Abstract
Acute and chronic venous disorders of the lower extremities affect millions of people and cause substantial disability. Long ago, surgeons and pathologists identified the presence of ‘spur-like’ abnormalities of the left common iliac vein; these abnormalities were hypothesized to result from compression and/or irritation from the adjacent crossing right common iliac artery. In the 1990s, physicians, starting to perform catheter-directed thrombolysis to treat extensive deep vein thrombosis (DVT), observed that about 50% of patients had an iliac vein stenosis. Vascular physicians have become aware of the occasional patient with otherwise-unexplained extremity swelling and/or pain but without a DVT history who is subsequently found to have an iliac vein abnormality. These ‘lesions’ have been hypothesized to elevate ambulatory venous pressures and thereby produce lower-extremity symptoms, increase the risk of initial and recurrent DVT episodes, and increase the risk of treatment failure with medical and endovascular therapies for thrombotic and non-thrombotic venous conditions. As a result, many practitioners now actively seek iliac venous obstructive ‘lesions’ when evaluating patients with known or suspected venous disease. However, for many patients, it continues to be unclear what degree of obstruction to venous blood flow is being caused by such lesions, how clinically significant they are, how much emphasis should be placed on identifying them, and when they should be treated. This article seeks to improve the knowledge base of vascular practitioners who make clinical decisions about the diagnosis and treatment of obstructive iliac vein lesions.
Introduction
Acute and chronic venous disorders of the lower extremities affect millions of people and cause substantial disability. Long ago, surgeons and pathologists identified the presence of ‘spur-like’ abnormalities of the left common iliac vein; known as Cockett’s deformity or iliac vein compression syndrome, these abnormalities were hypothesized to result from compression and/or irritation from the adjacent crossing right common iliac artery.1,2 In the 1990s, physicians starting to perform catheter-directed thrombolysis (CDT) to treat extensive deep vein thrombosis (DVT) observed that about 50% of patients had an iliac vein stenosis, heightening awareness of a previously suggested association with the occurrence of iliofemoral DVT (May–Thurner syndrome).2–4 Vascular physicians have become aware of the occasional patient with otherwise-unexplained extremity swelling and/or pain but without a DVT history who is subsequently found to have an iliac vein abnormality. These ‘lesions’ have been hypothesized to elevate ambulatory venous pressures and thereby produce lower-extremity symptoms, increase the risk of initial and recurrent DVT episodes, and increase the risk of treatment failure with medical and endovascular therapies for thrombotic and non-thrombotic venous conditions.
As a result, prompted also by advances in venous imaging and catheter-based treatment, many practitioners now actively seek iliac venous obstructive ‘lesions’ when evaluating patients with known or suspected venous disease. 5 However, for many patients, it continues to be unclear what degree of obstruction to venous blood flow is being caused by such lesions, how clinically significant they are, how much emphasis should be placed on identifying them (given that the etiology of venous disease is often multi-factorial), and when they should be treated. This article seeks to improve the knowledge base of vascular practitioners who make clinical decisions about the diagnosis and treatment of obstructive iliac vein lesions.
History, physical examination, and duplex ultrasound
When a patient with venous disease is encountered, a careful medical history should be obtained. The physician should consider whether the current consultation is during the up-front care (i.e. initial encounter) of an acute DVT, whether it has been prompted by continued or worsening symptoms of DVT despite use of anticoagulant therapy, or whether this is a patient with chronic venous disease not known to be related to thrombosis. The physician should query: (i) the type and location of symptoms, including whether they involve the calf, thigh, and/or pelvic regions; (ii) the anatomic distribution of any current DVT (if present); (iii) the number, dates, and involved limb for previous DVT episodes (if any), whether they occurred in association with reversible risk factors versus being unprovoked, and whether there are ongoing risk factors; and (iv) the type of therapy that was utilized, and the effect of therapy upon the course of symptoms.
The physical examination should include inspection of both lower extremities; the groins, buttock, and perineal regions; and the pelvis and lower abdomen. The location of superficial varicosities should be noted, both in the extremities as well as along the abdomen and lower body wall. In the extremities, specific attention should be paid to evaluating the presence, degree, and distribution of edema and any skin changes such as hyperpigmentation or lipodermatosclerosis. The presence, number, location, size, and status (i.e. open or healed) of any ulcer should be noted, and the patient should be asked how long the ulcer has been present.
In general, the physical examination should be routinely supplemented with duplex ultrasound of the lower-extremity veins. The extent of thrombus within the proximal veins should be clarified and compared with previous assessments, and the phasicity of the common femoral vein (CFV) spectral waveforms should be examined – if dampened on the symptomatic side to a greater degree than the contralateral CFV (especially if monophasic), heightened suspicion should be present. 6 For patients seeking an explanation for chronic lower-extremity symptoms, the superficial venous system should be assessed for valvular reflux with the extremity in a dependent position, per accepted testing standards. 7 The standing position may be preferred due to the increased reproducibility of measurements obtained in this position. 8
Overall, left-sided clinical findings are presumed to be more likely to be associated with an obstructive iliac vein lesion because these lesions appear to be more frequent on the left side. However, such lesions have also been reported near the confluence of both internal and external iliac veins (believed to result from compression by the crossing external iliac artery) – so right-sided findings can also indicate a pelvic source of venous obstruction. 9
To summarize, the following findings should prompt a higher level of suspicion for the presence of an obstructing iliac vein lesion: (i) known current or prior DVT location in the iliac and/or common femoral vein; (ii) pain or swelling of the entire limb (thigh and calf) as a feature of previous DVT, current daily symptoms (especially if unilateral or unexplained), or the physical examination; (iii) a history of ipsilateral DVT recurrence; (iv) presence of visible varicosities over the lower body wall, groin, or thigh; (v) dominance of venous claudication, as suggested by prior investigators 10 ; or (vi) on ultrasound, a CFV that is incompletely compressible or for which the Doppler waveform lacks phasicity compared with the contralateral CFV.
Advanced non-invasive diagnostic testing
A number of physiological tests and imaging modalities have been used to evaluate patients for obstruction of the iliac vein outflow tract. Trans-abdominal duplex ultrasound is often unable to visualize the iliac vein with sufficient diagnostic accuracy due to overlying bowel gas and the vein’s depth within the pelvis.
Air plethysmography, quantified by the venous outflow fraction (VOF), can effectively diagnose outflow obstruction, but a normal exam does not necessarily exclude significant disease.11–15 An arm/foot venous pressure differential of ≥ 4 mmHg and/or a reactive hyperemia-induced pressure increase of ≥ 8 mmHg have also been considered to represent hemodynamic criteria for venous obstruction.12,13 However, these tests are difficult to interpret when pressure differentials are normal or near-normal in symptomatic patients; neither criterion has demonstrated the ability to reliably exclude iliac vein obstruction.12,13 In modern clinical practice, these examinations are rarely performed.
Cross-sectional imaging is employed by some physicians to evaluate for iliac vein pathology. Computed tomographic (CT) venography can clearly identify the more pronounced stenotic lesions, and has the advantage of being able to identify discrete causes of extrinsic venous compression (e.g. pelvic tumors) that may be contributing to symptoms (Table 1). A core challenge faced by CT venography is the difficulty of timing the imaging sequence to optimal contrast opacification of the target vein. This problem, which is a universal technical consideration for CT studies in the vascular system, can be particularly vexing in the iliac venous system since both innate patient characteristics (e.g. cardiac output) and the presence of varying degrees of venous obstruction can impact contrast transit time to a variable degree. Hence, while lesions can be diagnosed, many CT venograms are likely to have a non-diagnostic result.
Causes of iliac vein obstruction.

One small retrospective study in a selected population of women < 45 years of age found the presence of decreasing iliac vein diameters on CT venography to correlate with a greater likelihood of experiencing an ipsilateral DVT. 16 In another study, 34 patients with diagnosed May–Thurner syndrome (Figure 1) were compared with 34 age- and sex-matched asymptomatic patients. The degree of left common iliac vein narrowing observed on CT venography was 45–100% (mean 74% ± 17%) in the May–Thurner syndrome patients versus 0–68% (mean 28% ± 20%) in the asymptomatic controls (p<0.001). 17 This study appropriately highlights the fact that there is a great deal of overlap in findings between normal and abnormal patients. Overall, we do not recommend CT venography for routine use due to its known limitations and the fact that it has not undergone rigorous, prospective validation studies for its ability to identify or exclude obstructive iliac vein lesions.
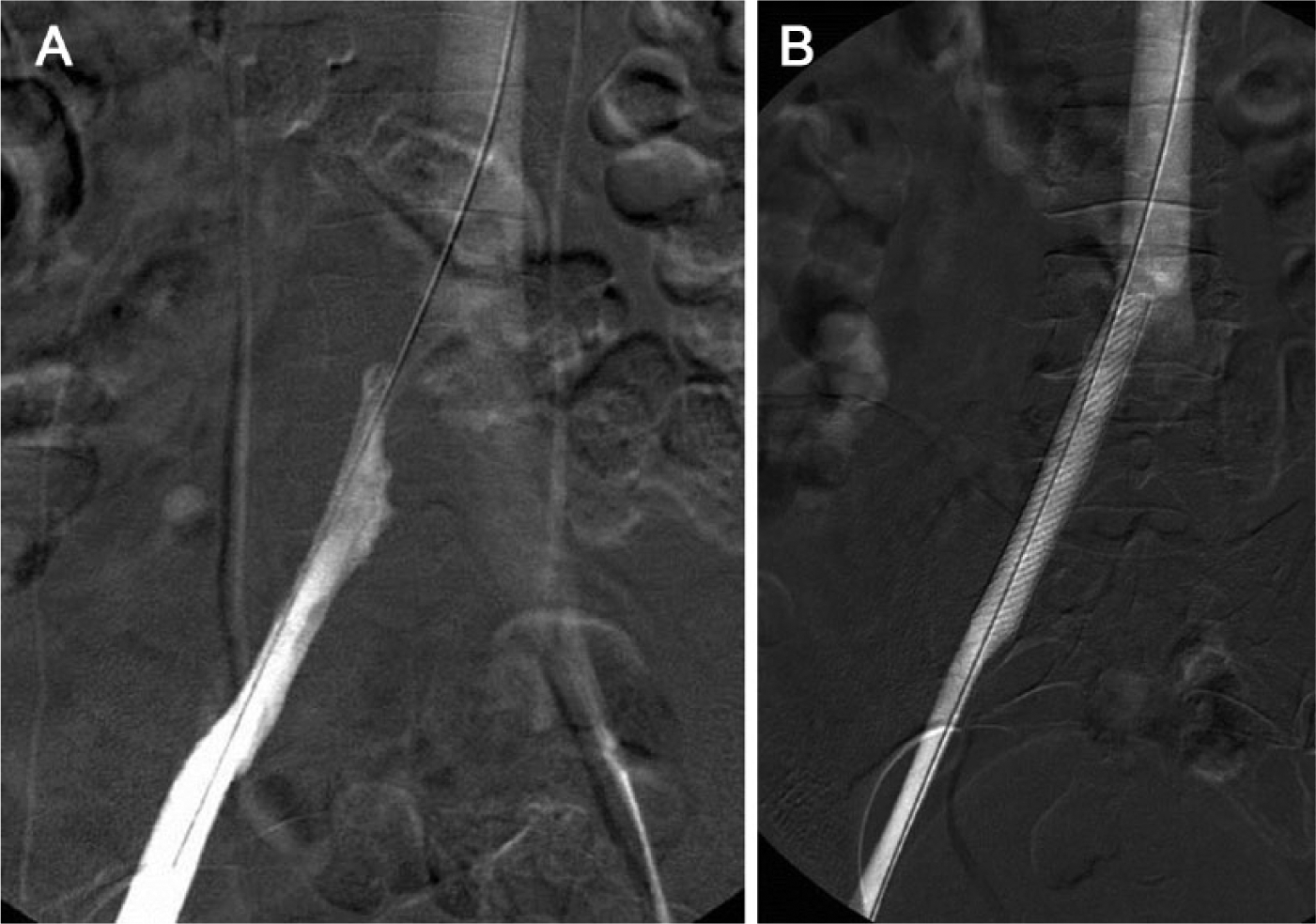
Classic May–Thurner syndrome in a 30-year-old woman with a left iliofemoral DVT who has undergone catheter-directed thrombolysis. This catheter venogram shows tight stenosis of the left common iliac vein (patient is prone) before
Magnetic resonance venography (MRV) has shown the potential to overcome the contrast timing limitation of CT scanning, since newer magnetic resonance imaging (MRI) contrast media persist in the vascular system for a longer period of time, increasing the likelihood that the vein will be opacified during imaging. However, MRV cannot be performed in patients with metallic implants, and it also has not undergone prospective validation for detection of iliac vein lesions. One small MRV study suggests that iliac vein diameter measurements that are obtained in the same patient several months apart exhibit broad variance (mean difference exceeding 20% diameter narrowing). 18 Perhaps more importantly, the resolution of both MRV and CT scanning for subtle lesions (e.g. webs) is limited. Hence, available cross-sectional imaging methods can miss venous pathology.
Transpedal venography can also be used to assess the luminal contours of the iliac venous system. However, except in cases of complete or near-complete iliac vein obstruction, its technical limitations are considerable. Even with optimal use of a tourniquet system, long experience, and a robust volume of contrast injected, the contrast is prone to being washed out by non-opacified blood from other inflow veins, greatly reducing the exam’s diagnostic capability.
In summary, an obstructive iliac vein lesion cannot be excluded with non-invasive assessments. However, it is important to realize that a 100% negative predictive value is not necessary to have for all presenting patients. In our practice, we tend to employ MRV for a small subset of patients with mild–moderate disease in whom the level of suspicion for a lesion is low but not zero (e.g. CEAP Clinical Classes 0–3, or patients with a history of right-sided DVT limited to the femoropopliteal veins), since in these patients (i) the pre-test probability of finding a lesion is low; (ii) the risk of harm from missing a lesion is low; and (iii) the benefit-to-risk ratio for catheter-based treatment of a subtle identified lesion (e.g. with implantation of a permanent stent) might be borderline anyway.
On the other hand, if the level of clinical suspicion for an iliac vein lesion is medium-to-high or if the patient has advanced (e.g. CEAP Clinical Class 4–6, or CEAP Class 3 with massive edema) manifestations of venous disease, then invasive testing (see below) is performed since (i) it enables catheter-based treatment to be performed; (ii) robust clinical suspicion would continue even with a negative MRV or CT venogram; and (iii) the clinical consequences of missing a lesion could be very significant (e.g. non-healing of an ulcer or progression of functional disability).
For any diagnostic and treatment plan, we encourage a transparent discussion of the expected risks, benefits, and costs with the patient prior to implementation.
Invasive testing
Catheter venography, sometimes supplemented by direct pressure measurements, has often been considered the most ‘definitive’ available imaging test for evaluation of obstructive venous pathology. In addition to depicting the contrast-outlined contours of the vein, venography enables real-time visual estimation of the pace of blood flow and visualization of peri-venous or other pelvic collateral veins that support the hemodynamic significance of a lesion. Although single-plane venography in the anteroposterior (AP) projection is routinely performed, this method suffers from the limitations of attempting to view a three-dimensional structure in two dimensions. This is especially important since external compression of the iliac vein, as seen in May–Thurner syndrome, often occurs in the AP plane which results in a ‘pancaked’ iliac vein that does not exhibit diameter narrowing viewable in the AP projection.
It can be quite challenging to determine the hemodynamic significance of a venous stenosis. In contrast to the arterial system, in which a hemodynamically significant stenosis does not occur until there is stenosis greater than 70–80% of the area of the vessel lumen, 19 a specific threshold has not been validated in the venous system. The compliance of veins may make a vessel appear stenotic in one dimension when there is no decrease in the actual lumen surface area compared to more rounded segments of the vessel. Factors such as the volume status of a patient can play a role in the degree of distension or collapse. No study has identified a clear threshold for what degree of stenosis will cause a patient to experience symptoms. Some authors have suggested that symptom relief is more likely with stent placement with stenosis of greater than 25% 20 or 50%.21,22 However, these values are empirically derived; none of these thresholds have been prospectively validated in methodologically rigorous studies.
Venous catheter placement affords the ability to directly measure venous pressures above and below a suspected lesion. Typically one would either insert a 5 French or larger catheter beyond the lesion and transduce a pressure reading during pullback across the lesion (ideally over a 0.018 inch guidewire to maintain access across the lesion), or for best temporal accuracy one can simultaneously transduce above and below the lesion with co-axial use of a larger (e.g. 8–10 French) guiding catheter or sheath. It has been noted that increased pressure peripheral to a lesion with augmentation of venous inflow may indicate a significant focal stenosis.22,23 Available studies suggest that an intraluminal pressure gradient across a perceived stenosis that is greater than 2–3 mmHg is likely to be hemodynamically significant.21,24 However, pressure measurements performed with the patient supine, a position in which most patients are asymptomatic, cannot exclude a hemodynamically significant lesion. Hence, we consider pressure gradients less than 4 mmHg to be non-diagnostic (not negative, not positive).
Intravascular ultrasound (IVUS) appears to have distinct advantages in evaluating subtle iliac vein pathology. Of greatest significance is the ability of IVUS, a catheter-mounted ultrasound probe that is placed directly within the vein being imaged, to bypass traditional ultrasound imaging barriers and permit high-resolution examination of the vein wall and internal architecture. 25 Preliminary studies suggest that IVUS is more sensitive than venography for the detection and qualitative characterization of iliac vein lesions. Specifically, studies comparing venography with IVUS suggest that many iliac vein lesions are either missed or underestimated in severity26–30 (Figure 2). In one study, approximately 25% of limbs determined to be ‘normal’ on single-plane venography were subsequently shown to have greater than 50% stenosis on IVUS. 31 In another study of 304 symptomatic limbs, the median degree of narrowing of iliac vein lesions visualized on venography was 50%, compared to 80% in the same vessels measured with IVUS. 26 The authors of this study noted that were IVUS to be considered the gold standard, the sensitivity of single-plane venography for stenosis of 70% or greater was only 45%, with a negative predictive value of 49%.
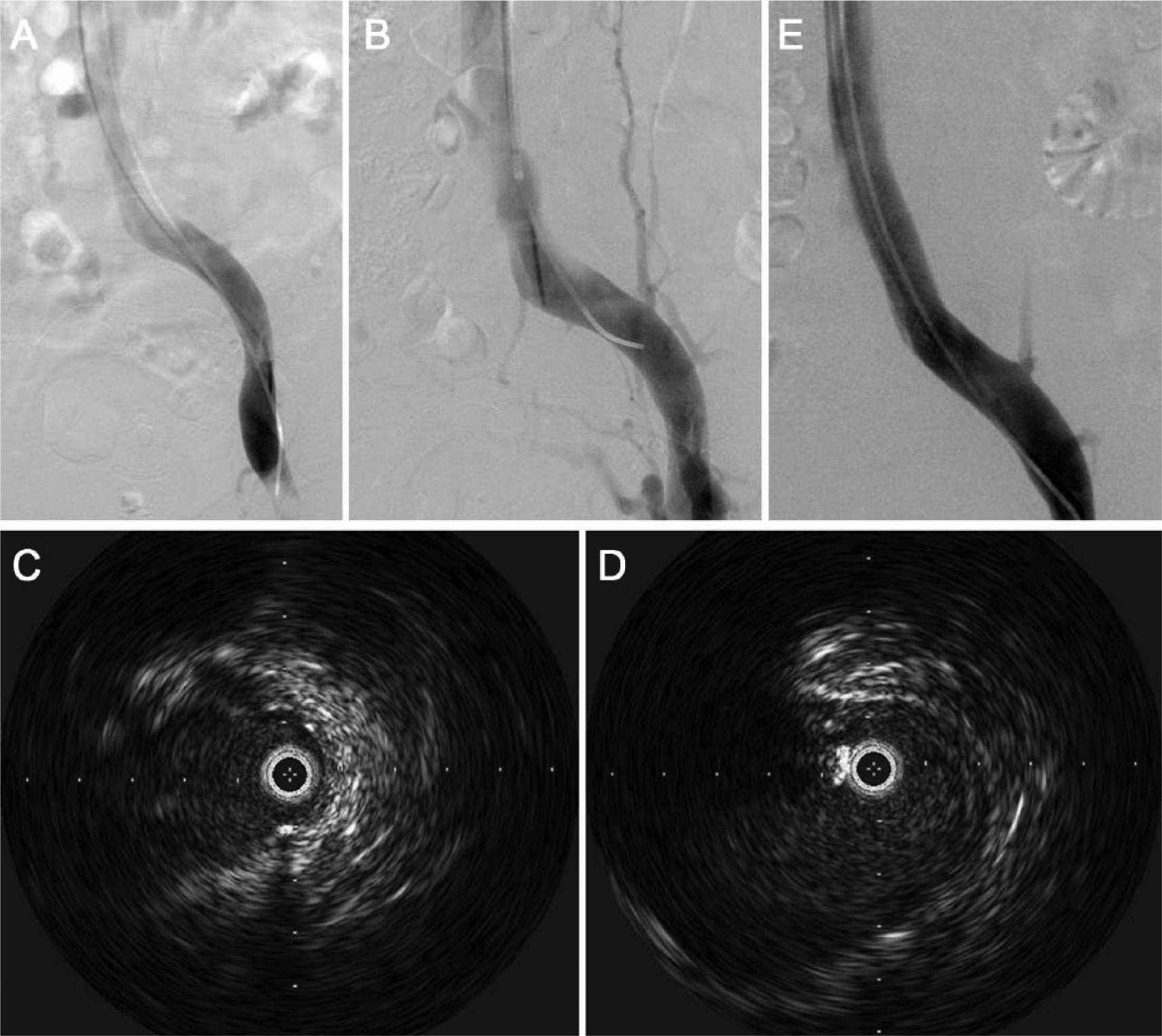
Complementary use of venography and IVUS to diagnose iliac vein stenosis in a 55-year-old man with a history of left lower-extremity DVT 5 years previously, with ongoing symptoms of left lower-extremity swelling and pain with even mild activity.
Proponents of IVUS further note the limitations of venography – it is poorly suited to evaluate subtle contour abnormalities that do not happen to be orthogonal to the imaging plane, as well as abnormalities of internal vein architecture (e.g. webs, septations, wall thickening) that may be partially permeable to contrast-containing blood (Figure 3). Structures adjacent to a vein, which may be relevant to the overall assessment of a lesion’s significance, such as a compressing right common iliac artery or peri-venous collaterals, may be better perceived with IVUS as well.20,26,28
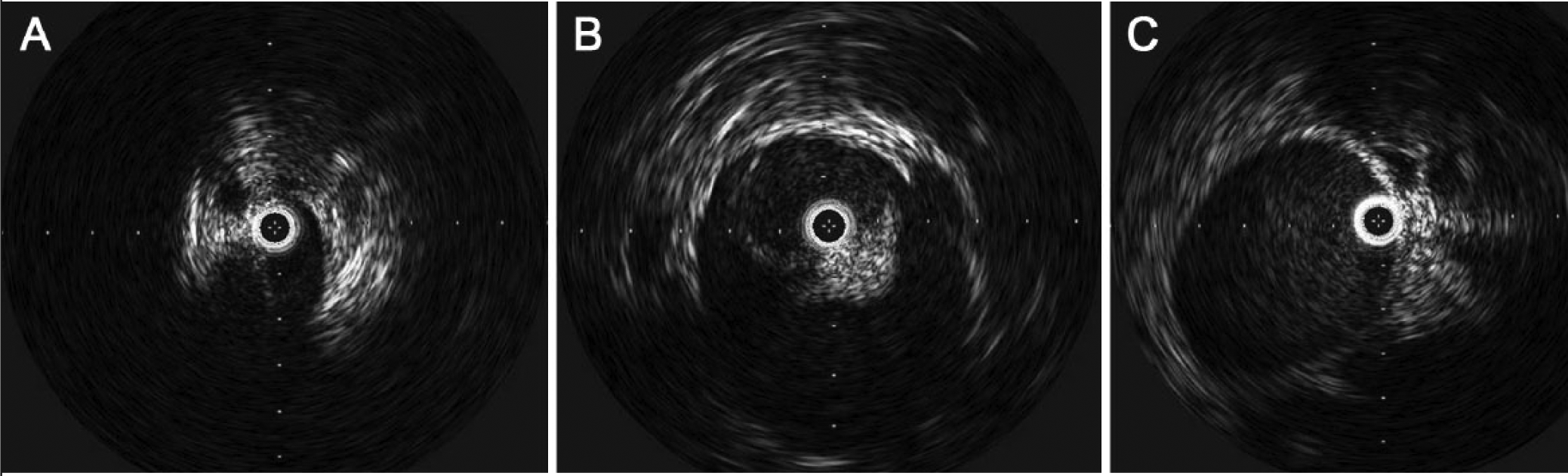
Non-stenotic venous abnormalities on IVUS may include:
However, IVUS skeptics cite the methodological limitations of the existing comparative studies that were not prospective investigations, the fact that they utilized single-plane venography as the comparator, the lack of data on the inter-observer reliability of IVUS measurements, and the fact that there is no gold standard measure to judge what truly constitutes a ‘significant’ stenosis in such studies. Comparisons are even more complex for lesions that do not constitute typical stenosis. Since vessels can be deformed with scarring or external compression from other structures and may extend longitudinally along the vein, simple measurements of diameter or area may not accurately estimate the degree of vessel compromise. IVUS can clearly identify intraluminal trabeculations and webs that can be occult on venography. 26 Circumferential or eccentric vein wall thickening or echogenicity, which may relate to chronic inflammation due to chronic compression from an adjacent structure, may also be better appreciated with IVUS. It should also be noted that residual intraluminal thrombus is a common ‘obstructive lesion’ in patients who have undergone thrombolysis for DVT: in one study, IVUS identified residual thrombus in 36% of patients who were felt to have normal post-thrombolysis venograms. 31
On the other hand, it is very important to note that the true clinical significance of many of the abnormalities that have been described on IVUS (especially those that are entirely occult on venography) has not been established. While lesions that clearly cause severe persistent narrowing or deform the internal architecture of the vein are likely to be significant, lesser degrees of stenosis may or may not account for an individual patient’s symptoms, especially when the structure of the vein wall and lumen appear otherwise normal.
We have observed a broad range of IVUS findings and a variable degree of concordance with venography. In reviewing the first 34 limbs in our practice for which IVUS and venography were performed to evaluate chronic symptoms thought likely to represent venous disease, we found 21 patients (61%) to have an abnormality on IVUS, of whom one-third had no abnormality seen on venography. Although the most common IVUS abnormality seen was venous narrowing, 14 patients (which is 67% of the abnormal IVUS exams and 41% of all limbs examined) had non-stenotic lesions including webs, pleats, wall thickening, and/or increased wall echogenicity, with multiple abnormalities seen in many patients. It should be noted that even for venous stenosis, there was a wide range of findings in terms of the degree of narrowing, the degree to which the narrowing varied with respirations, and the degree of associated vein wall and luminal abnormalities. Importantly, for some patients, the sole finding was narrowing of between 25% and 50% of the area of an otherwise normal-appearing vein, causing a significant dilemma in terms of whether or not endovascular treatment should be pursued.
For these reasons, as we have gained experience with IVUS over the years, we have come to the following conclusions.
It is important to mitigate the limitations of catheter venography by imaging the vein in multiple projections.
Even multiplanar catheter venography is limited in its ability to identify iliac vein lesions.
IVUS clearly identifies more pathology than venography, but also lacks defined criteria to adjudicate what findings are truly of clinical significance.
Therefore, in symptomatic patients who undergo venography ± IVUS, the possibility of seeing ‘indeterminate’ findings should be discussed
Treatment
The treatment of obstructive iliac vein lesions should be undertaken only after consideration of many factors, including the clinical context in which the lesion is discovered, the likelihood that it explains the patient’s symptoms and signs, the level of clinical severity of venous disease, and the patient’s life-expectancy, co-morbidities, and personal preferences. Below we outline three common scenarios in which treatment can be contemplated, and summarize medical, surgical, and endovascular treatment options.
A. Acute DVT associated with an iliac vein obstructive lesion
Although the mainstay of treatment for acute DVT is anticoagulant therapy, patients who remain severely symptomatic have been referred for thromboreductive therapies, previously surgical venous thrombectomy (which in modern practice tends to be limited to patients with thrombolytic contraindications in select centers with the needed surgical expertise), and in recent years CDT. When these interventions are performed, the finding of a left common iliac vein stenosis (May–Thurner syndrome), or occasionally a stenosis in another location, has prompted consideration for endovascular treatment for the purpose of aiding in alleviating initial DVT symptoms, preventing early post-procedure re-thrombosis, and preventing the post-thrombotic syndrome (PTS). While quality studies are limited, case series have reported short-term and long-term success.32–35 Although randomized controlled trials (RCTs) have not focused specifically upon the stent element of therapy, two retrospective studies of 106 patients with acute iliofemoral DVT have evaluated the use of stent placement for iliac vein obstructive lesions in patients who underwent concomitant surgical or endovascular venous thrombectomy/thrombolysis. In these studies, iliac vein stent placement was associated with low (12–14%) rates of postoperative early re-thrombosis. In contrast, in the larger study, a non-stented control group had a much higher rate (73%) of postoperative early re-thrombosis (p<0.01), suggesting that leaving stenotic lesions untreated after thrombus removal portends a poor immediate outcome.36,37
In a 473-patient multicenter prospective registry of DVT patients who underwent CDT, patients in whom stents were placed to treat an iliac vein stenosis or obstructive lesion had significantly greater 1-year venous patency than patients who did not receive a stent (73% versus 54%, p<0.001). 4 These were not identical patient cohorts (in that many of the non-stented patients presumably did not have a stenosis), significantly limiting the comparison, but at a minimum the stents did not appear to impair long-term venous patency.
Three RCTs have evaluated the use of CDT (incorporating venous stenting of iliac vein lesions) to treat patients with extensive DVT. All three RCTs found the CDT + stent + anticoagulation strategy to be associated with improved clinical outcomes in terms of prevention of PTS and/or valvular reflux, compared with anticoagulation alone.38–42 However, none of the studies included a control group in whom iliac vein lesions were not treated with stents; only one of these studies was a rigorously performed multicenter RCT, and even that study was limited by its moderate sample size (209 patients enrolled). 40 When it is completed, additional information may be gleaned from the much larger, ongoing, US-based, NIH-sponsored multicenter randomized ATTRACT Study, which is evaluating long-term outcomes of pharmacomechanical CDT (including stent placement for identified iliac vein lesions) for patients with proximal DVT. 43 However, at this stage, the consensus among endovascular practitioners is that failure to treat a known iliac vein obstructive lesion in a patient undergoing CDT for DVT would improperly put him/her at high risk for re-thrombosis, so a RCT specifically evaluating stent placement for this purpose is not likely to be conducted.
To date, no comparative studies have been performed to define the optimal type, dose, target range, or duration of anticoagulation therapy for use following endovascular DVT therapy. Current recommendations favor stratification of patients by the clinical scenario in which their DVT occurred, with at least 3 months of anticoagulation for patients with DVT associated with a major reversible risk factor, at least 6–12 months of anticoagulation for patients with unprovoked or recurrent DVT, and routine long-term use of low molecular weight heparins for patients with cancer-related DVT, irrespective of whether or not an iliac vein lesion is identified or treated.44–46 The concomitant use of anti-platelet therapy for at least several months (e.g. until the stents have endothelialized) in patients who have received stents is common, but the impact of this practice upon stent patency and bleeding complications has not been studied.5,22
Patients who undergo endovascular venous intervention (any scenario) are typically followed closely in clinic, most commonly at intervals of 1 month, 6 months, 12 months, and yearly thereafter unless their symptoms recur. Practices vary in terms of imaging follow-up: we do not recommend routine surveillance imaging for patients who are asymptomatic. If a patient has persistent or recurrent symptoms, duplex ultrasound is the first imaging test to be done.
B. Established post-thrombotic syndrome with iliac vein stenosis or occlusion
Surgical venous bypass has been occasionally used to provide symptom relief to patients with severe PTS in association with iliac vein obstruction, with widely varying results described in case series over a period of many years. 47 In modern practice, these procedures are performed with relative rarity, and predominantly by highly specialized venous surgeons. When venous bypass is performed, an arteriovenous fistula is created to enhance early patency. Overall, these techniques are invasive and involve a significant recovery period for patients, and have been associated with only modest 3-5-year patency rates. Surgical removal of complex intraluminal debris from the common femoral vein and its tributaries can also be used by some surgeons to improve inflow in conjunction with iliac vein stent placement. 48
Stent recanalization of chronically occluded iliac veins in patients with advanced PTS has been shown in multiple studies to reduce PTS symptoms, improve quality of life (QOL), and enable healing of venous ulcers.49–51 In the two largest studies, the initial reduction in lower-extremity pain and swelling occurred in greater than 95% of treated patients and was maintained at 3 years in 66–79% of patients.49,50 Scores on the Chronic Venous Insufficiency Questionnaire (CIVIQ), a validated venous disease-specific QOL measure, were significantly improved. Another large study (n=493) found that in patients with PTS, stent patency was good (84%), even when stents needed to be extended into the common femoral vein to treat venous inflow obstruction. 52 A number of additional studies comprising more than 200 patients have found concordant results, with about 80% of treated patients with moderate or severe PTS achieving major reduction in their dominant PTS symptom (pain or swelling) with stent recanalization of the iliac vein.53–58 These studies have demonstrated marked improvements in the clinical severity of chronic venous insufficiency on validated outcome measures (CEAP Classification, Venous Disability Scale, Venous Clinical Severity Score, Villalta PTS Scale, VEINES-QOL/Sym venous disease-specific QOL measure)46,47,50; reductions in lower-extremity pain and swelling45,46,48; healing of venous ulcers45,49; 1–3-year primary patencies ranging from 67% to 83%46,48; and 1–3-year secondary patencies ranging from 90% to 100%.48,49
The technique of stent placement can vary substantially per physician preferences. For patients with established PTS, typically ultrasound-guided access to the common femoral vein or internal jugular vein is used – the internal jugular vein has the advantage of permitting ready access into the profunda femoral vein which can be an important source of inflow. 5 A venogram and/or IVUS are performed, followed by guidewire recanalization of the obstructed segment – this is easy for stenosis and short-segment occlusions, but can be challenging for long-segment occlusions. Pre-dilatation with an angioplasty balloon is then performed, followed by stent implantation. Although no stent is FDA-approved for iliac vein implantation, typically self-expandable, longitudinally flexible stents of diameters from 12 mm to 18 mm are used. 50
Anti-thrombotic therapy is of particular importance in patients with established PTS who undergo stent placement due to the inherently pro-thrombotic nature of this population. These patients are at risk for early re-thrombosis (especially if the recanalization procedure was long or particularly traumatic), as well as late loss of patency from stenosis or thrombosis. The limited available data suggest that 40% of patients receiving stents for the treatment of established PTS will require an additional procedure to restore stent patency within 4 years.22,50 This argues in favor of a much more pro-active approach to the use of anticoagulation and anti-platelet therapy post-procedure, provided the patient’s expected risk of bleeding is low. There are no studies of anti-thrombotic strategies with which to guide practice in this area, so practices vary widely. For 1–3 months post-procedure, many operators prefer low molecular weight heparin over warfarin due to the more consistent level of anticoagulation provided by the former, though this can be expensive and poorly tolerated by some patients. Most endovascular physicians routinely employ concomitant anti-platelet therapy for these patients, often with a more potent agent such as clopidogrel. The institution or re-institution of aggressive anti-thrombotic therapy in this group of patients tends to be well-accepted since they have suffered major QOL impairment from their PTS and strongly wish to maintain their post-stenting clinical status. The ability to achieve sustained patency, major symptom reduction, and improvement in function and QOL in most treated patients is immensely valuable to patients suffering from PTS.
C. Symptomatic non-thrombotic chronic venous disease with iliac vein lesion
Stent placement may also be selectively employed to reduce venous hypertension and symptom severity in patients with established chronic venous disease of moderate or severe clinical intensity. Studies have shown that iliac vein obstructive lesions may serve as an undiagnosed source of venous hypertension in patients with diverse clinical phenotypes ranging from patients with venous ulcers, to patients with limb pain/swelling and concomitant saphenous vein reflux, to those who have limb pain/swelling that is otherwise entirely unexplained. For patients with severe clinical manifestations (i.e. CEAP Clinical Classes 4–6, or CEAP Clinical Class 3 with massive edema or pain causing significant functional limitations), stent placement may be used with confidence to reduce venous obstruction and improve symptoms. An interesting finding of two studies has been that patients with non-thrombotic iliac vein lesions tend to experience significantly better long-term stent patencies than patients with PTS. In one 982-patient study, primary and secondary patency rates at 72 months’ follow-up were 79% and 100%, respectively, for non-thrombotic vascular lesions; but were much lower (57% and 86%, respectively) for lesions in patients with a DVT history. 22 In another study of stents for non-thrombotic iliac vein lesions, a 98% primary stent patency was observed after 4 years’ follow-up. 58 Hence, most patients who receive stents for non-thrombotic lesions probably do not require anticoagulant therapy, since they constitute a population that is relatively ‘non-thrombotic’.
Notwithstanding the above-mentioned favorable results of iliac vein stent placement, it is important to recognize that stent placement carries known risks that include late re-thrombosis, re-stenosis, and migration (rare), as well as unknown risks that stem from the paucity of studies that have followed patients for more than 5 years. When clinical manifestations are severe, or when patients are elderly, these risks are more likely to be justified. However, we have seen two situations where decision-making is more challenging: (i) young or middle-aged patients who have concomitant saphenous reflux; and (ii) young or middle-aged patients who have only mild or moderate symptoms (e.g. moderate daily limb swelling without pain, and no skin changes) and in whom imaging demonstrates a lesion of indeterminate significance. In these situations, the patient’s preferences regarding the therapeutic approach should be discussed and understood by the physician. In general, when clinical manifestations are not severe, we suggest treatment of the saphenous reflux (if present) and a trial of graduated compression stockings (if not already done) as a first approach, with longitudinal follow-up. If symptoms progress to causing greater life impact, the decision to place a stent is better justified.
Conclusion
Overall, it is hoped that future research will provide improved imaging characterization of iliac vein lesions to enable discrimination of those whose treatment will produce symptom relief versus less significant abnormalities, as well as better ways to quantify venous obstruction.
Footnotes
Declaration of conflicting interest and Funding
The authors declare that their employer received research support from Volcano, Inc., for this study.
