular basement membrane is associated with pericyte recruitment to maturing microvessels. 34,35 In a PDGF-BB-deficient mouse model, pericytes were found to partial detach from microvessels. 36 Another study on mice with endothelial ablation of PDGF-B demonstrated a negative correlation between pericyte density and the retinal microvascular abnormalities in patients with diabetic. 37
Other Types of AGFs
Inflammation is an important trigger for angiogenesis in an ischemic tissue, and some inflammatory cytokines, such as tumor necrosis factor 38 and interleukin-6, 39 have been demonstrating an accelerating role in angiogenesis. The other types of molecules include platelet-derived EC growth factor/thymidine phosphorylase (PD-ECGF/TP), 40 angiopoietins, 41 TGF, 42 hepatocyte growth factor, 43 insulin-like growth factors (IGFs), 44 thrombospondin 1, and angiostatin, 45 erythropoietin, 46 granulocyte-macrophage colony-stimulating factor (GM-CSF), 47 G-CSF, 48 angiotensin II, 49 and endothelins. 50 In addition to these molecules listed, adrenomedullin, 51 leptin, 52 adiponectin, 53 resistin, 54 neuropeptide-Y, 55 vasoactive intestinal peptide, 56 tryptase, and chymase 57 have all shown to be associated with angiogenesis. However, early studies have only focused on VEGF and FGF in clinical trials (Table 1).
Angiogenic Growth Factors Potentially Applicable for Therapeutic Angiogenesis.

Delivery Systems of AGF
Although some delivery systems were developed, to include recombinant proteins or genes that encode the relevant AGFs, current research with a bolus injection of AGFs at the ischemic area have shown little efficacy in the patients with CAD. Problems such as tumor formation and angiogenesis at undesired sites either systemically or locally administered by the intra-arterial administration, intravenous, and direct intramuscular injection have surfaced. 6,7 Both the concentrations and the life span of AGFs have pivotal roles in angiogenesis. The AGFs such as VEGF, FGF, and PDGF bind to their homologous receptors with short half-lives (from minutes to hours), 58,59 and AGFs are needed at 10−9 to 10−11 M concentrations in a long time (from hours to days) that can play angiogenic roles. 60,61 However, the controlled delivery system has brought out a novel promising strategy in therapeutic angiogenesis.
Protein or Gene Therapy?
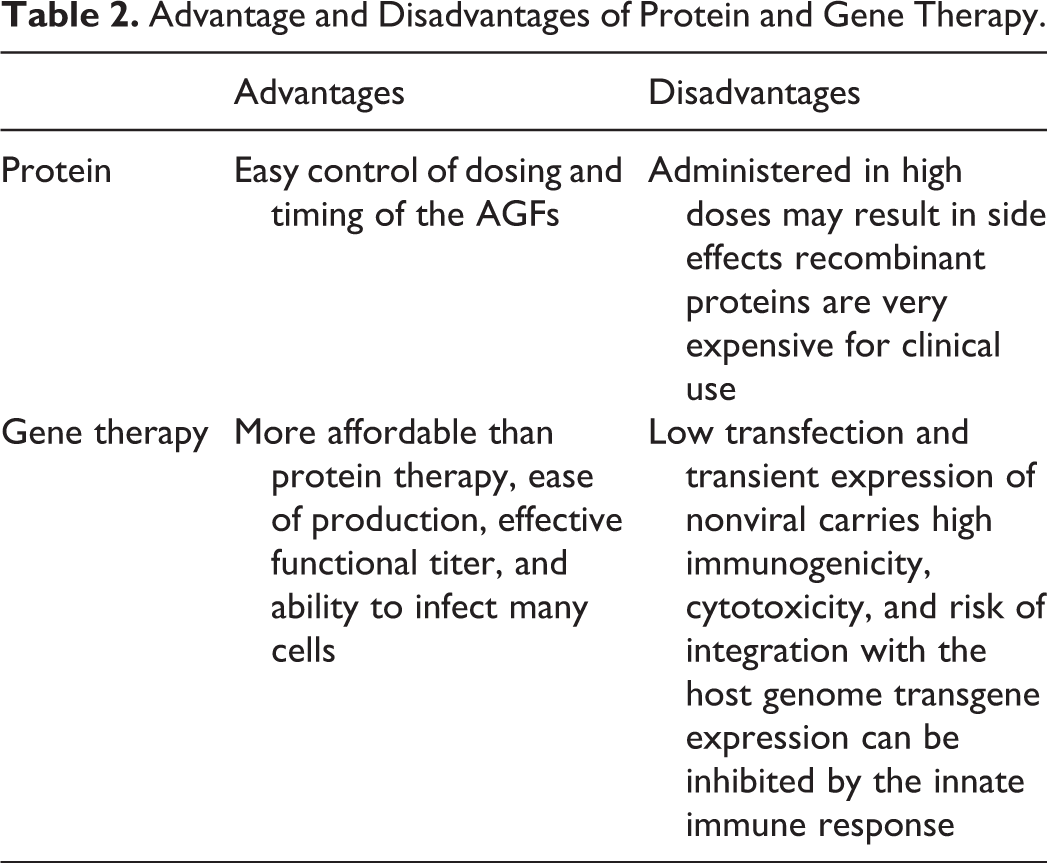
Exogenous administration of AGFs, such as recombinant and purified proteins, is a potent modality for therapeutic angiogenesis, which is the most practical and conventional ways to control dosing and timing of the AGFs. However, recombinant protein therapy has many limitations. First, AGFs are usually administrated systemically in a high plasma dose, which may result in significant side effect. Second, recombinant proteins are very expensive for clinical use.
Gene therapy for angiogenesis is more affordable over protein therapy,
62
but gene delivery carrier and vectors pose the greatest challenge.
63
The vector or carrier of gene therapy for angiogenesis should have following features: no toxicity, high efficiency, low immunogenicity, and nucleic acid–loading capacity
Advantage and Disadvantages of Protein and Gene Therapy.
Controlled Delivery System
To develop the controlled delivery system is a crucial issue for cardiac angiogenesis because of the instability and local abnormally high concentration of most AGFs. The increasing evidence demonstrates that ECM can be viewed as the biological material system, so understanding the biochemical and biophysical properties (eg, stiffness, degradability, and numbers of cell-binding sites or growth factor-binding sites) of ECM is useful for designing bioactive materials. Hydrogels are the most prevalent type of biomaterial that has been engineered to sequester and release AGFs with spatial and temporal cooperation. Different hydrogels were developed for angiogenic therapies, including synthetic and natural biomaterials. For the natural biomaterials, fibrin, dextran, alginate, hyaluronic acid, collagen, and gelatin are commonly considered. Among them, fibrin, collagen, and gelatin hydrogels are purer and provide cell-friendly backbones with native cell adhesion and protease-sensitive sequences. However, their applications are not popular because of their limited release to ECM; hyaluronic acid, alginate, and dextran are all cell-compatible polysaccharides with low immunogenicity, but their incorporation with cell-adhesive or cell-degradable peptides often result in their overexpression. Moreover, although the natural biomaterials are the highlight of current studies, they may potentially bring about immunogenicity, pathogen transmission, ethical concerns, and so on. The synthetic biomaterials including poly-lactide-co-glycolide (PLGA) and polyethylene glycol have an excellent controllability, and their biophysical and biochemical properties, to include stiffness, specificity of cell adhesion, 67 protease-sensitive sites, 68 and degree of cross-linking, can be regulated precisely for angiogenesis. 69 Similar to some natural materials, the complexity of incorporating multiple biomolecular into synthetic biomaterials can be substantially increased because of their lack of biological function and cell-stimulating capacities. 70
Preclinical Studies
Vascular Endothelial Growth Factor
Animal studies with VEGF focused primarily on the VEGF165. VEGF165-containing hydrogel injected into a perinfarcted area immediately after myocardial infarction is found to reduce the collagen content in the myocardial infarction area, inhibit cell apoptosis, and improve cardiac function when compared with the control group that used saline injection alone. 71 Implantation of a collagen membrane patch loaded with VEGF into infarcted rabbit myocardium improved left ventricle function and vascularization comparative to the control group. 72 In a rat model, Miyagi and colleagues replaced the right ventricular free wall defect with VEGF165-immobilized collagen scaffold and indicated that the covalently immobilized scaffolds improved tissue formation and increased blood vessel density in heart tissue of the patch area. 73 The treatment for 1 month with controlled delivery of VEGF165 by PLGA microparticles in a rat model with myocardial ischemia–reperfusion increased angiogenesis. 74 Sequential delivery of VEGF and IGF-1 from biodegradable gelatin microspheres in rats with heart failure (silk suture ligating the proximal left anterior descending coronary artery) was proven to attenuate myocardial remodeling. 75 Also, gene transfer by adenoviral VEGF-A165 and soluble form of VEGF-D injected intramyocardially into nonischemic pig hearts resulted in angiogenesis and was verified by alpha-smooth muscle actin staining in the transduced regions. 76
Fibroblast Growth Factor
Administration of microparticles containing FGF1 and neuregulin 1 improved cardiac function and inhibited cardiac remodeling with smaller infarct size and induced tissue angiogenesis in a myocardial infarction rat model. 77 Newzealand white rabbits were injected with intramyocardial FGF-1 and enoxaparin after ligation of the left anterior descending artery, which increased the regional myocardial blood flow and the microvessel density. 78 The FGF-2-impregnated gelatin hydrogel microspheres introduced into the myocardium improved ventricular and angiogenesis function in a rat myocardial infarction model. 79 The FGF-2 fused into fibrin glue improved myocardial cardiac and perfusion function and increased the vascular density in a canine model of acute infarction. 80 In a swine model of myocardial ischemia, a single intracoronary infusion of adenovirus 5 FGF-4 was found to improve regional blood flow and cardiac function in the ischemic region. 81
Clinical Trial
Vascular Endothelial Growth Factor
In a EUROINJECT-ONE randomized trial of 80 patients with severe stable ischemic heart disease, the VEGF-A165 plasmid (0.5 mg) was delivered by catheter-based intramyocardial injection demonstrating that 82 the myocardial perfusion evaluated by single-photon emission computed tomography (SPECT) after 3 months’ follow-up did not improve significantly as compared with placebo plasmid. There were 93 patients in the NORTHERN trial with refractory angina, which were treated with a VEGF-A165 plasmid (2 mg), and delivered via the endocardial route with an electroanatomical guidance catheter. After 6 months of follow-up, the patients showed no improvement in myocardial perfusion detected with SPECT. 83 A total of 67 patients with severe angina were randomized to receive AdVEGF121 gene transfer via mini-thoracotomy or to receive medical treatment in Revascularization study. It revealed that AdVEGF121 gene transfer improved total exercise duration and angina symptom as measured by Canadian Cardiovascular Society Angina Class compared with a control group who received medical treatment, but SPECT evaluation showed no improvements in myocardial perfusion. 84 These negative results mentioned above may be associated with many factors, such as incomplete blinding, catheter systems, and vector delivery.
The other clinical trials have also indicated that some different isoforms of VEGF could enhance angiogenesis. Phase I trial (GENESIS I) focused on assessing the safety and the efficacy of intramyocardial high-dose plasmid-VEGF-165 (pVEGF165) gene transfer in no-option patients with CAD demonstrated that high-dose intramyocardial pVEGF165 is safe and the angina functional class decreased. However, after following up for 2 years, it has been found that the stress ejection fraction did not change, which indicated that the efficacy of VEGF must be taken cautiously. 85 Also, a phase II trial was performed to evaluate the safety and effectiveness of catheter-mediated AdVEGF-D regenerative gene transfer in patients with refractory angina in whom revascularization cannot be performed. The primary outcome of this trial is that Canadian Cardiovascular Society class after follow-up for 6 months assessed the heart function and the clinical symptoms using a 6-minute walking (ClinicalTrials.gov Identifier: NCT03039751). The phase I KAT301 trial will now recruit 30 patients with CHD and that would be treated with an adenoviral vector encoding VEGF-D complementary DNA, in which, different vector doses will be injected at 10 different sites in the endocardium. The primary outcomes are safety and tolerability of the vector as well as the distribution of antiadenovirus antibodies and VEGF levels (ClinicalTrials.gov Identifier: NCT01002430).
Fibroblast Growth Factor
The Angiogenic Gene Therapy (AGENT)-1 trial was to evaluate the safety and anti-ischemic effect of 5 ascending doses of Ad5-FGF-4 in 79 patients with chronic stable angina Canadian Cardiovascular Society class 2 or 3. After following up for 4 weeks, the patients who received Ad5-FGF showed symptomatic clinical improvement. 86 Subsequently, a randomized, double-blind, placebo-controlled AGENT-2 trial was conducted to determine whether intracoronary administration of the adenoviral gene for FGF (Ad5-FGF-4) could improve myocardial perfusion. After 8 weeks of follow-up, the Ad5-FGF-4 treatment showed modest improvement in myocardial perfusion evaluated by SPECT. 87 The AGENT-3 and AGENT-4 trials recruited 532 patients with CAD. After a pooled data analysis of the 2 nearly identical trials, both trials ended due to no substantial differences in the primary end point (treadmill exercise time). 88 Another randomized controlled clinical trial in Russia to evaluate the efficacy and safety of Ad5-FGF-4 using adenosine myocardial perfusion SPECT imaging in 100 patients with stable angina pectoris is currently in the recruitment phase. 89 The VEGF-A165/bFGF-coronary heart disease (VIF-CAD) trials recruited 52 patients with refractory CAD who were randomized to receive VEGF/FGF plasmid or placebo plasmid in the myocardium, and the results of SPECT showed no improvement in cardiac perfusion following plasmid treatment after 5 months of follow-up. 90 In the first trial, 337 patients with chronic myocardial ischemia were randomized to receive a dose of recombinant FGF-2 (rFGF-2) at 0, 0.3, 3, or 30 µg/kg with a single intracoronary infusion indicating that the different dose of rFGF-2sx is safe and feasible, but the efficacy of rFGF-2 for therapeutic angiogenesis was doubtable 91 (Table 3).
Angiogenic Clinical Trials of VEGF and FGF-Based Therapy on Coronary Heart Disease.

Abbreviations: AGFs, angiogenic growth factors; CCS, Canadian Cardiovascular Society; FGF, fibroblast growth factor; rFGF-2, recombinant FGF-2; SPECT, single photon emission computed tomography; vps, viral particles; VEGF, vascular endothelial growth factor; VIF-CAD, VEGF-A165/bFGF-coronary heart disease; AGENT, Angiogenic Gene Therapy.
The GM-CSF
We randomized 21 patients not eligible for CABG but had CHD to receive an intracoronary injection of 40 μg of GM-CSF or placebo and subcutaneous GM-CSF (10 mg/kg) or placebo for 2 weeks, respectively. An invasive collateral flow index (CFI) significantly increased in the GM-CSF group. 92 The safety and efficacy of an exclusively systemic application of GM-CSF in patients with chronic stable CAD were tested in a randomized, double-blinded, placebo-controlled trial. CFI in all vessels increased significantly in the GM-CSF group compared to placebo group. However, the drug’s safety is questionable. 93 Twenty patients with AMI were randomized and divided into GM-CSF group (10 µg/kg, for 7 days) and control group (saline). The results showed that GM-CSF could successfully mobilize the CD34-positive cells and at the same time may raise the levels of plasma C-reactive protein in patients with AMI. 94 Two hundred and sixteen patients were randomized and assigned to receive conventional therapy or different doses of GM-CSF (5.0 μg/kg, 2.5 μg/kg, and 1.0 μg/kg; n = 54). The results demonstrated that semiquantitative perfusion score and left ventricular ejection fraction (LVEF) were significantly improved in the GM-CSF group. Additionally, the mortality and the incidence of side effects were also lower in GM-CSF group than that in the conventional group. 95
Granulocyte Colony-Stimulating Factor
Kang et al 96 reported treatment with G-CSF alone or control group in the patients with myocardial infarction who underwent coronary stenting. Cardiac function improved. However, an unexpectedly high rate of in-stent restenosis was noticed. Suzuki et al 97 showed G-CSF could increase the ejection fraction and peak creatine kinase compared with the control group in AMI group with no serious complications. A total of 114 patients with ST-segment elevation AMI who had successful PCI were randomly assigned to receive either a daily dose of 10 µg/kg of G-CSF or placebo for 5 days subcutaneously. The G-CSF therapy did not influence infarct size, left ventricular function, or coronary restenosis. 98 The Regenerate Vital Myocardium by Vigorous Activation of Bone Marrow Stell Cells (REVIVAL)-2 trial reported the subcutaneous daily dose of 10 µg/kg G-CSF or placebo in patients with AMI. The infarction size and LVEF were not significantly improved in G-CSF group. 99 Meier et al demonstrated that G-CSF improved signs of myocardial salvage by coronary collateral growth promotion in patients with chronic stable CAD. 100 Chih et al 101 reported that G-CSF did not affect myocardial ischemia by cardiac magnetic resonance imaging, exercise stress test, Seattle Angina and Utility Based Quality of Life Heart Questionnaire in patients with Canadian Cardiovascular Society class III-IV angina. Meier et al 102 found that the treatment with G-CSF increased the endothelial progenitor cells and peripheral monocytes in patients with symptomatic CAD eligible for PCI. Details of clinical trials with GM-CSF and G-CSF for CAD treatment are summarized in Table 4.
Clinical Trials of GM-CSF and G-CSF-Based Therapy on Coronary Heart Disease.
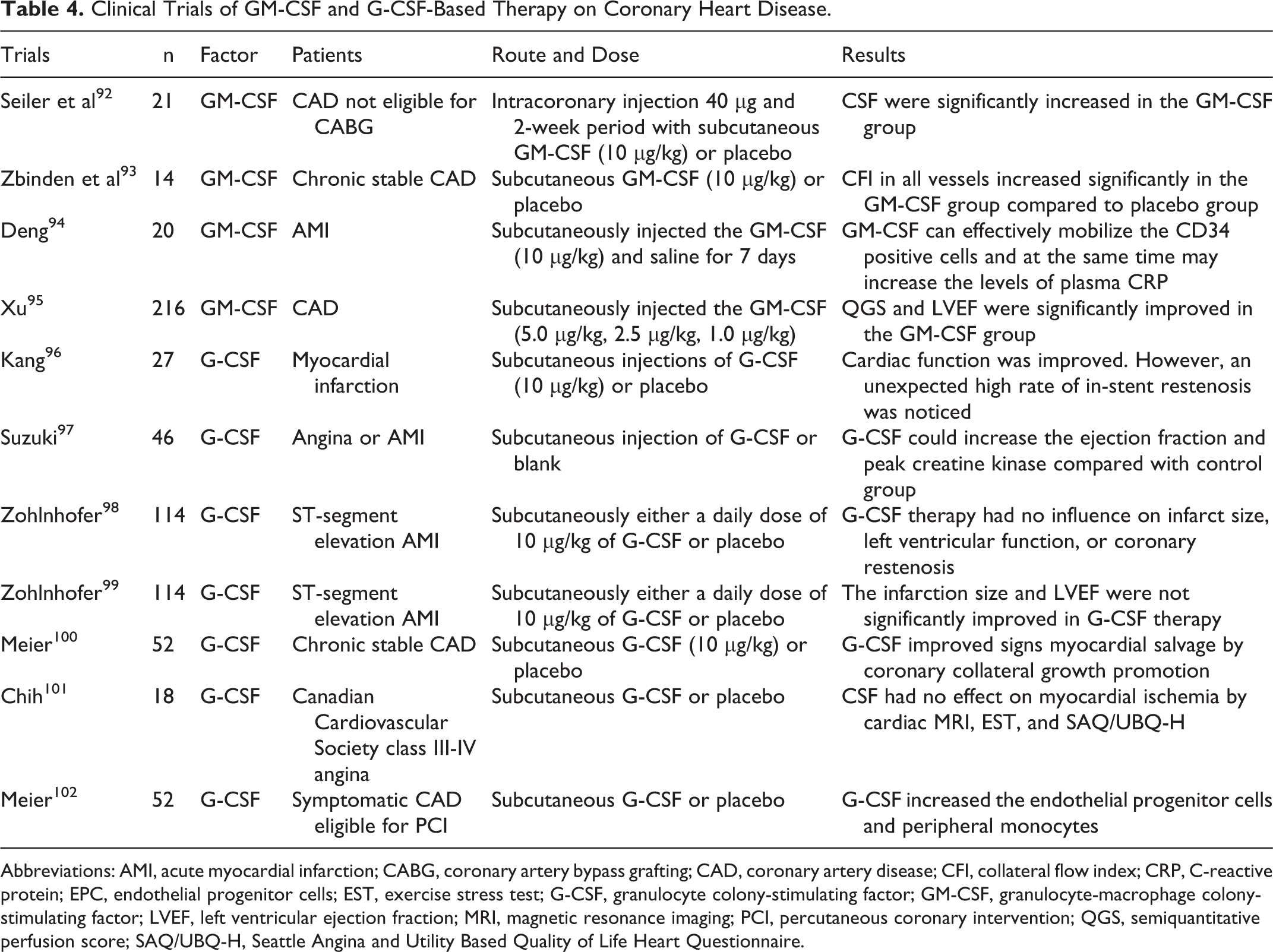
Abbreviations: AMI, acute myocardial infarction; CABG, coronary artery bypass grafting; CAD, coronary artery disease; CFI, collateral flow index; CRP, C-reactive protein; EPC, endothelial progenitor cells; EST, exercise stress test; G-CSF, granulocyte colony-stimulating factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; LVEF, left ventricular ejection fraction; MRI, magnetic resonance imaging; PCI, percutaneous coronary intervention; QGS, semiquantitative perfusion score; SAQ/UBQ-H, Seattle Angina and Utility Based Quality of Life Heart Questionnaire.
Summary and Perspectives
In this review, we highlighted the roles of AGFs in cardiac angiogenesis, especially VEGFs, FGFs, and PDGFs. The different AGF delivery systems have been developed recently to improve protein-based therapy, and some AGFs can be delivered into the ischemic heart tissue efficiently in the experimental studies or clinical studies.
Although cardiac
Some experimental research and clinical trials have shown that nonphysiological delivery of AGFs failed to establish sustained therapeutic angiogenesis, so improving the materials that can be applied to therapeutic angiogenesis is a great challenge. For designing the novel materials, the following issues should be taken into consideration.
First, the ECM, where angiogenesis naturally takes place, exhibits pivotal biophysical and biochemical signals that can be incorporated into angiogenic materials. Second, cell-adhesion and cell-degradable sites should be included in angiogenic biomaterials to facilitate cell migration and matrix remodeling, which are the central mechanisms of angiogenesis. Third, the network of angiogenic materials (such as synergistic receptor signaling) should be strengthened to reduce the delivered dose of AGFs. Finally, the angiogenic materials should be feasible in the clinical applications.
As the recovery potential of an animal model may differ from that of human patients, the enhanced capillary might not be enough to improve the recovery of human tissues. The mitogenic properties of some AGFs besides angiogenesis may help to explain some positive clinical results. Also, the combination of gene and protein should be considered. It can be argued that angiogenesis does not improve myocardial tissue ischemia. The dysfunction of blood vessels may contain many undiscovered features which could help us develop novel therapies for CAD. At last, preclinical models must accurately resemble the features of human disease to evaluate the treatment of humans.
Footnotes
Authors’ Note
This manuscript has not been published or presented elsewhere in part or in entirety and is not under consideration by another journal. We have read and understood your journal’s policies, and we believe that neither the manuscript nor the study violates any of these.
Authors’ Contributions
Da-zhuo Shi conceived the topic and helped to draft the paper. Ming Guo and Jun-he Shi collected references and wrote the paper. Pei-li Wang contributed to developing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and publication of this article: This work was supported by the National Natural Science Foundation of China (No. 81473528).
