Abstract
Introduction
Diabetic retinopathy (DR) is an ocular complication associated with diabetes mellitus, accounting for approximately 30%–40% of diabetic individuals. 1 Among DR cases, approximately 5%–10% of patients endure severe visual impairment. 2 At the advanced stage of DR, neovascularization followed by fibrovascular changes occur, leading to vitreous hemorrhage or tractional retinal detachment. 2 The sight-threatening disease becomes the leading cause of irreversible blindness in developed countries. 3 Fortunately, a large portion of visual loss arising from DR is preventable, accompanied by a decrease in its incidence in recent years. 4 The vitreous is an almost avascular and acellular gel-like body, composed of 97%–99% water, that can exhibit changes reflecting underlying retinal diseases. 5 In the eyes of diabetic patients, the vitreous humor constitutes a complex and extensive interactive arena for cytokines. 5 At present, studies focused on vitreous humor not only shed insights into the etiology of DR but also highlight the importance of molecular biomarkers.6–8
Extracellular matrix metalloproteinase inducer (EMMPRIN), also known as CD147 or Basigin, participates in the regulation of extracellular matrix remodeling during pathological and physiological processes such as inflammation, wound healing, and cancer.9,10 EMMPRIN was found to be upregulated in vitreous samples from patients with proliferative DR. 11 Importantly, the previous article indicated that EMMPRIN is implicated with proliferative DR angiogenesis by regulating matrix metalloproteinases (MMPs) and vascular endothelial growth factor (VEGF). 11 The study collected rat vitreous samples and further explored the influence of EMMPRIN on inflammation, angiogenesis, accumulation of MMPs, and blood-retinal barrier injury in DR.
Clinical studies have validated that serum levels of inflammatory factors, such as tumor necrosis factor α (TNFα), interleukin 6, and C-reactive protein, were increased in diabetics. The finding highlights the importance of inflammatory processes in the pathogenesis of DR. 12 Furthermore, the expression of angiogenic cytokines including VEGF and endothelin 1 was augmented in the vitreous from patients with advanced DR. 13 It is widely acknowledged that there is a close relationship between aggravated inflammation and enhanced angiogenesis. 14 MMPs act as active players in VEGF-mediated cell proliferation and new vasculature formation. 15 Particularly, it was reported that MMP2 and MMP9 can directly regulate angiogenesis. 16 Patients with proliferative DR showed high activities of MMP9 and MMP2 in the epiretinal neovasculature membrane. 17 Moreover, the catalytic activity of MMP9 not only directly affects angiogenesis but also leads to proteolysis of cellular junctional molecules at the blood-retina barrier. 18 Therefore, in light of previous studies, we put forward the hypothesis that EMMPRIN may affect angiogenesis and blood-retinal barrier injury in DR by regulating the accumulation of MMPs.
SP-8356 is an inhibitor for EMMPRIN and has been demonstrated to mitigate pathological fibrosis in Alkali-burned rat cornea. 19 Kisoo Pahk et al. found that SP-8356 can effectively lower MMP activity by hindering EMMPRIN dimerization. 20 In the current study, SP-8356 was administered to rats to silence EMMPRIN in vivo.
In this study, a rat model of DR was established, and the functions of EMMPRIN in regulating molecules related to inflammation, angiogenesis, and blood-retinal barrier in rat vitreous samples were investigated. The study further elucidates the role of EMMPRIN during DR progression and highlights the potential application of SP-8356 for DR treatment.
Materials and methods
Animal model
Animal experiments were approved by the Ethics Committee of Hubei Province Disease Prevention Control Center (No 202320005; 6th, January, 2023). Animals were maintained in line with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Male Sprague Dawley rats (8–9 weeks, 220–250 g) were purchased from Beijing Vital River Animal Technology (Beijing, China). All rats were randomly divided into four groups (n = 20/group): Sham + saline, Sham + SP-8356 (an inhibitor for EMMPRIN), DR + saline, and DR + SP-8356.
To establish the rat model for DR, rats were fasted overnight and then intraperitoneally injected with a high dose of streptozotocin (HY-13753, 65 mg/kg, Medchem Express, USA) in 10 mM sodium citrate buffer (C9999, pH = 6.0, Sigma Aldrich, St. Louis, USA). Three days after STZ injection, body weight and blood glucose concentrations were measured. Blood was collected from the tail vein, and rats having a glucose level exceeding 250 mg/dl were identified as diabetic.
Rats in sham groups were administered with an equal volume of citrate buffer.
Rats in sham + SP-8356 and DR + SP-8356 groups were intraperitoneally injected with SP-8356 (50 mg/kg in saline) once daily for 4 weeks.
Four weeks later, rats were deeply anesthetized with an overdose of sodium pentobarbital (100 mg/kg body weight). An eye speculum was utilized to open the rats’ eyelids. Then, vitreous samples were collected using 20 G needles 4 mm posterior to the limbus and stored at −80°C.21,22
Enzyme-linked immunosorbent assay (ELISA)
To determine the concentration of EMMPRIN, TNFα, IL-6 and IL-1β in rat vitreous samples or serum samples, rat EMMPRIN ELISA kit (orb780849, biorbyt, Cambridge, UK), rat TNFα ELISA kit (ab236712, Abcam), rat IL-6 ELISA kit (ab234570, Abcam), and rat IL-1β ELISA kit (ab255730, Abcam) were used. A microplate reader (Diatek, Wuxi, China) was used to identify ELISA plate reading.
Reverse transcription quantitative polymerase chain reaction (RT-qPCR)
Sequences of primers used for reverse transcription-quantitative PCR.

Western blotting
An equal volume of vitreous samples (15 μL) was boiled in Laemmli’s sample buffer (1:1, v/v) under reducing conditions for 10 min and analyzed as previously described.11,23 Rat serum samples were collected after centrifugation of coagulated blood samples at 2000 g for 10 min. The protein content was quantified using bicinchoninic acid method. The samples were subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis and then transferred to polyvinylidene fluoride membranes (Millipore, Billerica, USA). The membranes were blocked followed by incubation with primary antibodies at 4°C overnight. The primary antibodies in this study include anti-EMMPRIN (ab230921, Abcam), anti-MMP1 (orb446976, 1 mg/mL, biorbyt), anti-MMP2 (orb11061, 1 mg/mL, biorbyt), anti-MMP9 (orb11064, 1 mg/mL, biorbyt), anti-VEGF (orb11553, 0.5 mg/mL, biorbyt), anti-FGF2 (orb10186, 1 mg/mL), anti-TGFβ1 (ab215715, 1:1000), anti-zonula occludens-1 (ZO-1; ab96587, 1:500), anti-Occludin (ab216327, 1:1000), anti-Claudin-1 (ab307692, 1:1000), and anti-β-actin (ab6276, 1:5000). After further incubation with secondary antibodies, membranes were loaded with high-performance chemiluminescence kit (Vazyme, Nanjing, China) to visualize the blots. ImageJ software was applied to quantify protein intensity. 24
Statistical analysis
Data were processed using GraphPad Prism (La Jolla, USA) and are presented as the mean ± standard deviation. The comparison among multiple groups was evaluated by one-way analysis of variance followed by Tukey’s post hoc test. The value of p less than 0.05 was regarded as the threshold for statistical significance.
Results
EMMPRIN is highly expressed in serum and vitreous samples of model rats
After establishment of animal models and subsequent sampling, ELISA and western blotting were carried out to assess EMMPRIN level in serum and vitreous samples of four groups. In sham-treated rats, ELISA revealed that EMMPRIN concentration was not significantly altered by SP-8356 (Figure 1(a) and (d)). Compared with EMMPRIN level in the sham + saline group (serum: 34.26 ± 3.11; vitreous: 44.52 ± 4.2), DR rats had high serum and vitreous levels of EMMPRIN (serum: 325.69 ± 28.15 pg/mL; vitreous: 415.36 ± 36.58 pg/mL). Additionally, the DR-induced increase in EMMPRIN level was reversed by SP-8356 treatment (serum: 145.63 ± 12.47 pg/mL; vitreous: 182.46 ± 16.41 pg/mL) (Figure 1(a) and (d)). The results of western blotting were in accordance with those of ELISA, which disclosed the high protein expression of EMMPRIN in rat serum and vitreous samples as well as the successful knockdown efficacy of SP-8356 in DR rats (Figure 1(b), (c), (e), (f), p < 0.001). EMMPRIN is highly expressed in rat serum and vitreous humor samples. Rats in DR + SP-8356 or DR + saline group were intraperitoneally injected with 65 mg/kg streptozotocin to induce diabetes and then administered with 50 mg/kg SP-8356 (an inhibitor for EMMPRIN) or saline. Rats in sham + SP-8356 or sham + saline group received injection of 65 mg/kg saline followed by administration of 50 mg/kg SP-8356 or saline. (a) ELISA was performed to measure the content of EMMPRIN in rat serum samples of each group. (b) The protein level of EMMPRIN in rat serum samples was measured by western blotting. (c) Relative protein level of EMMPRIN in rat serum was calculated with normalization to β-actin. (d) EMMPRIN level in rat vitreous samples was measured using ELISA. (e) Western blotting was conducted to measure EMMPRIN protein level in rat vitreous samples of each group. (f) Relative protein level of EMMPRIN in vitreous humor was normalized to β-actin level. Significance among multiple groups was evaluated by one-way analysis of variance followed by Tukey’s post hoc test. Data are shown as the mean ± standard deviation. ***p < 0.001 versus sham + saline group, ###p < 0.001 versus DR + saline group. DR: Diabetic retinopathy. EMMPRIN: extracellular matrix metalloproteinase inducer. ELISA: Enzyme-linked immunosorbent assay.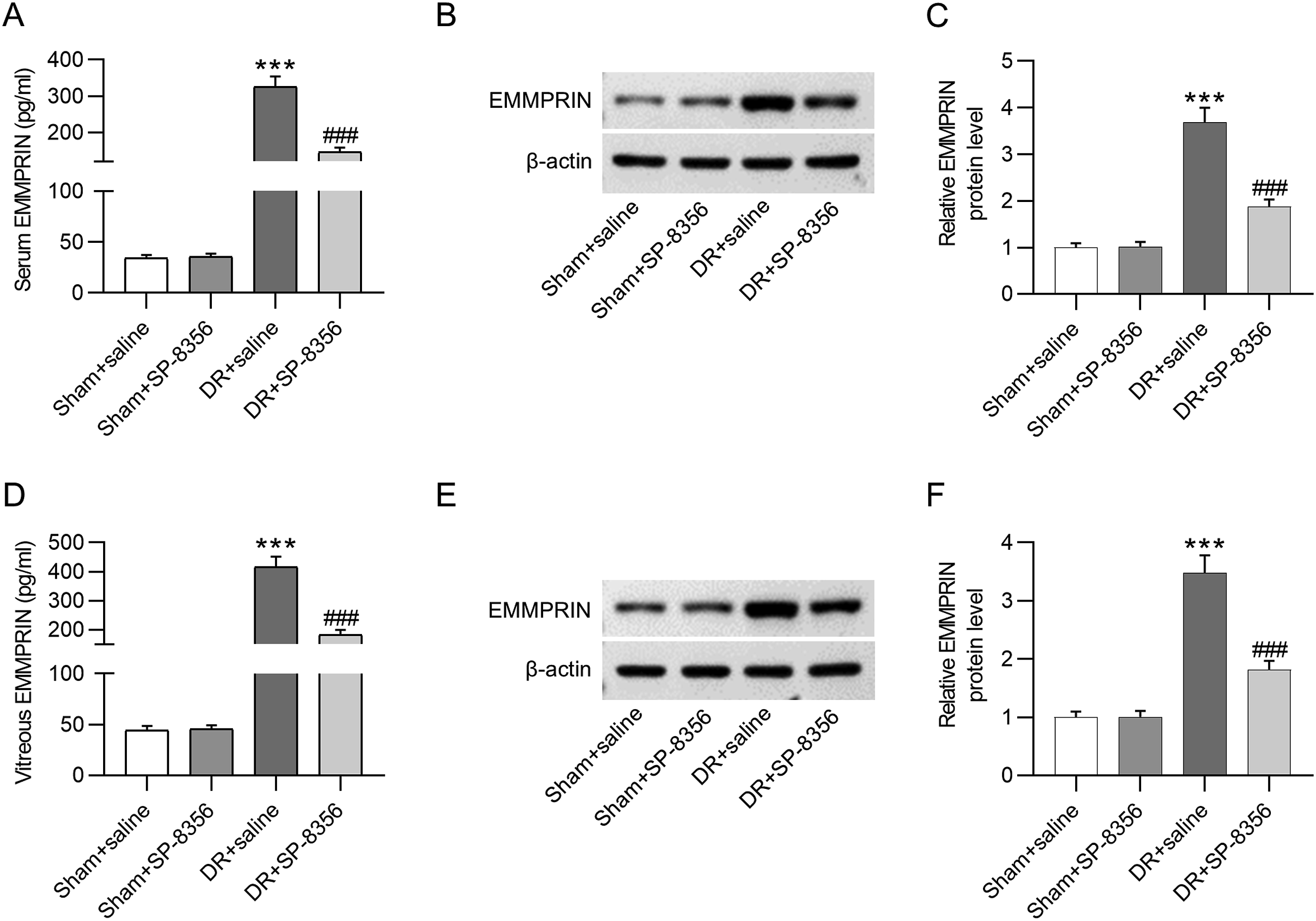
Inhibition of EMMPRIN restrains the release of inflammatory cytokines
ELISA was conducted to assess the concentration of proinflammatory cytokines in rat vitreous samples. The concentrations of TNF-α, IL-6 and IL-1β were markedly increased in DR rats (65.28 ± 6.14, 76.85 ± 7.21, 83.65 ± 7.85 pg/mL) compared with their levels in sham + saline group (12.85 ± 1.16, 22.65 ± 2.05, 18.87 ± 1.63 pg/mL) (Figure 2(a)–(c)). Inhibition of EMMPRIN using SP-8356 significantly repressed the release of proinflammatory cytokines (22.63 ± 2.02, 36.23 ± 3.28, 39.58 ± 3.51 pg/mL) in vitreous samples (Figure 2(a)–(c)). Consistently, results of RT-qPCR revealed that mRNA levels of TNF-α, IL-6 and IL-1β were markedly increased in response to DR modeling compared to those in the context of sham treatment (3.85 ± 0.35 vs. 1 ± 0.09, 4.21 ± 0.38 vs. 1 ± 0.1, 4.62 ± 0.42 vs. 1 ± 0.09) (Figure 2(d)–(e)). While DR-induced high proinflammatory cytokine levels were counteracted through inhibition of EMMPRIN using SP-8356 treatment (1.49 ± 0.12, 2.33 ± 0.2, 1.85 ± 0.15) (Figure 2(d)–(e)). SP-8356 treatment alone did not alter inflammatory cytokine levels in vitreous samples of sham rats (Figure 2(a)–(e)). The finding indicates that inhibition of EMMPRIN leads to the alleviation of inflammatory response in DR rats. The silencing of EMMPRIN diminishes proinflammatory cytokine levels. (a-c) ELISA was performed to measure concentrations of inflammatory cytokines, including TNF-α (a), IL-6 (b) and IL-1β (b), in rat vitreous samples. (d–e) RT-qPCR was conducted to assess mRNA levels of inflammatory factors (TNF-α, IL-6, and IL-1β) in rat vitreous humor. Significance among multiple groups was evaluated using one-way analysis of variance followed by Tukey’s post hoc test. Data are displayed as the mean ± standard deviation. ***p < 0.001 versus sham + saline group; ###p < 0.001 versus DR + saline group. EMMPRIN: extracellular matrix metalloproteinase inducer. ELISA: Enzyme-linked immunosorbent assay. TNF-α: Tumor Necrosis Factor alpha. IL-6: Interleukin-6. IL-1β: Interleukin-1 beta. RT-qPCR: reverse transcription quantitative polymerase chain reaction. DR: Diabetic retinopathy.
Inhibition of EMMPRIN prevents the overproduction of MMPs
Previous studies elucidate that EMMPRIN can promote angiogenesis by enhancing the production of MMPs and VEGF in psoriatic arthritis, and knocking down EMMPRIN expression reduces the angiogenic potential in colon carcinoma.25,26 Therefore, subsequent experiments were conducted to evaluate the effect of EMMPRIN inhibitor (SP-8356) on the production of MMPs and angiogenic factors in DR. Western blotting was performed to measure MMP1, MMP2, and MMP9 protein levels in sham or DR model rats in the presence or absence of SP-8356 treatment. As shown in Figure 3(a)–(d), MMP1, MMP2, and MMP9 protein levels were surged in vitreous samples of DR rats (3.45 ± 0.3, 3.36 ± 0.3, 3.05 ± 0.27), and the alterations were strongly dampened by SP-8356 (1.85 ± 0.15, 1.56 ± 0.13, 1.36 ± 0.11). In addition, mRNA levels of the three MMPs in rat vitreous humor were all increased by DR modeling (6.36 ± 0.6, 4.81 ± 0.44, 6.3 ± 0.6), and their high mRNA expression was reversed by the deficiency of EMMPRIN (3.29 ± 0.28, 2.3 ± 0.2, 3.26 ± 0.28) (Figure 3(e)–(g)). MMP accumulation in sham rats was not significantly altered by SP-8356 (Figure 3(a)–(g)). In conclusion, MMPs were accumulated in vitreous samples of DR rats, and the silencing of EMMPRIN significantly ameliorates the excessive generation of MMPs in DR rats. Inhibition of EMMPRIN attenuates the overproduction of MMPs. (a) Western blotting was performed to quantify protein levels of MMP1, MMP2, and MMP9 in rat vitreous samples. (b–d) Relative MMP1 (b), MMP2 (c), and MMP9 (d) protein levels were quantitated and normalized to β-actin. (e–g) RT-qPCR was performed to assess mRNA levels of MMP1, MMP2, and MMP9 in rat vitreous humor. Comparisons among multiple groups was evaluated by one-way analysis of variance followed by Tukey’s post hoc test. Data are shown as the mean ± standard deviation. ***p < 0.001 versus sham + saline group, ###p < 0.001 versus DR + saline group. EMMPRIN: extracellular matrix metalloproteinase inducer. MMPs: matrix metalloproteinases. RT-qPCR: reverse transcription quantitative polymerase chain reaction. mRNA: messenger RNA. DR: Diabetic retinopathy.
Deficiency of EMMPRIN leads to upregulation of tight junction proteins
Changes of tight junction complex in response to inflammation can result in alterations in retinal vascular permeability, leading to retinal dysfunction.
27
Protein levels of tight junction factors in vitreous samples were quantified by western blotting. ZO-1, Occludin, and Claudin-1 levels were markedly reduced in the context of DR (0.28 ± 0.02, 0.31 ± 0.03, 0.25 ± 0.02), and the alterations were significantly reversed in response to EMMPRIN deficiency (0.85 ± 0.08, 0.9 ± 0.09, 0.88 ± 0.08) (Figure 4(a)–(d)). The mRNA expression levels of ZO-1, Occludin, and Claudin-1 were also diminished in the DR + saline group relative to that in the sham + saline group (0.33 ± 0.03 vs. 1 ± 0.1, 0.45 ± 0.04 vs. 1 ± 0.09, 0.28 ± 0.02 vs. 1 ± 0.09) (Figure 4(e)–(g)). The downregulation of tight junction proteins in DR rats was rescued by SP-8356 administration (0.62 ± 0.06, 0.57 ± 0.08, 0.92 ± 0.09) (Figure 4(e)–(g)). Inhibition of EMMPRIN in sham rats did not lead to the dysfunction of tight junctions (Figure 4(a)–(g)). The results show that inhibition of EMMPRIN contributes to the recovery of tight junction proteins in DR. EMMPRIN depletion increases levels of tight junction proteins. (a) Levels of tight junction proteins (ZO-1, Occludin, and Claudin-1) were identified by western blotting. (b–d) ZO-1 (b), Occludin (c), and Claudin-1 (d) levels were quantified by ImageJ software with normalization to β-actin. (E-G) The mRNA levels of tight junction proteins (ZO-1, Occludin, and Claudin-1) were assessed using RT-qPCR. Significance among multiple groups was analyzed by one-way analysis of variance followed by Tukey’s post hoc test. Data are displayed as the mean ± standard deviation. ***p < 0.001 versus sham + saline group; ###p < 0.001 versus DR + saline group. EMMPRIN: extracellular matrix metalloproteinase inducer. ZO-1: zonula occludens-1. RT-qPCR: reverse transcription quantitative polymerase chain reaction. mRNA: messenger RNA. DR: Diabetic retinopathy.
EMMPRIN knockdown suppresses protein and mRNA levels of angiogenic factors
Vitreous samples from DR rats showed high protein levels of angiogenic factors including VEGF, FGF2, and TGFβ1 (2.88 ± 0.25, 3.32 ± 0.3, 3.85 ± 0.35) (Figure 5(a)–(b)). The inhibition of EMMPRIN using SP-8356 successfully dampened the protein expression of these angiogenesis-related factors (1.83 ± 0.15, 1.86 ± 0.15, 1.74 ± 0.15) (Figure 5(a)–(d)). Similarly, DR modeling in rats led to an increase in mRNA expression of VEGF, FGF2, and TGFβ1 compared to corresponding expression levels in the sham group (4.22 ± 0.38 vs. 1 ± 0.1, 3.82 ± 0.35 vs. 1 ± 0.09, 4.63 ± 0.42 vs. 1 ± 0.1), and these changes were reversed by the inhibition of EMMPRIN (1.54 ± 0.13, 1.74 ± 0.15, 1.68 ± 0.14) (Figure 5(e)–(g)). Expression levels of angiogenetic factors in sham rats were not significantly changed by SP-8356 administration (Figure 5(a)–(g)). The finding suggests the potential role of EMMPRIN in regulating angiogenesis during DR development. EMMPRIN knockdown suppresses protein levels of angiogenic factors. (a) Protein levels of angiogenic factors, including VEGF, FGF2, and TGFβ1, in rat vitreous humor were quantified using western blotting. (b–d) Relative VEGF, FGF2, and TGFβ1 protein levels in each group were calculated and normalized to β-actin level. (e–g) RT-qPCR was performed to measure mRNA expression of angiogenic factors (VEGF, FGF2, and TGFβ1) in rat vitreous humor. Significance among multiple groups was evaluated by one-way analysis of variance followed by Tukey’s post hoc test. Data are shown as the mean ± standard deviation. ***p < 0.001 versus sham + saline group; ###p < 0.001 versus DR + saline group. EMMPRIN: extracellular matrix metalloproteinase inducer. VEGF: vascular endothelial growth factor. FGF2: Fibroblast growth factor 2. TGFβ1: Transforming growth factor beta 1. RT-qPCR: reverse transcription quantitative polymerase chain reaction. mRNA: messenger RNA. DR: Diabetic retinopathy.
Discussion
The pathogenesis of DR extends beyond the retina, with the vitreous serving as a reservoir for inflammatory cytokines, chemokines, and angiogenesis mediators. This explains why vitrectomy provides long-lasting benefits. 28 This study validated the high EMMPRIN level in rat vitreous samples and summarized that EMMPRIN depletion suppressed levels of inflammatory factors, MMPs, tight junction proteins and angiogenic factors in vitreous samples of DR rats.
The blood-retina barrier is important for the maintenance of optimum retinal function. 29 The breakdown of this barrier increases vascular permeability and leads to vision loss. 30 Nearly all retinal diseases are accompanied by the disruption of the blood-retina barrier. The outer blood retina barrier is formed by tight junctions among retinal pigment epithelial cells, while the inner barrier consists of tight junctions among retinal capillary endothelial cells. 29 Tight junctions comprise over 40 proteins including the claudin family, the transmembrane proteins occludin, peripheral membrane-related proteins such as ZO family, and the junction adhesion molecule family. 27 In the present study, the pathogenesis of DR contributed to the loss of tight junctions, suggesting the disruption of the barrier integrity. EMMPRIN deficiency upregulated mRNA and protein levels of tight junctions including ZO-1, Occludin, and Claudin-1. A previous study revealed that EMMPRIN was expressed at a high level on the ocular surface of dry eye patients and interfered with epithelial barrier function depending on MMP9-mediated Occludin cleavage. 31 In cholangiocarcinoma, a reduction of Claudin-1 and E-cadherin levels was observed in the context of EMMPRIN overexpression. 32 The findings from previous studies were consistent with ours. Despite its role in blood retina barrier, EMMPRIN is also a transmembrane molecule in neural blood-brain barrier endothelial cells 18 and is regarded as an upstream regulator of MMPs in central nervous system. 33
Moreover, it was reported that EMMPRIN promotes angiogenesis by enhancing the accumulation of MMPs and VEGF in psoriatic arthritis. 25 The promoting effect of EMMPRIN on angiogenesis has also been verified in renal cell carcinoma. 34 Consistent with these findings, we found that EMMPRIN depletion reduced protein levels of MMP1, MMP2, MMP9, VEGF, FGF2, and TGFβ1 in vitreous samples. Akarii Saiki et al. elucidated that EMMRPIN suppressed FGF2-caused IL-6 secretion in an osteoblastic cell line MC3T3-E1. 35 This may explain the reason why inflammatory cytokine IL-6 was also reduced in vitreous humor in the present study. The promoting effect of EMMPRIN on TGFβ1 level was in line with a previous study that revealing the involvement of EMMPRIN in a positive ERK1/2-dependent loop of the TGFβ1/Smad signaling. 36 Another study validated that EMMPRIN is positively associated with TGFβ1 production; however, the inhibition of EMMRPIN using AC-73 did not significantly affect the activated TGFβ1 protein. 37 This study concluded that EMMRPIN knockdown using SP-8356 could diminish the TGFβ1 protein level in vitreous samples. In spite of VEGF, FGF2, and TGFβ1, another angiogenic factor endothelin-1 was found to be increased in DR vitreous. 5 However, the impact of EMMPRIN on the endothelin-1 level was not explored in this study.
The limitations of the study were that only single experiments were performed to explore the role of EMMPRIN in vitreous samples. The upstream or downstream molecules of EMMPRIN were not illustrated in the present study. According to previous studies, the blocking of EMMPRIN was shown to inhibit the Wnt/β-catenin signaling in periodontitis. 38 Cao et al. reported that inhibition of AMPK, MAPK and Protein Kinase C pathway led to the downregulation of EMMPRIN in macrophages. 39 Atorvastatin reduces EMMPRIN expression to attenuate plaque vulnerability by inhibiting cyclooxygenase-2/prostaglandin E2 pathway. 40 In addition, the Wnt/β-catenin, AMPK, MAPK, and cyclooxygenase-2/prostaglandin E2 pathways were all reported to be involved in DR progression.41–44 In the future, these pathways shall be further investigated to comprehensively disclose the mechanism of EMMPRIN in DR. Moreover, clinical samples should be collected for further investigation of EMMPRIN. A previous study showed that EMMPRIN expression was upregulated in vitreous samples collected from patients with proliferative DR, and the vitreous level of EMMPRIN correlates with those of MMP1, MMP9, and VEGF. 11 Our findings obtained from experimental animal models are consistent with those in the previous study based on clinical samples. Therefore, it can be suspected that similar findings to our study might be anticipated in human DR. Nevertheless, more clinical experiments are supposed to be performed in the future for further validation.
In conclusion, the inhibition of EMMPRIN alleviates angiogenesis and blood-retina injury in the rat model of DR by regulating MMPs. The study further highlights the contributory role of EMMPRIN in vitreous samples to the progression of DR, and the inhibition of EMMPRIN using SP-8356 has the potential to be a therapeutic method for DR.
Footnotes
Author Contributions
Jie Zhong and Yingzi Guo were the main designers of this study. Jie Zhong, Yingzi Guo and Wang Xin performed the experiments and analyzed the data. Jie Zhong, Yingzi Guo and Wang Xin drafted the manuscript. All authors read and approved the final manuscript.
Declaration of conflicts of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
