Abstract
Given the very ill nature of patients with critical limb ischemia (CLI), the use of percutaneous vascular interventions (PVIs) for limb salvage may or may not be efficacious; in particular, for those with polyvascular arterial disease. Herein, we reviewed large, multi-institutional outcomes of PVI in polyvascular and peripheral arterial disease (PAD) patients with CLI. An 18-hospital consortium collected prospective data on patients undergoing endovascular interventions for PAD with 6-month follow-up from January 2008 to December 2011. The patient cohort included 4459 patients with CLI; of those, 3141 patients had polyvascular (coronary artery disease, cerebrovascular disease and PAD) disease, whereas 1318 patients suffered from only PAD. All patients were elderly and with significant comorbidities. The mean ankle–brachial index (ABI) was 0.44 and was not different between those with and without polyvascular disease. Polyvascular patients had more femoropopliteal and infra-inguinal interventions and less aortoiliac interventions than PAD patients. Pre- and post-procedural cardioprotective medication use was less in the PAD patients as compared with polyvascular patients. Vascular complications requiring surgery were higher in PAD patients whereas other access complications were similar between groups. At 6-month follow-up, death was more common in the polyvascular group (6.7% vs 4.1%, p<0.001) as was repeat PVI, but no difference was found in the amputation rate. Considering the group as a whole at the 6-month follow-up, predictors of amputation/death included age (HR=1.01; 95% CI=1.002–1.02), anemia (HR=2.6; 95% CI=2.1–3.2), diabetes mellitus (HR=1.6; 95% CI=1.3–1.9), congestive heart failure (HR=1.6; 95% CI=1.4–1.9), and end-stage renal failure (HR=1.9; 95% CI=1.5–2.3), while female sex was protective (HR=0.7; 95% CI=0.6–0.8). In conclusion, from examination of this large, multicenter, multi-specialist practice registry, patients with polyvascular disease had higher 6-month mortality than PAD patients, but this was not a factor in 6-month limb amputation outcomes. This study also underscores that PAD patients still lag in cardioprotective medication use as compared with polyvascular patients.
Introduction
Peripheral arterial disease (PAD) is associated with significant morbidity and mortality, potential limb loss, and negatively affects quality of life.1–3 PAD has variable manifestations, ranging from asymptomatic to non-critical limb ischemia, to intermittent claudication and atypical limb symptoms including fatigue to rest pain and tissue less consistent with critical limb ischemia (CLI). The risk of major limb amputation is less than 1% per year in claudicants compared to the 14–45% major amputation rate in CLI patients.4–6
The literature on PAD is extensive, and outcomes associated with specific procedures and techniques have been published.7–14 An aggressive strategy is often used in patients with CLI, such as surgical debridement of necrotic tissue and target vessel revascularization (surgical bypass or percutaneous) to improve perfusion to avoid amputation.12,15,16 Over the last few decades, dramatic changes have occurred in the management of PAD patients, including defining objective performance goals 17 after revascularization and increased catheter-based percutaneous vascular interventions (PVIs).15,17–19 Although associated with low morbidity and mortality,4,20 whether PVI confers salutary short-term outcomes in CLI patients is not widely published outside of single-institution studies or administrative data.12,15,21
Patients with CLI all have significant systemic atherosclerotic burden, but some primarily manifest PAD while others have PAD with manifestations of other symptomatic arterial beds, including cerebrovascular disease (CVD) and coronary artery disease (CAD). These patients have so-called ‘polyvascular disease’. Their long-term survival is worse than non-polyvascular patients, particularly after an incident coronary event, and many may be undertreated with cardioprotective medications.22–24 Whether polyvascular patients fair worse with PVI than ‘PAD only’ patients is unclear, but important when determining what therapy to offer patients.
Given the high morbidity and mortality of the polyvascular disease CLI population, the benefit of PVI in terms of amputation-free survival (AFS) may be questioned. Herein, we compared patient outcomes and variables associated with amputation/death for patients undergoing PVI in those with polyvascular disease as compared to those with PAD only, in a large prospectively collected patient data registry from an 18-hospital consortium. We hypothesized that patients with polyvascular disease would fare worse than those with ‘PAD only’ in patients with CLI.
Methods
An 18-hospital consortium collected prospective data on patients undergoing elective PVI for PAD with 6-month follow-up from January 2008 to December 2011. Of 6285 cases with CLI (Rutherford ≥4), 4459 had 30-day or 6-month follow-up information (71%), and formed the basis of this study. Of these patients, 2806 had both 30-day and 6-month follow-up, 1042 had only 30-day, and 60 had only 6-month follow-up. Polyvascular disease patients were defined as those with a history of clinically manifest disease in other vascular beds, including cerebral vascular occlusive disease (CVOD) and CAD vascular beds in addition to PAD. ‘PAD only’ patients were defined as those without a history of clinically manifest CVOD or CAD – acknowledging that patients with PAD have systemic atherosclerosis.
The details of the BMC2 PVI (Blue Cross/Blue Shield of Michigan Cardiovascular Consortium Peripheral Vascular Intervention) program have been described previously.25,26 Supported by Blue Cross/Blue Shield of Michigan, BMC2 PVI is a prospective, multicenter observational registry designed to collect information on patients undergoing PVI in an effort to evaluate evidence-based disease management.
The registry currently collects detailed information on consecutive patients who have undergone PVI for clinical syndromes related to PAD. A data form is compiled for each patient, including demographic information, past medical history, laboratories pre- and post-PVI, patient history, presenting symptoms, procedural indications, medication details, details of procedure, and associated complications if present. Data quality and the inclusion of consecutive procedures are ensured by ad hoc queries, random chart review, and a series of diagnostic routines included in the database. A list of standard definitions determined by the American College of Cardiology Data Standards Committee has been used as a reference. CAD was defined as a history of myocardial infarction, percutaneous coronary intervention (PCI) or coronary artery bypass surgery (CABG). CVD was defined as a history of stroke or transient ischemic attack (TIA).
All data undergo a three-step validation process, including a manual review for completeness and a face validity review of rejected data forms during the import process, and review of forms that fail diagnostic inquiries. Additionally, a database manager checks all data forms for completeness, and a random sample of data forms is checked for face validity and clinical consistency. Twice yearly, sites are audited by a nurse monitor from the coordinating center where all cases associated with severe complications and a randomly selected 5% of cases are audited for accuracy.
CLI was defined as ischemic rest pain and/or tissue loss in the lower extremity requiring emergent or urgent procedure where intervention was done to save the limb/tissue or to aid in healing (Fontaine stage III or above; Rutherford category 4 or above). Technical success was defined as successful access and deployment of the device and ≤30% residual stenosis of the treated area. 19 Procedural success was defined as technical success and freedom from procedural complication. Baseline demographic information, preprocedural clinical variables, procedural variables, and in-hospital and singular 6-month outcomes were collected, analyzed and compared in both groups. Endoluminal intervention procedural variables included location, access type, device type, and outcome measures of in-hospital major complications (mortality rates, stroke, transfusion, access bleeding, etc.), and 6-month amputation and mortality were determined. Repeat PVI was captured in hospital and at 6 months, and included contralateral as well as ipsilateral procedures and in other vascular beds.
Statistical analysis
All 4459 cases were distinct patients and all analyses were done on a per patient basis. Differences between the groups were evaluated by the Student t-test for continuous variables and by the chi-square or Fisher exact tests for categorical variables. Some continuous variables were also presented as median with interquartile ranges, and were compared using the Kruskal–Wallis test. Patients were included if they had one follow-up at either 30 days, 6 months, or both. Thus, more patient data are available for early post-procedure outcomes. Six-month follow-up outcomes were analyzed using time-to-event methodology, and presented as Kaplan–Meier estimates and compared with the log-rank test. Cox proportional hazard regression analysis was performed to evaluate the impact of preprocedural and procedural variables on the 6-month rates of death or amputation in the entire cohort. This was done by using preprocedural variables (age, female sex, body mass index, history of congestive heart failure (CHF), hypertension (HTN), CAD, CVD, anemia, glomerular filtration rate (GFR)<60, renal failure requiring hemodialysis and previous peripheral intervention), which were entered into the model. The proportional hazards assumption was checked for each variable by using log-log plots. The hazard ratios, corresponding two-sided 95% confidence intervals (CIs) and probability values for predictor variables are presented. All reported p-values are two-sided, and p<0.05 was considered statistically significant. SAS version 9.2 (SAS Institute, Inc., Cary, NC, USA) was used for statistical analyses.
Data collection in the BMC2 PVI registry was approved by the institutional review board or ethics committee at each participating center. Trained nurse coordinators performed follow-up via telephone calls and using a standardized questionnaire.
Role of sponsor
The BMC2 registry is funded by Blue Cross Blue Shield of Michigan and Blue Care Network. The funding agency had no role in study design, analysis or decision to publish.
Results
Of the 4459 patients with CLI who had 6-month follow-up, 3141 were defined as polyvascular disease patients (those with documented CAD, CVOD and PAD) and 1318 patients had the diagnosis of PAD only. The breakdown of polyvascular patients was: CAD and PAD = 1719; PAD and CVD = 379; PAD and CAD and CVD = 1015. The clinical presentations were similar, with rest pain in approximately 40% and tissue loss in ~60%; there was no significant difference between the groups (p=0.4). About half of the patients had a prior PVI (n=2165), 48% had both prior endovascular and open surgery, 13% had prior open vascular surgery, and 38% had no prior interventions. Comparison of those patients in our registry of CLI patients without follow-up (n=1826) with those with follow-up showed few significant baseline differences (Supplemental Tables 1–3).
Demographics showed markedly different baseline comorbidities between the polyvascular group as compared with the PAD only group (Table 1). Patients with PAD only were younger, more commonly women, currently smoking, but were less likely to have diabetes, hypertension, CHF, COPD, hyperlipidemia, anemia and end-stage renal failure (ESRF) requiring hemodialysis, or a history of prior PVI. Within the cohort, the total cholesterol (mean±SD) was 160±48 mg/dl (n=1840). The mean ABI was similar in those with PAD only as compared with those with polyvascular disease, both within the severe range of ~0.45 (p=0.1), and were available for 46% of the cohort. The median ABI (IQR) was 0.48 (0.34–0.58). Length of hospital stay was also similar between the PAD only and polyvascular disease groups (2.7±5.3 versus 2.7±4.5 days, respectively; p=0.7).
Demographics and clinical characteristics.
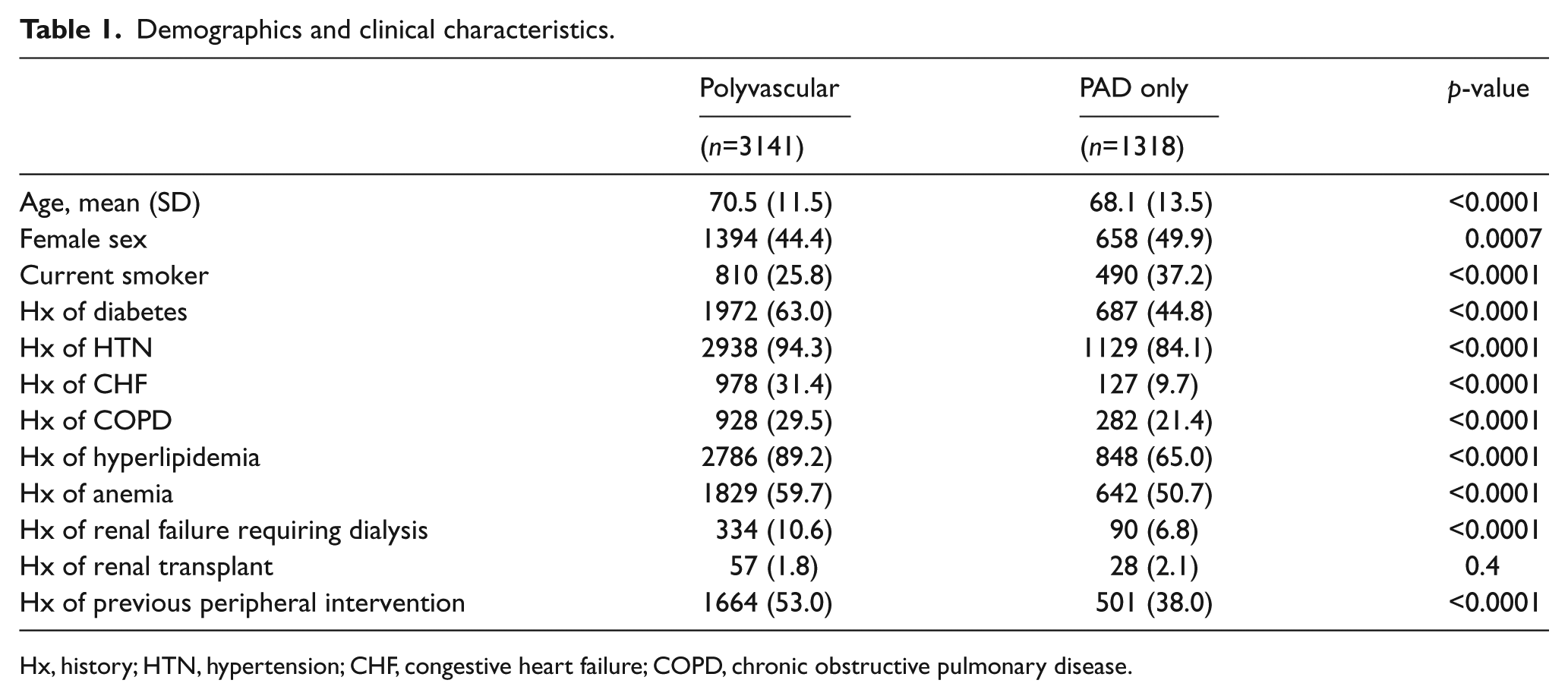
Hx, history; HTN, hypertension; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease.
Comparison of preprocedural cardioprotective medication use showed those with polyvascular disease were more likely prescribed angiotensin-converting-enzyme inhibitors (ACEIs) or angiotensin-receptor blockers (ARBs), beta-blockers, statin agents, mono or dual antiplatelet therapy; or anticoagulants than PAD only patients. Post-procedural medication rates, which are a statewide quality improvement focus, showed an increase in both groups. However, in all cases, the PAD only patients were less likely to be prescribed cardioprotective medications as compared with polyvascular disease patients (Table 2).
Post-procedural medication usage for polyvascular (PV) versus PAD only patients.
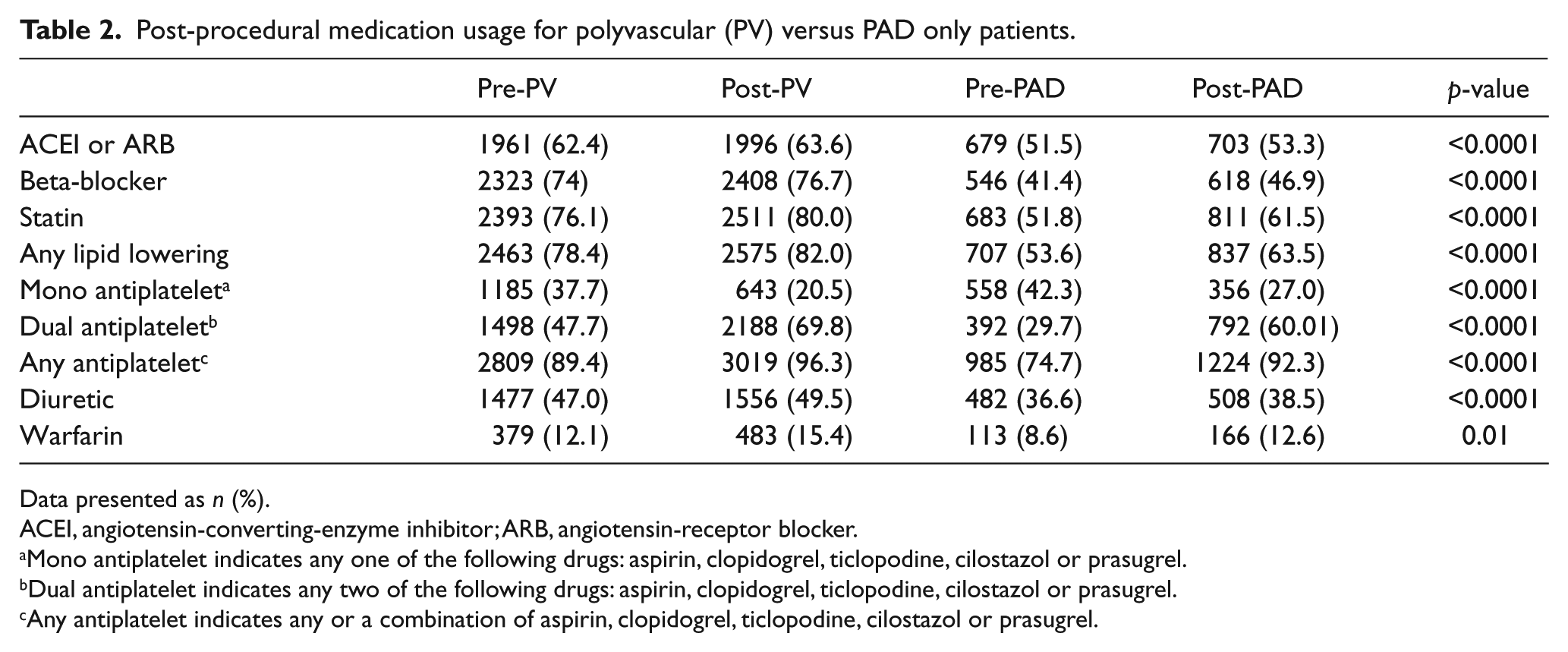
Data presented as n (%).
ACEI, angiotensin-converting-enzyme inhibitor; ARB, angiotensin-receptor blocker.
Mono antiplatelet indicates any one of the following drugs: aspirin, clopidogrel, ticlopodine, cilostazol or prasugrel.
Dual antiplatelet indicates any two of the following drugs: aspirin, clopidogrel, ticlopodine, cilostazol or prasugrel.
Any antiplatelet indicates any or a combination of aspirin, clopidogrel, ticlopodine, cilostazol or prasugrel.
For the entire cohort, interventions included aorto iliac (n=689), femoral·popliteal (n=1782), below knee (n=634) and mixed (n=1354). Significant differences were noted in the location of treated vascular territory/segments (Table 3). Polyvascular patients had more infra-inguinal (femoropopliteal and below knee) interventions, while PAD only patients had significantly more aortoiliac segment interventions. Procedural techniques varied between the groups as well. Balloon angioplasty as the primary intervention was performed equally in polyvascular and PAD only patients, while stenting was more common in PAD only patients. Atherectomy and cutting balloon angioplasty was more common in polyvascular patients. Antegrade or retrograde arterial approaches were not different between groups. However, mean contrast total volume and fluoroscopy times were similar. Access-related complications were low, at less than 3%, in both groups. However, the need for surgical arterial repair was higher in those with PAD only as compared with those with polyvascular disease (0.8% vs 0.34%, p=0.02).
Intervention location (vascular bed).
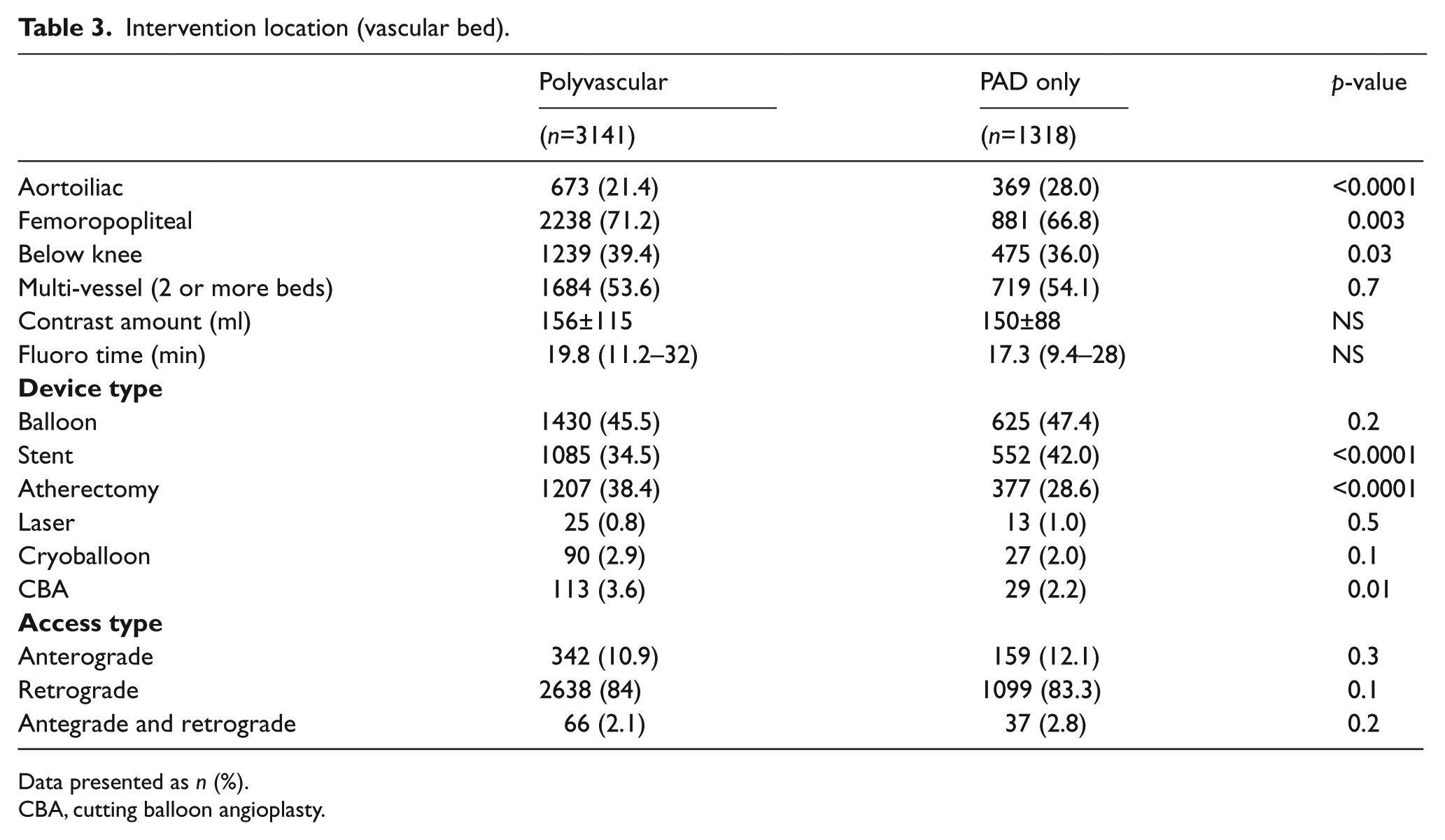
Data presented as n (%).
CBA, cutting balloon angioplasty.
In-hospital adverse events were low in both groups. Only periprocedural myocardial infarction (MI) was higher in the polyvascular group as compared with the PAD only group (Table 4). In-hospital amputation rates were ~5% in both groups and the need for repeat PVI was <1% in each group. Adverse events at 6 months showed a ~7% death rate in those with polyvascular disease as compared to 4% in those with PAD only (p<0.001). MI, amputation, and repeated PVI was ≤10% in both groups and not significantly different. The need for repeat PVI in those with follow-up was higher in polyvascular patients as compared with the PAD only group, but was less than 10% overall, acknowledging less than ideal follow-up and included any vascular bed, not just the ipsilateral index limb. The primary composite endpoint of amputation or death was 16% in those with polyvascular disease compared with 14% in the PAD only group (p=0.07).
In-hospital and 6-month morbidity and mortality.
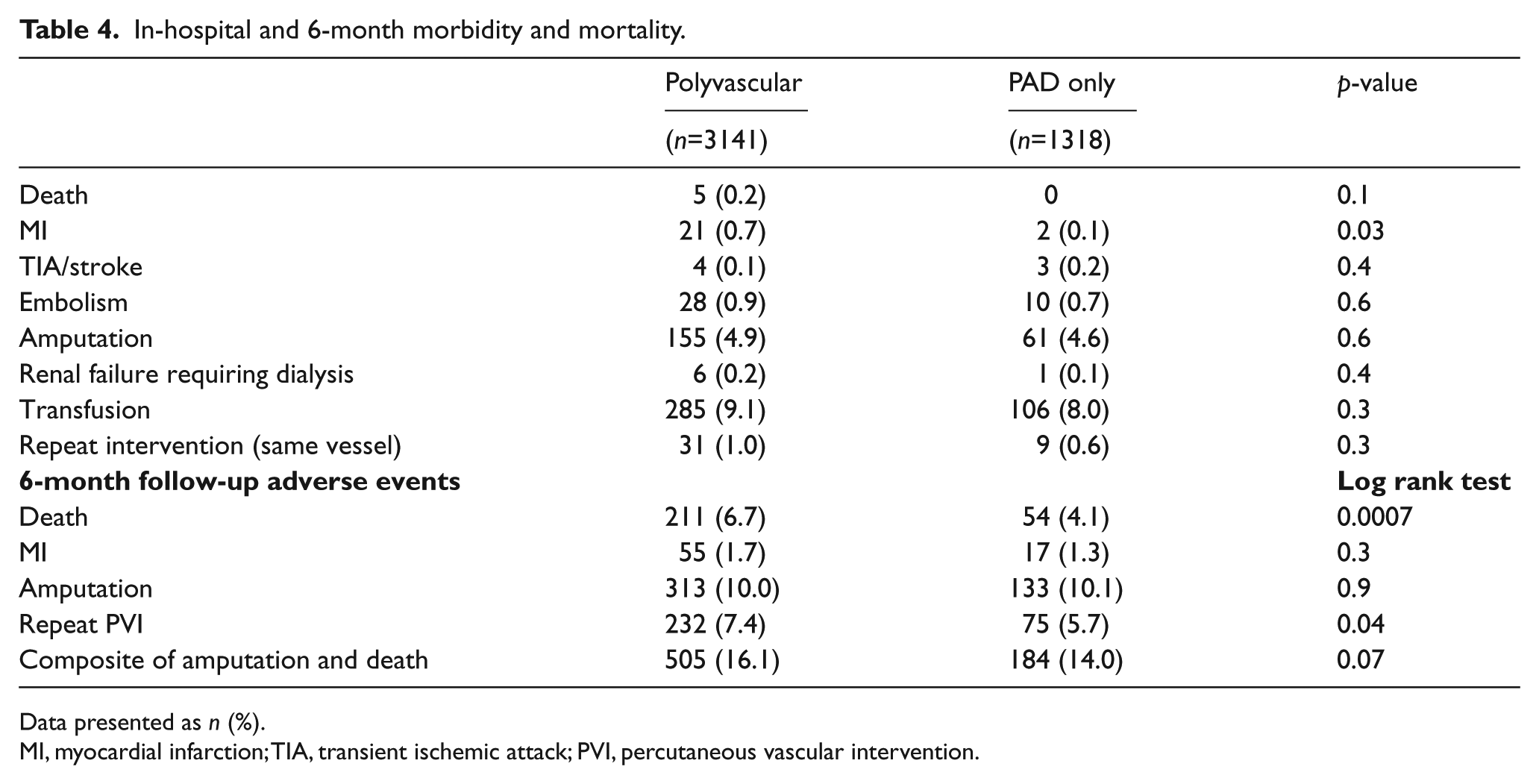
Data presented as n (%).
MI, myocardial infarction; TIA, transient ischemic attack; PVI, percutaneous vascular intervention.
Kaplan–Meier plots show the relationships of amputation, death, and combined outcome in the polyvascular and PAD only groups (Figures 1–3). Death was more common in polyvascular patients as compared with PAD only patients (7% vs 4.1%, p<0.001), but no difference in amputation rate was observed. A Cox proportional hazard model identified variables that independently predicted amputation or death at the 6-month follow-up. Factors associated with amputation or death included increased age, diabetes, anemia, hemodialysis-dependent renal failure (HDRF), and CHF, while female sex was protective (Table 5). For example, the mean±SD hematocrit of PAD only patients alive (37.4±6.1) was greater than those who died (30.8±4.9) (p<0.001), and was similar in the polyvascular group (36.6±6.2 vs 32.4±4.9, p<0.001). A similar relation was found in renal function. In the polyvascular group, 78% had a GFR <60 ml/min/1.73 m2 (low GFR) who died as compared to only 54% with a low GFR in those who survived to follow-up (p<0.001). In the PAD only group, 61% had a low GFR who died as compared to only 44% in those who survived (p<0.001).
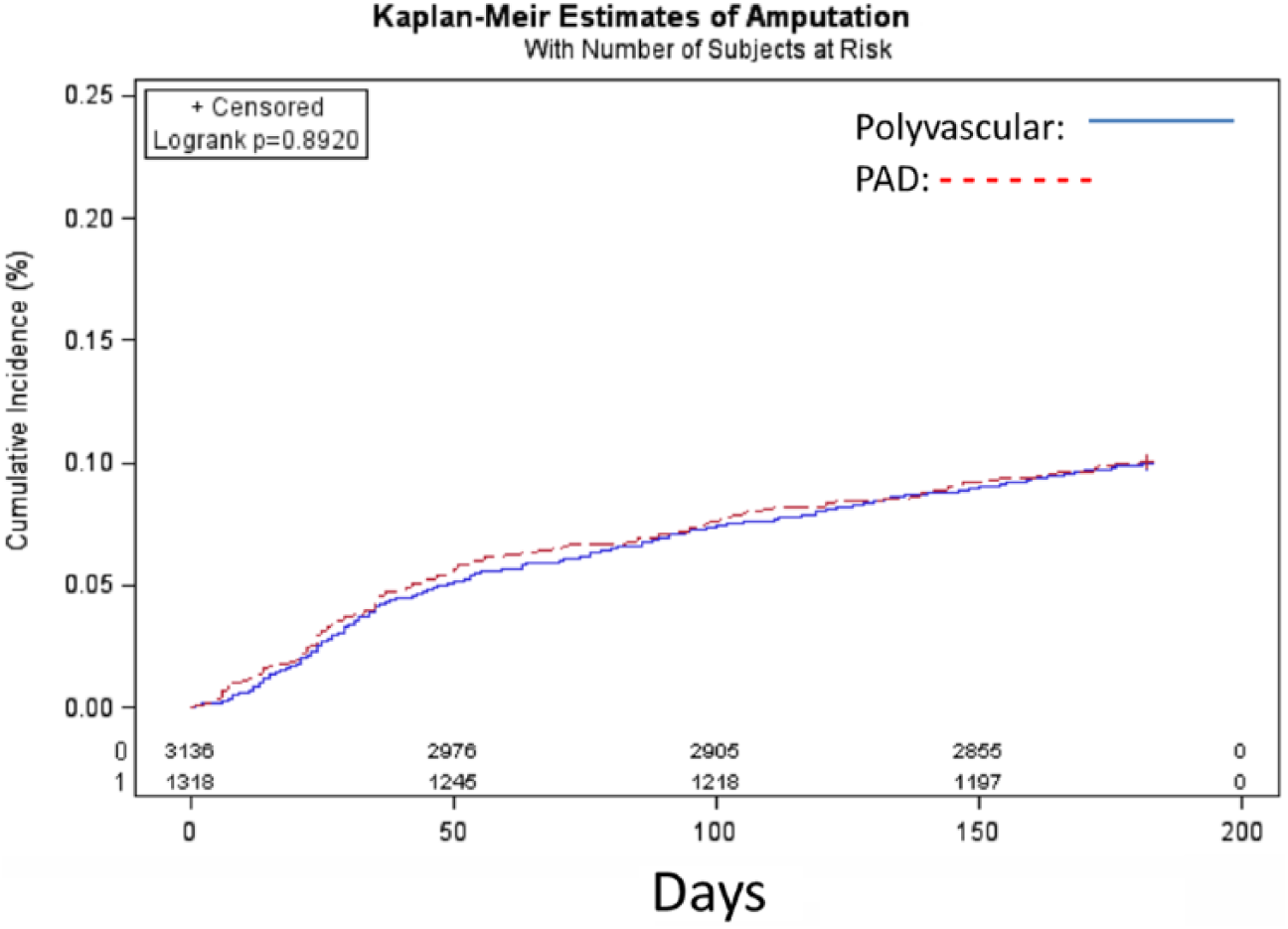
Kaplan–Meier plot of amputation in polyvascular disease (PAD) and PAD only patients over time.
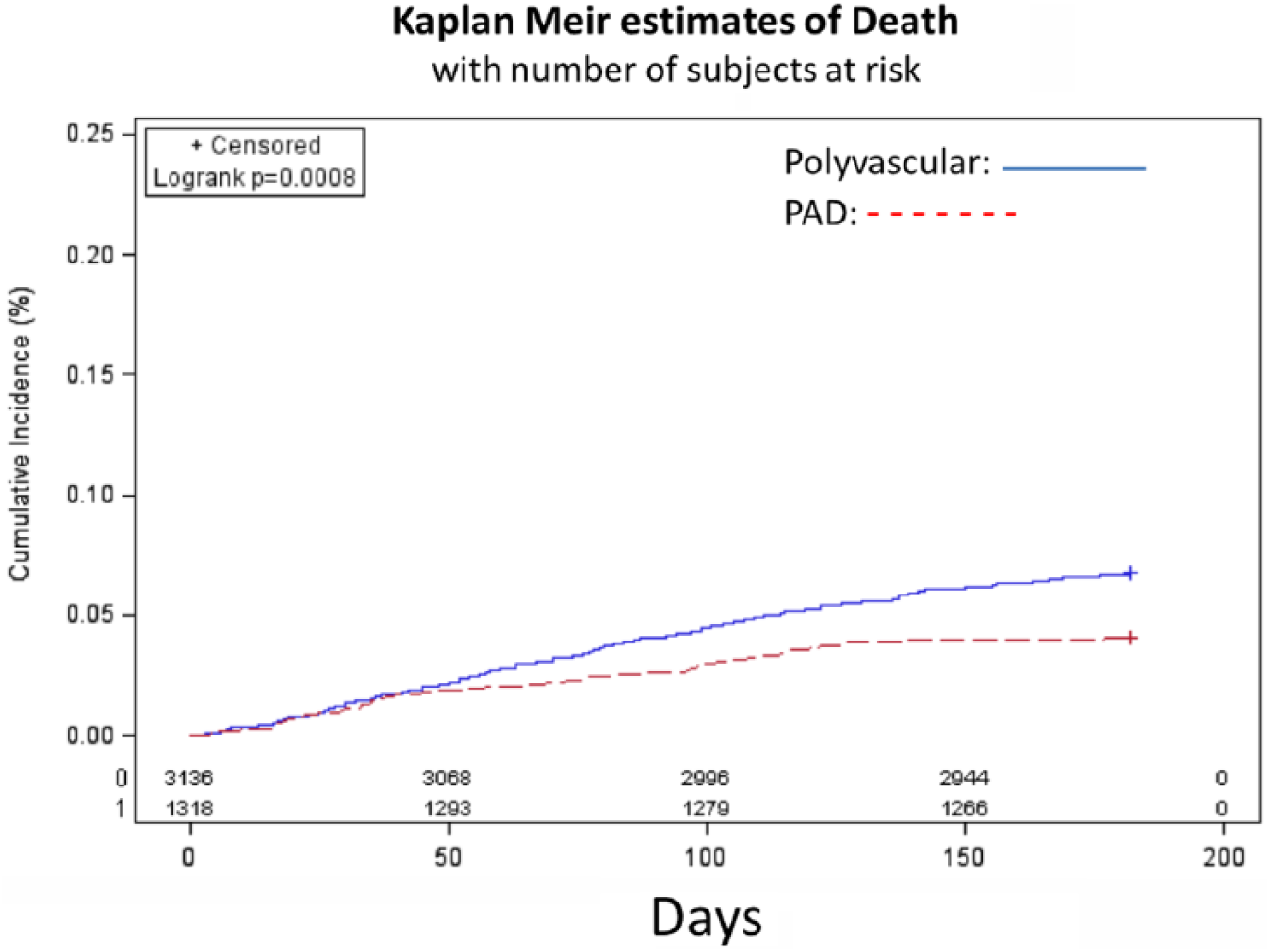
Kaplan–Meier plot of death in polyvascular disease (PAD) and PAD only patients only over time.
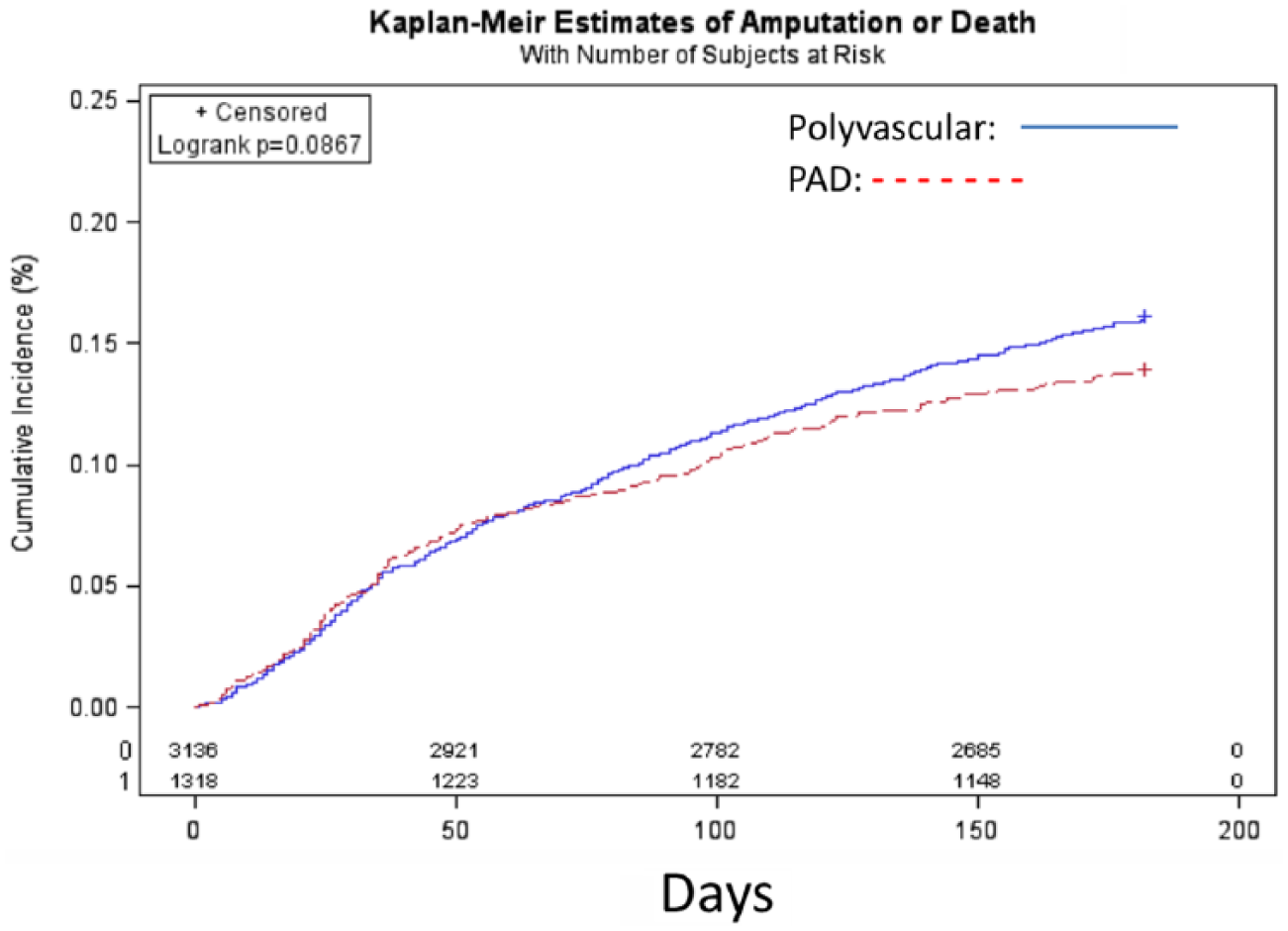
Kaplan–Meier plot of amputation/death in polyvascular disease (PAD) and PAD only patients over time.
Proportional hazard regression model for amputation or death at 6-month follow-up.
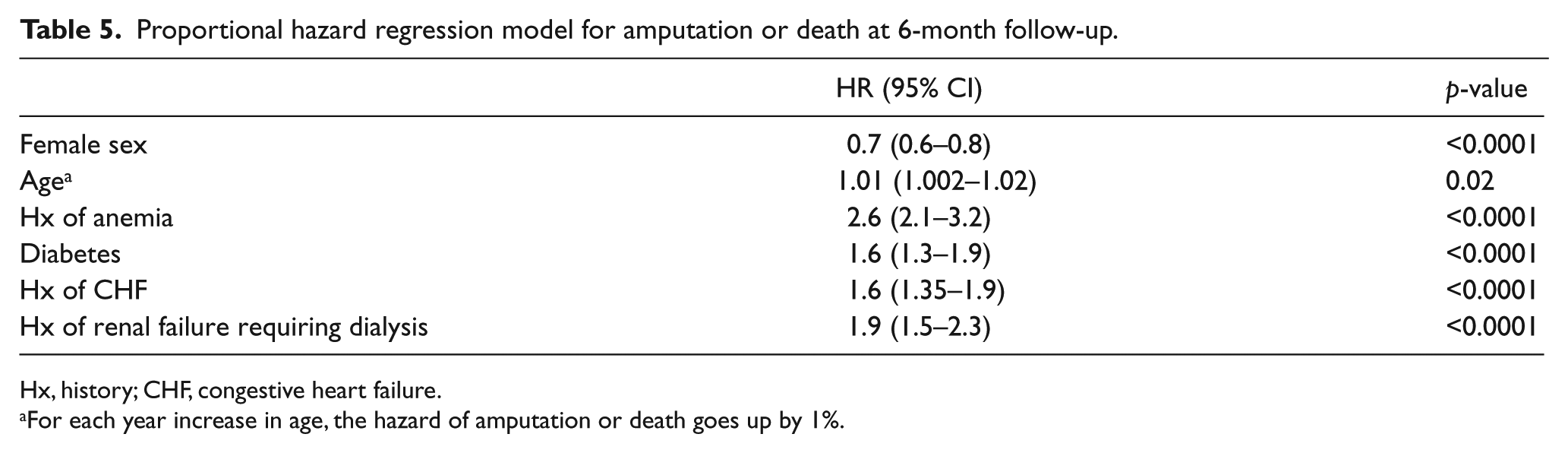
Hx, history; CHF, congestive heart failure.
For each year increase in age, the hazard of amputation or death goes up by 1%.
Several baseline comorbidities were not associated with amputation or death, including smoking, diabetes, hypertension, CAD history, CVD history, COPD or stroke, a history of prior vascular surgery or prior intervention, and PAD only.
Discussion
CLI patients have a significant atherosclerotic burden and high long-term mortality.6,12,21 The main findings of this study are that the presence of polyvascular disease as compared with PAD only confers a slightly increased death rate at 6 months, but does not impact limb salvage. Second, cardioprotective medication use was higher in the polyvascular patients than in PAD only patients, but none were independently associated with the composite outcome of amputation/death. Third, the major comorbidities that impact outcome in this high-risk CLI cohort is age, anemia, diabetes, CHF and ESRF; these factors should be highly considered when offering or holding aggressive interventions. Conversely, CAD and CVD (as part of the polyvascular definition), as well as smoking, and prior procedures were not associated with worse outcomes at discharge or 6 months. These similar outcomes also underscored that patients who manifest ‘PAD only’ clinically have a high burden of atherosclerosis and have occult CVD and CAD. To our knowledge, this analysis is the largest series providing a direct comparison of preprocedural and procedural variables on morbidity, amputation, and mortality in those with CLI undergoing PVI.
The distribution of critical lesions causing limb ischemia is reflected by the target sites that were treated in our study. A high frequency of infra-inguinal (femoropopliteal and infra-popliteal) disease and multi-segmental disease treatment in the CLI group reflects the severity and extensive nature of disease in CLI patients.7,11,27,28 Despite the observed differences in endoluminal intervention among the two-limb ischemia groups, vascular access complications were uncommon in both groups. Interestingly, patients with polyvascular disease as compared with PAD only patients had more infrainguinal interventions, likely due to a greater prevalence of diabetes and ESRF, while PAD only had more aortoiliac interventions and arterial stenting. However, this did not affect limb salvage, as amputation rates were less than 15% at 6 months and were not different between groups, despite repeat PVI being more common in the polyvascular group. However, wound care and non-major amputation data were not available, which may affect outcomes. 29
Most in-hospital complications were similar in the polyvascular group as compared with the PAD only group. Post-procedure myocardial infarction was more common in polyvascular patients, and was not surprising given that CAD history was part of the polyvascular disease definition. The presence of diabetes, CHF, and ESRF was associated with significantly higher rates of in-hospital and 6-month death/amputation. These diseases underscore much of systemic atherosclerosis and are only treatable, but not curable. Others have shown that these same factors correlate with an increased risk of early amputation/death in CLI patients, with similar odds ratios to this series.4,6 These particular variables were also independent predictors of amputation/death at 6 months in this series, and are known independent risk factors for amputation and complications in CLI patients.30–36 Given this population of ill patients, a reasonable amputation rate was achieved in our cohort of CLI patients following PVI, comparable to published reports in the literature.3,10,37
The association of preprocedural anemia with worse outcomes in PAD patients has been described in open bypass patient cohorts.6,14 Anemia was fairly common in our study, and was an independent predictor of both death and amputation in hospital and at 6 months. Anemia was not simply a reflection of ESRF patients, as the factor of those requiring hemodialysis was independently associated with death/amputation. It is not clear whether treating the anemia would improve outcomes, or whether this finding was simply a marker of greater illness burden, frailty, or reduced bone marrow reserve. Data on treating anemia by transfusion are mixed, and transfusions themselves may contribute to poor outcomes in many scenarios.38,39 From a similar state registry, anemia was associated with significantly worse outcomes in patients undergoing PCI.40,41 Thus, anemia is important when considering the aggressiveness of intervention, given that the in-hospital risk of death/amputation was increased fourfold.
The prevalence of various comorbid diseases such as CAD, hypertension, diabetes, COPD, and CVD was significant in both groups, but more prevalent in polyvascular patients, yet not related to worse outcomes. The comorbidity rates herein are consistent with other published studies such as REACH (Reduction of Atherothrombosis for Continued Health) survey 23 and others.27,42,43 This suggests a high prevalence of CAD/CVD, which may not correlate with the severity of limb ischemia or the success of PVI. The relationship between smoking and PAD has been well established. 44 Similarly, a history of smoking was widely prevalent among these patients in our study (average >30% patients), and while polyvascular patients were less likely to be current smokers, this factor had no impact on amputation or death. Lastly, although women fare worse with surgical cardiovascular interventions, 45 this was not the case in this series; rather, females had a lower death/amputation at 6 months.
Cardioprotective medication use is strongly indicated for PAD patients, 3 including statins and antiplatelet therapy. For example, the Heart Protection Study (HPS) demonstrated that in patients with PAD (even in the absence of a prior MI or stroke), aggressive low-density lipoprotein lowering was associated with significant reductions in mortality and morbidity. 46 In our cohort, the use of statins was noted in ≥65% of patients in both groups, although the use at baseline was significantly higher among the polyvascular patients as compared with PAD only patients. Similarly, antiplatelet drugs are clearly indicated in the overall management of symptomatic PAD. Post-procedural use of cardioprotective medications improved in both groups, but remained higher in the polyvascular patients. Others have observed cardioprotective medication prescription is often better in polyvascular disease patients, 22 possibly due to better specialist care. 43 This process measure has been a quality initiative in the BMC2 PVI hospital consortium for many years and represents a major opportunity to improve outcomes. 43 In our consortium, the prescription of the cardioprotective medication at discharge is more important in affecting long-term use than is the specialist doing the procedure (manuscript in preparation). Overall use of cardioprotection medications in the polyvascular patient group compares favorably with other large population databases, such as the National Health and Nutrition Examination Survey (NHANES), 47 the REACH registry, 23 and better than our own institution >10 years ago. 48 However, in the PAD only group, only 63% were on a statin agent and only 48–53% were on a beta-blocker or ACEI/ARB for hypertension, illustrating a significant potential for improvement.25,49 Lastly, we acknowledge that many of the PAD only patients likely had subclinical CAD and likely contributed to cardiovascular morbidity.
Limitations
This study was not a randomized trial of therapies between polyvascular disease patients and PAD only patients. However, PVI was performed at multiple hospitals with multiple operators from different training backgrounds (vascular surgery, interventional cardiology and interventional radiology) in a quality registry-based improvement consortium, thus allowing a close look at real-world contemporary practice. 20 Our study is limited by only 6-month follow-up and the reliance on selective data available through chart documentation. Multiple operators, procedural biases, and hospital expertise and preferences were not taken into account during analysis of the data. Nevertheless, the BMC2 PVI uses trained data abstractors with routine audits and intensive standardization of procedural definitions and education.25,26 The database is also limited by less detailed follow-up of quality of life outcomes (e.g. did these procedures in both groups benefit patients outside of the hard endpoints?). We cannot exclude that the patients without follow-up may have biased the data, but the large numbers and consistency with published literature suggest this is less likely. Importantly, we found few significant demographic, ABI, or procedural details in those without follow-up as compared with the study cohort (Supplemental data). We do acknowledge that we were unable to ascertain the reasons for lack of follow-up and potentially these may have been different between the groups and selected for more healthy patients. Lastly, we cannot say if the patients treated by PVI for CLI would have been better treated with open surgical bypass. Early outcomes were similar in the BASIL trial, with later follow-up demonstrating benefit with bypass over percutaneous procedures. 21 However, similar independent predictors exist between those with CLI treated by PVI and those by open surgery, 14 and similar limb salvage rates are reported. 50
Conclusions
From this large, multicenter, real-world practice registry, patients with polyvascular disease have an increased death rate, but no increased risk of amputation at 6 months as compared with PAD only patients, which suggests the fate of the limb is independent of the atherosclerosis burden in the other vascular beds. Increased age, anemia, CHF and ESRF independently predict amputation and death in these patients. These factors should be strongly considered when offering intervention versus primary amputation or only palliative care in this very ill group of patients. Nonetheless, PVI is associated with good outcomes, with >80% of CLI patients maintaining limb salvage to at least 6 months.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest related to this work.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
