Abstract
We investigated treatment with a receptor for advanced glycation endproduct (RAGE) blocking antibody on angiogenic response to hind limb ischemia in diabetic mice. Streptozotocin treated C57BL/6 mice received either murine monoclonal anti-RAGE F(ab′)2 intraperitoneally (n=10) or saline (n=9) for 9 weeks. Diabetic plus 10 non-diabetic C57BL/6 mice underwent left femoral artery ligation and 5 days later angiogenesis imaging with 99mTc-Arg–Gly–Asp (RGD) nanoSPECT/CT. Twenty-four days later, hind limb blood flow was measured with ultrasound, the mice were euthanized, and tissue was taken for immunohistochemistry. The angiogenic imaging signal in ischemic limbs was higher in RAGE-ab treated versus saline treated mice at day 5 (3.1±1.4 vs 1.68±0.35, p=0.02) and blood flow was higher at day 24 (1.49±0.5 vs 0.61±0.39, p=0.04). Immunohistochemistry of ischemic muscles showed greater capillary density in the RAGE-ab treated group versus the vehicle-treated group (p<0.001) (NS from non-diabetic mice). In conclusion, treatment with anti-RAGE F(ab′)2 in diabetic mice improves neovascularization in the ischemic leg.
Introduction
Peripheral artery disease (PAD) is a common condition producing symptomatic ischemic leg pain with exertion (claudication). 1 In diabetic individuals, this can be a particularly malignant disease, with resting limb ischemia and non-healing ulcers requiring amputation. There are currently no effective drugs to treat PAD in diabetes, leaving only surgical revascularization and interventional catheter-based approaches, and these can fail leading to limb loss. Developing an effective drug therapy to prevent or ameliorate the symptoms of PAD represents an unmet need.
The receptor for advanced glycation endproducts (RAGE) plays an important role in the development and progression of vascular disease. It is constitutively expressed in low levels on smooth muscle cells and endothelial cells in vascular endothelium and increases in response to hyperlipidemia and hyperglycemia.2–5 In diabetics, increased RAGE expression attenuates the angiogenic response to tissue hypoxia and thereby contributes to long-term complications including poor collateral formation in the lower extremities, poor wound healing, and ulcers. 4 Mechanisms for this adverse effect of RAGE include maladaptive regulation of vascular endothelial growth factor (VEGF) ligand signaling, impaired release of endothelial progenitor cells from the bone marrow, and defective function of the released cells.6,7
We developed an antibody that binds a unique peptide on the extracellular domain of RAGE for radiolabeling as a tool to localize and quantify RAGE expression in live animals.8–10 We demonstrated increased RAGE expression in the ischemic hind limbs of diabetic mice compared to control legs and to ischemic hind limbs in non-diabetic mice.8–10 In a human pathology paper, Ritthaler et al. in 1995 documented prominent enhancement of endothelial RAGE expression in small and medium size arteries in patients with occlusive peripheral vascular disease both with and without diabetes. 11
For imaging, microgram quantities of antibody are linked to the radionuclide and this amount has no biological effect. We hypothesized that if the anti-RAGE antibody has blocking properties it may have therapeutic potential. After positive results from blocking experiments in cell culture we designed a placebo-controlled treatment study in live animals to test the hypothesis that pretreatment with RAGE antibody improves angiogenesis at 5 days following femoral artery ligation (peak time for angiogenesis in this model) documented by uptake of 99mTc-HYNIC-RGD on single-photon emission computed tomography (SPECT) scans and improves blood flow at 24 days to the hind limbs of diabetic mice with femoral artery ligation.
Materials and methods
Cell culture
To test the inhibitory effect of RAGE antibody on phosphorylated protein kinase B (p-AKT), vascular smooth muscle cells (VSMCs) were maintained in DMEM medium supplemented with 10% fetal bovine serum (Gibco-Life Technologies, Carlsbad, CA, USA) and 1% penicillin/streptomycin (Gibco) in a 5% CO2 incubator at 37°C. 12 For experiments, cells were plated at a density of 1 × 105 cells/ml in a 60 mm dish. Cells were serum-starved overnight after they reached 70–80% confluency and the next day cells were pre-treated with 10 µg/ml non-immune immunoglobulin G (IgG) F(ab′)2 or anti-RAGE F(ab′)2 for 1 hour followed by treatment with 10 µg/ml of advanced glycation endproducts (AGEs) for 20 minutes.
Western blot analysis
Cells were lysed and total cellular lysates were immunoblotted and probed with p-AKT (Ser473) and AKT antibodies (Cell Signaling, Danvers, MA, USA). Briefly, proteins (25 µg) from cell lysates were denatured and resolved by 4–12% Bis-Tris gels and then transferred on nitrocellulose membrane. Membranes were blocked in 5% non-fat dry milk and then incubated with primary antibodies: p-AKT, total AKT. Horseradish peroxidase (HRP)-conjugated anti-rabbit IgG (Amersham, GE, Pittsburg, PA, USA) was used to identify sites of primary antibody binding. Membranes were stripped of bound primary antibodies and re-probed with total AKT antibody. The p-AKT/t-AKT ratios were quantified and normalized using ImageJ software (NIH, Bethesda, MD, USA).
Animals
All animal experiments were performed in accordance with the approval of the Institutional Animal Care and Use Committee of Columbia University. Male wild-type (WT) C57BL/6 mice (n=30) were obtained from The Jackson Laboratory.
Induction of diabetes
At 6–8 weeks of age, mice (n=20) were injected intraperitoneally with five consecutive daily doses of streptozotocin (STZ; Sigma, St Louis, MO, USA) dissolved in citrate buffer (55 mg/kg, pH 4.5). One week after the first dose, fasting blood glucose levels were assessed by glucometer (FreeStyle Flash; Abbott, Alameda, CA, USA). The criterion of two consecutive glucose levels >250 mg/dL was used to indicate diabetes.
Antibody
We initially produced an antibody in rabbits against the V-domain of RAGE designed to display immunoreactivity in mice, pigs, and humans. Based on Genbank sequences, a peptide was prepared and used to immunize mice and subsequently hybridomas were produced. Monoclonal antibodies (mouse IgG2a kappa) were produced in vitro and purified by protein A and low endotoxin units (less than 3 endotoxin units/mg of purified antibody) (Strategic Diagnostics or SDIX, Newark, DE, USA). The monoclonal whole IgG was fragmented into F(ab′)2 fragments by pepsin digestion.
Two weeks after the STZ treatment, half of the diabetic mice were treated intraperitoneally with 100 µg/day (three times per week) of monoclonal anti-RAGE F(ab′)2 (n=10) and nine received normal saline intraperitoneally for 9 weeks when they underwent left femoral artery ligation. Ten additional age-matched WT non-diabetic mice also underwent femoral artery ligation. The fasting blood glucose levels and body weights were measured weekly on mice from all three groups.
Femoral artery ligation
Unilateral femoral artery ligation was performed in both diabetic and non-diabetic mice as previously described.8,9 Briefly, mice were anesthetized via intraperitoneal injection of ketamine (100 mg/ml) and xylazine (5 mg/kg). Under sterile conditions, a skin incision was made on the upper thigh of the mouse. The inguinal ligament and the upper half of the femoral artery were exposed in both legs. On the left leg, the femoral artery was ligated with two sterile 8/0 non-absorbable silk sutures below the inguinal ligament proximally and just above the bifurcation into the superficial and deep femoral arteries distally and then excised. The vascular bundle on the right leg was isolated without further intervention. The skin incision was closed with sterile 5/0 nylon suture.
Preparation of radiotracer
A radiolabeled RGD probe targeting αvβ3 integrin expression on budding capillaries is a well-described imaging probe to document tissue angiogenesis in response to hypoxia, including hind limb ischemia following femoral artery ligation (FAL).9,13–18 Aliquots of 5 µg of HYNIC-RGD were incubated with 0.5 ml of tricine solution (70 mg/ml in distilled water), 99mTcO4− solution (50 mCi = 1850 MBq) and 20 µl of tin (II) solution (10 mg of SnCl2.2H2O in 10 ml of nitrogen-purged 0.1 N HCl for 20 min) at room temperature for 30 min.
SPECT/CT imaging
Five days after femoral artery ligation, each mouse was re-anesthetized for placement of a jugular vein catheter (Braintree Scientific, Braintree, MA, USA) and injected with 12.2±2.8 MBq 99mTc-HYNIC-RGD and 2–3 hours later (blood pool clearance) underwent SPECT/CT (computed tomography) imaging on nanoSPECT/CT (Mediso, Boston, MA, USA).
A topogram (sequence of 2D side view X-ray projections) was used to determine the axial scan range for SPECT and CT imaging. CT images were acquired with an integrated CT scanner using an X-ray tube at 45 kVp and an exposure time of 1000 ms per view. Following CT acquisition, helical SPECT scans were acquired using dual-headed detectors each outfitted with nine pinhole apertures. Each pinhole has a diameter of 1.4 mm with each collimator providing a transaxial field-of-view (FOV) of 30 mm and an axial FOV of 16 mm, extendable through helical scanning to 270 mm. SPECT data were acquired with the following parameters: step and shoot rotation, 30° step in 360° rotation using 24 projections, 60 s per projection, 256 × 256 frame size with 1.0 mm pixels, and 140 keV with 10% energy window. The obtained projection data were reconstructed by an ordered-subsets expectation maximization algorithm with subset and iteration numbers set to 16 and 8, respectively, a voxel size of 300 µm, and SPECT and CT datasets fused.
Image analysis
The SPECT/CT scans were reconstructed and processed using InVivoScope software (Invicro, Boston, MA, USA). Regions of interest (ROIs) were drawn on serial 5-voxel thick transverse sections from below the hip joints to distal hind limbs, and using a calibration factor the tracer uptake in mCi was summed for each limb and subsequently divided by the injected dose (ID).
Ultrasound Doppler imaging
A Power-Doppler image system (Vevo 2100; VisualSonics, Toronto, Ontario, Canada) was used to measure blood flow perfusion 24 days after femoral artery ligation. At this time point, blood flow recovery is observed in this model. 18 The MS-550D probe with a center operating frequency of 32 MHz and axial resolution of 40 µm was used to acquire all images. Briefly, mice were anesthetized using isofluorane (1.5–2.0%) and secured to a heated platform in the prone position. The hair on the hind limbs was removed using a depilatory cream followed by application of ultrasound gel. Moving the probe around the proximal and distal hind limbs, Doppler signals from vascular structures in multiple views in both ischemic and non-ischemic hind limbs were obtained and the pulsatility index was determined as: (peak systolic velocity – end diastolic velocity) / velocity time integral. 19
Quantitative immunohistology
Twenty-four days after left femoral artery ligation, mice were sacrificed and tibialis anterior muscles were harvested and fixed in 10% formalin for 48 h. Specimens were embedded in paraffin and sectioned (5 µm). Serial sections were stained for hematoxylin and eosin (H&E) for morphology. Capillary density was determined using monoclonal antibody against von Willebrand factor (vWF; clone F8/86, DAKO, Agilent Technologies, Carpinteria, CA, USA) to identify endothelial cells. Serial sections were also stained for RAGE (mouse monoclonal anti-RAGE antibody; 50 µg/ml) and VEGFR-2 (mouse monoclonal antibody, 1:1500 dilution; Cell Signaling Technology, Danvers, MA, USA). Secondary stains were performed using avidin-biotin HRP visualization systems (Vectastain ABC Kit; Vector Laboratories, Burlingame, CA, USA) or HRP-conjugated secondary antibody. The number of capillaries was counted in four to five randomly selected fields in three to four serial sections, averaged, and expressed as the number of capillaries per ×400 field. Positive staining for RAGE was quantified using color recognition software (Image ProPlus; Media Cybernetics, Bethesda, MD, USA). Chromagen-positive cells were summed and expressed as a percentage of the total cell number in each section for three to four serial tissue sections and averaged. The images were processed using a Nikon microscope equipped with a digital camera (Nikon Co., Tokyo, Japan).
Statistical analysis
Results are shown as mean ± SD. Comparisons between treated and control groups were made using Student’s t-test. All statistical tests were two-tailed. A p-value less than 0.05 was considered statistically significant.
Results
Western blot analysis
Western blot analysis demonstrated a marked reduction of p-AKT (Ser473) in smooth muscle cells pretreated with 10 µg/ml of RAGE-antibody F(ab′)2 antibody compared to cells pretreated with control non-immune IgG F(ab′)2. RAGE-antibody pretreatment reduced p-AKT expression by 44% (Figure 1).
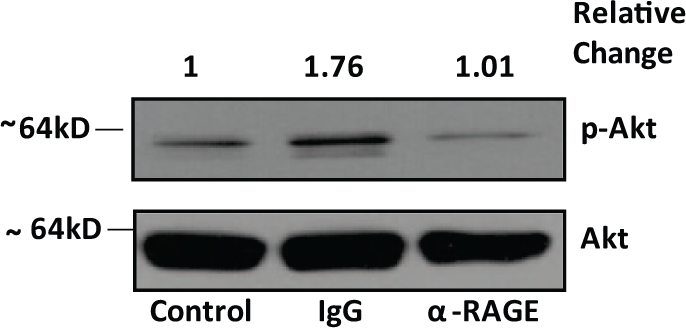
Levels of phosphorylated (Ser473)/total Akt in vascular smooth muscle cells shown by western blotting upon stimulation with 10 µg/ml of advanced glycation endproducts for 20 minutes after pre-treatment with 10 µg/ml of IgG and α-RAGE F(ab′)2 antibodies for 1 hour. Results shown are representative of three independent experiments.
Blood glucose and body weight
All mice treated with streptozotocin became diabetic with the exception of one mouse, which was excluded. Two diabetic mice died before imaging, leaving 17 mice: nine in the antibody treated group, eight in the saline treated group, and 10 non-diabetic controls. The mean fasting blood glucose levels for the RAGE-antibody treated diabetic group (329±37.9 mg/dL) were not significantly different from the vehicle-treated diabetic group (342.3±33.8 mg/dL; p=NS) (Figure 2A). The mean fasting blood sugar for the non-diabetic controls was 93.1±8.0 mg/dL. The mean body weights for both the RAGE-antibody treated diabetic mice (23.9±1.76 g) and the vehicle-treated diabetic mice (23.78±1.38 g) were lower than for the non-diabetic mice (30.55±1.2 g; p<0.0001 for both). There was no significant difference in body weight between the RAGE-antibody treated diabetic mice and the vehicle-treated diabetic mice (Figure 2B).
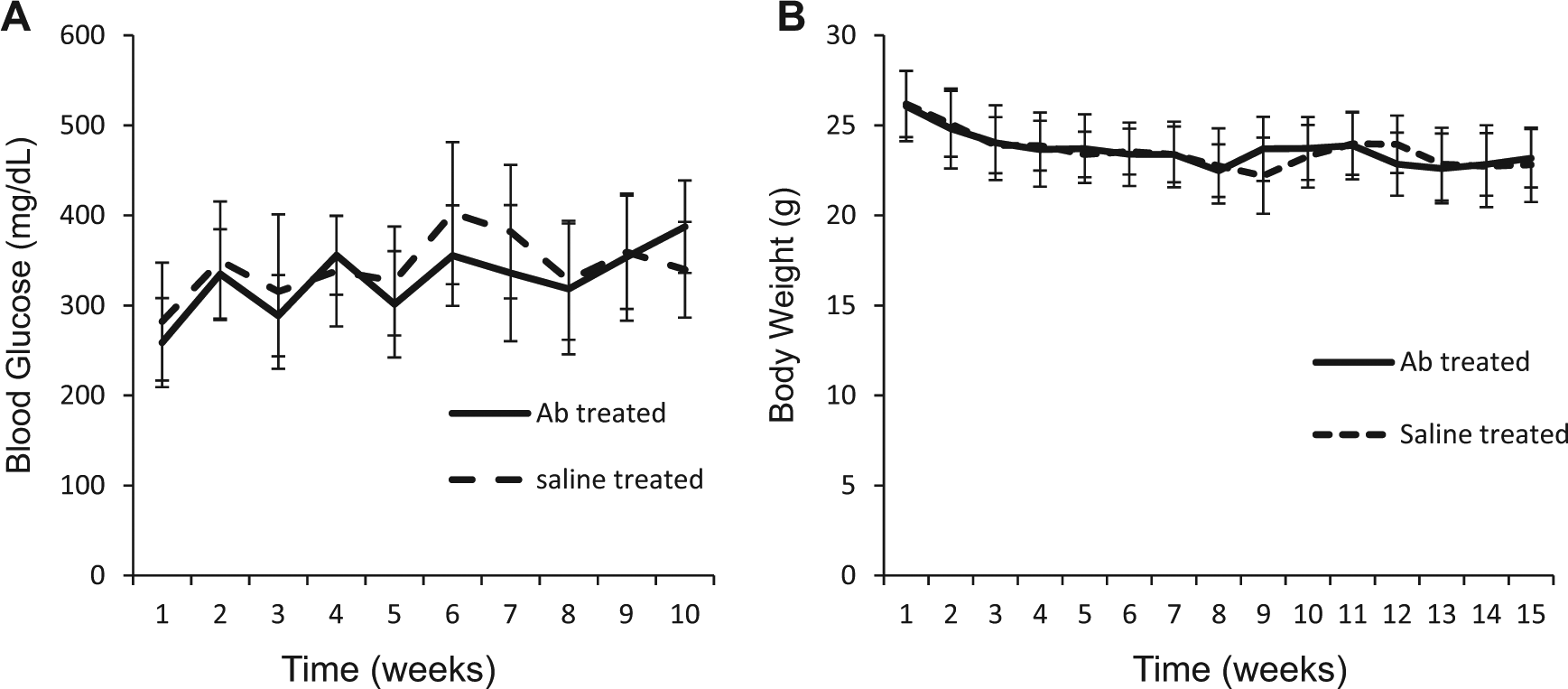
(A) Mean weekly blood glucose and (B) body weight measurements in saline-treated diabetic mice and RAGE-antibody treated diabetic mice. Data are expressed as average ± mean ± standard deviation.
SPECT/CT scan analysis
Five days after left femoral artery ligation, SPECT/CT scans following injection of 99mTc-HYNIC-RGD showed greater uptake of the tracer in the ischemic left limbs of RAGE-antibody treated diabetic mice compared to uptake in the ischemic limbs of saline treated diabetic mice (Figure 3: A–C). The quantitative tracer uptake as %ID in the RAGE-antibody treated diabetic ischemic left limbs (3.1±1.4 × 10−3) was significantly higher than the uptake in the contralateral right (non-ischemic) limb (1.81±0.62; p=0.04) or saline treated diabetic ischemic left limbs (1.68±0.35 × 10−3; p=0.02) (Figure 3D). For the RAGE-antibody treated diabetic group, the mean count ratio for the left over right (L/R) limbs (1.7±0.52) was also significantly higher than the saline treated diabetic group (1.15±0.12; p=0.01). The mean count ratio for the RAGE-antibody treated diabetic group was not statistically different from the control non-diabetic group (1.99±0.86; p=NS) (Figure 3E).
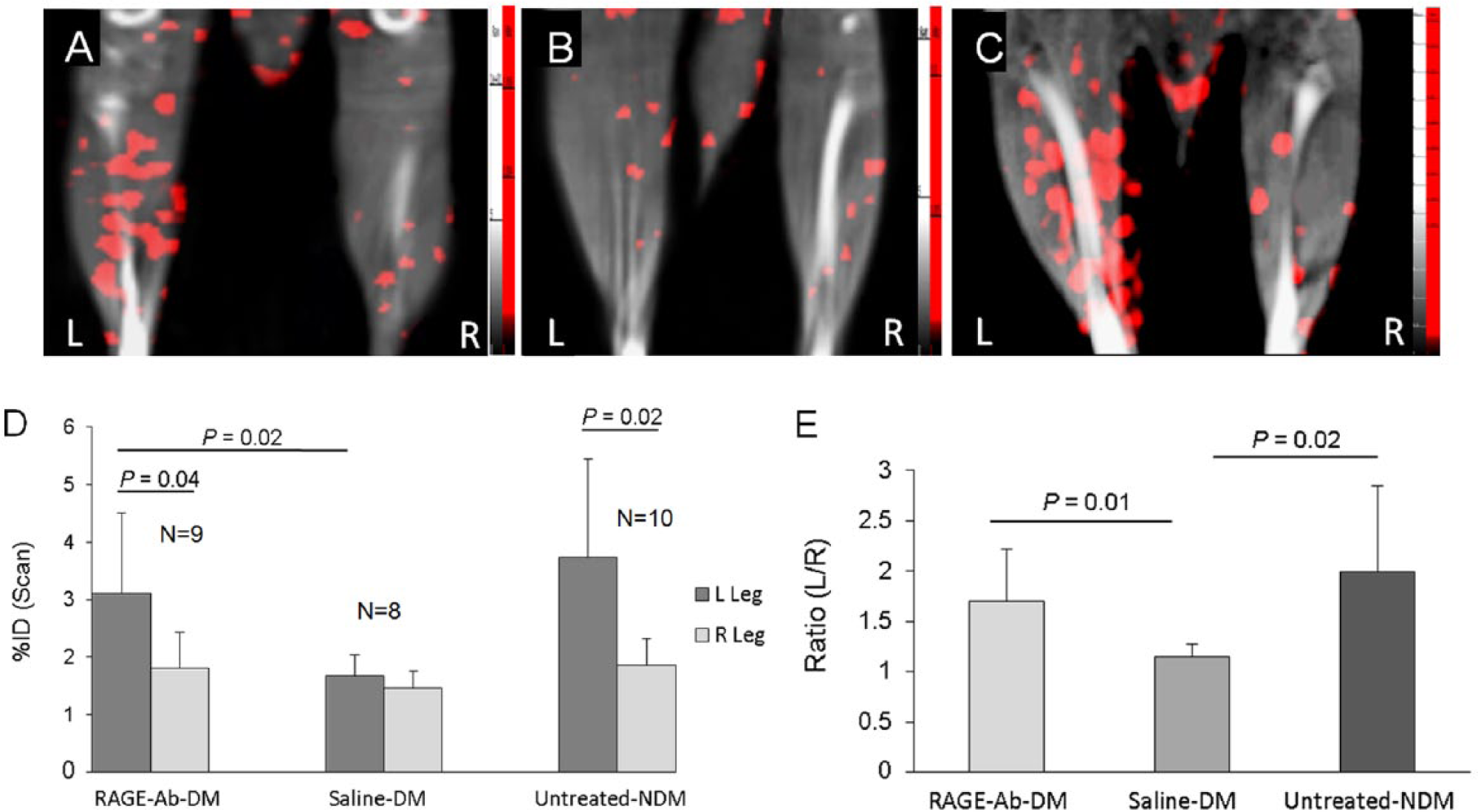
SPECT/CT imaging. Representative coronal slices from SPECT/CT scans following injection of 99mTc-HYNIC-RGD 5 days after left FAL for: (A) RAGE-antibody treated diabetic mouse; (B) vehicle-treated diabetic mouse; and (C) control non-diabetic mouse. Tracer uptake is shown in red for the L (ligated) and R (sham-operated) legs. (D) Bars represent mean ± standard deviation values for %ID for both left and right hind limbs for RAGE-antibody treated diabetic mice (left set of bars), saline treated diabetic mice (center bars), and non-diabetic mice (right set of bars) 5 days after FAL. (E) Bars represent mean ± standard deviation values for the ratios of left/right (L/R) hind limbs from scan count data. (DM, diabetes mellitus; NDM, non-diabetes mellitus; FAL, femoral artery ligation.)
Ultrasound Doppler imaging
Twenty-four days after femoral artery ligation, an index of hind limb blood flow was measured with Doppler ultrasound (Figure 4A). The left distal limb arterial pulsatility index for the RAGE-antibody treated diabetic group (1.49±0.5) was significantlly higher than the vehicle-treated diabetic group (0.61±0.39; p=0.04) (Figure 4B).
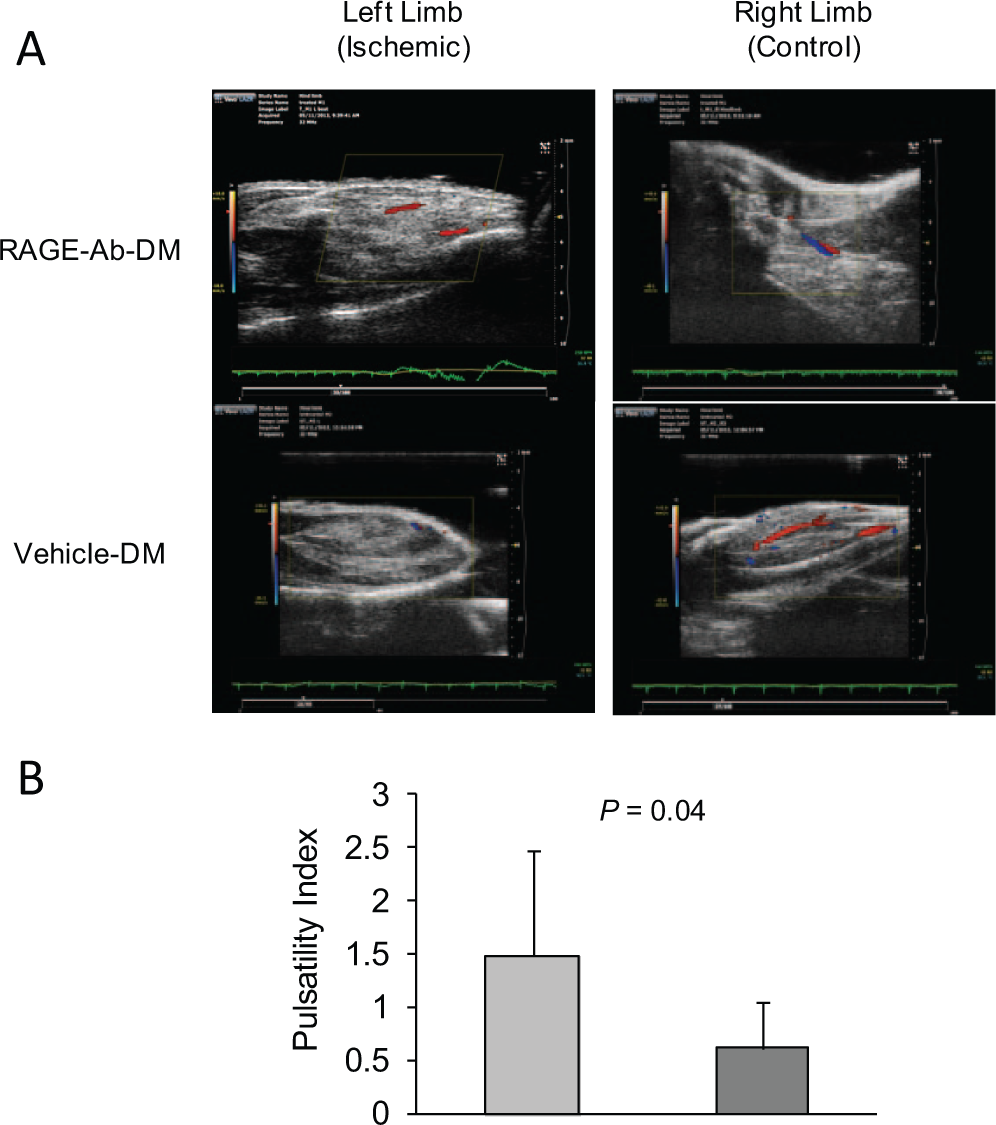
Doppler ultrasound imaging. (A) Representative Power-Doppler images of ischemic (left) and contralateral right hind limbs of RAGE-antibody treated diabetic mice (top) and saline treated diabetic mice (bottom) 24 days after left femoral artery ligation. (B) Bar graph for pulsatility index, where each bar represents mean ± standard deviation. Light gray bar = RAGE-ab-DM, dark gray = vehicle-DM.
Histological findings
Representative photomicrographs of tissue sections stained for H&E and vWF with bar graphs showing capillary densities are shown in Figure 5; representative photomicrographs stained for RAGE and bar graphs for RAGE-positive staining cells are shown in Figure 6. Quantitative immunohistological analysis showed treatment with RAGE antibody significantly increased capillary density in the ischemic hind limbs compared with saline treated diabetic ischemic hind limbs and reduced RAGE expression.
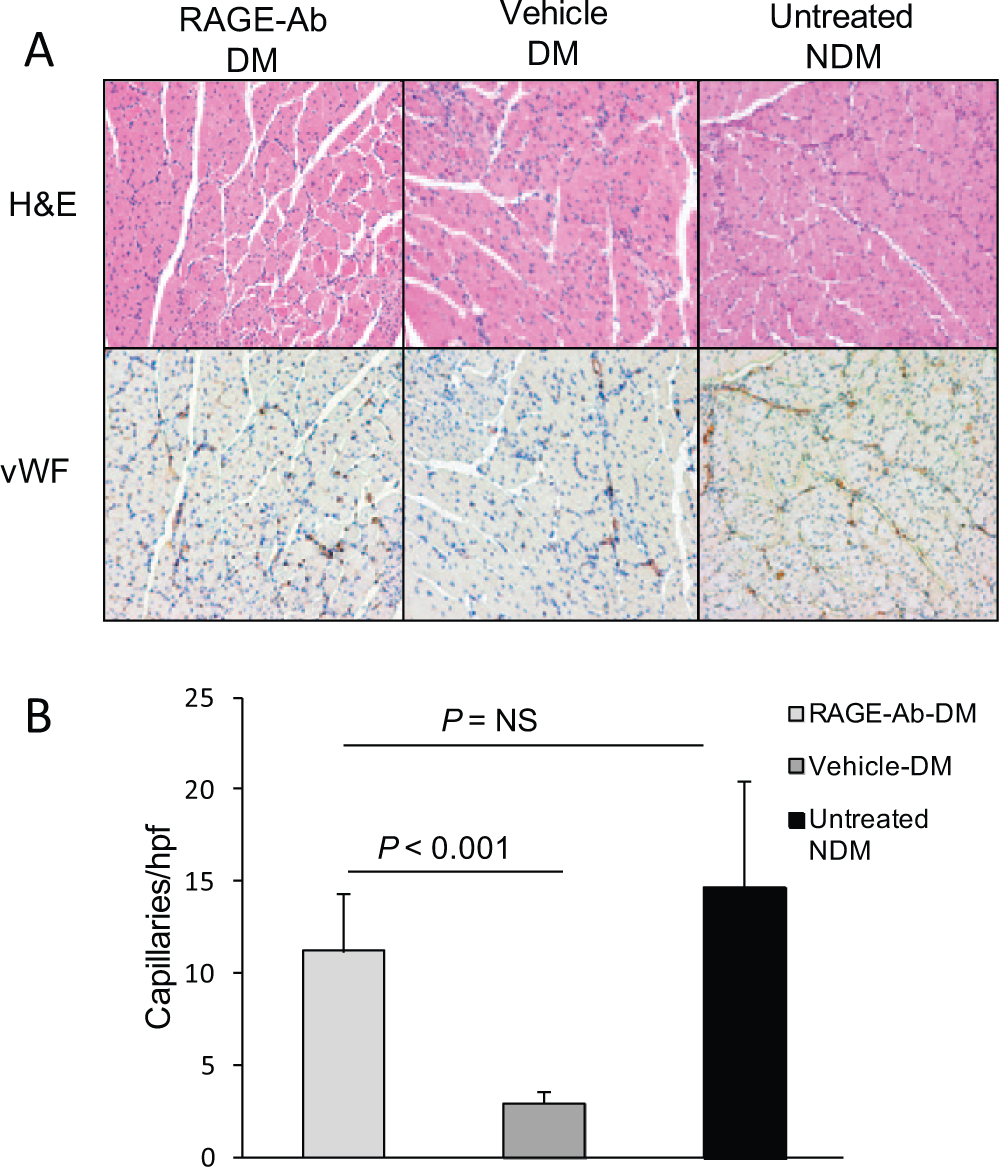
Capillary density. (A) Representative photomicrographs show immunostaining of ligated hind limb sections for H&E and vWF 24 days after surgery (magnification ×200). (B) The capillary density measured by quantifying the vWF staining was significantly higher in the RAGE-antibody treated diabetic group than in the saline treated diabetic group. There was no significant difference (NS) in the capillary density between the RAGE-antibody treated diabetic group and the control non-diabetic group. Bars represent average ± standard deviation. (DM, diabetes mellitus; NDM, non-diabetes mellitus; hpf, high-power field.)
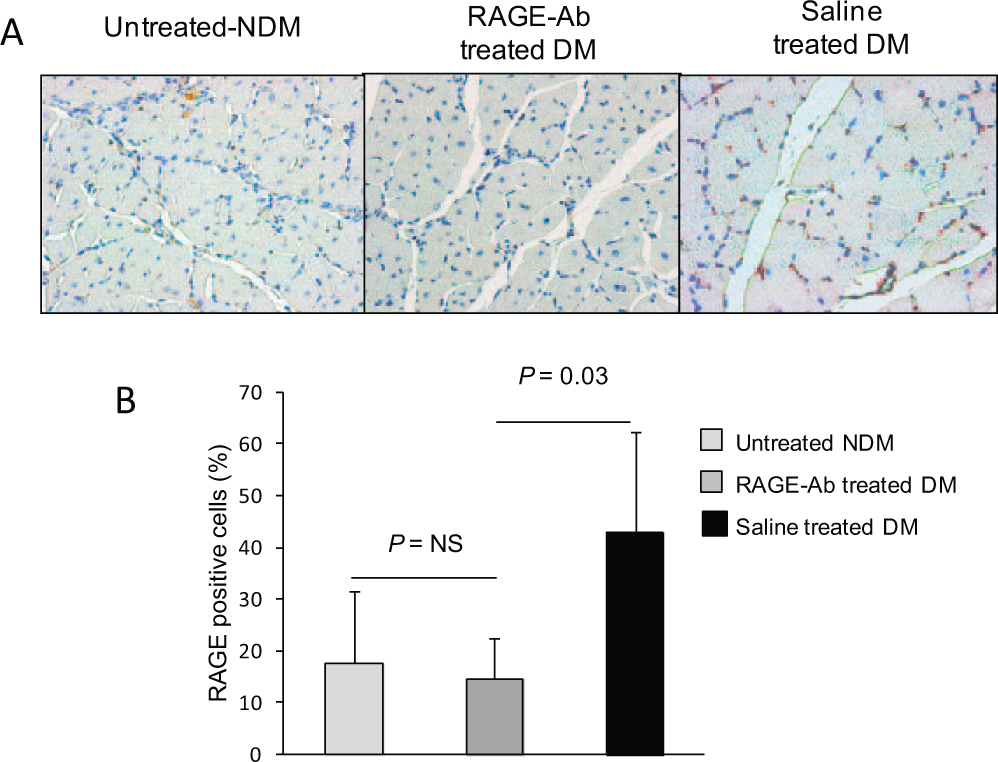
RAGE expression. (A) Representative photomicrographs show immunostaining of ligated hind limb sections. (B) The percent RAGE positive cells in the ischemic hind limbs were lower in the RAGE-antibody treated diabetic group compared with the vehicle-treated diabetic group.
Discussion
Initially developed as a diagnostic imaging agent, the anti-RAGE F(ab′)2 fragments were coupled to 99mTc via DTPA and, using this imaging probe, we have shown in live animals the extent of RAGE expression in ischemic hind limbs in diabetic mice and in the hind limbs of hyperlipidemic pigs.8–10 The extent of RAGE expression and its contribution to the progression of limb ischemia through inhibition of angiogenesis, indicates that blocking RAGE is a potential therapeutic approach to restore blood flow and reduce symptoms. We tested the blocking properties of the antibody against VSMCs in cell culture and the promising results of these experiments prompted us to design the present placebo-controlled treatment study. Our results indicate that in this model, pretreatment with RAGE antibody improves angiogenesis, as documented by in vivo molecular imaging at 5 days following femoral artery ligation (the peak time for angiogenesis in this model), and subsequently improves limb blood flow.
Diabetics have an attenuated angiogenic response to tissue hypoxia which contributes to long-term complications including poor collateral formation in the lower extremities which is further aggravated by poor wound healing and ulcers. Several mechanisms have been identified that contribute to this reduced angiogenic response in diabetics.2–7 The binding of ligand AGEs to its receptor initiates downstream pathways shown to impair angiogenesis, including suppressing metalloproteinase activity, attenuating the chemotactic response of monocytes to VEGF-A, and inhibiting the bone marrow-derived cell contribution to angiogenesis.6,7
Approaches to blocking RAGE have been reported using either a decoy protein copy of the extracellular two-thirds of the receptor (s-RAGE) that binds circulating ligands (particularly AGEs), thereby reducing receptor expression or administering a blocking or neutralizing antibody. Treatment of apoE−/− mice with s-RAGE as a decoy to bind circulating ligands reduced atherogenesis compared to placebo, improved wound healing, sepsis, tumor growth and metastasis, and treatment of diabetic mice with limb ischemia showed improved limb blood flow compared to placebo.20–23 Blocking or neutralizing antibodies to RAGE have been shown to reduce or ameliorate the detrimental effects of RAGE expression in diabetic renal disease. 24 This is the first study to report a therapeutic effect of a RAGE blocking antibody in limb ischemia. Therapeutic monoclonal antibodies are now approved for clinical use in several diseases, making this approach a possibility to pursue.
To document angiogenesis in live animals we used a radiolabeled RGD (Arg–Gly–Asp) peptide as an imaging probe that targets increased expression of αvβ3 integrin on endothelial cells in regions of capillary sprouting. Radionuclide probes to image angiogenesis in hind limb ischemia include SPECT and PET-labeled RGDs or peptomimetic proteins.9,14–17 Experimental studies using these probes have shown that quantitative estimates of tracer uptake on scans in the affected limbs correlate well with capillary sprouting. We therefore chose to estimate angiogenesis at 5 days after FAL (peak time for capillary sprouting) with imaging. We estimated mature capillary density at 24 days after FAL by quantitative immunohistology of positive vWF stained cells. At this later time point after femoral artery ligation in mice new capillary growth has declined.15,16
While imaging integrin expression indicates the extent of neovessel growth at 5 days after FAL, it is important to translate these results into arterial blood flow or tissue perfusion at the later time point. Doppler ultrasound is used to assess lower limb blood flow in humans. Operating in the Power-Doppler mode, small animal ultrasound machines can now obtain Doppler signals from arteries in murine hind limbs. The pulsatility index, also known as the ‘resistive index’, is determined from the peak systolic velocity, end-diastolic velocity, and velocity time integral and was first described by Dr Gosling in 1974. 19 While this index is currently used mostly in obstetrics and neurology (transcranial Doppler), application in vascular disease and PAD have been reported, as well as for flow assessment though vein grafts to coronary arteries intraoperatively.25–27 Compared to laser Doppler which assesses tissue perfusion, the pulsatility index more closely indicates blood flow in arterial vascular collaterals.
Summary
PAD is a common condition producing symptomatic ischemic leg pain for which there are currently no effective drug therapies. We developed a monoclonal antibody that targets a unique peptide sequence on the extracellular domain of the RAGE receptor initially for diagnostic applications and subsequently found to have blocking properties. Because of the importance of RAGE expression in vascular disease and in suppressing angiogenesis in limb ischemia, especially in diabetics, we hypothesized that treatment with this antibody in an established small animal model would show beneficial effects on angiogenesis and limb blood flow. We documented these beneficial effects with molecular imaging of angiogenesis at 5 days and by ultrasound Doppler flow at 24 days. We supported the in vivo imaging results with quantitative immunohistology showing lower RAGE staining and higher staining for vWF (capillary density) in the antibody treated diabetic mice compared to the saline treated mice. These results suggest that further testing and development of this antibody and imaging approach towards potential clinical trials is indicated.
Footnotes
Acknowledgements
The authors thank Dr Alexander Shekhtman (University at Albany, NY) for providing the AGEs.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
