Abstract
Although risk factors have been identified for the cross-sectional prevalence of venous disease, few studies have investigated risk factors for venous disease progression. Therefore, the aim of this study was to investigate the relationship between foot and ankle characteristics and the progression of venous disease. A total of 1025 participants from the San Diego Population Study were assessed at baseline and at follow-up 11 years later. Risk factors were assessed by questionnaire and physical measurements, while venous disease was determined by physical examination and Duplex ultrasound. Change in venous disease from baseline to 11-year follow-up was characterized as stable or progression. Those with venous disease progression were less likely to spend increased time lying per day, more likely to have a history of hypertension, lie supine for a surgical procedure greater than an hour, and report an occupation that was professional, technical, administrative, or management. Those with a normal arch reported the greatest degree of plantar flexion. In multivariable logistic regression, including adjustment for weight-bearing arch characteristics, greater dorsiflexion (per 5 degrees) was significantly associated with progression of venous disease (OR = 1.11, p = 0.01). A weight-bearing flat arch compared to a weight-bearing normal arch was of borderline significance as a protective factor against progression of venous disease with adjustment for dorsiflexion (OR = 0.56, p = 0.07). Our results indicate that the ability to have higher levels of dorsiflexion is a risk factor for the progression of venous disease, and suggest a role for connective tissue laxity in the pathogenesis of venous disease.
Introduction
In the western world, venous disease is a common vascular disorder and cause of significant morbidity. 1 It has been estimated that venous disease accounts for over $3 billion in annual healthcare expenditures in the United States. 2
Prior studies have identified several risk factors for chronic venous insufficiency (CVI). These include older age, male sex, Caucasian ethnicity, family history of venous disease, smoking, and obesity.3 –8 Studies have also reported foot characteristics and ankle mobility to be associated with chronic venous disease. Specifically, in cross-sectional analyses, the presence of flat feet has been associated with varicose veins and chronic venous insufficiency.3,9 –11 Uhl et al. reported a positive association between the presence of flat feet and increased severity of CVI. 12 In the San Diego Population Study, and in cross-sectional analyses, flat feet were associated with the presence of trophic changes and deep venous system obstruction in women. 3 Previous studies have also demonstrated a relationship between decreased ankle mobility and chronic venous disease.9,13 –15 For example, Cavalheri et al. found that a decrease in ankle range of motion was associated with worsening venous hemodynamics. 16 Poor ankle mobility is postulated to promote venous disease through decreased foot and calf muscle pump function. 17
While most prior studies have investigated risk factors for prevalent venous disease, few studies have researched markers for disease progression. Therefore, in the current study, we investigated the relationship between foot characteristics and ankle mobility with the progression of chronic venous disease.
Methods
Study population
From 1994 to 1998, the San Diego Population Study (SDPS) enrolled current and retired employees of the University of California, San Diego (UCSD). Random selection was made by stratum within age, sex and ethnic groups. Women and ethnic minorities were over-selected to allow statistical power for contrasts by sex and ethnicity. Spouses and significant others were invited to participate, while volunteers who heard about the study could also be enrolled.
There were 2404 persons in the baseline visit for the SDPS. Eleven years later, a follow-up visit for incident venous disease was conducted between 2008 and 2011. With the use of the Social Security Death Index, we determined that 199 had died since baseline. The remaining 2205 persons were contacted, and 1103 participated in the follow-up (incidence) visit. For those who chose to enroll and following a detailed explanation of the study, signed informed consent was provided by participants. The study was approved by the Committee on Investigations Involving Human Subjects of UCSD.
Risk factors
At both study visits, trained interviewers utilized a standardized protocol to collect information on patient demogra-phics, lifestyle, and personal history. At the baseline visit, participants defined their ethnicity as ‘Non-Hispanic White’, ‘Hispanic/Chicano’, ‘African American’, ‘Asian/Pacific Islander’, ‘Native American’, or ‘Other’. Occupation was categorized by the interviewer as ‘Professional’, ‘Technical/Administrative/Managerial’, ‘Skilled/Clerical’, ‘Semi-Skilled’, or ‘Laborer’.
At both visits, participants answered questions regarding personal habits including hours spent lying down, sitting, walking, and standing, level of physical activity and smoking habits. Participants also answered questions regarding history of cardiovascular disease. Specific questions regarding a history of vein abnormalities, hernia surgery, surgical procedures, serious lower limb injury, and immobility were also assessed. Participants self-reported the presence of flat feet and hypermobile joints. Family history (among first-degree relatives) of venous disease, including telangiectasias, varicose veins, blood clots in lower limb veins, phlebitis, venous ulceration, pulmonary embolism, or other venous problems, was obtained.
Also collected at both study visits were height, weight, waist, and hip measurements (measured with the participant wearing light clothes and no shoes). Body mass index (BMI) was calculated as body mass (kilograms) divided by the square of height (meters). Certified registered vascular technologists conducted the foot exam in which participants were asked to remove their shoes, slippers, and socks. They then proceeded to stand on one foot, and foot arches were visually assessed and scored as flat, small, or normal to high.18,19 A goniometer was used to measure maximal plantar flexion and dorsiflexion. These measurements were reported as the maximal deviation of the sole of the foot from the reference point, which was set as perpendicular to the leg.
Venous disease evaluation and classification
Certified registered vascular technologists performed the venous examination at both visits. Simple and reticular telangiectasias, simple and reticular varicosities, edema, and trophic changes were recorded on each leg with the participant standing. Shoes and socks were removed during the examination. Duplex ultrasonography was performed to evaluate reflux and obstruction. Reflux was defined as flow greater than 10 cm/second for greater than 0.5 seconds in the vein of interest. This was tested by having the patient either Valsalva for the common femoral vein or compression of the calf for distal veins. Obstruction was defined as duplex ultrasound non-compressibility of the vein being interrogated, with or without the presence of echogenic material in the lumen. 20 Partial and complete venous obstruction was determined by the degree of compressibility of the venous walls with the participant on a tilt table in a 15° reverse Trendelenburg position with the legs slightly flexed in minimal external rotation. Details of the study protocol have been published previously. 3
Using data from the venous evaluation, each leg was categorized into three mutually exclusive, hierarchical categories: normal, moderate, or severe venous disease. Normal included persons without varicose veins or trophic changes (lipodermatosclerosis, hyperpigmentation, or healed or active ulceration on visual inspection), and without obst-ruction or venous insufficiency determined by duplex ultrasound. This follows with the C0 or C1 with Pn in the Clinical-Etiologic-Anatomic-Pathophysiologic (CEAP) classification. Moderate venous disease included persons with varicose veins or reticular varices without the presence of trophic changes on visual inspection, but with obstruction or insufficiency of the superficial venous system or perforating veins and not the deep venous system. This follows with the C2, As, or Ap in the CEAP classification. Severe venous disease includes persons with trophic changes on visual inspection, or obstruction or insufficiency of the deep venous system. This follows with the C4, C5, C6, or Ad in the CEAP classification. Edema, or C3, was not used as a venous disease criterion in this study because edema can occur in the absence of venous disease. 20
Classification of the progression of venous disease was as follows: participants were categorized into three mutually exclusive categories: regression, stable, or progression. Regression included persons characterized as severe at baseline who changed to moderate or normal at follow-up, as well as those who were moderate at baseline and normal at follow-up. Progression included persons characterized as normal at baseline and moderate or severe at follow-up, or moderate at baseline and severe at follow-up. Stable included persons who did not change classification. Of the 1103 participants, 78 regressed, 340 progressed, and 685 were stable. Owing to small numbers, those participants who regressed were not included in this particular study, and only those characterized as stable or with progression of venous disease were compared.
Statistical analysis
Analysis of co-variance was used to compare age-, sex- and ethnicity-adjusted means/prevalence of cohort characteristics for categories of venous disease. Logistic regression was used to determine independent associations of ankle mobility and foot characteristics with progression and regression of venous disease compared to stable disease. An initial model was unadjusted, while subsequent models adjusted for age, sex, ethnicity, history of cardiovascular disease, occupation, loose/hypermobile joints, hernia surgery, supine for surgical procedure, current time spent lying, current time spent standing, history of hypertension, family history of venous disease, smoking history, BMI, and dorsiflexion (for small and flat arches) or weight-bearing arch measurements (for dorsiflexion and plantar flexion). A p-value <0.05 was considered significant. Statistical analyses were performed with SPSS, Version 21 (SPSS Inc., Chicago, IL, USA).
Results
Among the 1025 study participants, the mean (SD) age at the baseline visit was 59 (10) years, the mean BMI was 27 (5), 33% were male, 60% were non-Hispanic white, 15% were Hispanic, 12% were African American, and 12% were Asian (Table 1). With weight bearing, 68% had a normal arch, 25% had a small arch, and 7.3% had a flat arch. The mean dorsiflexion and plantar flexion values (in degrees) were 13 and 72, respectively.
Cohort characteristics by change in venous disease.
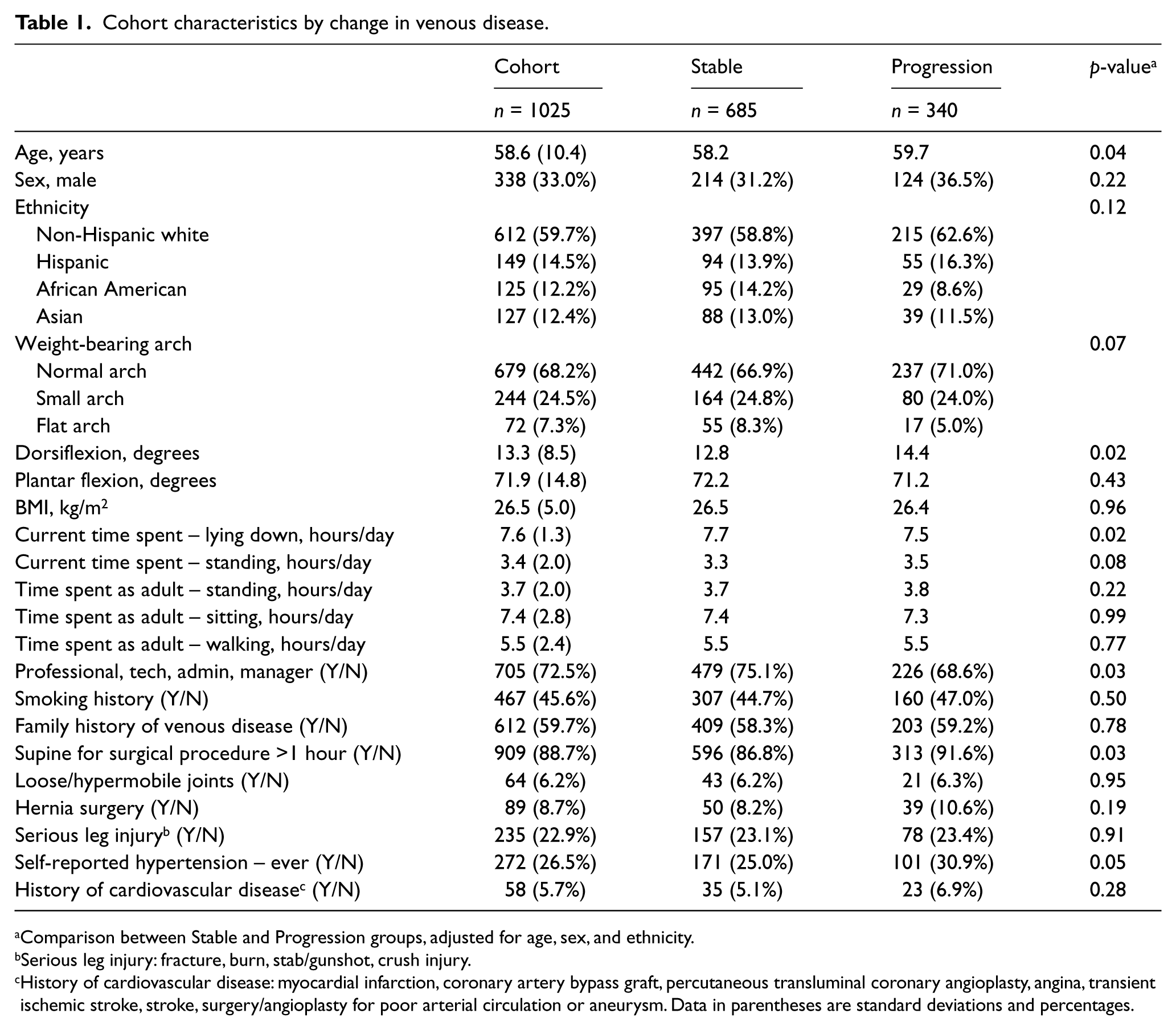
Comparison between Stable and Progression groups, adjusted for age, sex, and ethnicity.
Serious leg injury: fracture, burn, stab/gunshot, crush injury.
History of cardiovascular disease: myocardial infarction, coronary artery bypass graft, percutaneous transluminal coronary angioplasty, angina, transient ischemic stroke, stroke, surgery/angioplasty for poor arterial circulation or aneurysm. Data in parentheses are standard deviations and percentages.
The characteristics were then stratified by change in venous disease (Table 1). With adjustment for age, sex, and ethnicity, there were significant differences for dorsiflexion, current time spent lying, occupation, lying supine for a surgical procedure greater than an hour, and a history of hypertension. Those who progressed had a higher degree of dorsiflexion and were more likely to have a history of hypertension and lying supine for a surgical procedure greater than 1 hour. Those with stable venous status were more likely to report an occupation that was professional, technical, administrative or management.
Cohort characteristics were stratified by weight-bearing arch to identify the relationship with other variables and identify potential confounders (Table 2). Those with a flat weight-bearing arch had an increased BMI and were less likely to report lying supine for a surgical procedure greater than an hour compared to those with a normal or small arch. They also reported decreased time spent lying and sitting, and increased time walking compared to those with a normal or small arch. Those with a normal arch had increased plantar flexion compared to those with a small or flat arch.
Weight-bearing arch characteristics.
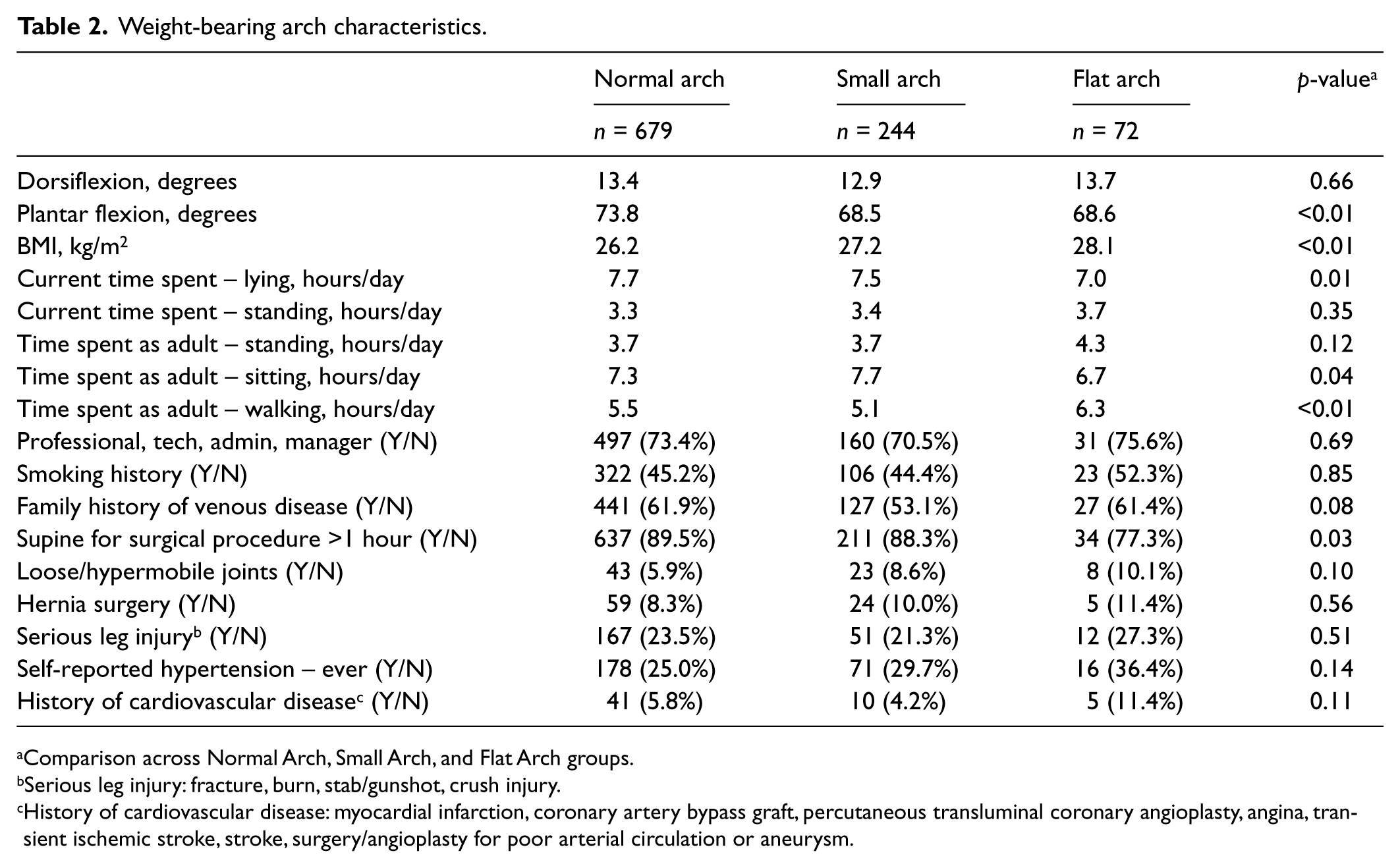
Comparison across Normal Arch, Small Arch, and Flat Arch groups.
Serious leg injury: fracture, burn, stab/gunshot, crush injury.
History of cardiovascular disease: myocardial infarction, coronary artery bypass graft, percutaneous transluminal coronary angioplasty, angina, transient ischemic stroke, stroke, surgery/angioplasty for poor arterial circulation or aneurysm.
The results of multivariable logistic regression are shown in Figure 1. Adjustment was made for age, sex, ethnicity, history of cardiovascular disease, occupation, loose/hypermobile joints, hernia surgery, supine for surgical procedure, current time spent lying, current time spent standing, history of hypertension, family history of venous disease, smoking history, BMI, and dorsiflexion (for arch measurements) or weight-bearing arch measurements (for dorsiflexion and plantar flexion). In these analyses, higher dorsiflexion (per 5 degrees) was associated with progression of venous disease (odds ratio (OR) = 1.11, p = 0.01). A flat weight-bearing arch compared to a normal arch was borderline significant (OR = 0.56, p = 0.07). Plantar flexion (per 5 degrees) and a small weight-bearing arch compared to a normal arch were not significantly associated with venous disease progression.
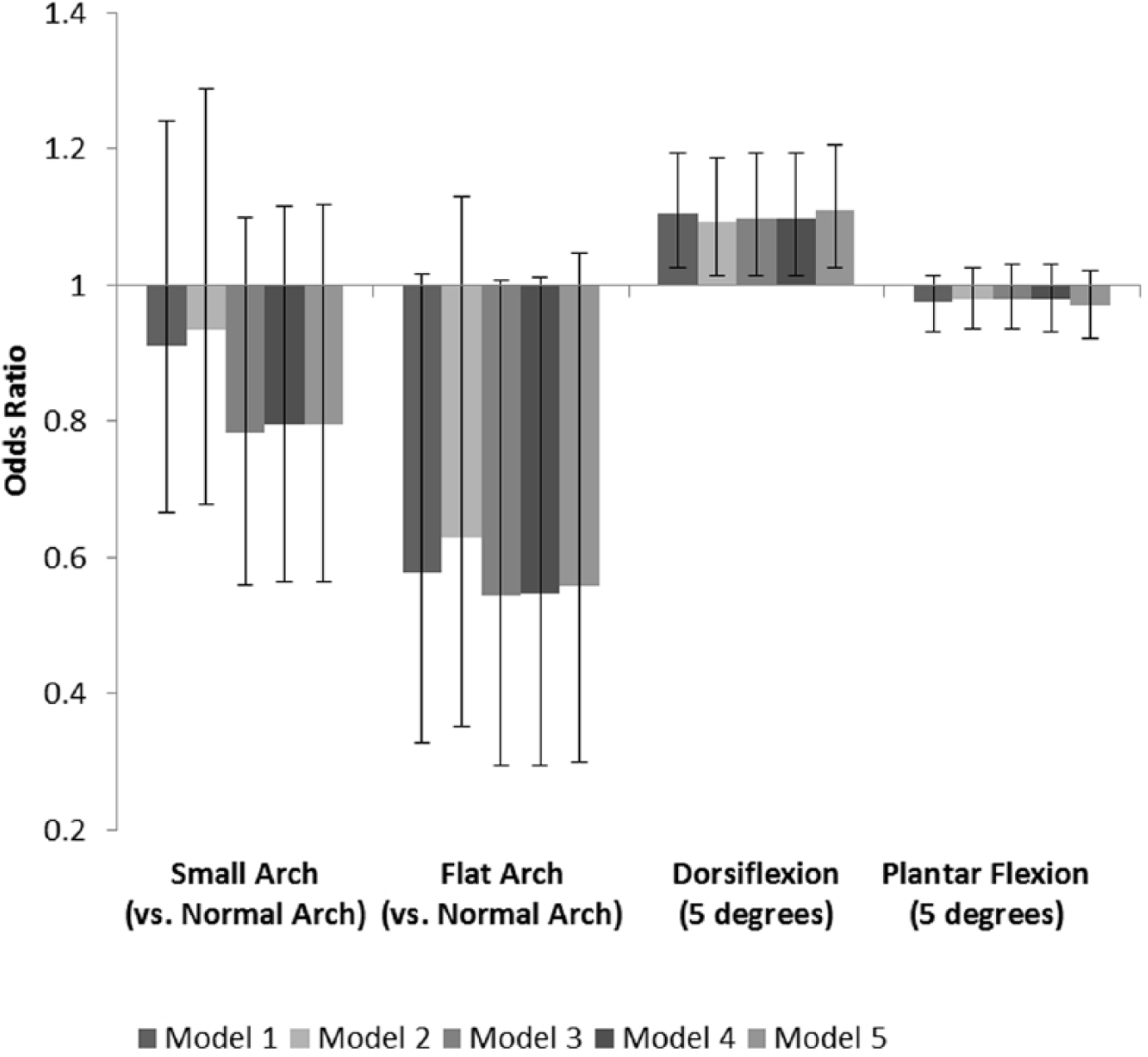
Odds ratio of the progression of venous disease compared to the stable group. (Model 1: unadjusted; Model 2: age, sex, and ethnicity; Model 3: Model 2 + covariates (history of cardiovascular disease, occupation, hernia surgery, supine for surgical procedure, current time spent lying, current time spent standing, history of hypertension); Model 4: Model 3 + family history of venous disease, smoking history, and BMI; Model 5: Model 4 + dorsiflexion (for weight-bearing arch measurements) or weight-bearing arch measurements (for dorsiflexion and plantar flexion)).
Secondary analysis was performed limiting the stable category to persons categorized as normal at both baseline and follow-up (i.e. those characterized with moderate or severe venous disease at baseline were excluded from the stable group). Multivariable logistic regression was performed, comparing the progression group against this ‘stable’ group. The model adjusted for age, sex, ethnicity, history of cardiovascular disease, occupation, loose/hypermobile joints, hernia surgery, supine for surgical procedure, current time spent lying, current time spent standing, history of hypertension, family history of venous disease, smoking history, BMI, and dorsiflexion (for weight-bearing arch measurements) or weight-bearing arch measurements (for dorsiflexion and plantar flexion). In these sensitivity analyses, the odds ratios for progression of disease with dorsiflexion and plantar flexion remained similar to the results provided above. Specifically, dorsiflexion remained significant while plantar flexion became borderline significant (OR = 1.16 and 0.95, p = <0.01 and 0.09, respectively). The presence of a weight-bearing flat arch was protective for progression of disease, but this association was not statistically significant (OR = 0.72, p = 0.40).
Discussion
In a cohort of community dwelling individuals, we found that a greater degree of dorsiflexion was significantly associated with the progression of venous disease. To our knowledge, no previous clinical studies have investigated the link between connective tissue laxity and the progression of venous disease. An earlier study of the San Diego Population Study found that connective tissue laxity was a risk factor for the prevalence of moderate and severe venous disease. 3 El-Shahaly and El-Sherif also reported an increased prevalence of varicose veins in women with joint hypermobility. 21 Dorsiflexion has been shown to be directly related to the length of the Achilles tendon. 22 This is in contrast with plantar flexion, which has been found to be directly related to calf muscle pump function, and not the Achilles tendon or connective tissue laxity.15,23 Thus, increasing dorsiflexion reflects increased lengthening of the Achilles tendon and may be a surrogate of increasing levels of connective tissue laxity.
Several studies have found an association between de-creased ankle range of motion and severity of venous disease, postulating that decreased ankle range of motion inhibits the function of the calf muscle pump, causing impaired venous hemodynamics.16,24–28 However, several of these studies looked only at total range of motion, and did not examine dorsiflexion and plantar flexion separately. Furthermore, one of these studies did not find a significant association between dorsiflexion and venous disease, while the other that did was limited by a small sample size of 26 patients.25,26 Total ankle range of motion was not significantly associated with a change in venous disease in this study.
Foot disorders are a common problem, found in up to 23% of the general population. 12 In our longitudinal study, the presence of a flat arch at the baseline visit, compared to a normal arch, was borderline significant and protective against the progression of venous disease. This is in contrast with our initial hypothesis and previous data, which reported a positive association between flat feet and the prevalence of varicose veins.3,9,12 The pathophysiology of acquired flat feet most commonly involves damage to the posterior tibialis or the spring ligament, leading to inefficient venous return of the lateral plantar veins.17,29 Pooling of the blood in the veins leads to venous hypertension and stasis, and, subsequently, venous inflammation.8,30–32
However, our results suggest that this hypothesis may not be sufficient in fully explaining the pathogenesis of venous disease, and that other less defined mechanisms may also play a role. In this regard, studies have shown that flat feet affect walking biomechanics through various mechanisms.33 –39 To compensate for changes in foot structure, flat feet require a different balance of muscle strength to maintain strength and stability. In those with flat feet, the posterior tibialis, anterior tibialis, and vastus medialis have increased muscle activation while walking compared to a normal arch.33,34 Larger lower leg muscles in persons with flat feet may enhance the muscle pump action of the leg, and facilitate venous return to the heart. 40 Studies have also found that those with flat feet have increased pressure in the midfoot while walking. 41 The plantar veins, located within the midfoot, drain 20–30 ml of venous blood with each step while walking. 12 Thus, increased pressure may facilitate improved venous return in the foot. These physiologic changes may facilitate venous circulation in a manner that is independent of walking. To our knowledge, no studies have examined how venous blood flow is affected by muscle activation among those with flat feet compared to those with normal arches. Thus, although ligamentous laxity and connective tissue disorders have been associated with flat feet, changes in muscle function and physiology may be protective against progression of venous disease. Notably, previously described protective factors against venous disease including mobility and exercise did not confound our results.42,43 Thus, these findings suggest that a flat arch may be protective against venous disease and that the understanding of the pathogenesis of venous disease may not be fully understood.
It has been proposed that arch support orthotics may help those with flat feet and concurrent venous disease by improving venous circulation. 12 However, the results of this study question the validity of this theory. If a flat arch is protective against the progression of venous disease, then correction with orthotics may counteract these effects. If altered biomechanics and muscle activation improve venous circulation, then orthotics may not be beneficial. Although orthotics have improved pain in those with flat feet, there has not been a trial that examined the effects of arch support orthotics on venous blood flow or progression of venous disease.44,45 The findings described in this study may aid clinicians in identifying those at increased risk of worsening venous disease who may require more intensive monitoring and treatment.
The strengths of our study include the size of the population and the thorough assessment of venous disease by imaging and inspection at both visits. Limitations include the reliance on a patient’s self-reporting of medical history and lifestyle factors. In this particular study, only the worst leg of each patient was included in the analysis. The inclusion of both legs may provide additional insight into the role of various risk factors and venous disease. The arch of the foot was also determined via visual examination. Other more precise measures including a footprint and Clarke’s angle could have been utilized to provide a more accurate determination of the arch.
Conclusion
The results of the current study indicate that greater dorsiflexion is a risk factor for the progression of venous disease. Conversely, a flat arch was found to be of borderline significance in protecting against the progression of venous disease. Further studies are needed to understand how ankle and foot characteristics are related to chronic venous disease.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research was supported by the National Institute on Aging T35 grant AG26757, and the National Institutes of Health – National Heart, Lung, and Blood Institute grant 5RO1HL84229.
