Abstract
Catheter-based thrombolysis (CBT) is emerging as an option for acute pulmonary embolism (PE). Although prior studies have demonstrated improvement in right ventricular function, little data is available regarding clinical patient outcomes. Our institution adopted CBT as an option for patients with submassive and massive PE and we evaluated its effect on patient outcomes. Two hundred and twenty-one patients who presented to our institution with submassive and massive PE were analyzed over three years by time period; 102 prior to the use of CBT and 119 during the time CBT was performed. The primary outcome was in-hospital major adverse clinical events (a composite of death, recurrent embolism, major bleeding, or stroke). Secondary outcomes were overall and ICU length of stay and individual components of the composite outcome. Mean age was 56.3±16 years with high rates of central PE (57.9%), RV dysfunction (37%), and myocardial necrosis (26%). Mean RV/LV ratio was 1.2. Thirty-two patients were treated with CBT. The composite endpoint occurred more frequently in the CBT era vs the pre-CBT era (21.0% vs 14.7%, p=0.23). After multivariate adjustment, CBT treatment demonstrated no effect on major adverse clinical events (OR 0.84, CI 0.22–3.22, p=0.80). CBT era patients had an unadjusted 37% increase in ICU days and 54% increase in total length of stay (p<0.001). Within the CBT era, CBT treatment resulted in an adjusted 190% increase in overall length of stay (p<0.001). CBT did not demonstrate improvement in hospital outcomes, despite adjustments of PE severity, and was associated with a significant increase in overall and ICU length of stay.
Introduction
Patient presentations for acute pulmonary embolism (PE) vary widely. Traditional prognostic indices for hemodynamically stable patients with PE include evidence of right ventricular (RV) dysfunction,1,2 presence of RV thrombus,1,3 elevated brain natriuretic peptide (BNP) levels,4 –6 and elevated cardiac biomarkers.7,8 While most patients receive anticoagulation as the cornerstone of medical therapy, systemic thrombolysis is recommended for patients who are in shock or are hemodynamically unstable. One of the major limitations of systemic thrombolysis is a high incidence of bleeding. 9 Limited small trials have evaluated outcomes of thrombolytic therapy for acute PE with variable results, but pooled analysis demonstrates significantly decreased composite endpoints of death and recurrent thromboembolism. 10
Patients with submassive PE represent a unique subset of all PE patients. While systemic thrombolysis may not be indicated, mortality remains high as they have evidence of right ventricular (RV) dysfunction or myocardial necrosis without features of overt shock.
In the last several years, variations in fibrinolytic dosing have been evaluated in the intermediate risk population. Low-dose tissue plasminogen activator (up to 50% of the full dose) in the MOPETT (Moderate Pulmonary Embolism Treated with Thrombolysis) study demonstrated a large reduction in clinical events without an increase in bleeding. 11 However, enthusiasm has been tempered by the PEITHO (Pulmonary Embolism Thrombolysis) study which resulted in significant increases in major bleeding. 12 Given that the potential benefits of fibrinolytic therapy may be offset by concerns about adverse bleeding rates, further large-scale studies are needed to define the role of thrombolysis further.
The use of catheter-based thrombolysis (CBT) has emerged over recent years as a potential treatment option for patients with submassive PE. Recently, randomized studies have examined the use of this technology demonstrating improvements in RV function and dimension with low rates of bleeding. 13 However, these studies have not evaluated clinical outcomes or compared treatment strategies of CBT to standard medical therapy in a real world setting. 13 Our institution recently adopted CBT as a treatment option for patients with submassive and massive PE, and we sought to evaluate our experience.
Methods
Sample
The Institutional Review Board at the University Of Kentucky approved this study. Patients included in the study were selected using current procedural terminology codes for PE as well as by reviewing the University of Kentucky Catheterization Laboratory database. The sample consisted of 337 consecutive patients treated for acute PE between 1 March 2010 and 30 March 2013.
Individual computed tomography (CT) scans were reviewed in order to confirm the presence of definite PE and to define RV and LV (left ventricular) dimensions. Patients with PE as identified by ventilation/perfusion scanning were not included as part of the cohort. All pulmonary embolisms were classified further as central or segmental. Central pulmonary embolism was selected when the thrombus was located in either the right, left, or main pulmonary artery. In individuals with radiographic evidence of PE, the clinical investigators determined the RV:LV ratio by individually reviewing chamber diameters. Ventricular diameters were measured as the maximum distance from the interventricular septum to the endocardial border perpendicular to the long axis of the heart. 14 All patients with a CT-determined RV:LV ratio ⩾0.9 were included in the study. Patients with RV:LV ratios of ⩾0.7 to ⩽0.9 were further investigated for other signs of RV dysfunction, as measured by echocardiogram or signs of myocardial necrosis. Massive and submassive groups were determined according to American Heart Association guidelines. 15
A total of 114 patients were excluded as they did not have radiographic evidence of having a PE or did not meet criteria for submassive PE. In circumstances where a patient had multiple admissions for a submassive PE, only the most recent was included. The final sample consisted of 221 patients with acute submassive and massive PE. The study design was a retrospective cohort analysis in which outcomes were analyzed based on treatment strategy. Consultation to the interventional service was left to the discretion of the referring physician for consideration of CBT therapy. Systemic thrombolysis was administered with an institutional full dose alteplase infusion protocol when clinically indicated.
Catheter-based thrombolysis procedure
Patients were administered therapeutic unfractionated or low molecular weight heparin prior to the procedure. Femoral venous access was obtained with ultrasound guidance. In the majority of cases, when bilateral catheters were planned, two 6 French sheaths were placed in the common femoral vein. The right and left pulmonary arteries were then selectively engaged with diagnostic catheters and next exchanged over a long wire for the infusion catheters. An angled pigtail, Bernstein, or balloon tipped wedge catheter were most commonly used. Catheterization laboratory standards were followed. 16 The procedure was considered a technical success if the desired catheter was able to be delivered in stable position with acceptable distal run off. Next, the alteplase infusion was initiated at 1 mg/hour and ultrasound-assisted thrombolysis begun with the patient transferred to the intensive care unit (ICU). The infusion was continued for 12 hours in the case of bilateral catheters or 24 hours in unilateral placement for a total dose of 24 mg. At the completion of the alteplase infusion, pulmonary pressures were obtained and the catheters were removed, including the access sheaths regardless of clinical response. No additional systemic thrombolysis was administered. The patient was continued on standard anticoagulation and patient disposition, including level of care, was left to the discretion of the primary service.
Outcomes
Personnel, blinded to the study design, extracted pre-specified demographics and clinical data from our institutional medical records. Data included patient demographics, medical comorbidities, baseline laboratory studies, baseline vital signs, and management strategies. Repeat RV imaging by echocardiography and CT were collected when available. Clinical events that had occurred by the time of discharge included death, length of stay, stroke or transient ischemic attack, renal failure, urgent surgery, recurrent PE, vascular complications, need for transfusion, and other major bleeding events. For the purposes of our study, any bleeding determined to have significantly altered patient care was determined to be a major bleeding event (Bleeding Academic Research Consortium [BARC] classification 2–5). 17 Any events needing adjudication were determined by two independent authors, who were blinded to the treatment strategy, with 100% agreement.
The primary outcome was a composite of major adverse clinical events (MACE) including death, recurrent PE, major bleeding, or stroke. Secondary outcomes focused on in-hospital and ICU length of stay.
Statistical analysis
Outcomes were compared by treatment era, pre-CBT (from March 2010 to September 2011) versus CBT (from September 2011 to March 2013). In the CBT era, patients were compared by treatment strategy (CBT versus medical therapy). Dichotomous outcomes were summarized by percentages, with Fisher’s exact test used for comparisons. Normally-distributed outcomes are summarized by means and standard deviations with t-tests used for comparisons. Numeric, but non-normally-distributed outcomes are summarized by medians and interquartile ranges with rank sum tests used for comparisons.
Associations between MACE and individual patient factors were estimated and tested for significance with univariable logistic regression models. Patient factors that were tested for potential association with future events were selected based upon previously documented associations with adverse outcomes including age, gender, diabetes, hypertension, chronic kidney disease, known coronary artery disease, prior myocardial infarction, history of stroke, tobacco abuse, obesity (BMI>30), and prior PE. The potential cardiac risk factors were included in a multivariable logistic regression model if they showed univariable associations with a p<0.20. Results from logistic regression modeling were summarized by estimated odds ratios (OR) with corresponding 95% confidence intervals (CI).
The secondary outcomes of in-hospital and ICU length of stay were also analyzed using univariable and multivariable modeling, similar to that described above, except that linear regression replaced logistic regression, and estimated regression coefficients (betas) replaced OR as measures of effect. Because the secondary outcomes were non-normally distributed, they were subjected to a transformation before linear regression modeling: add one, and then take the natural logarithm; the offset of one was necessary because the logarithm of zero is undefined. Thus, exponentiation of beta gives approximately the factor by which the mean level of the (offset) outcome is multiplied when the risk factor increases by one unit. A p-value less than 0.05 was considered statistically significant. Version 9.3 of Statistical Analysis Software was used for data analysis.
Results
The baseline demographics of the study population are displayed in Table 1. Overall, the two populations were comparable with only subtle differences including a higher proportion of patients with diabetes being treated in the pre-CBT era. Also, the CBT era saw more patients with a history of hypercoagulable states and patients who had surgery within the last three months or a history of prolonged immobilization.
Patient demographics.
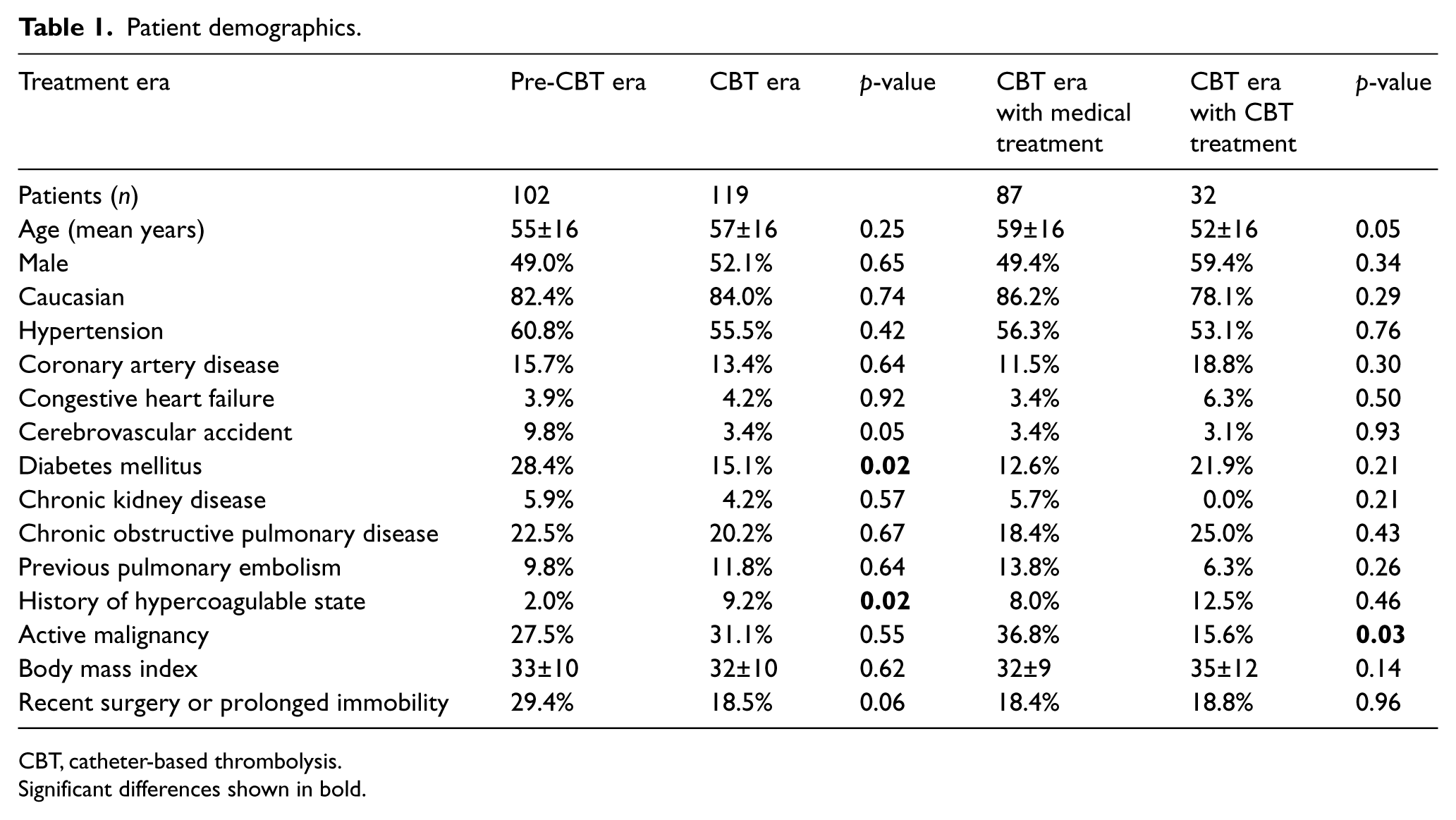
CBT, catheter-based thrombolysis.
Significant differences shown in bold.
Table 2 describes clinical characteristics at the time of presentation, displayed by era and by treatment strategy in the CBT era. Overall, the majority of patients were tachycardic (mean of 101 beats/minute) with low rates of shock: 3% overall (4% of patients in the pre-CBT compared to 2% in the CBT era, p=NS) with minimal differences between the groups. The most common enrollment criterion for submassive PE was RV/LV ratio>0.9, with average RV/LV ratios being 1.1–1.2. A substantial percentage of patients had evidence of RV dysfunction on echo and/or myocardial necrosis, with no significant differences between the groups. Notably, there were high rates of centrally located PE in the population, with significantly more in the CBT era population (66.4% versus 48.0%, p<0.01); no significant differences were noted between those in the CBT era stratified by treatment (medical therapy versus CBT).
Clinical characteristics pre-treatment.
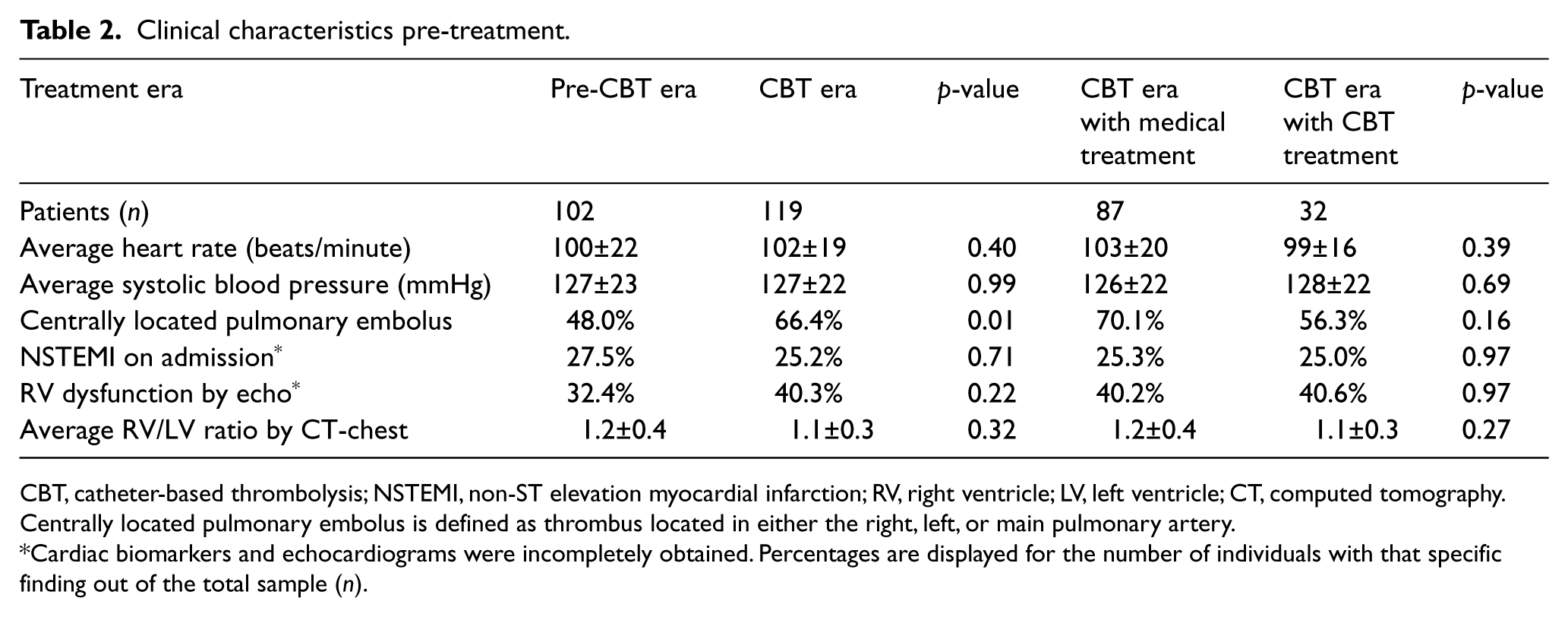
CBT, catheter-based thrombolysis; NSTEMI, non-ST elevation myocardial infarction; RV, right ventricle; LV, left ventricle; CT, computed tomography.
Centrally located pulmonary embolus is defined as thrombus located in either the right, left, or main pulmonary artery.
Cardiac biomarkers and echocardiograms were incompletely obtained. Percentages are displayed for the number of individuals with that specific finding out of the total sample (n).
In those receiving CBT therapy, the procedure was successfully completed in 100% of cases, without complications during catheter placement or infusion. Overall, the composite endpoint of death, recurrent PE, major bleeding, or stroke (MACE), occurred numerically, but not statically, more frequently in the CBT era than the pre-CBT era (21.0% versus 14.7%, p=0.23). Severe bleeding occurred six times in each era with intracranial bleeding occurring only once in each cohort. Minor access site bleeding occurred in one patient late after CBT. Patients in the CBT era were more likely to require ICU stay (40.3% versus 23.5%, p<0.01) or to require intubation (34.5% versus 13.7%, p<0.001) than those in the pre-CBT era. In addition, there were numerically higher rates of death and recurrent PE that were not statistically significant. Within the CBT era alone, the composite outcome was higher in those receiving CBT therapy versus medical therapy alone (23.0% versus 15.6%, p=0.38). The remaining secondary endpoints are seen in Table 3.
Outcomes.
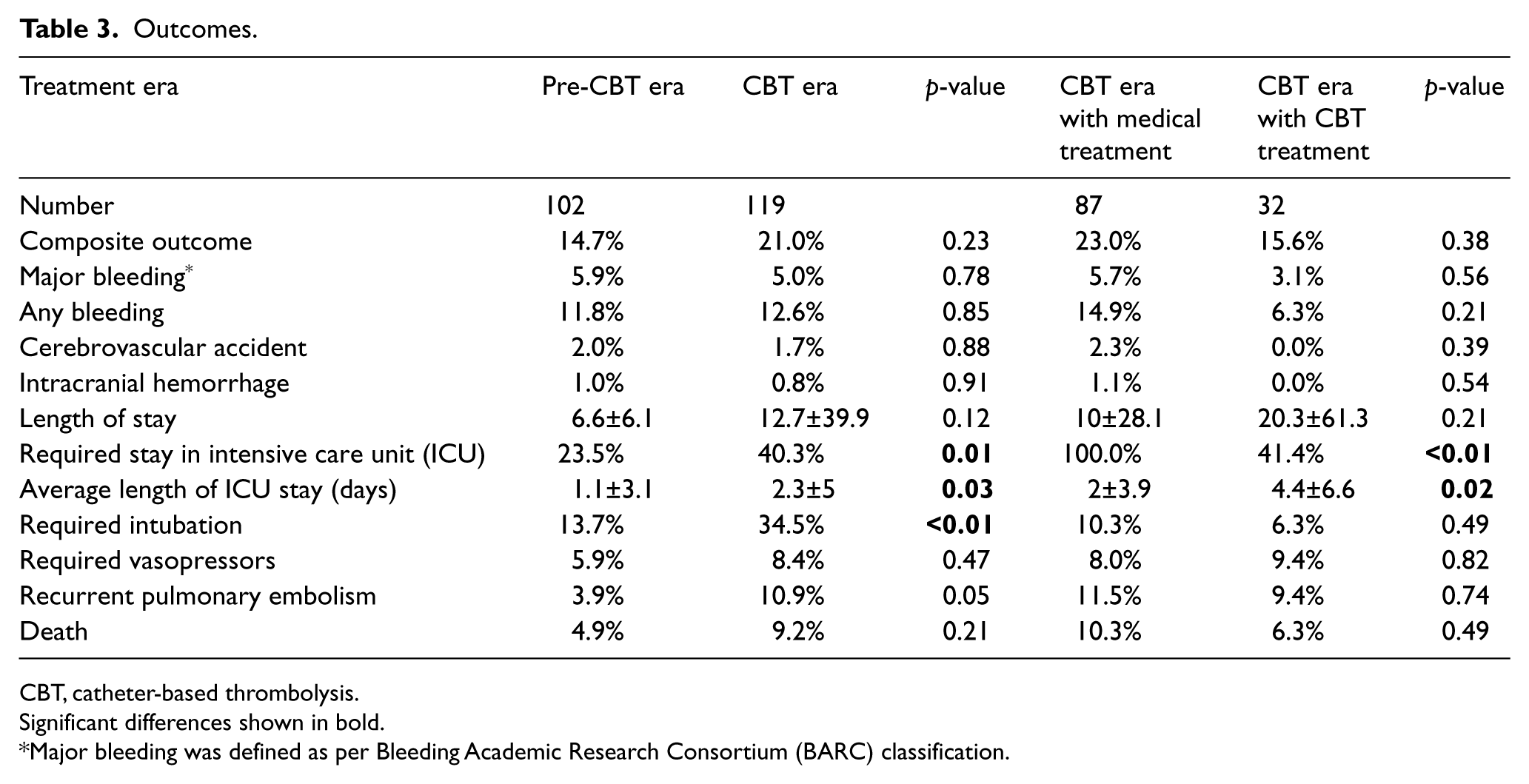
CBT, catheter-based thrombolysis.
Significant differences shown in bold.
Major bleeding was defined as per Bleeding Academic Research Consortium (BARC) classification.
Univariate predictors for major adverse events are listed in Table 4 with associated estimated odds ratio (OR). As shown, prior PE was the only significant predictor of our composite outcome amongst our total population with an OR of 7.4 (CI 9.0–18.3, p<0.001) and demonstrated an even stronger association within the CBT era alone (OR 23.8, CI 5.1–96.2, p<0.001). In addition, the RV/LV ratio also predicted the composite outcome (OR 0.34, CI 0.12–0.95, p=0.04). Overall, treatment era had no effect on outcome (OR 1.54, CI 0.76–3.11, p=0.23). In multivariable models, the only predictor of major in-hospital events, was prior PE (OR 8.87, CI 3.37–23.3, p<0.001). No treatment effect was seen between the two eras (OR 1.26, CI 0.56–2.80, p=0.58, see Table 5). Within the CBT era, CBT therapy did not demonstrate a treatment benefit (OR 0.84, 95% CI 0.22–3.22, p=0.80).
Univariate predictors for major adverse events.
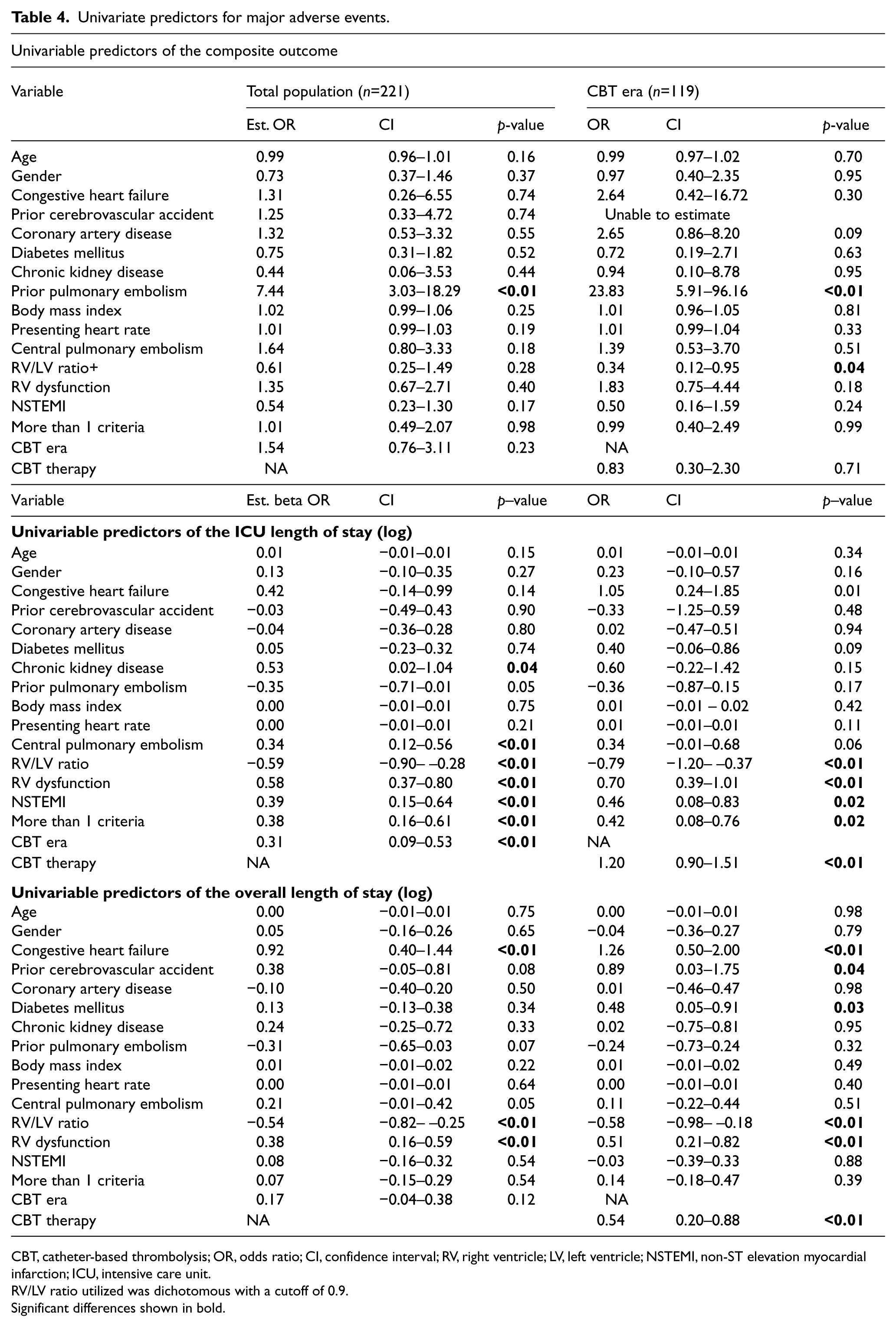
CBT, catheter-based thrombolysis; OR, odds ratio; CI, confidence interval; RV, right ventricle; LV, left ventricle; NSTEMI, non-ST elevation myocardial infarction; ICU, intensive care unit.
RV/LV ratio utilized was dichotomous with a cutoff of 0.9.
Significant differences shown in bold.
Multivariable predictors.
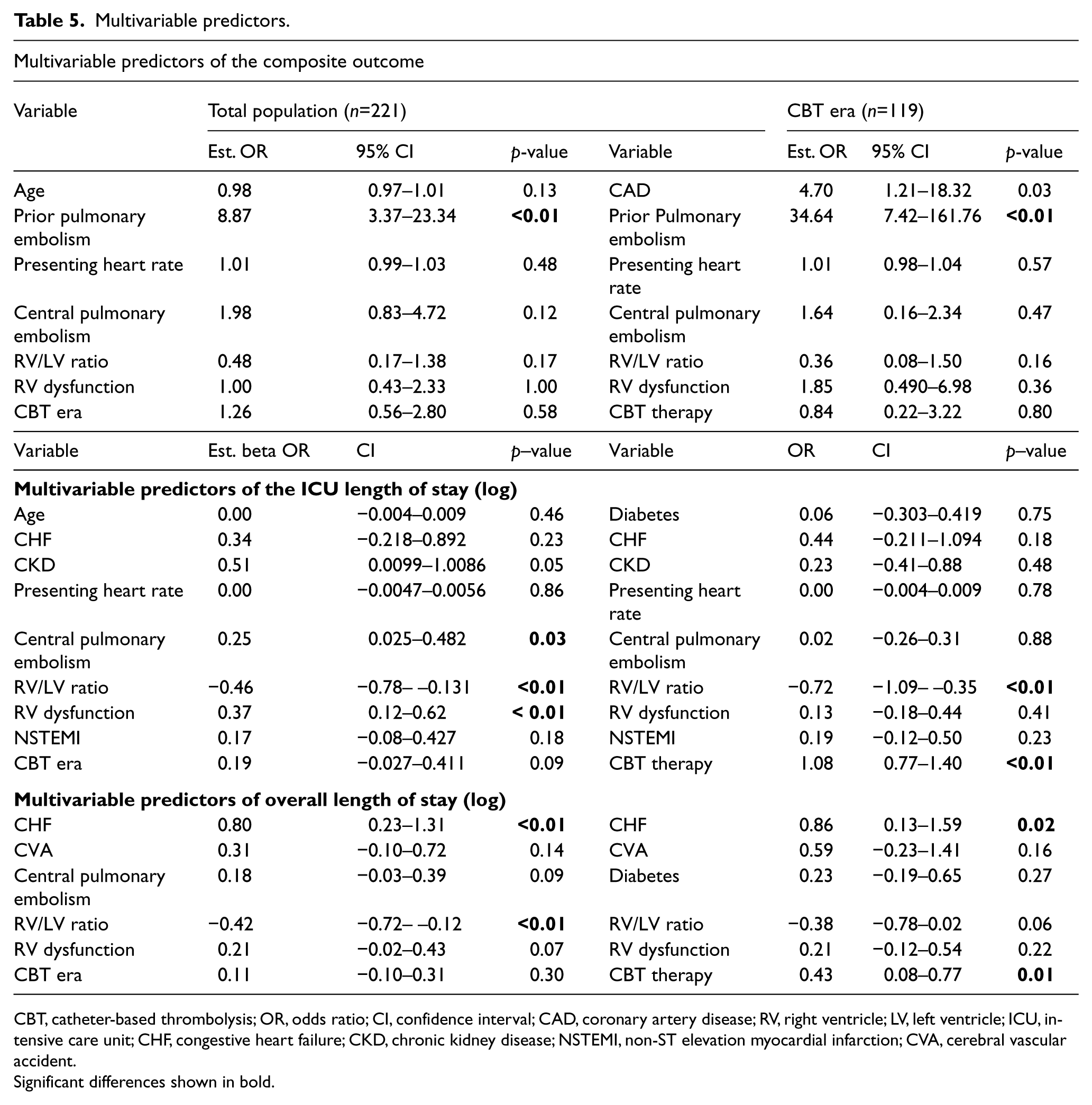
CBT, catheter-based thrombolysis; OR, odds ratio; CI, confidence interval; CAD, coronary artery disease; RV, right ventricle; LV, left ventricle; ICU, intensive care unit; CHF, congestive heart failure; CKD, chronic kidney disease; NSTEMI, non-ST elevation myocardial infarction; CVA, cerebral vascular accident.
Significant differences shown in bold.
The effect of individual characteristics on ICU length of stay (LOS) is displayed in Table 4. In the overall study population, chronic kidney disease, RV/LV ratio, evidence of RV dysfunction, evidence of myonecrosis, and number of submassive criteria were all significantly associated with ICU length of stay. Patients in the CBT era spent on average 37% more time in the ICU than patients did prior to the introduction of CBT therapy. Within the CBT era alone, those receiving CBT therapy spent, on average, over 200% more time in the ICU. The (offset) ICU length of stay was therefore increased, on average, by an estimated 190% (95% CI, 116% to 305%, p<0.001) with CBT therapy in the CBT era, after adjusting for other model variables. The remaining multivariable predictors are shown in logarithmic scale in Table 5.
Analysis of the overall length of stay demonstrated that congestive heart failure, increasing RV/LV ratios, and evidence of RV dysfunction was associated in univariable modes with increased LOS, while the era was not. After adjusting for model variables, only CHF and RV/LV ratios remained significant. Within the CBT era, CBT therapy was independently associated with prolonged hospitalization, requiring on average 53.7% (95% CI, 8.3% to 115.9%, p<0.001) more time in the hospital than for standard medical therapy, after adjustment for medical co-morbidities. Table 5 displays the remaining multivariable results.
Discussion
Patients with acute PE span a wide spectrum of clinical outcomes. Risk stratification of this population is important and can impact treatment decisions. Patients are considered low risk if they are hemodynamically stable, are without evidence of RV dysfunction, and have normal cardiac biomarkers. The mortality rates for such low-risk patients receiving standard therapy is around 3%. 18 However, over a third of normotensive patients with acute PE may have evidence of RV dysfunction or myocardial necrosis, placing them at higher risk for in-hospital death or clinical deterioration with mortality rates as high as 40%.19,20
To date, the cornerstone of therapy for PE remains anticoagulation alone, even in patients with submassive PE. Thrombolytic therapy is a treatment option for patients with high-risk features, but patients with submassive PE who would benefit from systemic thrombolysis are poorly described.15,21 Compared with anticoagulation therapy alone, systemic thrombolysis has not been demonstrated to reduce mortality or recurrence of PE. In fact, data supports the notion that major bleeding is increased, including intracranial hemorrhage, thereby offsetting any potential benefit. 22 To date, this data has limited the expansion of systemic thrombolysis therapy beyond the highest risk patients. 23 By instituting lower doses and targeting the thrombolysis therapy to the pulmonary bed, CBT has the potential to offset the previously noted bleeding complications and provide clinical benefit. Of note, the efficacy of low-dose thrombolysis, which exhibits similar efficacy as higher-dose thrombolytics, has not been directly compared to a CBT strategy. 24
One key consideration in the adoption of new technologies is demonstration of their overall safety and ease of adoption. In general, placement of pulmonary infusion catheters is not technically challenging. Several recent trials have demonstrated high procedural success rates and appear to be associated with low rates of adverse events (ULTIMA and SEATTLEII).13,25 Similar to these early reports, we demonstrated high procedural success rates (100%) without any immediate complications. In our cohort, we evaluated in-hospital safety outcomes including major bleeding, and no differences were noted between the two treatment eras. A numerically higher, but not statistically significant, difference in bleeding was noted in those receiving CBT therapy in the CBT era versus anticoagulation alone (5.7% versus 3.1%).
The overall effectiveness of CBT therapy is still not well understood with early data demonstrating improvement surrogate outcomes such as RV function. In the ULTIMA trial, intermediate-risk patients with PE, randomized to receive ultrasound-accelerated thrombolysis, had significant improvement of RV dilation with RV/LV ratios improving by 0.30 versus 0.03 (p<0.001) when treated with heparin alone within the first 48 hours. Improvement was also seen in RV function, and estimated pulmonary artery pressures. 13 Reassuringly, these results were durable, sustaining these improved RV/LV ratios at 90 days. However, a catch up was noted in those receiving heparin alone (RV/LV improvement from initial diagnosis 0.35 versus 0.24 in those treated with CBT versus heparin respectively, p=0.07). Furthermore, the SEATTLEII trial, which was a single-arm study evaluating the effectiveness of CBT therapy, also showed a RV/LV ratio improvement of 0.42 at 48 hours. However, no long term data is available at this time. 25 In our study, follow-up echocardiogram and CT scan rates were inadequate for formal analysis of the RV function.
Despite the emerging data focused on surrogate outcomes, there are no studies evaluating whether initiation of CBT for intermediate- to high-risk PE can improve patient clinical outcomes. In our study, we selected a population that consisted entirely of submassive or massive PE patients and evaluated the outcomes of these patients in an era when advanced treatments with catheter-based ultrasound therapies were available. Patients with submassive PE represented 42.1% of our institutional acute PE population. In our sample, we did not demonstrate a significant short-term improvement in our composite outcome when CBT was introduced as a treatment modality for our high risk population. In fact, our composite outcome occurred numerically more frequently in those in the CBT era than those prior to CBT initiation. However, clinical differences existed between the two eras, notably the presence of centrally located PE, which could affect event rates. Therefore, in multivariable models, we adjusted for clinical markers of PE severity and known predictors of adverse events. After adjustment, the increased rates of adverse events were minimized and outcomes were nearly neutral (OR 1.25, p=0.58). To assess these findings further, a separate analysis was performed on those within the CBT era only, evaluating the effect of CBT compared to medical therapy alone. After adjusting for RV function, dilation, heart rate on presentation, and central PE, CBT therapy still was not associated with a difference in patient outcomes (OR 0.84, p=0.80).
Clinical benefits beyond major adverse events may also be seen with CBT therapy, including differences in overall LOS and ICU LOS. The effect of CBT therapy on LOS has not been compared in previous CBT studies. Despite a theoretical reduction in PE burden, and therefore quicker improvement in hemodynamics with potential shorter critical care time, ICU LOS was actually longer in the CBT group. In fact, on average, patients spent nearly triple the time in the ICU when receiving CBT therapy. This association remained significant after adjustment for baseline demographics and PE severity (OR 2.9, 95% CI, 0.16 to 4.05, p<0.001). There are significant limitations to this data, as ICU LOS was largely driven by our institutional CBT protocol. For example, patients receiving CBT therapy were routinely admitted to the ICU for post-procedural monitoring with central access in place while fibrinolysis was administered. While this only necessitated 12–24 hours, we can conclude, however, that any benefit on ICU LOS was far less than the time necessary to complete the CBT therapy. Given the inherent limitations of an ICU LOS endpoint, we further evaluated for any potential benefits on overall LOS. Similarly, CBT therapy increased our hospital LOS on average by over 50%, even after adjustment for other patient variables.
Our study has several limitations worth discussing. Firstly, our study was conducted at a single center where there is no dedicated service for PE management. Therefore, not all patients in the CBT era were considered for CBT therapy or selected in a consistent manner across the institution. Secondly, confounders will always exist in patient treatment decisions which cannot be adjusted for retrospectively despite our best attempts to collect all data pertinent to PE severity. This specifically includes the subtle clinical differences that may exist between patients with high-risk submassive or massive PE in whom full-dose thrombolytics may have prohibitively excess risk. We attempted to mitigate this bias by evaluating patient outcomes both during and prior to our institutional availability of CBT therapy. Thirdly, our sample had a relatively small proportion of patients undergoing CBT therapy. This limited our ability to assess subgroups of patients in whom CBT may potentially improve in-hospital outcomes. However, the study still remains the first to analyze this unique group by clinically-driven outcomes. Lastly, long-term clinical outcomes were not assessed in this analysis. It is possible that the benefits of CBT therapy extend beyond the immediate hospitalization.
Going forward, this unique population warrants further investigation into new technologies to help improve both acute and long-term outcomes. CBT therapy is one such direction that needs to be investigated further, but the rather rapid adoption of such innovative therapy is concerning given the lack of clinical outcomes data. In fact, a large clinical study evaluating the effect of CBT therapy in deep venous thrombosis confirmed our findings, demonstrating no improvement in major outcomes with increased rates of adverse events and LOS. 26 Together, these observational findings highlight the need for a large trial focused on clinical outcomes, not just surrogate endpoints, to move the approach of acute PE forward with the help of emerging technologies.
Conclusion
Our study found that the introduction of catheter based therapy for acute submassive and massive PE at our institution was safe, but did not demonstrate appreciable improvements in in-hospital outcomes. In addition, CBT therapy was associated with increased ICU and overall length of stay.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
